Alcohols, Phenols and Ethers : Notes and Study Materials -pdf
Notes and Study Materials
- Concepts of Alcohols, Phenols and Ethers
- Master File Alcohols, Phenols and Ethers
- NCERT Solutions for – Alcohols, Phenols and Ethers
- NCERT Exemplar Solutions for – Alcohols, Phenols and Ethers
- Mind Map of Alcohols, Phenols and Ethers
- Concept Map of Alcohols, Phenols and Ethers
- Past Many 12th Board Years of Alcohols, Phenols and Ethers
Examples and Exercise
CBSE Class 12th Chemistry Notes: Alcohols, Phenols and Ethers
Get important notes on CBSE Class 12 Chemistry: Alcohols, Phenols and Ethers (Part – I). These notes will give you a quick glance of the chapter and are important for revision purpose.
Get revision notes on CBSE Class 12 Chemistry: Chapter 11 – Alcohols, Phenols and Ethers. These notes are based on the latest CBSE syllabus and very helpful for revision purpose.
The main topics covered in this part are:
• Alcohols
o Classification
o Nomenclature
o Preparation
o Physical properties
o Chemical properties
• Test for distinguishing 1o, 2o and 3o alcohols
• Commercially Important Alcohols
o Methanol: preparation, properties and uses
o Ethanol: preparation, properties and uses
• Denaturation of alcohol
The key notes of the chapter are as follows:
Alcohols
An alcohol is any organic compound in which the hydroxyl functional group (–OH) is bound to a saturated carbon atom. For example: methyl alcohol, CH3OH; ethyl alcohol C2H5OH, etc.
Classification of alcohols
(i) On the basis of number of carbons attached to the α-carbon, i.e., the carbon to which the OH group is attached, alcohols are classified as:
Primary (1o) alcohol:
This type of alcohol has only one carbon atom attached to the α-carbon atom.
For example:
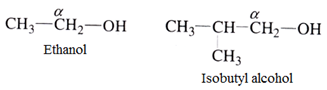
Secondary (2o) alcohol:
Here α-carbon has two other carbon atoms attached to it.
For example:
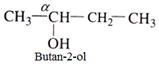
Tertiary (3o) alcohol:
In such alcohol three other carbon atoms are attached to the α-carbon atom.
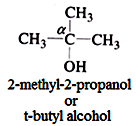
(ii) On the basis of number of hydroxyl groups (−OH) present, alcohols can be divided into the following categories:
Monohydric alcohols:
They contain only one OH group. They have a general formula CnH2n+2O.
For example: CH3OH, CH3CH2OH, CH3CH2CH2OH, etc.
Dihydric alcohols:
Such alcohol contains two OH groups.
For example:
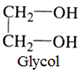
Trihydric alcohols:
This type of alcohol contains three OH groups.
For example:
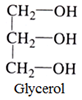
Polyhydric alcohols:
these are the alcohols that contain more than three OH groups.
Nomenclature of alcohols
IUPAC naming system:
(i) Select the longest possible chain that contains the α-carbon
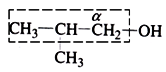
(ii) Number the carbon atoms in the chain in such a way that α-carbon gets the minimum number
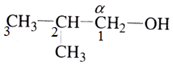
(iii) Then identify the hydrocarbon group and name it as per the IUPAC naming system.
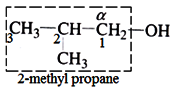
(iv) Now replace the ‘e’ in the end of the parent hydrocarbon name by ‘ol’.
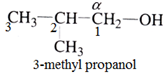
Common naming system:
o The common name of alcohol is alkyl alcohol.
o Depending upon the upon the structure of the alkyl group, prefix n, iso, sec, tert or neo is added to the common name.
‘n’ is added when α-carbon and other carbon atoms make a straight chain.
For example:
![]()
‘Iso’ is added when the ─OH group is attached to
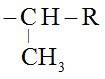
For example:
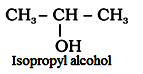
‘sec’ is added when the α-carbon is attached to two other carbon atoms.
For example:
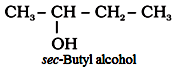
‘tert’is added when the α-carbon is attached to three other carbon atoms.
For example:
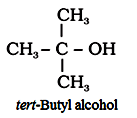
‘Neo’ is used when quaternary carbon is present.
For example:
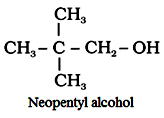
Preparation of alcohols
General methods of preparation of alcohols are described below:
(a) From alkenes:
(i) By acid catalysed hydration of alkenes in accordance with Markownikoffs rule.
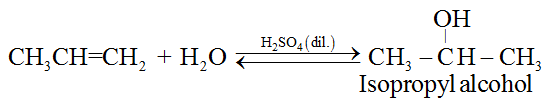
(ii) By hydroboration-oxidation of alkenes in accordance with anti-Markownikoffs rule.
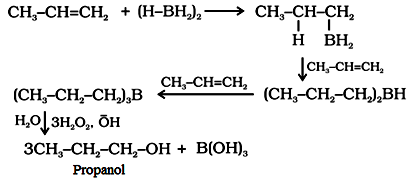
(b) From carbonyl compounds:
(i) By catalytic hydrogenation of aldehydes and ketones.
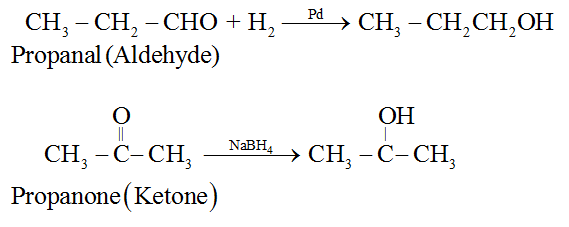
Note : Aldehydes yield primary alcohols whereas ketones give secondary alcohols.
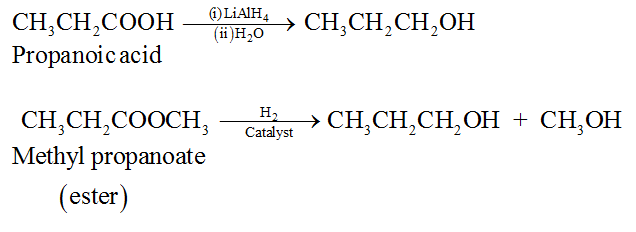
(ii) By catalytic reduction of carboxylic acids and esters.
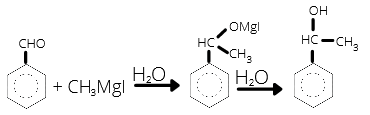
(c) Form Grignard reagent, RMgX:
It gives R group as a nucleophile to attack at the partrially positive carbon atom of carbonyl (–C=O) group.
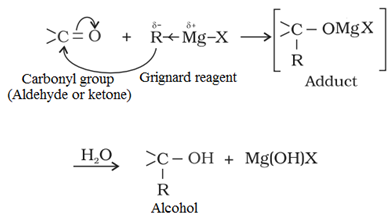
Physical properties of alcohols
• Lower alcohols are liquid at room temperature while higher ones are solid.
• The boiling point of alcohols is higher than haloalkanes with the same number of carbon atoms. This is due to the fact that alcohols are associated via intermolecular hydrogen bonding. For the same alcohol, as the branching increases, boiling point decreases due to decrease in surface area.
• Lower alcohols are completely miscible in water due to their ability to form hydrogen bonds with water. Solubility in water decreases with increase in molecular mass due to a decrease in the extent of intermolecular hydrogen bonding.
• Alcohols are weaker acids than water due to the lower polarity of O – H bond in alcohols. The acidic strength of alcohols decreases as the number of electron donating groups increase at carbon.
Chemical properties of alcohols
(a) Reactions of alcohols involving cleavage of O – H bond
• Reaction with metals:
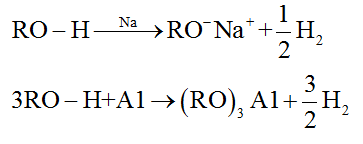
• Reaction with carboxylic acid (Esterification):
![]()
• Reaction with Grignard reagent:
![]()
(b) Reactions involving cleavage of C – O bond
For the reaction involving cleavage of C – O bond, the reactivity order is
![]()
• Formation of halides:
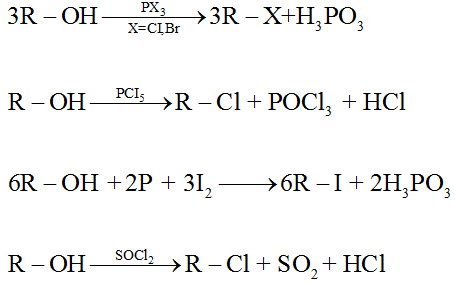
(c) Oxidation of alcohols
Depending upon the structure of the alcohol and the type of oxidising agent used, oxidation of alcohol gives different products.
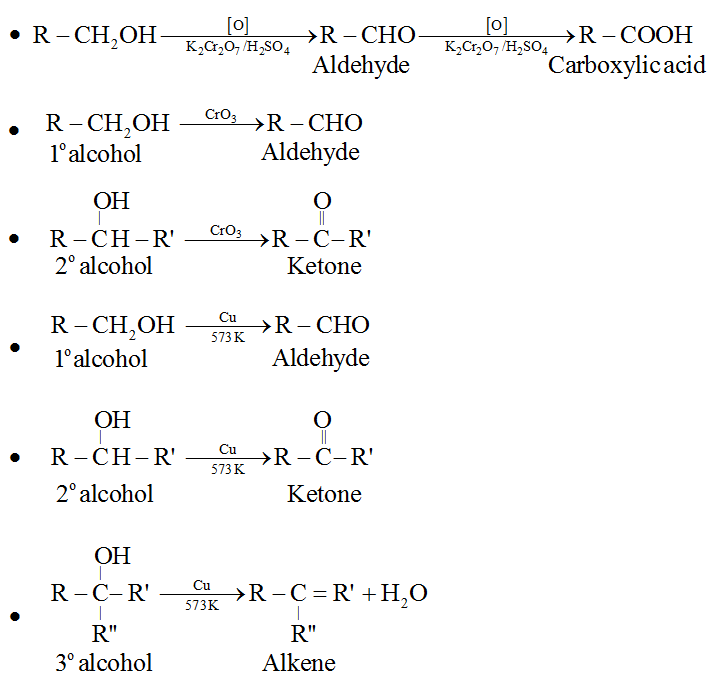
(d) Dehydrogenation of alcohols
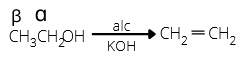
Dehydration of alcohols takes place in acidic medium. Intra-molecular dehydration leads to the formation of alkene while inter molecular dehydration forms ether.
Order of ease of dehydration is: 3° > 2° > 1°

(e) Dehydration of alcohols
Dehydration of alcohols with conc. H2SO4 at different temperatures yields different products.
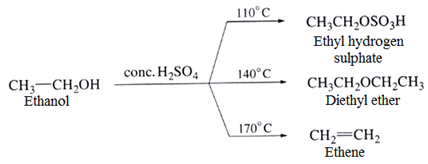
The ease of dehydration of three alcohols increases in the order
1o ROH < 2o ROH < 3o ROH
Test for distinguishing 1o, 2o and 3o alcohols
Lucas reagent which s a mixture of conc. HCI and anhyd. ZnCl2 is used to distinguish and alcohols.
• 3o alcohol gives cloudiness or turbidity with Lucas reagent immediately.
• 2o alcohol gives cloudiness or turbidity after 5 – 10 min.
• 1o alcohol does not give cloudiness or turbidity at room temperature.
Commercially Important Alcohols
Two commercially important alcohols are methanol and ethanol.
1. Methanol, CH3OH
Preparation:
Methanol is mainly produced by catalytic hydrogenation of carbon monoxide at high pressure and temperature and in the presence of ZnO – Cr2O3 catalyst.
![]()
Properties:
• It is a colourless liquid and highly poisonous.
• It is completely soluble in water.
Uses:
• It is used as a solvent for paints, varnishes, and for making formaldehyde.
2. Ethanol, C2H5OH
Preparation:
Ethanol is mainly obtained commercially by fermentation of sugars. The sugar sugarcane or fruits such as grapes is converted to glucose and fructose, in the presence of an enzyme, invertase. Glucose and fructose on fermentation in the presence of another enzyme, zymase, yield ethanol.
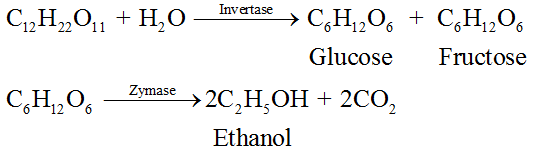
Properties:
• Ethanol is a colourless liquid.
• The boiling point of ethanol is higher than methanol.
Uses:
• It is used as s solvent in paint industry.
• It is also used in the preparation of a number of carbon compounds.
Denaturation of alcohol
The commercial alcohol is made unfit for drinking by mixing in it some copper sulphate and pyridine. This is known as denaturation of alcohol.
In Part-I you got acquainted with the alcohols, their classification, preparation, properties, etc.
In Part-II, you will get to know about the phenols, their preparation, properties and uses. These CBSE revision notes are based on the latest CBSE syllabus for CBSE Class 12th Chemistry.
The main topics covered in this part are:
o Phenols
• Classification
• Nomenclature
• Preparation
• Physical properties
• Chemical properties
• Kolbe’s reaction, Reimer-Tiemann reaction, Fries rearrangement
• Uses of Phenol
The key notes of the chapter are as follows:
Phenols
Phenols are the compounds formed when a hydrogen atom in an aromatic ring, is replaced by a hydroxyl group (-OH group).
For example:
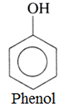
Classification of phenols
On the basis of number of hydroxyl groups (−OH) present, phenols can be divided into the following categories:
• Monohydric phenols:
They contain only one OH group.
For example:

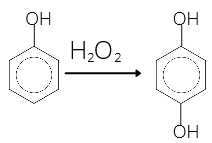
• Dihydric phenols:
Such phenols contains two OH groups.
For example:
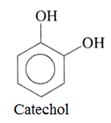
• Trihydric alcohols:
This type of phenol contains three OH groups.
For example:
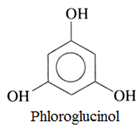
Nomenclature of Phenols
Common naming system:
• The simplest hydroxy derivative of benzene is a phenol. Phenol is the common name as well as an accepted IUPAC name of the compound.
• In the case of substituted phenol compounds, the terms ortho (1,2- disubstituted), meta (1,3-disubstituted) and para (1,4-disubstituted) are often used as prefixes in the common names.
For example:
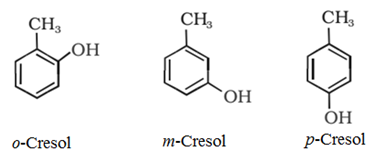
IUPAC naming system:
IUPAC names for some important phenolic compounds are given below
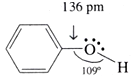
Structure of phenol
In phenols, the –OH group is attached to sp2 hybridised carbon of an aromatic ring. The C–O bond length (136 pm) in phenol is slightly less than that in methanol. This is due to (i) partial double bond character caused by the conjugation of unshared electron pair of oxygen with the aromatic ring and (ii) sp2 hybridised state of carbon to which oxygen is attached. The C−O−H bond angle in alcohols is slightly less than the tetrahedral angle (109°-28′) due to the repulsion between two lone pairs of electrons present on oxygen.
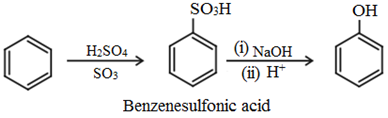
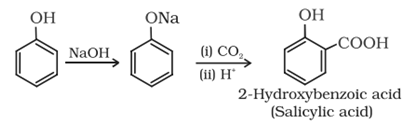
Preparation of phenol
General methods of preparation of phenols are described below:
(a) From benzene sulphonic acid:
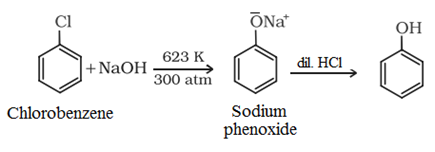
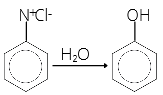
(b) From haloarenes:
(c) From diazonium salt:
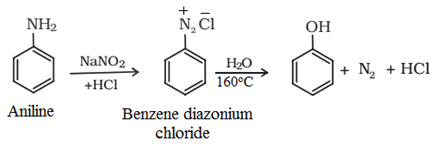
Commercial Preparation:
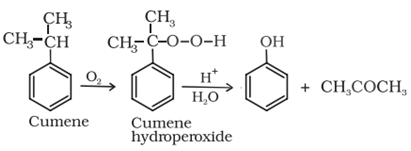
Phenol is prepared commercially by cumene-phenol process in which cumene (isopropylbenzene) is oxidised to cumene hydroperoxide which on treating with dilute acid yields phenol and acetones.
Physical properties of Phenols
• Phenols are colourless liquids or crystalline solids but become coloured due to slow oxidation with air.
• Due to the presence of strong intermolecular hydrogen bonding, phenols have a higher boiling point than the corresponding hydrocarbon or aryl halides.
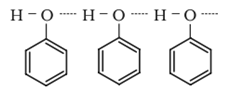
• Due to their ability to form hydrogen bonds with water, phenols are moderately soluble in H2O.
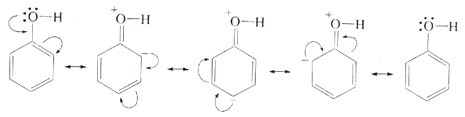
• The phenols are acidic in nature and stronger acids than alcohols. This is due to the fact that the sp2 hybridised carbon of phenol to which −OH is attached, is highly electronegative which causes a decrease in electron density on oxygen. This Increases the polarity of O−H bond and results in an increase in ionisatlon of phenols than that of alcohols.
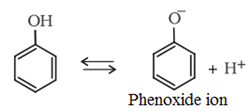
Moreover, the phenoxide ion so produced is stabilised by the delocalization of charge in phenol.
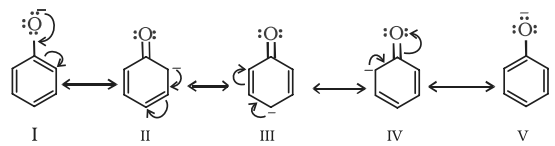
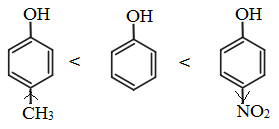
Note: The presence of electron withdrawing group like NO2 group, increases the acidic strength whereas the electron donating groups like an alkyl group decreases the acidic strength. Therefore, the acidic strength order is
Chemical properties of phenols
Eelectrophilic substitution reactions
The presence of OH group on benzene increases the electron density on the benzene ring making it more susceptible to attack by an electrophile. The reactions involving benzene ring are electropnilic substitution reaction. The presence of OH group makes the orthoand para carbon of benzene more electron rich than meta position. The OH group is called o ‒, p ‒ directing group.
Reactions of phenol involving the cleavage of O–H bond
• Kolbe’s reaction:
• Reimer-Tiemann reaction:
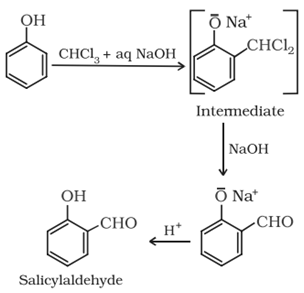
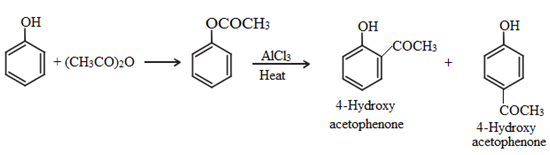
• Fries rearrangement:
Esters of phenol gives phenolic ketones on rearrangement in the presence of anhydrous AlCl3. This reaction is called fries rearrangement.
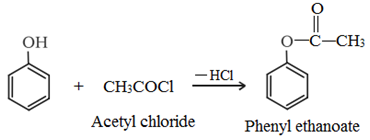
• Acetylation:
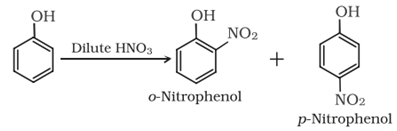
• Nitration:
Reaction with dilute HNO3:
• Reaction with conc. HNO3:
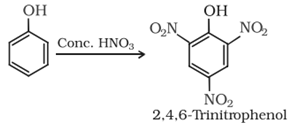
Halogenation:
Bromination in solvents of low polarity like CS2:
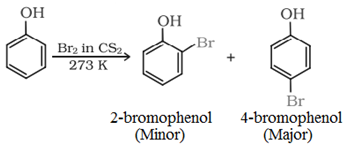
Reaction of phenol with bromine water:
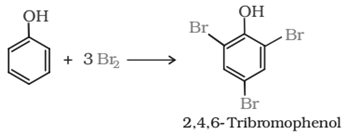
Reactions of phenol involving cleavage of C−O bond
• Reaction with zinc dust:
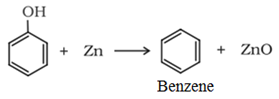
• Reaction with ammonia:
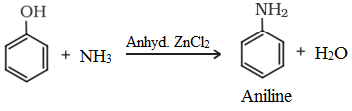
Uses of phenol
• It is used as an antiseptic.
• It is used as a disinfectant in household cleaners.
• It is used in the preparation of resins, dyes, explosives, lubricants, pesticides, plastics, drugs, etc.
Alcohols, Phenols and Ethers Class 12 Chemistry MCQs
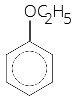
1. The heating of phenyl methyl ether with HI produces
(a) Iodobenzene
(b) Phenol
(c) Benzene
(d) Ethyl chloride
Answer/Explanation
Answer: b
Explaination: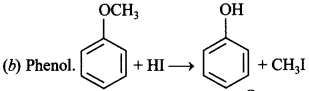
2. Which of the following gives positive iodoform test?
Answer/Explanation
Answer: c
Explaination: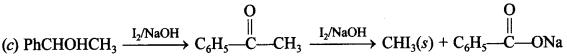
3.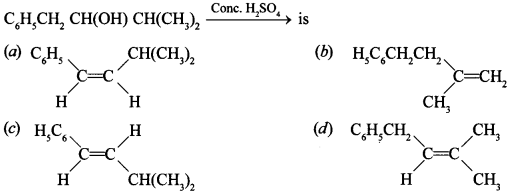
Answer/Explanation
Answer: c
Explaination: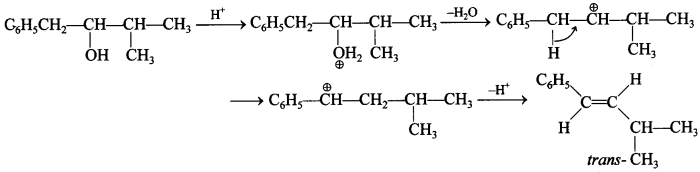
4. CH3—O—CH (CH3)2 + HI → Products is/are
Answer/Explanation
Answer: d
Explaination: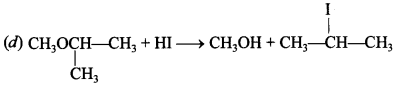
5.![]()
The product ‘Z’ is
(a) Benzaldehyde
(b) Benzoic acid
(c) Benzene
(d) Toluene
Answer/Explanation
Answer: b
Explaination: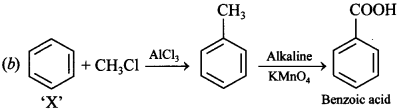
6. Which one of the following compounds has the most acid nature?
Answer/Explanation
Answer: d
Explaination:
(d) Phenol is most acidic in nature because phenoxide ion is stabilized by resonance.
7. Among the following sets of reactants which one produces anisole?
(a) CH3CHO : RMgX
(b) C6H5OH; NaOH, CH3I
(c) C6H5OH, neutral FeCl3
(d) C6H5 —CH3; CH3COCl; AlCl3
Answer/Explanation
Answer: b
Explaination: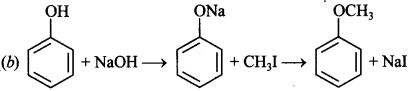
8. The electrophile involved in Riemer-Tiemann reaction of phenol with CHCl3 in presence of NaOH![]()
Answer/Explanation
Answer: a
Explaination:![]()
9. Arrange the following in decreasing order of acidic character: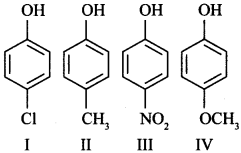
(a) IV > III > I > II
(b) II > IV > I > III
(c) I > II > III > IV
(d) III > I > II > IV
Answer/Explanation
Answer: d
Explaination:
(d) – Since, N02 and Cl are electron withdrawing but NO2 > Cl.
– OCH3 is more electron releasing due to +R effect than —CH3 group, therefore, IV is least acidic.
10.![]()
(a) Etard reaction
(b) Gattermann Koch reaction
(c) Williamson synthesis
(d) Esterification
Answer/Explanation
Answer: c
Explaination:
(c) Williamson synthesis.
11. Identify ‘C’ in the following: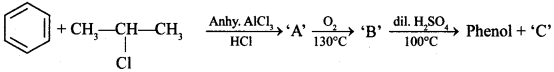
(a) Water
(b) Ethanol
(c) Propanone
(d) Cumene hydroperoxide
Answer/Explanation
Answer: c
Explaination: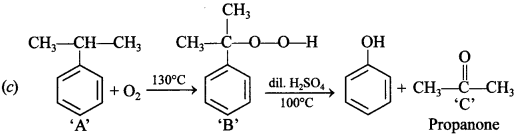
12.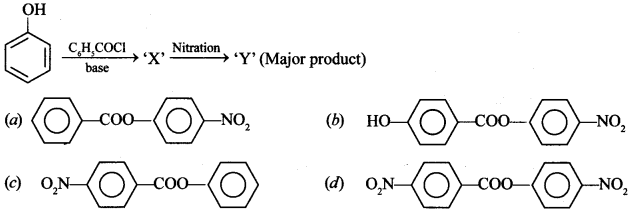
Answer/Explanation
Answer: a
Explaination: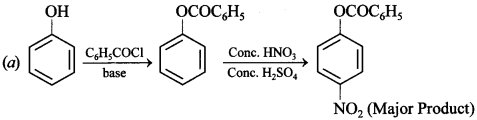
13. Find the product of the given reaction: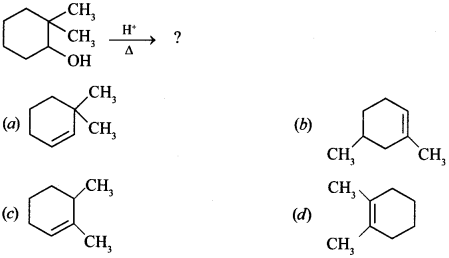
Answer/Explanation
Answer: a
Explaination: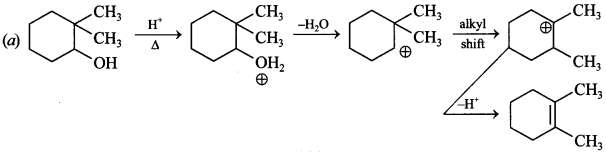
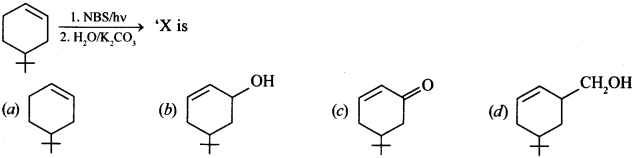
14.
Answer/Explanation
Answer: b
Explaination: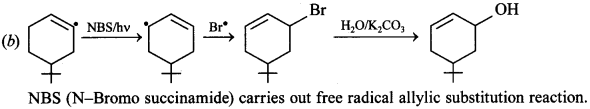
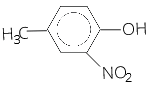
15. Monochlorination of toluene in sunlight followed by hydrolysis with aq. NaOH yields. [NCERT Exemplar]
(a) o-Cresol
(b) m-Cresol
(c) 2, 4-Dihydroxytoluene
(d) Benzyl alcohol
Answer/Explanation
Answer: a
Explaination: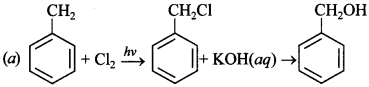
16. How many alcohols with molecular formula C4H10O are chiral in nature? [NCERT Exemplar]
(a) 1
(b) 2
(c) 3
(d) 4
Answer/Explanation
Answer: a
Explaination: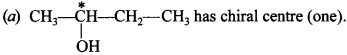
17. What is the correct order of reactivity of alcohols in the following reaction? [NCERT Exemplar]![]()
(a) 1° > 2° > 3°
(b) P < 2° > 3°
(c) 3° > 2° > 1°
(d) 3° > 1° > 2°
Answer/Explanation
Answer: c
Explaination:
(c) 3° >2° > 1° is order of stability of carbocation .’. Reactivity follows the same order.
18. CH3CH2OH can be converted into CH3CHO by __________ . [NCERT Exemplar]
(a) catalytic hydrogenation
(b) treatment with LiAlH4
(c) treatment with pyridinium chlorochromate
(d) treatment with KMnO4
Answer/Explanation
Answer: c
Explaination:![]()
19. IUPAC name of the compound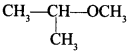
is _________ . [NCERT Exemplar]
(a) 1-methoxy-l-methylethane
(b) 2-methoxy-2-methylethane
(c) 2-methoxypropane
(d) isopropylmethyl ether
Answer/Explanation
Answer: c
Explaination:
(c) 2-methoxypropane
20. Which of the following species can act as the strongest base?
Answer/Explanation
Answer: b
Explaination:
(b) ∵ ROH is weakest acid,
∴ RO⊖ is strongest conjugate base.
21. The correct order of boiling point of primary (1°), secondary (2°) and tertiary (3°) alcohols is
(a) 1° > 2° > 3°
(b) 3° > 2° > 1°
(c) 2° > 1° > 3°
(d) 2° > 3° > 1°
Answer
Answer: a
22.
The above reaction is known as
(a) Williamson’s reaction.
(b) Hofmann’s synthesis.
(c) Mendies reaction,
(d) Darzen’s reaction.
Answer
Answer: d
23. The correct acidic strength order of the following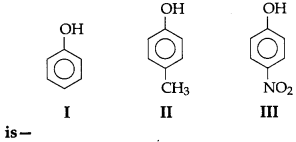
(a) I > II > III
(b) III > I > II
(c) II > III > I
(d) I > III > II
Answer
Answer: b
24. Which compound is predominantly formed when phenol is allowed to react with bromine in aqueous medium?
(a) Picric acid
(b) O-Bromophenol
(c) 2, 4, 6-Tribromophenol
(d) p-Bromophenol
Answer
Answer: c
25. Phenols are more acidic than alcohols because
(a) Phenoxide ion is stablised by resonance
(b) Phenols are more soluble in polar solvents
(c) Phenoxide ion does not exhibit resonance
d) Alcohols do not lose H atoms at all
Answer
Answer: a
26. The compound B is formed in the sequence of the reaction given below:![]()
The compound B is
(a) Salicylaldehyde
(b) Benzoic acid
(c) Salicylic acid
(d) Cinnamic acid
Answer
Answer: c
27. Which of the following reagents cannot be used to distinguish between phenol and benzyl alcohol?
(a) FeCl3
(b) Litmus soln
(c) Br2/CCl4
(d) All of these
Answer
Answer: c
28. Identify Z in the series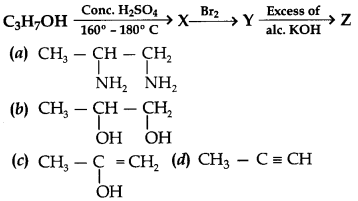
Answer
Answer: d
29. 1-propanol and 2-propanol can be best dis¬tinguished by
(a) Oxidation with KMnO4 followed by reaction with Fehling solution.
(b) Oxidation with acidic dichromate followed by reaction with Fehling solution.
(c) Oxidation by heating with copper followed by reaction with Fehling solution.
(d) Oxidation with concentrated H2SO4 followed by reaction with Fehling solution.
Answer
Answer: c
30. ![]()
In the above sequence Z is
(a) Toluene
(b) Cresol
(c) Benzene
(d) Benzol
Answer
Answer: c
31. The major organic product in the reaction, CH3 — O — CH(CH3)2 + HI → product: is/are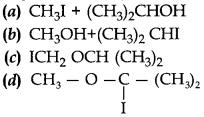
Answer
Answer: a
Note: In the following questions two or more options may be correct. (Q.21 to Q.23)
32. Which of the following are used to convert RCHO into RCH2OH? [NCERT Exemplar]
(a) H2/Pd
(b) LiAlH4
(c) NaBH4
(d) Reaction with RMgX followed by hydrolysis
Answer/Explanation
Answer:
Explaination:
(a), (b), (c) will convert RCHO into RCH2OH.
33. Which of the following reactions will yield phenol? [NCERT Exemplar]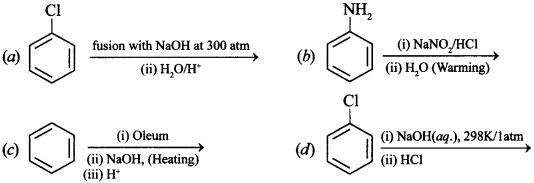
Answer/Explanation
Answer:
Explaination:
(a), (b), (c) will give phenol, (d) will not give phenol because high temperature is needed.
34. Which of the following reagents can be used to oxidise primary alcohols to aldehydes? [NCERT Exemplar]
(a) CrO3 in anhydrous medium.
(b) KMnO4 in acidic medium.
(c) Pyridinium chlorochromate.
(d) Heat in the presence of Cu at 573K.
Answer/Explanation
Answer:
Explaination:
(a), (c), (d) can be used. KMnO4 converts primary alcohol to carboxylic acids.
35. Match the structures of the compounds given in Column I with the name of the compounds given in Column II. [NCERT Exemplar]
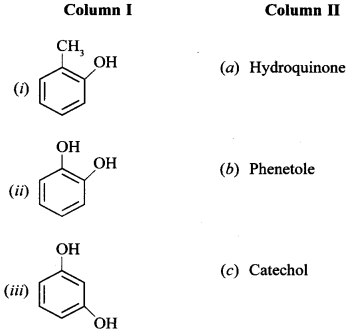
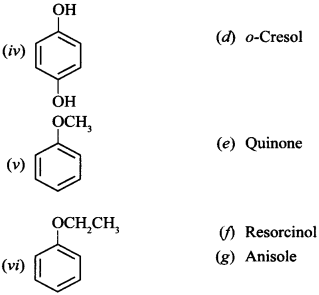
Answer/Explanation
Answer:
Explaination:
(i) (d) (ii) (c) (iii) (J) (iv) (a) or quinol (v) (g) (vi) (b)
36. Match the starting materials given in Column I with the products formed by these (Column II) in the reaction with HI. [NCERT Exemplar]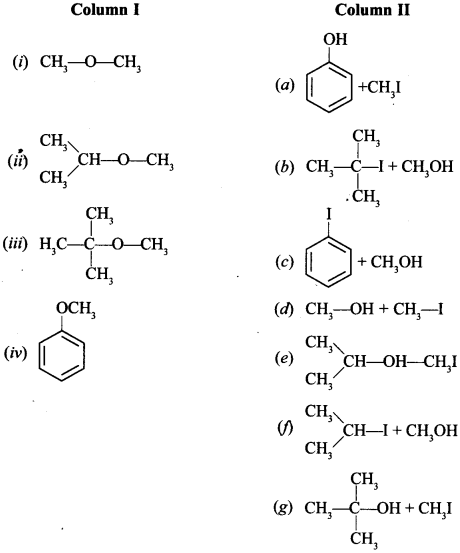
Answer/Explanation
Answer:
Explaination:
(i) (d) (ii) (e) (iii) (b) (iv) (a)
In cleavage of ethers, higher alcohol and lower alkyl halide is formed except in tert. butyl methyl ether, in which (CH3)3 C+ is more stable so (CH3)3C-I and CH3OH is formed. Anisole with HI gives phenol and CH3I because phenoxide ion is stabilised by resonance.
37. In the following question a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices.
(a) Assertion and reason both are correct and reason is correct explanation of assertion.
(b) Assertion and reason both are wrong statements.
(c) Assertion is correct statement but reason is wrong statement.
(d) Assertion is wrong statement but reason is correct statement.
(e) Both assertion and reason are correct statements but reason is not correct explanation of assertion.
Assertion: Bond angle in ethers is slightly less than the tetrahedral angle.
Reason: There is a repulsion between the two bulky (—R) groups. [NCERT Exemplar]
Answer/Explanation
Answer:
Explaination:
(d) Assertion is wrong statement but reason is correct statement.
38. Phenol reacts with Br2 in CS2 to give _________ as major product.
Answer/Explanation
Answer:
Explaination: p-bromophenol
39. Phenol gives o and p-nitrophenol with _________ .
Answer/Explanation
Answer:
Explaination: dil. HNO3
40. o-nitrophenol has _________ melting point than /j-nitrophenol.
Answer/Explanation
Answer:
Explaination: lower
41. Diethyl ether has dipole moment because they are bent molecule. [True/False]
Answer/Explanation
Answer:
Explaination: True
42. Ethers have lower boiling point than alcohol. [True/False]
Answer/Explanation
Answer:
Explaination:
True. It is due to weak van der Waals’ forces of attraction in ethers but intermolecular H-bonding in alcohol.
43. Dettol contains phenolic compound. [True/False]
Answer/Explanation
Answer:
Explaination: True, phenols are antiseptic in low concentration.
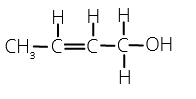
44. Write the IUPAC name of the given compound: [Foreign 2015]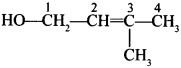
Answer/Explanation
Answer:
Explaination: 3-methyl but-2-en-1-ol.
45. Write the IUPAC name of the given compound: [AI2015]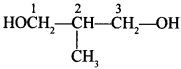
Answer/Explanation
Answer:
Explaination: 2-methyl propane 1, 3-diol.
46. Write the IUPAC name of the following compound: [Foreign 2012]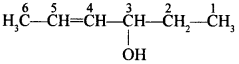
Answer/Explanation
Answer:
Explaination: Hex-4-en-3-ol.
47. Write IUPAC name of the following: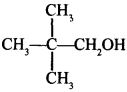
Answer/Explanation
Answer:
Explaination: 2, 2-Dimethyl propan-l-ol.
48. Write the IUPAC name of the following: [AI 2015 Bhubneshwar]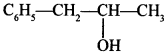
Answer/Explanation
Answer:
Explaination: l-Phenylpropan-2-ol.
49. Write IUPAC name of the compound: [Delhi 2015(C)]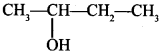
Answer/Explanation
Answer:
Explaination: Butan-2-ol
50. Write IUPAC name of the given compound: [AI 2015 Patna]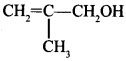
Answer/Explanation
Answer:
Explaination: 2-methyl prop-2-en-l-ol.
51. Draw the structural formula of 2-methylpropan-2-ol molecule. [Delhi 2012]
Answer/Explanation
Answer:
Explaination: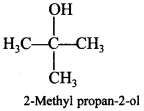
52. Draw the structure of hex-l-en-3-ol compound.
Answer/Explanation
Answer:
Explaination: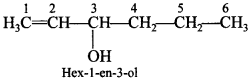
53. How is ethanol obtained from 2-butene? [Delhi 2012]
Answer/Explanation
Answer:
Explaination: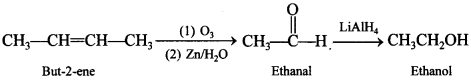
54. Write the IUPAC name of the given compound: [Delhi 2015]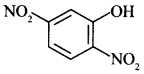
Answer/Explanation
Answer:
Explaination: 2, 5-Dinitro phenol.
55. Write IUPAC name of the given compound: [AI 2015 Guwahati]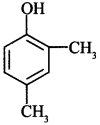
Answer/Explanation
Answer:
Explaination: 2, 4-Dimethyl phenol.
56. Draw the structure of 2, 6-dimethylphenol. [AI 2011]
Answer/Explanation
Answer:
Explaination: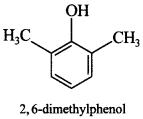
57. Write the IUPAC name of the given compound: [AI 2015]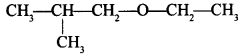
Answer/Explanation
Answer:
Explaination: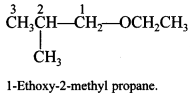
58. Write IUPAC name of the given compound: [AI 2015 Chennai and Trivandrum]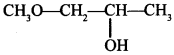
Answer/Explanation
Answer:
Explaination: 1-Methoxypropan-2-ol.
59. Write the structure of the following compound: 2-ethoxy-2-methyl pentane
Answer/Explanation
Answer:
Explaination: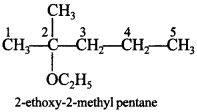
60. How is /-butyl alcohol obtained from acetone?
Answer/Explanation
Answer:
Explaination: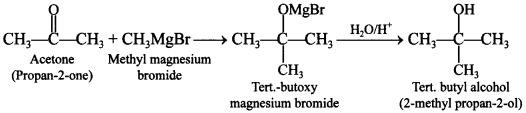
61. Write the chemical equation for the preparation of phenol from benzene using oleum and sodium hydroxide. [Delhi 2011(C)]
Answer/Explanation
Answer:
Explaination: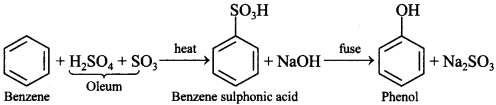
62. Why alcohols and phenols are soluble in water?
Answer/Explanation
Answer:
Explaination: This is due to their ability to form hydrogen bonds with water molecules.
63. Which of the following isomers is more volatile: o-nitrophenol or p-nitrophenol? [Delhi 2014]
Answer/Explanation
Answer:
Explaination: o-nitrophenol is more volatile, cone. H2SO4
64. How would you convert ethanol to ethene? [AI 2011]
Answer/Explanation
Answer:
Explaination: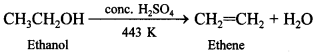
65. Name a compound which is used as antiseptic as well as disinfectant.
Answer/Explanation
Answer:
Explaination:
0.2% solution of phenol is used as antiseptic, 2% solution of phenol is used as disinfectant.
66. Arrange the following in decreasing order of their acidic character: [Similar to AI 2017]![]()
Answer/Explanation
Answer:
Explaination: (ii) > (iii) > (i)
67. Illustrate with examples the limitations of Williamson’s synthesis for the preparation of certain types of ethers.
Answer/Explanation
Answer:
Explaination:
Di-tertiary butyl ether cannot be prepared by Williamson’s synthesis because tertiary halides will undergo elimination reaction instead of substitution reaction.
68. What is the cause of large difference in boiling points of alcohols and ethers?
Answer/Explanation
Answer:
Explaination:
This is due to the presence of hydrogen bonding in alcohols but ether have weak van der Waals’ forces of attraction.
69. Convert anisole to p-bromoanisole. [Delhi 2015(C)]
Answer/Explanation
Answer:
Explaination: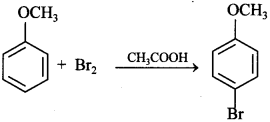
70. How will you synthesise the following from appropriate alkene: [Uttarakhand 2019]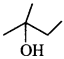
Answer/Explanation
Answer:
Explaination: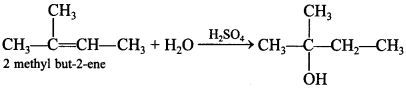
71. Show how you will synthesise the following alcohol prepared by reaction of a suitable Grignard reagent on methanal? [Uttarakhand 2019]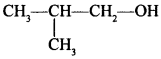
Answer/Explanation
Answer:
Explaination: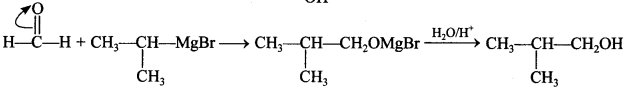
Alcohols Phenols and Ethers MCQ (1-10)
- Phenol, when it first reacts with concentrated sulphuric acid and then with concentrated nitric acid, gives
- 2,4,6-Trinitrobenzene
- o-Nitrophenol
- p-Nitrophenol
- Nitrobenzene
- The number of stereoisomers possible for a compound of the molecular formula
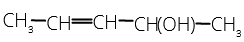
1 . 3 2. 2 3. 4 4. 6
- The compound which reacts the fastest with Lucas reagent at room temperature is
- Butan-1-ol
- 2-methyl-propan-2-ol
- 2-methyl-1-propan-1-ol
- Butan-2-ol
- Which of the isomers of nitrophenol is steam volatile?
- Ortho
- Para
- Meta
- None of these
- A compound develops red colour with a solution of ceric ammonium nitrate. The compound may be
- An alkene
- An alcohol
- A ketone
- An aldehyde
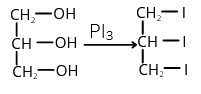
- Glycerol reacts with phosphorus triiodide to form
- Allyl alcohol
- Allyl iodide
- Acrolein
- 1,2,3-triiodopopane
- Choose the reagent to carry out the following reaction
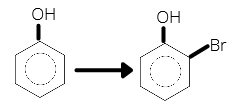
1 . Bromine water 2. Bromine in CCl4
3. Either 1 or 2 4. Reaction is not possible
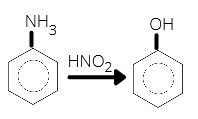
- Benzylamine reacts with nitrous acid to form
- Azobenzene
- Benzene
- Benzyl alcohol
- Phenol
- Which of the following will not be soluble in sodium bicarbonate?
- 2,4,6-Trinitrophenol
- Benzoic acid
- o-Nitrophenol
- Benzenesulphonic acid
- Which of the following gives alkene with ethylene glycol?
- PCl3
- PI3
- PBr3
- None of these
Answers (1-10)
1 . (2)
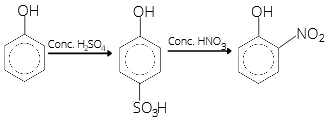
-OH group is the ortho and para directing group, but -SO3H group being bigger preferably occupies the para position. As the temperature is not mentioned, ortho nitrobenzene will be more stable.
2. (3)
One chiral carbon means two enantiomers and two geometrical isomers (cis and trans) are also possible. So, a total of four isomers are possible.
3. (2)
Lucas test is used to distinguish between primary, secondary and tertiary alcohol. As tertiary alcohol is the most stable, it reacts the fastest with the Lucas reagent and causes turbidity. Then comes the secondary alcohol and at last, the primary alcohol. As 2-methyl-propan-2-ol is a tertiary alcohol, it will react the fastest with the Lucas reagent.
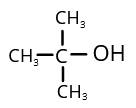
4. (1)
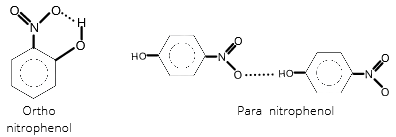
Ortho nitrophenol will be more volatile due to the presence of intramolecular hydrogen bonding. Para nitrophenol has intermolecular hydrogen bonding which is comparatively forms much stronger bonds, due to which the boiling point will also increase.
5. (2)
An alcoholic group gives red colour with ceric ammonium nitrate.
6. (2)
7. (2)
Bromine water
8. (4)
9. (3)
The reaction moves in forward direction if the reactants are more acidic than NaHCO3. As o-nitrophenol is less acidic than NaHCO3, it wont react.
10. (2)
Ethylene glycol + PI3 → Ethylene
Alcohols Phenols and Ethers MCQ (11-20)
- Which of the following is not true in the case of reaction with heated copper at 300℃?
- Phenol → Benzyl alcohol
- Primary alcohol → Aldehyde
- Secondary alcohol → Ketone
- Tertiary alcohol → Olefin
- 1-phenylethanol can be prepared by the reaction of benzaldehyde with
- Methyl bromide
- Ethyl iodide and magnesium
- Methyl iodide and magnesium
- Methyl bromide and aluminium bromide
- What is the reagent required in the following reaction
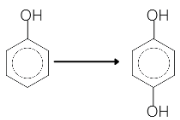
1 . Hydrogen peroxide 2. CrO3
3. Chromyl chloride 4. All of these
- In the following reaction, P is
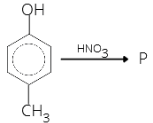
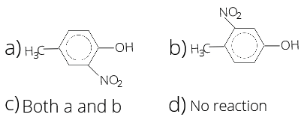
1 . (a) 2. (b) 3. (c) 4. (d)
- Dichloroacetic acid on hydrolysis gives
- Ethane-1,2-dione
- Formylmethanoic acid
- 2-Hydroxyethanoic acid
- Ethane-1,2-diol
- Identify X in the following reaction
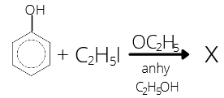
1 . C2H5OC2H5 2. C6H5OC6H5
3. C6H5I 4. C6H5OC2H5
- Which of the following reagent convert the propene to 1-propanol?
- H2O, H2SO4
- Aqueous KOH
- MhSO4, NaBH4/H2O
- B2H6, H2O2, OH–
- Which of the following can not be formed by hydrolysis of alkenes?
- Ethanol
- Propanol
- Methanol
- None of these
- Decolourisation of KMnO4 solution takes place when it reacts with ethene. The final product of this reaction which is used as antifreeze is
- Propylene glycol
- Ethanol
- Propanol
- Ethylene glycol
- 1-Bromopentane on boiling with alcoholic potassium hydroxide gives
- Pentan-1-ol
- Pent-1-ene
- Pentan-2-ol
- Pent-2-ene
Answers (11-20)
11. (1)
Phenol can not be converted to benzyl alcohol because the oxidation of phenol can’t give benzyl alcohol.
12. (3)
When benzaldehyde reacts with methyl iodide and magnesium followed by hydrolysis, the formation of 1-phenylethanol takes place.
13. (1)
14. (1)
15. (2)
Formylmethanoic acid
16. (4)
17. (2)
Aqueous KOH
18. (3)
Hydrolysis of alkene mainly gives alcohol simplest alkene that is ethane on hydrolysis gives ethanol. So methanol can not be formed by hydrolysis of alkene.
19. (4)
Hydrolysis of ethene takes place in the presence of cold KMnO4 to form ethylene glycol (HOCH2-CH2OH).
20. (2)
When haloalkane is treated with conc. alcoholic solution of KOH, a molecule of hydrogen halide is eliminated to form alkene. The eliminated hydrogen atom comes from the B-carbon atom, so it is called B-elimination. Most highly substituted alkene is a major product.
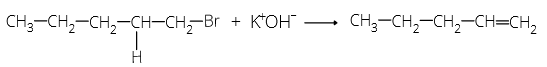
Alcohols Phenols and Ethers MCQ (21-30)
- Among ethanol, dimethyl ether, methanol and propanol, the isomers are
- Ethanol, dimethyl ether, methanol and propanol
- Ethanol and methanol
- Ethanol, dimethyl ether and methanol
- Ethanol and dimethyl ether
- Ether is obtained from ethyl alcohol in presence of H2SO4 at?
- 113 K
- 443 K
- 413 K
- 213 K
- Alkyl halides are converted into ether through
- Frankland reaction
- Williamson synthesis
- Fittig reaction
- Grignard reaction
- The compound having the lowest boiling point is
- H2O
- C2H5OH
- CH3OH
- CH3OCH3
- Which if the following pairs show ideal behavior?
- C6H5CH3 + C6H6
- CH3OH + H2O
- CH3COCH3 + CHCl3
- H2O + HCl
- One of the following which can not undergo dehydro-halogenation is
- Iso-propyl bromide
- Ethanol
- Ethyl bromide
- None of these
- Dehydration of alcohol is an example of
- Substitution reaction
- Elimination reaction
- Rearrangement reaction
- Addition reaction
- Which of the following is the most soluble in water?
- n-Butyl alcohol
- Isobutyl alcohol
- Tertiary alcohol
- Secondary butyl alcohol
- Which of the following is not a character of alcohol?
- They are lighter than water
- Their boiling points rise fairly uniformly with increasing molecular weight
- Lower members are insoluble in water and organic solvents but solubility regularly increases with molecular weight
- Lower members have pleasant smell and burning taste, while higher members are odourless and tasteless
- Enzymes used in the fermentation of cane sugar to alcohol are
- Diastase, invertase
- Invertase, zymase
- Diastase, zymase
- Maltase, zymase
Answers (21-30)
21. (4)
Ethanol (C2H5OH) and dimethyl ether (CH3-O-CH3) have the same molecular formula but different functional groups, so they are isomers.
22. (3)
Ether is obtained from ethyl alcohol at 413 K.
23. (2)
Alkyl halides are converted into ether through Williamson synthesis.
24. (4)
Due to less amount of hydrogen bonding in ether, boiling point will be the less.
25. (1)
C6H5CH3 (Toluene) + C6H6 (Benzene)
26. (2)
Ethanol doesn’t have a halogen group attached. Hence, it can’t undergo dehydrohalogenation.
27. (2)
Dehydration of alcohol is an example of ꞵ-elimination.
28. (3)
Solubility decreases with an increase in size of the alkyl group which is a hydrophobic group, which makes the alcohol less hydrophilic.
In the case of isomers, the order of solubility is 3°>2°>1° due to increase in polar character.
29. (1)
Lower members are soluble in water and solubility decreases with increasing molecular mass because the length of hydrocarbon chain increases.
30. (2)
Invertase and zymase are used in the fermentation of cane sugar to alcohol.
Alcohols Phenols and Ethers MCQ (31-40)
- Cyclohexanol is a
- Primary alcohol
- Secondary alcohol
- Tertiary alcohol
- Phenol
- Which of the following substances can not be used for the replacement of -OH group in organic compounds by Cl?
- S2Cl2
- SOCl2
- PCl3
- PCl5
- The formation of phenol from benzene diazonium chloride is a
- Pyrolysis reaction
- Photosynthesis reaction
- Hydrolysis reaction
- Combustion reaction
- Sodium benzene sulphonate reacts with NaOH and then on acidic hydrolysis, it gives
- Benzoic acid
- Benzene
- Disodium benzaldehyde
- Phenol
- Wood spirit is known as
- Methanol
- Ethanol
- Acetone
- Benzene
- Carbinol is a trivial name of
- C2H5OH
- CH3OH
- HCOOH
- CH3COOH
- Which among the following reactions does not give methyl alcohol?
- The reaction of water gas with hydrogen at high temperature
- Alkaline hydrolysis of methyl bromide
- The reaction of ethylene with H2SO4 at 80℃
- Both 1 and 2
- The other name of syngas is
- Fuel gas
- Tear gas
- Producer gas
- Water gas
- 4-chloro-3,5-dimethyl phenol is called
- Chloramphenicol
- Paracetamol
- Barbital
- Dettol
- IUPAC name of m-cresol is
- 3-methylphenol
- 3-chlorophenol
- 3-methoxyphenol
- Benzene-1,3-diol
Answers (31-40)
31. (2)
In cyclohexanol, -OH is attached to a secondary carbon in the ring.
32. (1)
S2Cl2 can’t be used.
33. (3)
34. (4)
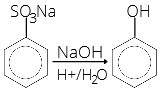
35. (1)
Methanol is known as wood spirit.
36. (2)
Carbinol is a trivial name of methanol (CH3OH).
37. (3)
The reaction of ethylene with H2SO4 at 80℃ gives
CH2CH2 + H2SO4 → CH3CH2OSO2OH
Water gas at high temperature gives
CO + 2H2 → CH3OH
Alkaline hydrolysis of methyl bromide gives methanol
CH3Br + H2O → CH3OH + HBr
38. (4)
The other name of syngas is water gas.
39. (4)
Dettol
40. (1)
3-methylphenol (CH3)C6H5OH
Alcohols Phenols and Ethers MCQ (41-50)
- IUPAC name of picric acid
- M-nitrobenzoic acid
- 2,4,6–trinitrophenol
- Trinitrotoluene
- Trinitroaniline
- Diethyl ether dissociates when it reacts with
- NaOH
- H2O
- HI
- KMnO4
- Cresols are
- Hydroxy toluenes
- Dihydric phenols
- Trihydric phenols
- Trihydric alcohols
- IUPAC name of sec butyl alcohol is
- 1-butanol
- 2-butanol
- 2-methyl-1-butanol
- 2-methyl-2-butanol
- Methyl phenyl ether can be produced by reacting
- Phenolate ions and methyl iodide
- Bromobenzene with methoxide ions
- Methanol and phenol
- Bromobenzene and methyl iodide
- Ortho-nitrophenol is less soluble in water than p- and m-nitrophenol because
- Melting point of o-nitrophenol is lower than of m- and p- isomers
- O-nitrophenol is more volatile in steam than those of m- and p- isomers
- O-nitrophenol shows intramolecular H-bonding
- O-nitrophenol shows intermolecular H-bonding
- Primary alcohols can be prepared from alkenes by
- Mercuration and demercuration of alkene
- Direct hydration of alkenes
- Hydroboration of alkenes
- All of the above
- The reaction of alkoxide ion with alkyl halide to form ether is called
- Wurtz reaction
- Kolbe’s reaction
- Perkins reaction
- Williamsons synthesis
- In which case would a Williamson ether synthesis fail?
- Sodium ethoxide + iodomethane
- Sodium ethoxide + iodoethane
- Sodium ethoxide + 2-iodopropane
- Sodium ethoxide + 2-iodo-2-methylpropane
- Grain alcohol is the common name of
- Ethyl alcohol
- Amyl alcohol
- Methanol
- None of these
Answers (41-50)
41. (2)
2,4,6-Trinitrophenol
42. (3)
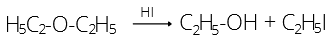
43. (1)
Cresols are hydroxy toluenes
44. (2)
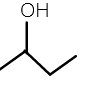
2-butanol
45. (1)
Methyl phenyl ether (anisole) can be produced by reacting phenolate ions and methyl iodide.
46. (3)
Ortho-nitrophenol is less soluble in water than p- and m-nitrophenol because o-nitrophenol shows intramolecular H-bonding.
47. (3)
Primary alcohols can be prepared from alkenes by hydroboration of alkenes.
48. (4)
Williamsons synthesis
49. (4)
2-iodo-2-methylpropane is a tertiary alcohol which will undergo dehydrohalogenation.
50. (1)
Grain alcohol is the common name of ethyl alcohol.
Assertion And Reasoning MCQs
Codes
(a) Both A and R are true and R is the correct explanation of A
(b) Both A and R are true and but R is not a correct explanation of A
(c) A is true but R is false
(d) A is false, but R is true
1. Assertion (A) (CH3)3-CONa and CH3CH2Br react to form (CH3)3C-O-CH2CH3.
Reason (R) Good yields of ethers are obtained when teri-alkyl halides are treated with alkoxide.
2. Assertion (A) Ortho and para nitrophenols can be separated by steam distillation.
Reason (R) Ortho isomer associates through intermolecular hydrogen bonding while Para isomer associates through intramolecular hydrogen bonding.
3. Assertion (A) In Lucas test, 3° Alcohol react immediately.
Reason (R) An equimolar mixture of anhyd. ZnCI2 and conc. HCI is called Lucas reagent.
4. Assertion (A) The water solubility of the alcohols follow the order: tert-butyl alcohol > sec-butyl alcohol > n butyl alcohol .
Reason (R) Alcohols form H-bonding with water to show solution nature.
5. Assertion (A) Tert-butyl alcohol undergoes acid catalysed dehydration readily than propanol .
Reason (R) 3° Alcohol do not give Victor-Meyer’s test.
6. Assertion (A) Phenol is less acidic than p-nitrophenol.
Reason (R) Phenolate ion is more stable than p-nitrophenolate ion.
7. Assertion (A) Reimer-Tiemann reaction of phenol with CCI4 in NaOH at 340 K gives salicylic acid as the major product.
Reason (R) The reaction occurs through intermediate formation of dichlorocarbene.
8. Assertion (A) The C-O-C bond angle in ethers is slightly less than tetrahedral angle.
Reason (R) Due to the repulsive interaction between the two alkyl groups in ethers.
9. Assertion (A) Phenol undergo Kolbe reaction, ethanol does not.
Reason (R) Phenoxide ion is more basic than ethoxide ion.
10. Assertion (A) Etherates are coordination complexes of ethers with Lewis acids.
Reason (R) Ethers are easily cleaved by mineral acids such as HCI and H2SO4 at 373K.
11. Assertion (A) Boiling point of alcohols are higher than that of ethers of comparable molecular mass.
Reason (R) Alcohols can form intermolecular hydrogen bonding while ethers can not.
Assertion and Reasoning MCQ Answers
1. (c)
(CH3)3-CONa and CH3CH2Br react to form (CH3)3C-O-CH2CH3. Good yeild of ether are obtained when primary alkyl halides are treated with alkoxides derived from any alcohol.
2. (c)
Ortho and para iomers of nitro phenol can be separated be steam distillation because of nearby same boiling point of both and ortho ieomers associated by intramolecular hydrogen bonding and para isomers by hydrogen bonding.
3. (b)
In Lucas test, tertiary alcohols react immediately because of the formation of the more stable tertiary carbocation.
4. (b)
The tendency to show H-bonding decreases with increasing hydrophobic character of carbon chain. The hydrophobic character of carbon chain increases with the length of carbon chain.
5. (b)
Alcohols which form the most stable carbocations undergo dehydration more readily. Since tert-butyl alcohol forms more stable tert-butyl cation, therfore, it undergoes dehydration more readily than propanol.
6. (c)
p-Nitrophenolate ion is more stable than phenolate ion.
7. (c)
Nucleophilic attack of phenolate ion through the ortho-carbon atom occurs on CCl4 to form an intermediate which on hydrolysis gives salicylic acid.
8. (d)
In ethers, bond angle around oxygen has deviation caused due to repulsive interaction between bulkier alkyl groups.
9. (b)
On using tert-butyl bromide and sodium ethoxide as reactants, the major product would be 1-methylpropene and ethanol.
10. (c)
Ethers being Lewis bases form etherates with Lewis acids. Ethers are not easily cleaved by H2SO4.
11. (a)
Alcohols have high boiling point than ethers because intermolecular H-bonding is found in alcohols.
