About this unit
Group 15 elements: General introduction, electronic configuration, occurrence, oxidation states, trends in physical and chemical properties; preparation and properties of ammonia and nitric acid, oxides of nitrogen (structure only); Phosphorous- allotropic forms; compounds of phosphorous: preparation and properties of phosphine, halides (PCI3 , PCI5 ) and oxoacids (elementary idea only).
Group 16 elements: General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemical properties; dioxygen: preparation, properties and uses; classification of oxides; ozone. Sulphur – allotropic forms; compounds of sulphur: preparation, preparation, properties and uses of sulphur dioxide; sulphuric acid: industrial process of manufacture, properties and uses, oxoacids of sulphur (structures only).
Group 17 elements: General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemical properties; compounds of halogens: preparation, properties and uses of chlorine and hydrochloric acid, interhalogen compounds oxoacids of halogens (structures only).
Group 18 elements: General introduction, electronic configuration, occurrence, trends in physical and chemical properties, uses.
| p-Block Elements – Nitrogen Family |
| p-Block Elements – Oxygen Family |
| p-Block Elements – Halogens |
p-BLOCK ELEMENTS – OXYGEN FAMILY
GENERAL CHARACTERISTICS
ELECTRONIC CONFIGURATION
Elements | At.No. | Electronic confg. | Valence shell electronic confg |
Oxygen | 8 | ||
Sulphur | 16 | ||
Selenium | 34 | ||
Tellurium | 52 | ||
Polonium | 84 |
The oxygen differs from the rest of the elements due to its-
- small size
- higher electronegativity
- absence of d atomic orbitals in valence shell
- tendency to form multiple bonding
METALLIC AND NON METALLIC CHARACTER
Metal (Radio active) t1/2 138.4 days
ABUNDANCE
DENSITY
MELTING POINT AND BOILING POINT
OXIDATION STATE
O | S | Se | Te | Po |
–1,–2 | –2 to +6 | –2 to +6 | –2 to +6 | –2 to +6 |
IONISATION ENERGY
ELECTRON AFFINITY
ELECTRONEGATIVITY
CATENATION
ATOMICITY
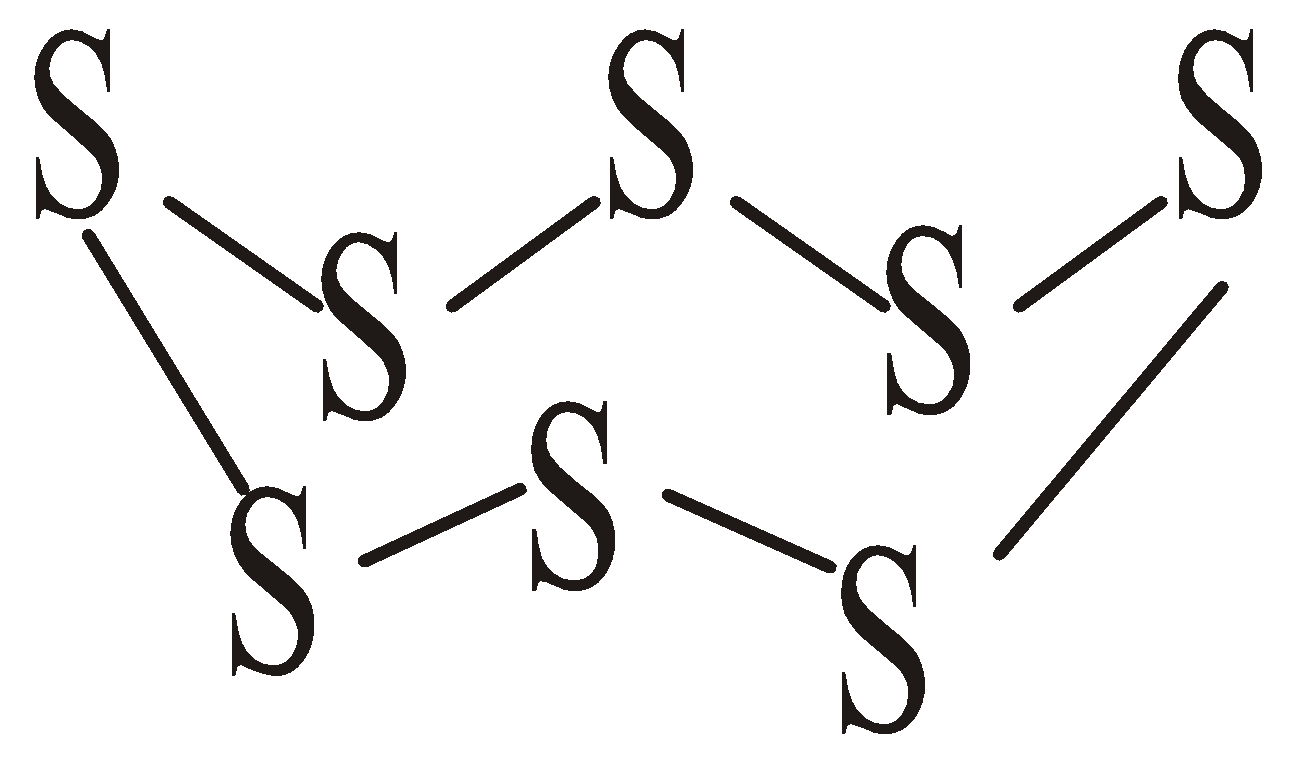
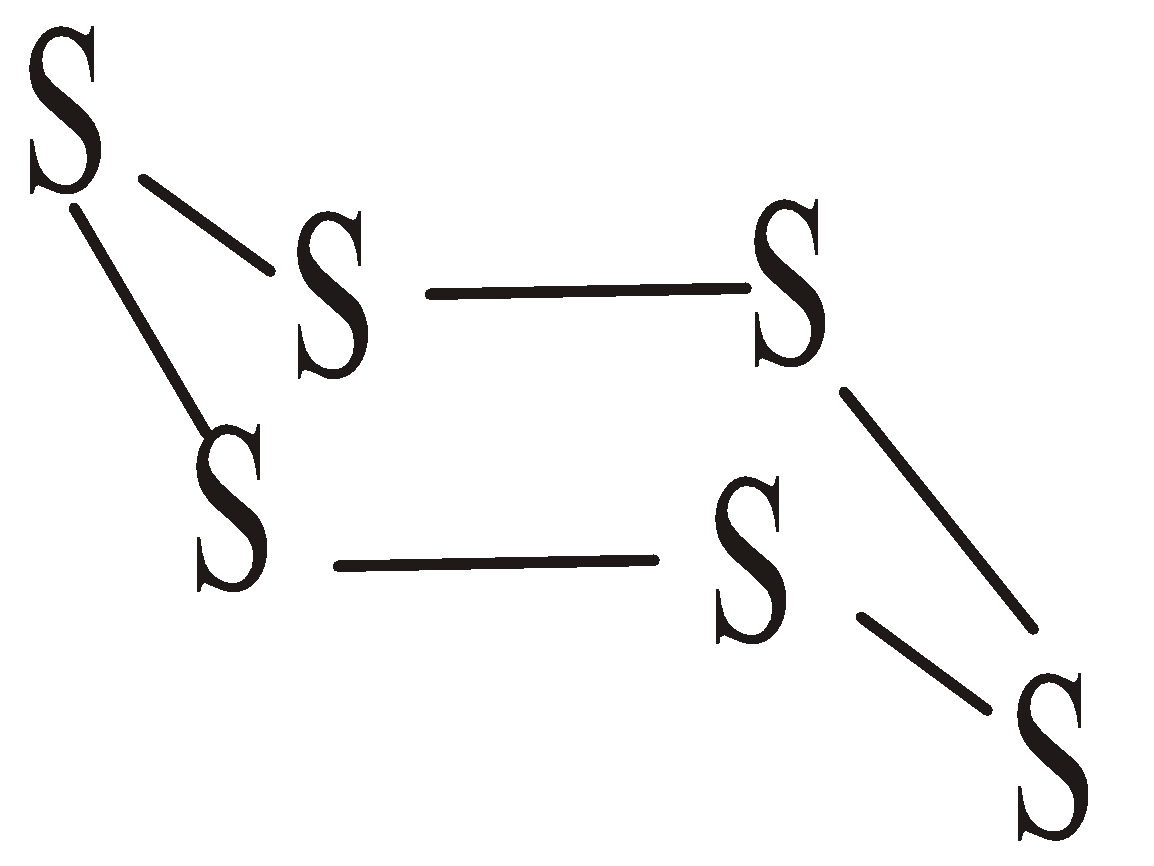
ALLOTROPY
ATOMIC RADII
IONIC RADII
ATOMIC VOLUME
MULTIPLE BOND FORMATION
COMPOUNDS OF SIX GROUP ELEMENTS
HYDRIDES
HALIDES
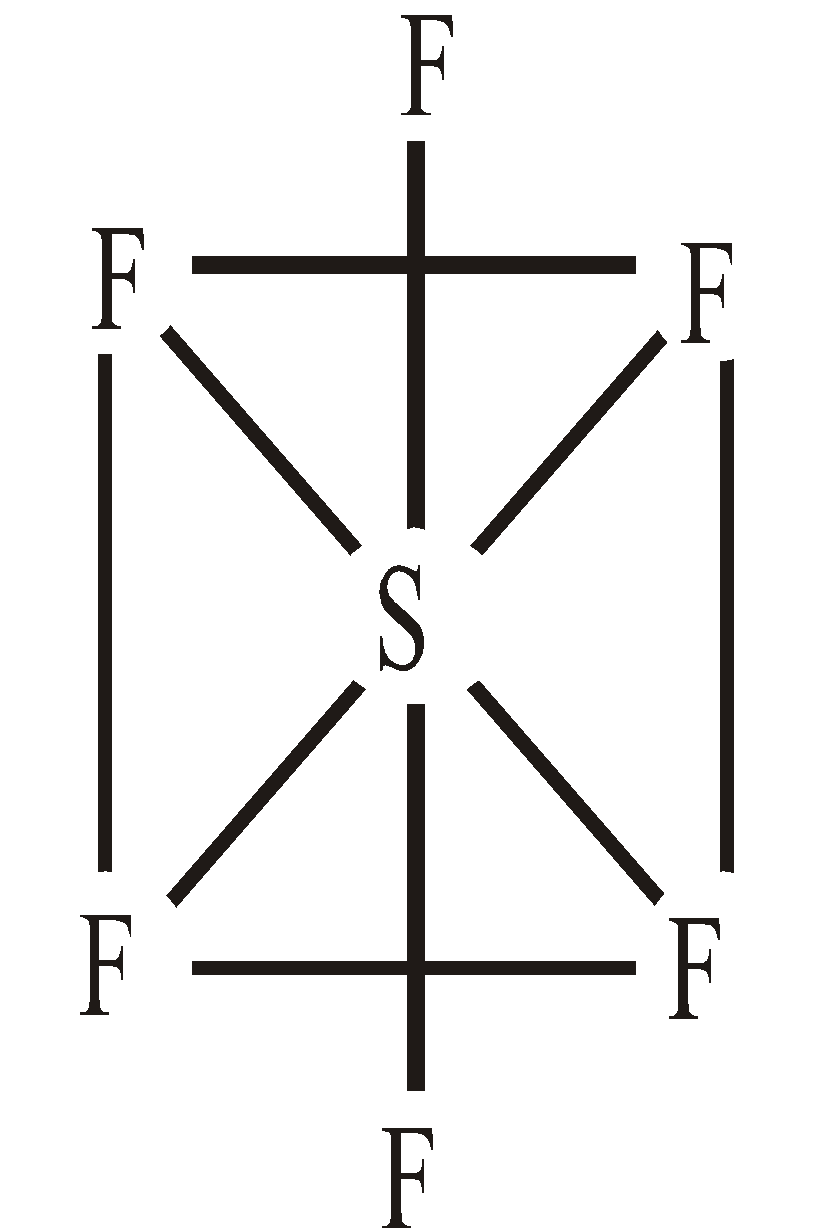
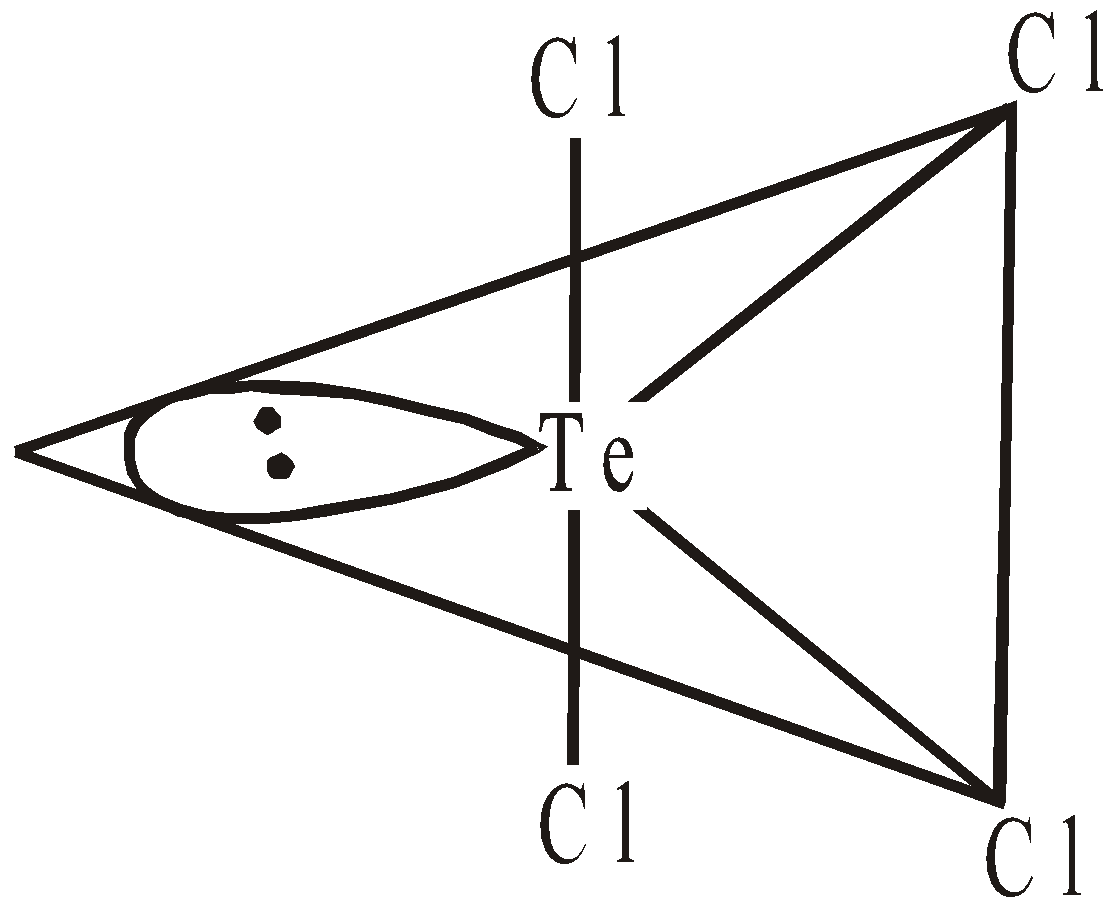
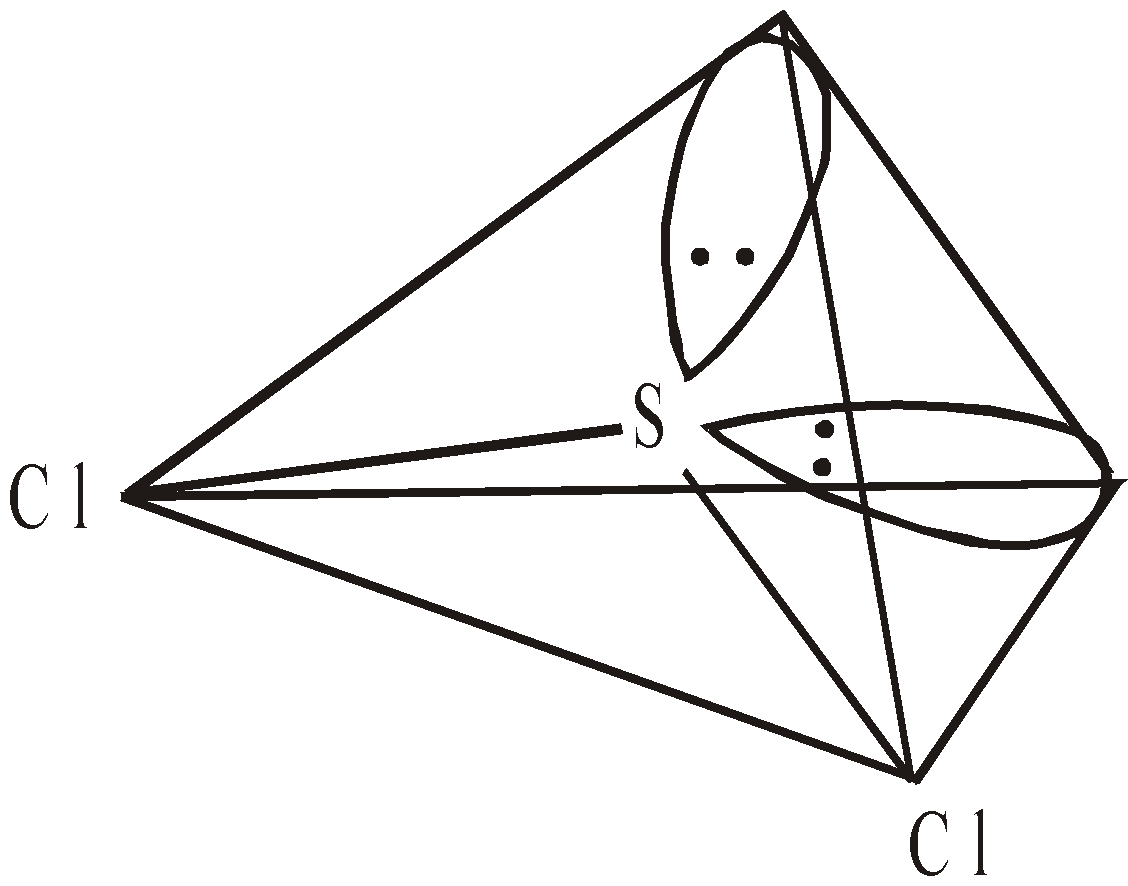
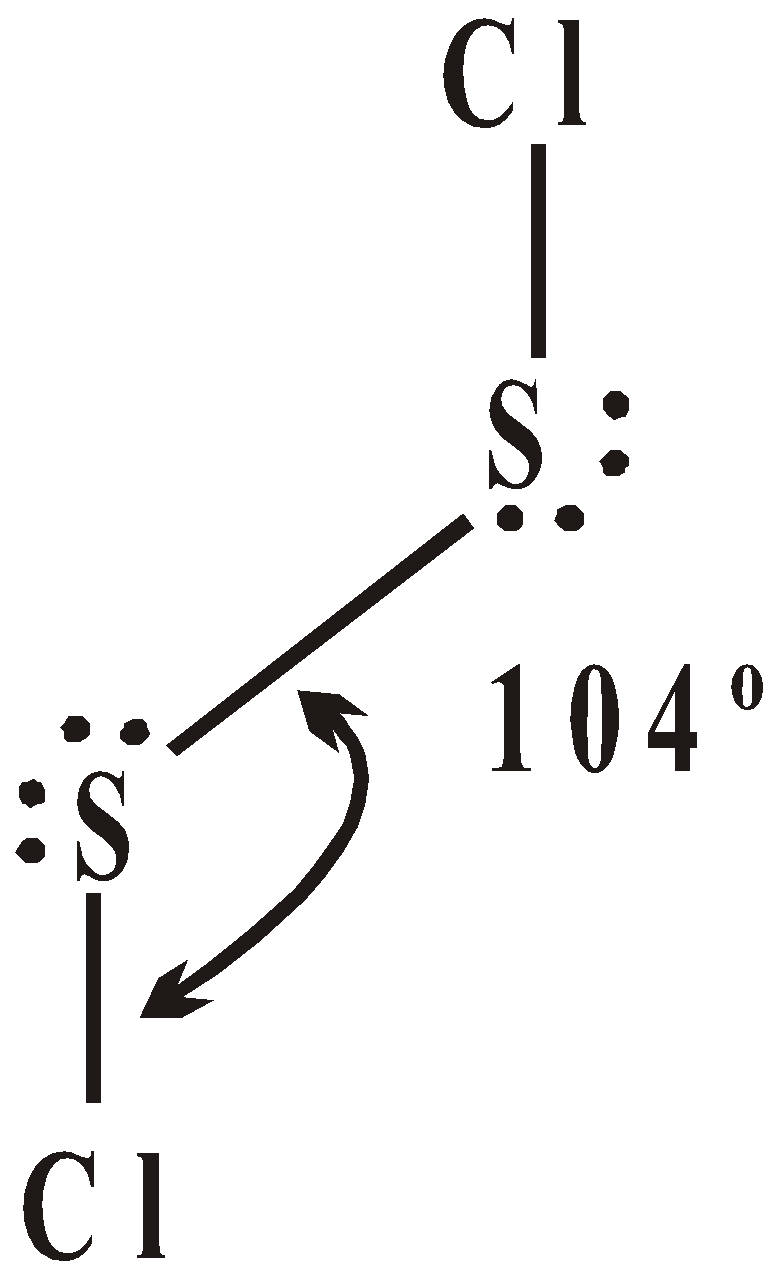
OXIDES
Element | Mono Oxide | Dioxide | Tri Oxide |
S | SO | SO2 | SO3 |
Se | – | SeO2 | SeO3 |
Te | TeO | TeO2 | TeO3 |
Po | PoO | PoO2 | – |
SO2 is a gas having sp2 hybridisation and V-shape.
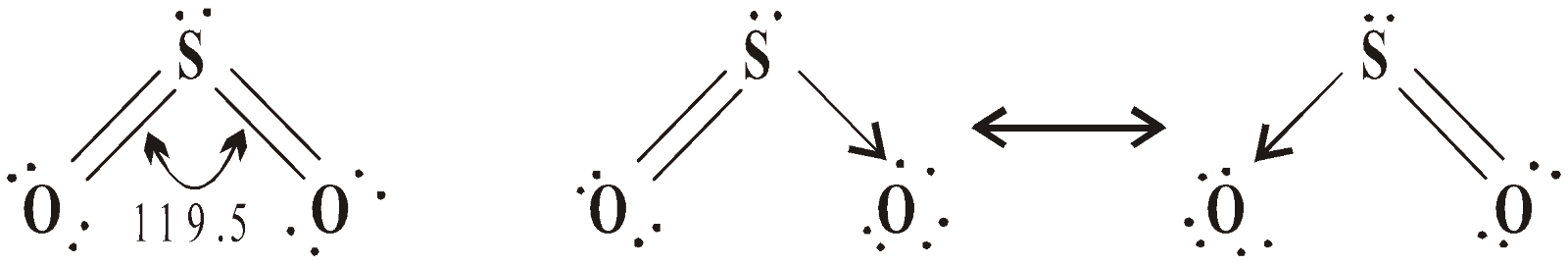
SO3 is a gas, sp2 hybridised and planar in nature.
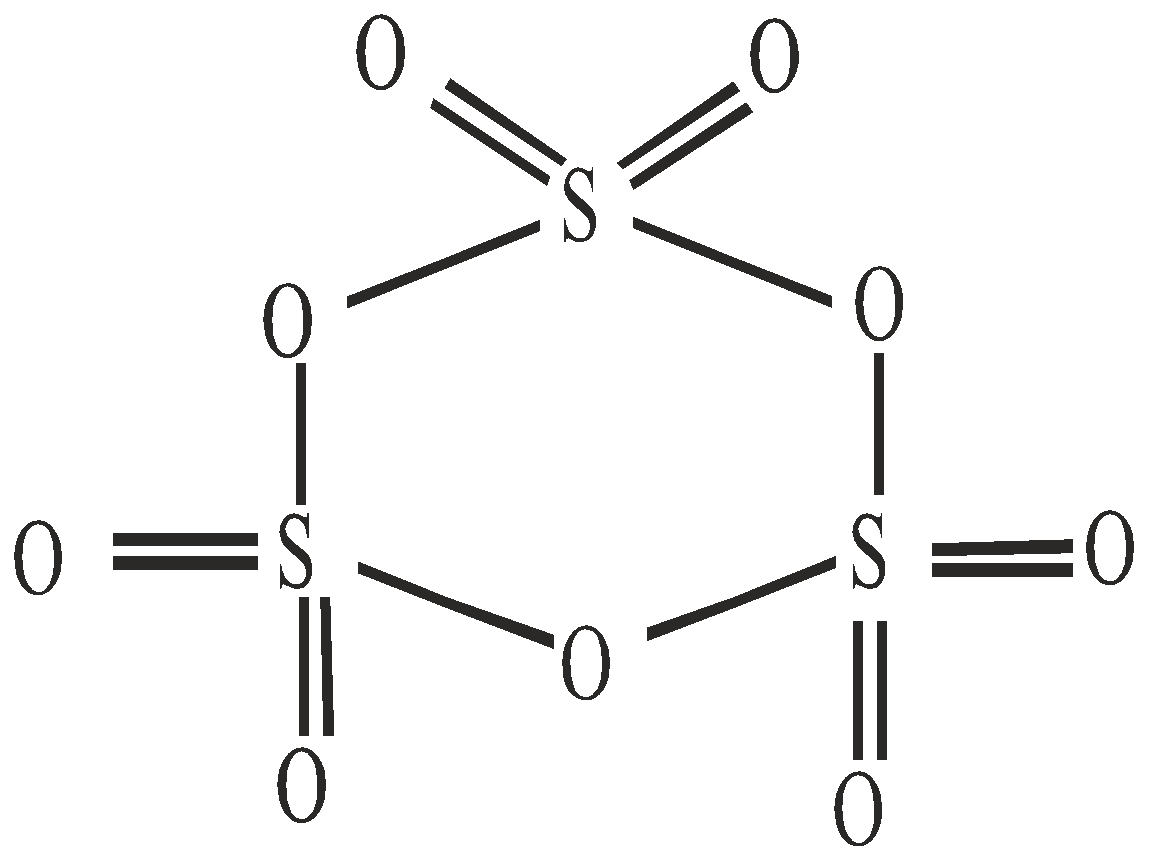
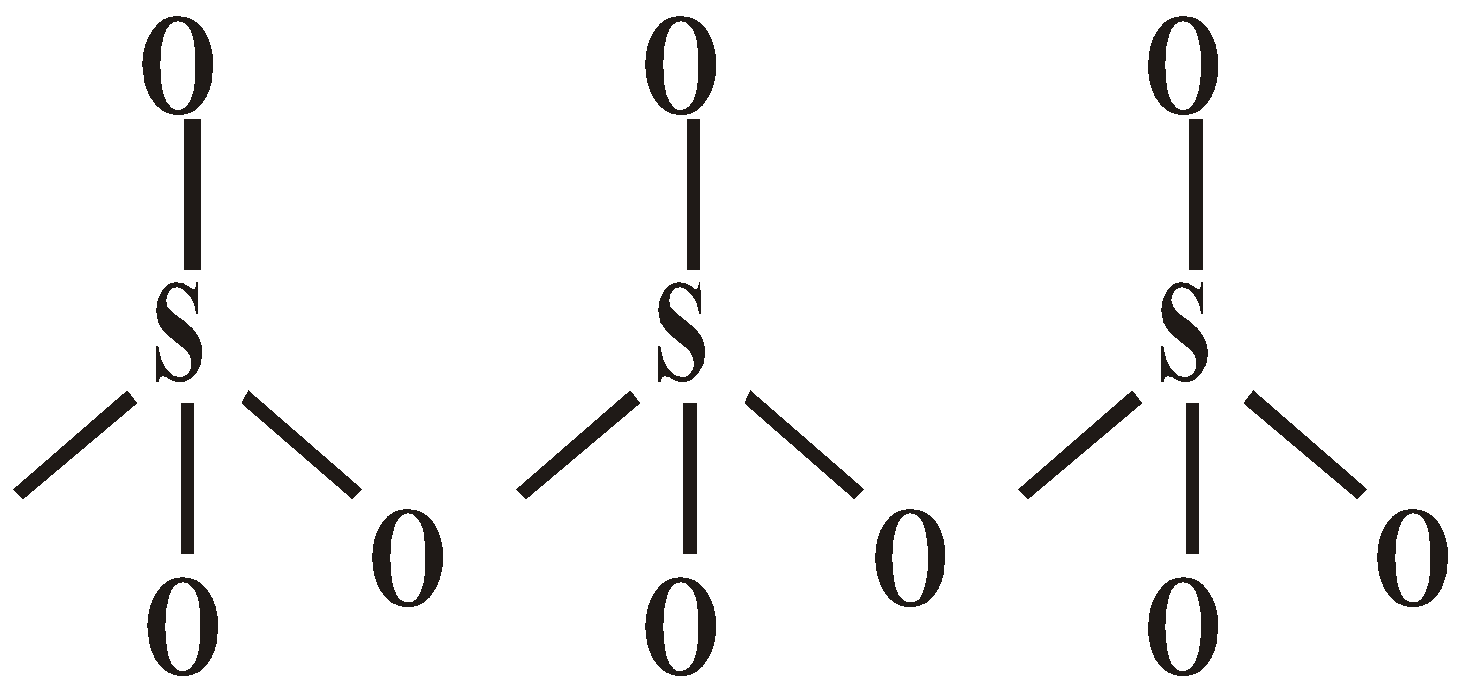
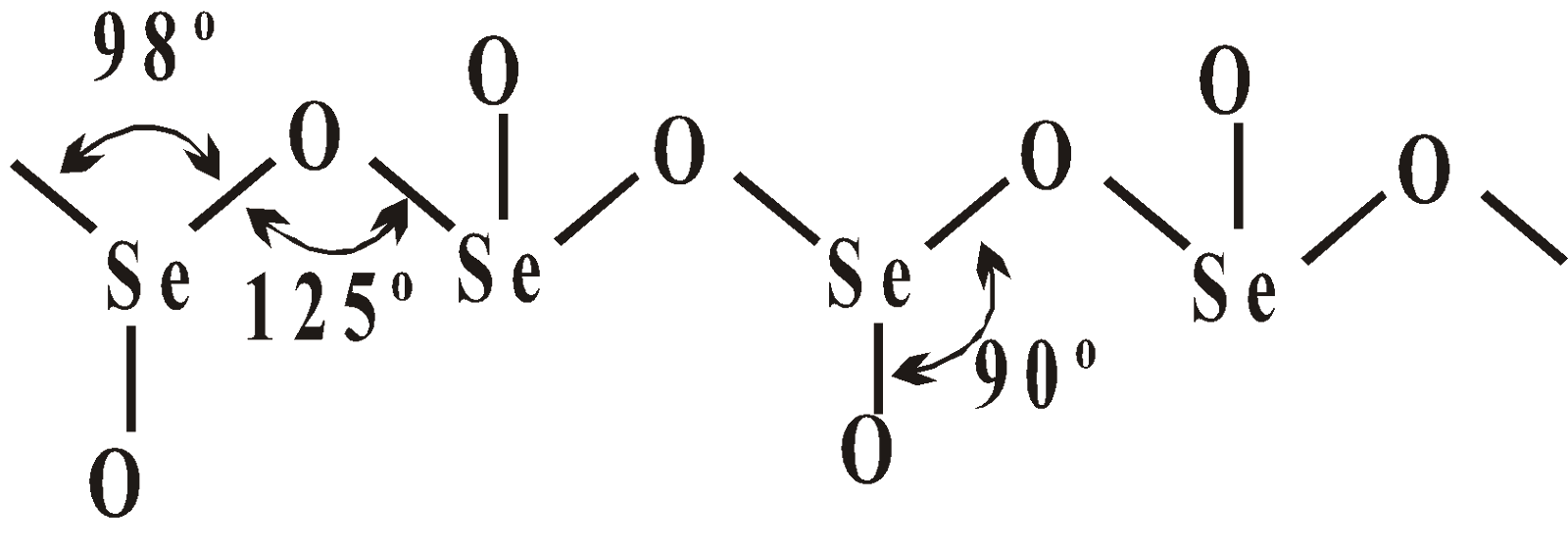
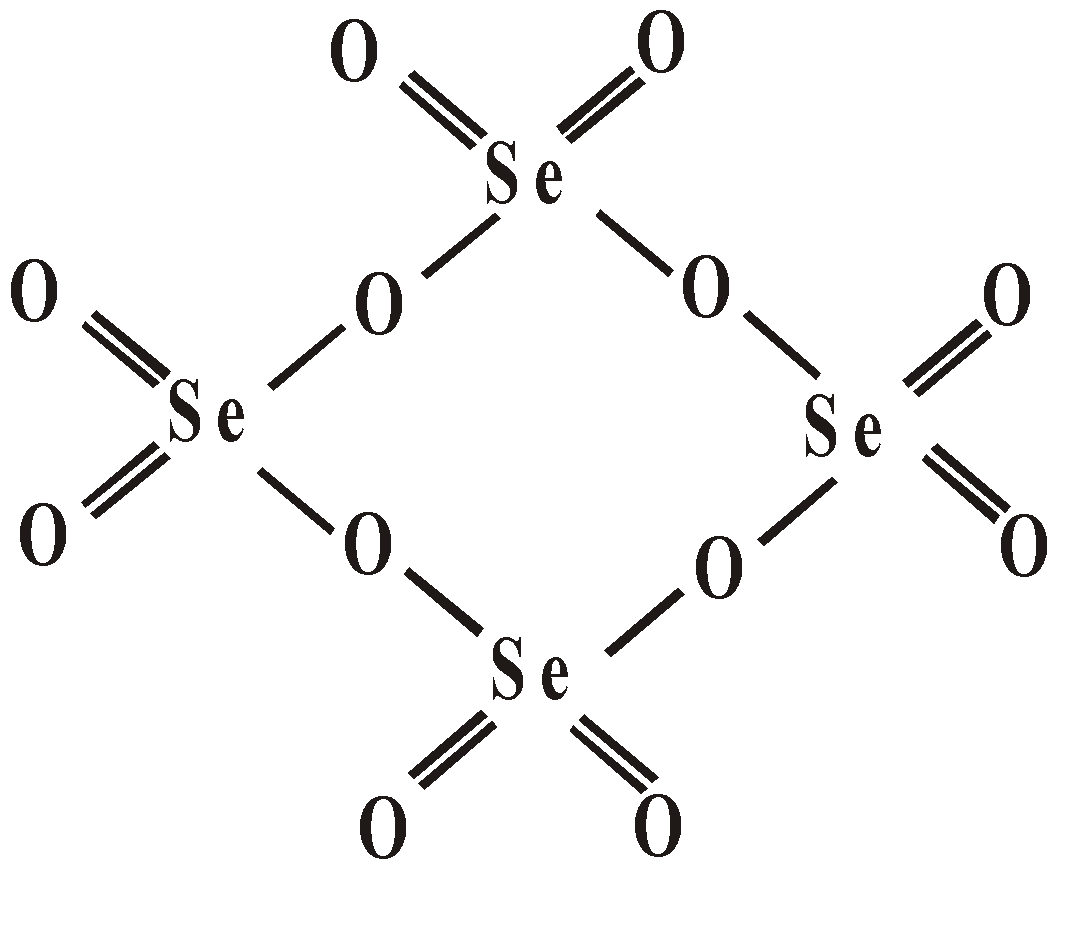
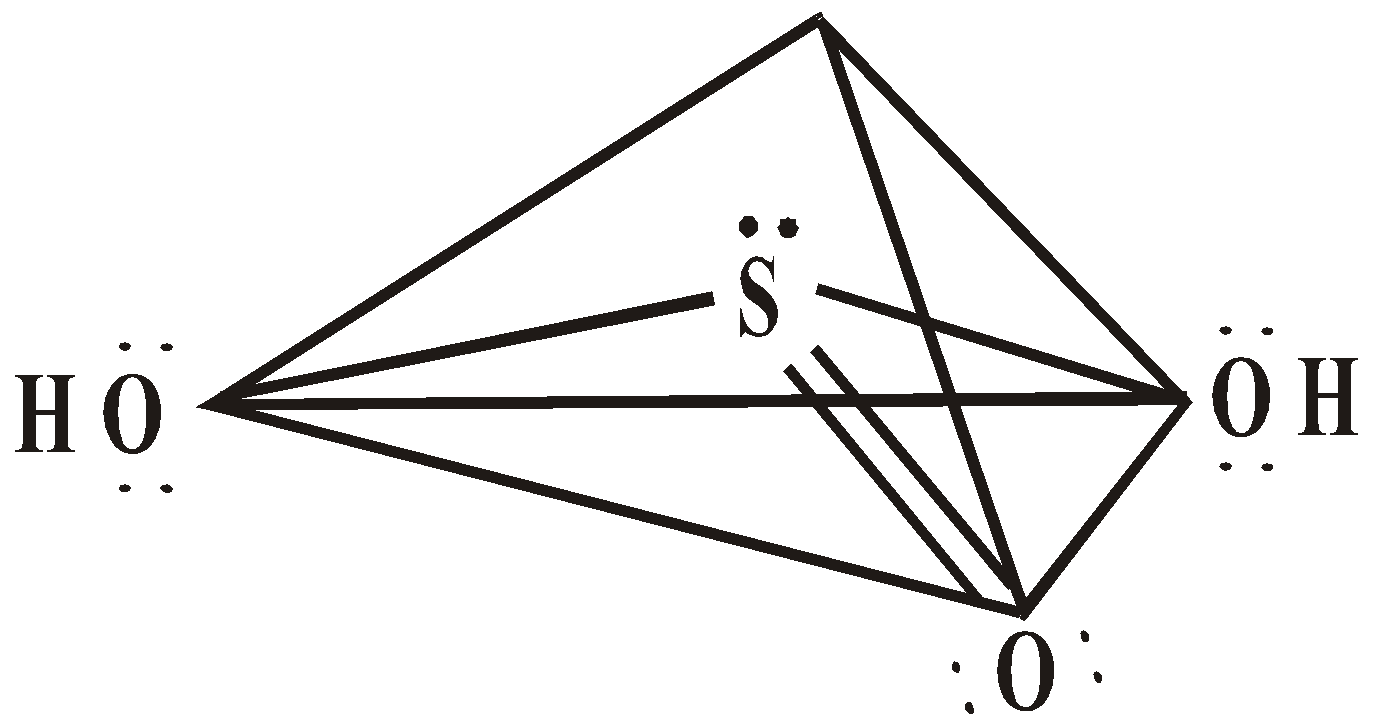
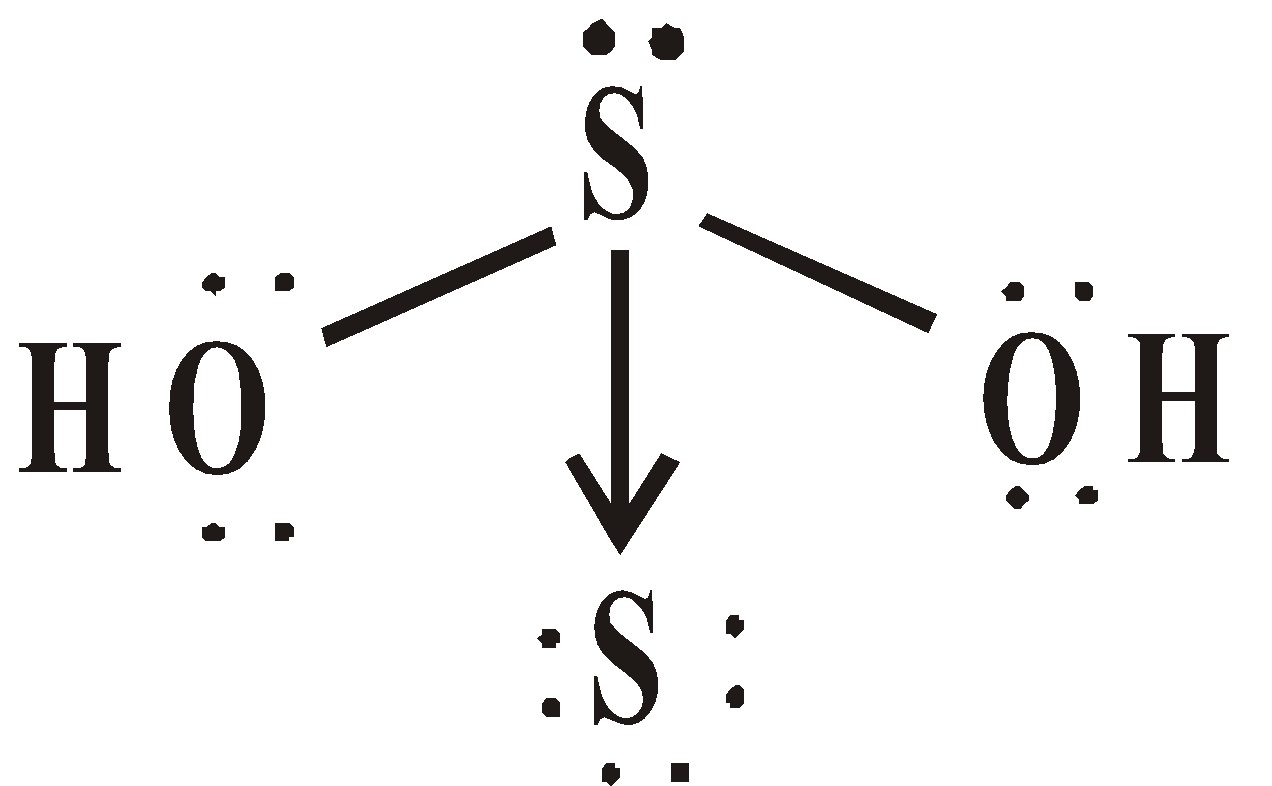
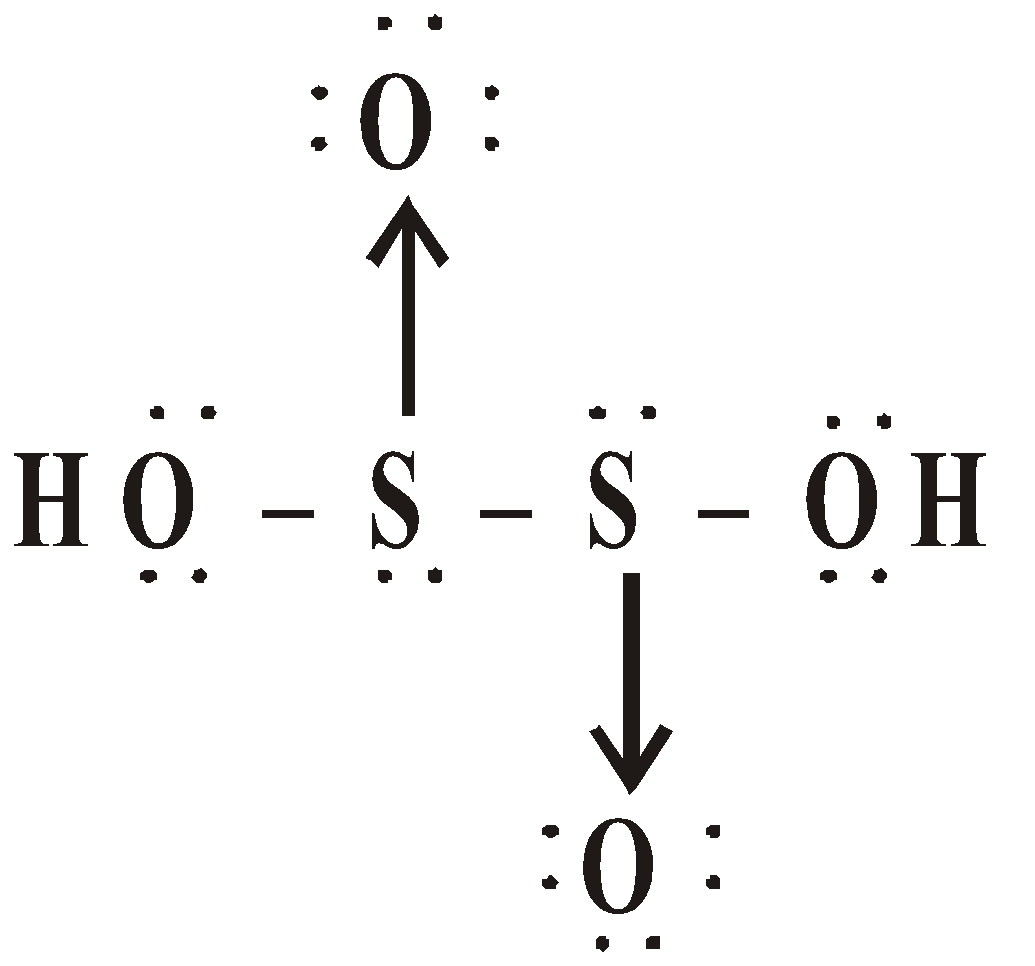
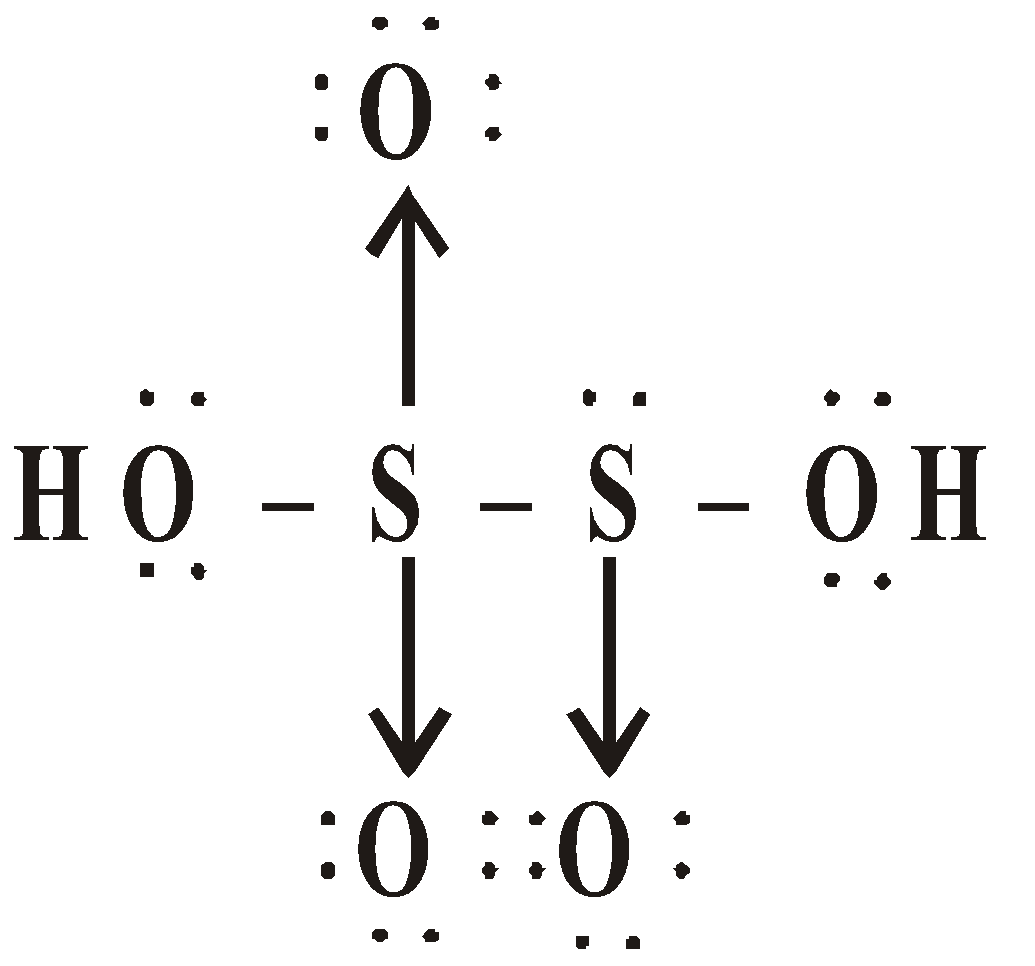
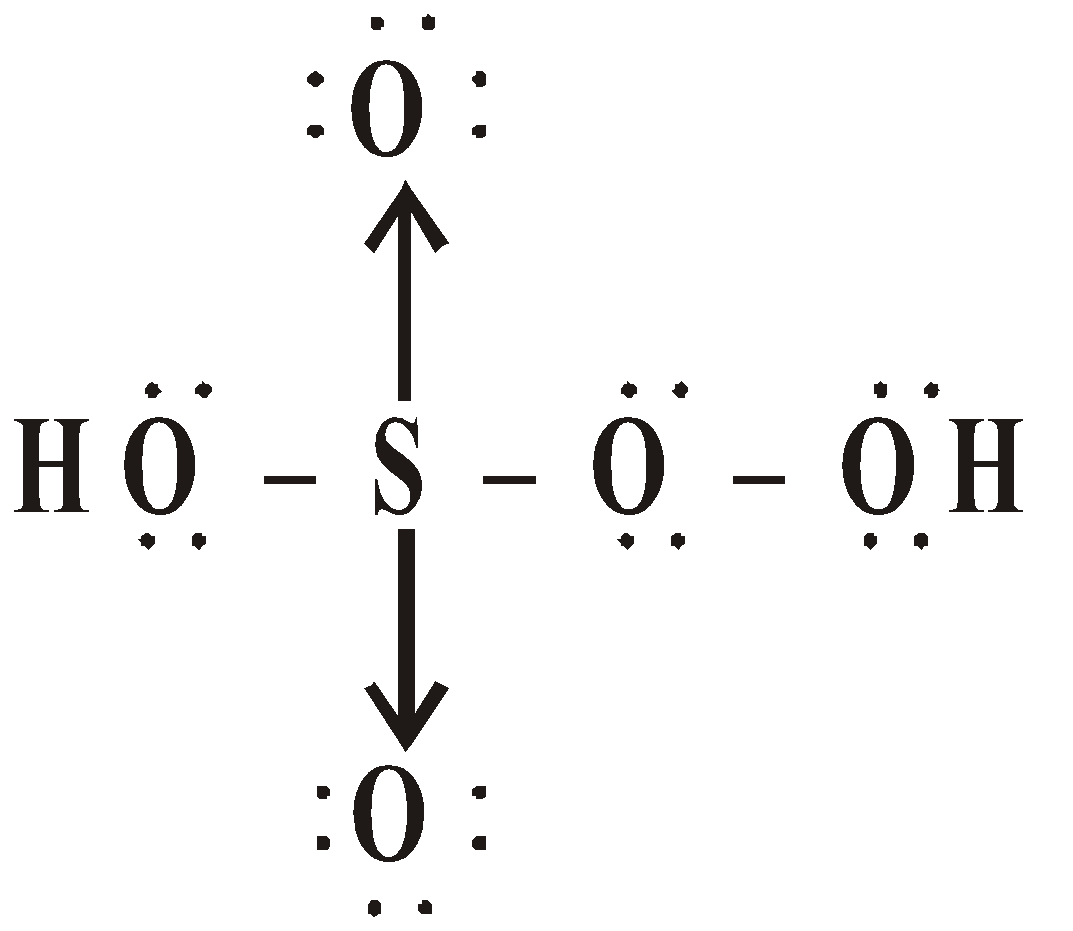
OXY ACIDS
 \
\
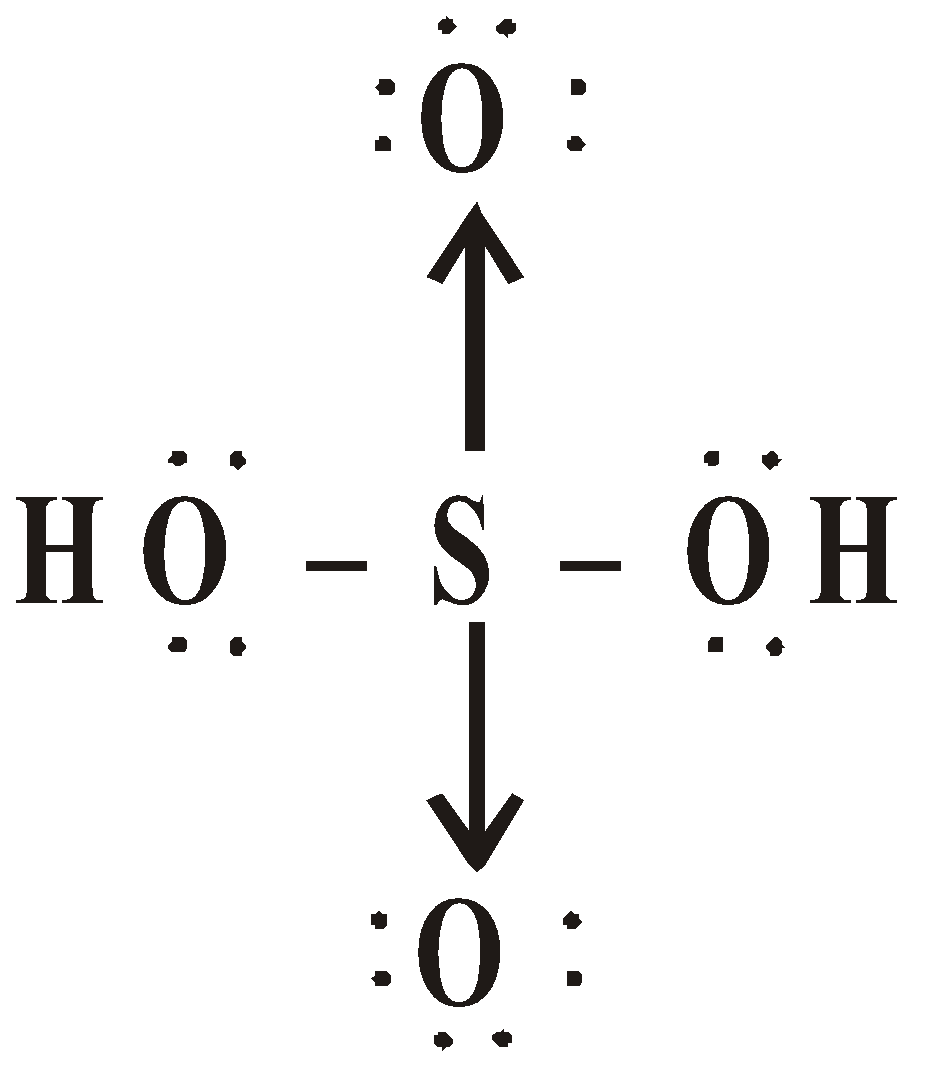
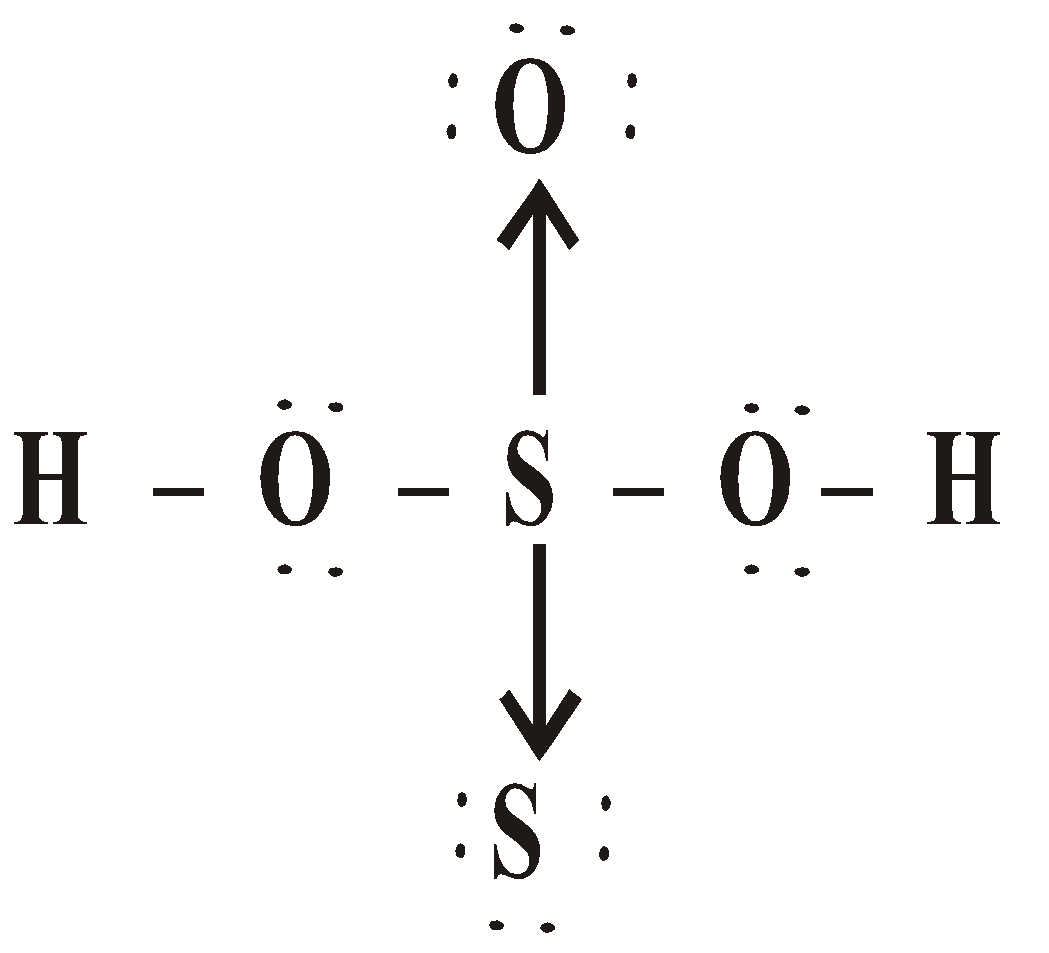 or
or 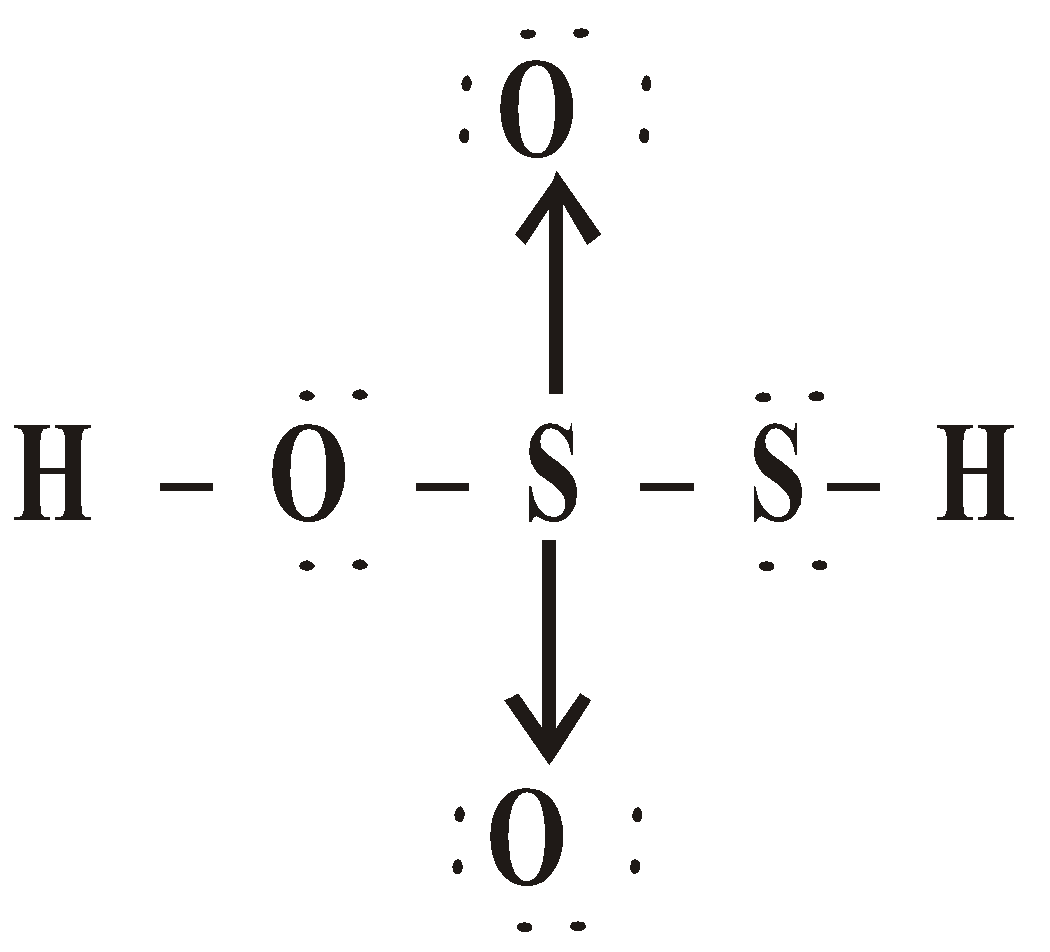
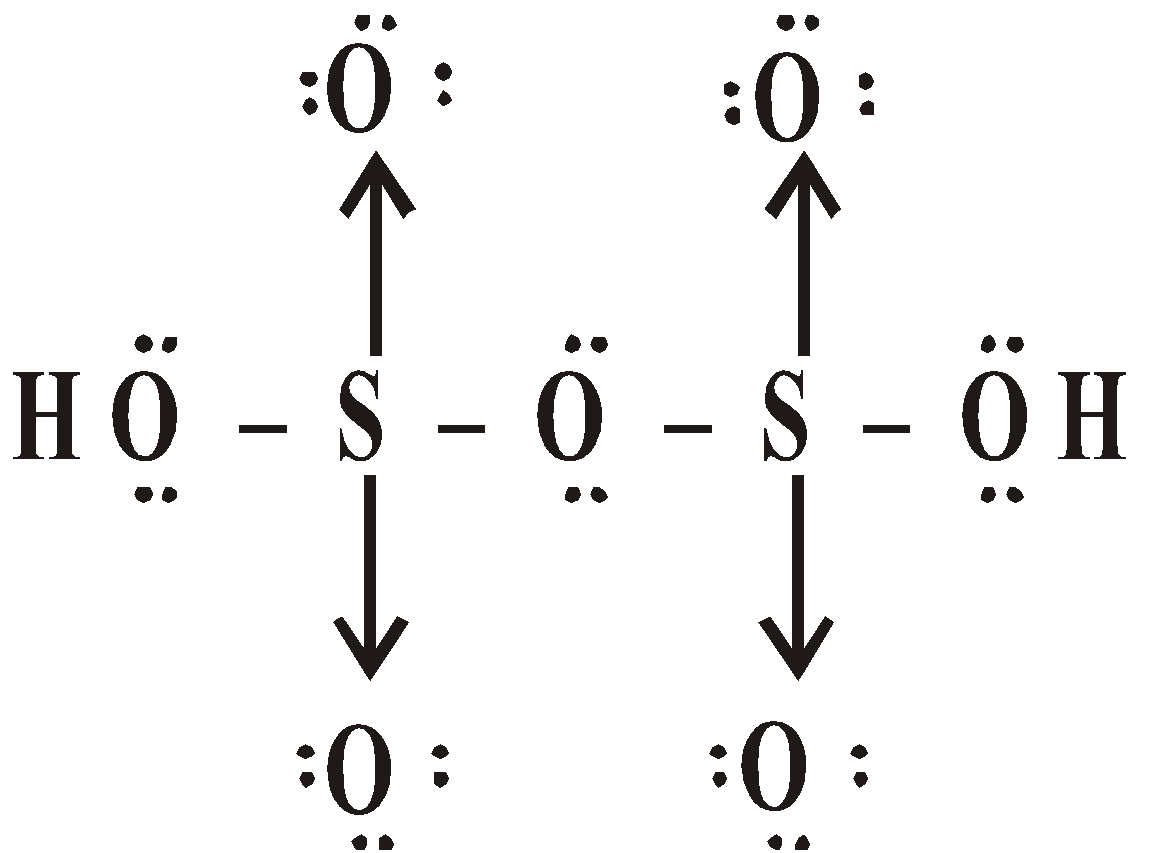
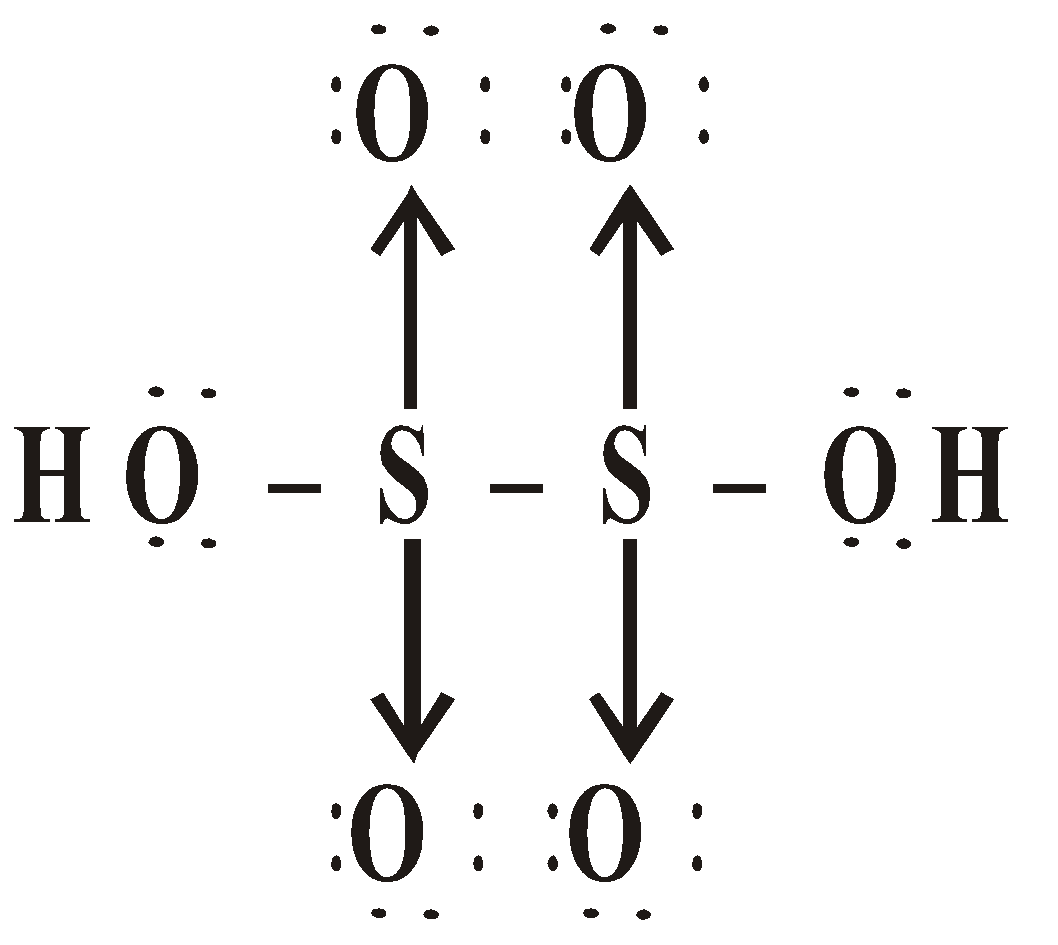
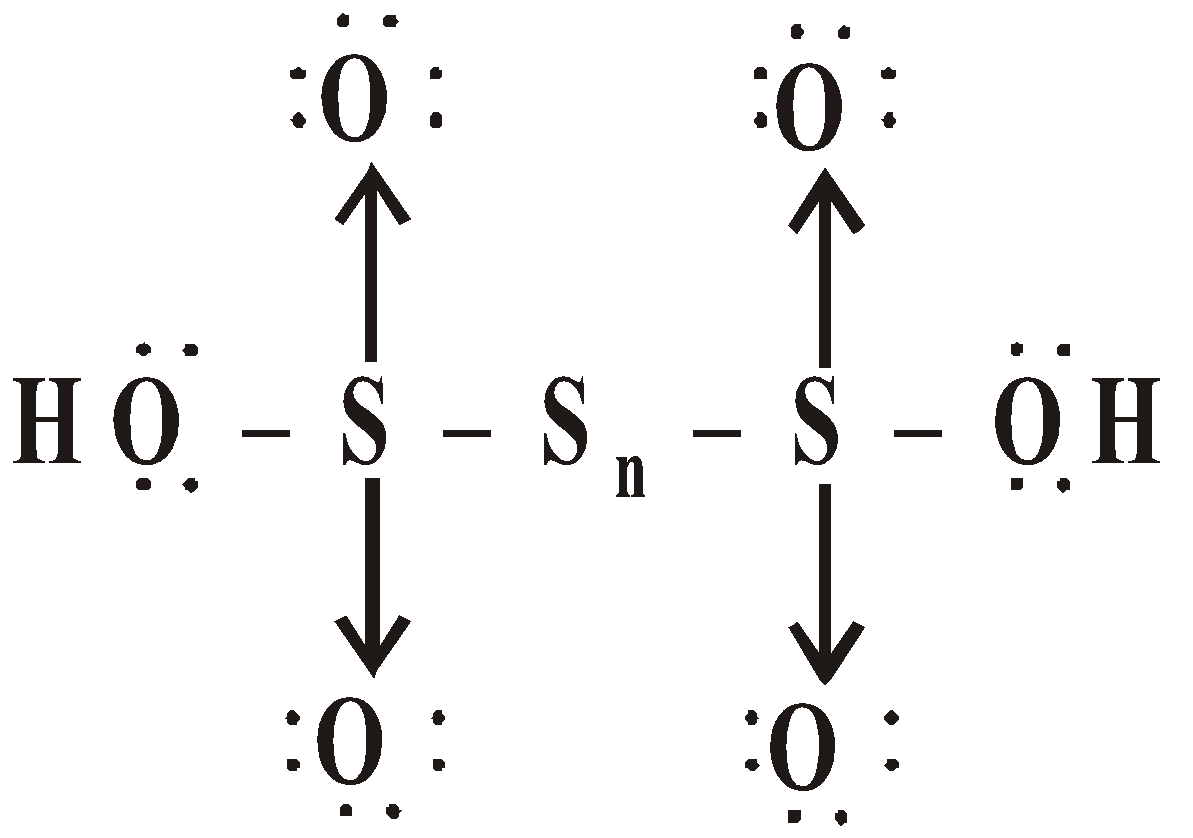
Peroxodisulphuric acid (H2S2O8)
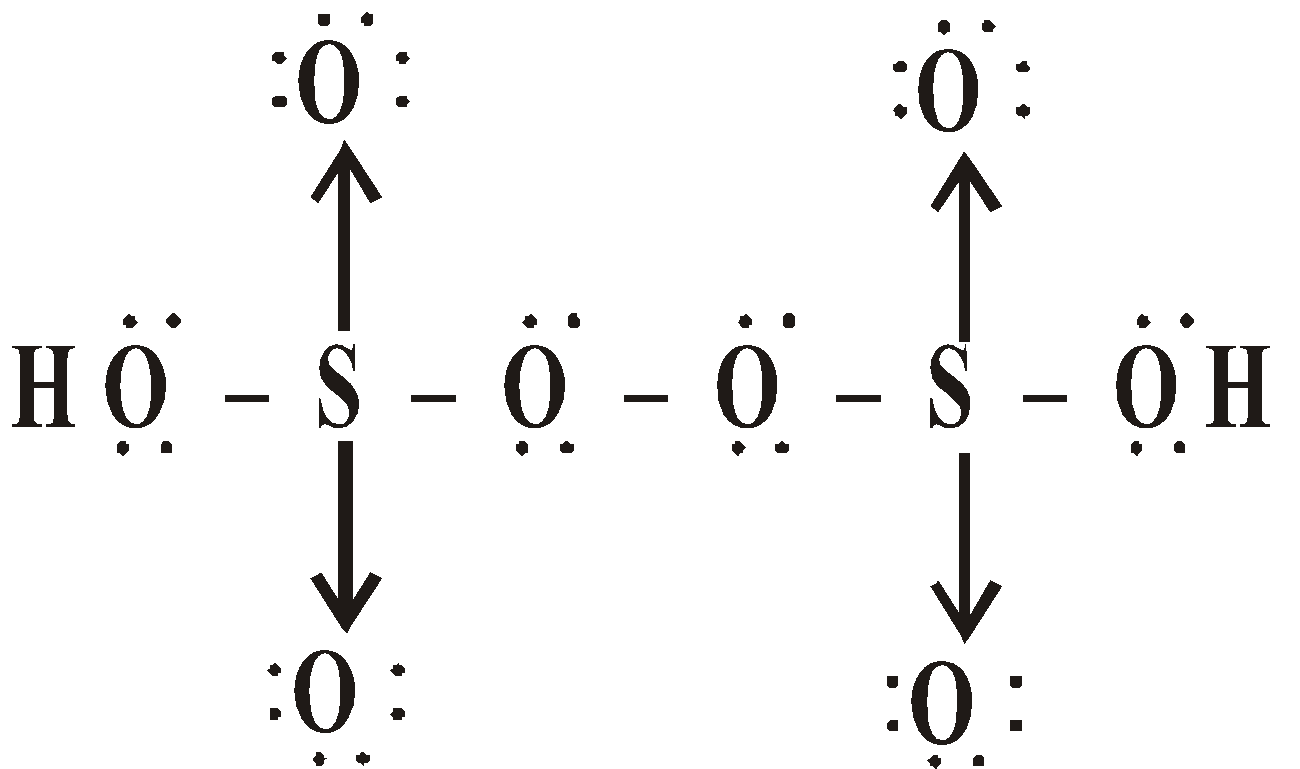
OZONE (O3)
- slow oxidation of phosphorus in air
- Reaction of fluorine with water at low temperature
- Electrolysis of water
- SO2 reacts with H2O2
PREPARATION
By passing silent electric discharge through cold, dry oxygen in ozoniser
- Siemens ozoniser
- Brodie’s ozoniser
MANUFACTURE
PROPERTIES
- Pale blue gas, dark blue liquid and violet black solid with characteristic strong smell, slightly soluble in water but more soluble in turpentine oil, glacial acetic acid and carbon tetrachloride.
- Decomposition
- Oxidising action
- Reducing action
- Addition reactions

USES
- Bleaching ivory, oils, flour etc.
- As germicide and disinfectant, for sterilising water
- For improving atmosphere in crowded places
- Manufacture of KMnO4 and artificial silk.
TESTS
- Turns starch iodine paper blue.
- Tailing of Hg – Mercury loses its meniscus in contact with O3 and sticks to the surface of glass due to formation of Hg2O
- Clean silver foil blackened by O3
- It turns benzidine paper brown and tetramethyl base violet
STRUCTURE
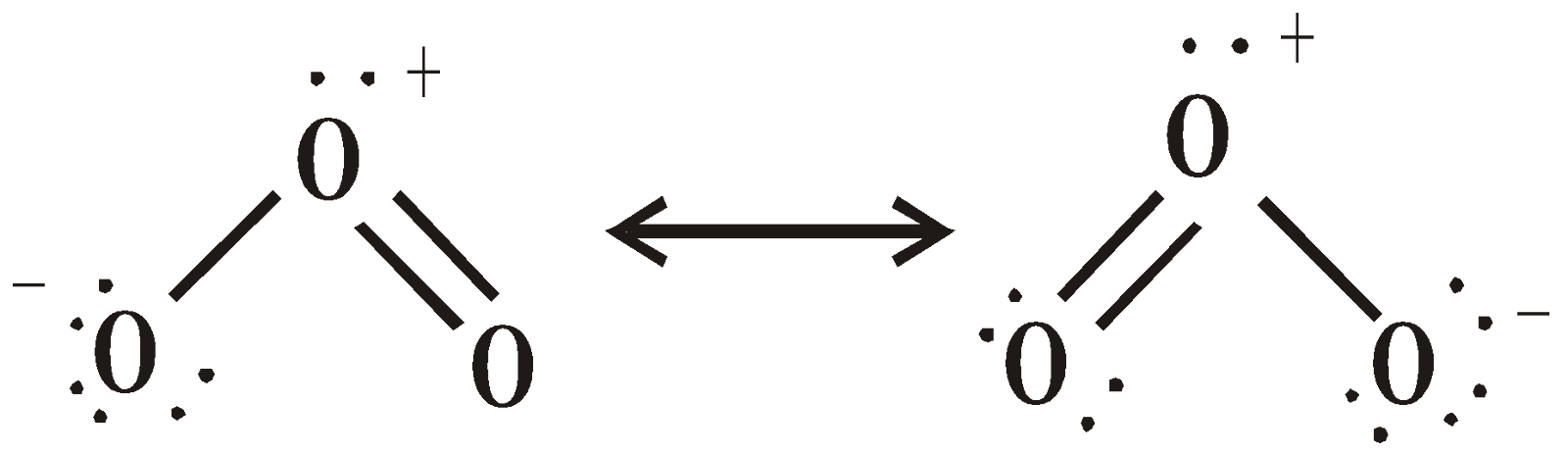
OXIDES
NORMAL OXIDES
POLYOXIDES
PEROXIDES
They contain ion, produce hydrogen peroxide with dil. acids and O2 with concentrated acids eg BaO2, Na2O2
SUPER OXIDES
They contain ion. With water they give hydrogen peroxide and oxygen
DIOXIDES
They give chlorine with conc HCl and oxygen with Conc H2SO4 eg. MnO2, PbO2 etc.
They contain lower percentage of oxygen eg. N2O, C3O2
Formed by the combination of two simple oxides
eg. Red lead, Pb3O4 (PbO2.2PbO), Fe3O4 (FeO+Fe2O3)
CLASSIFICATION ON THE BASIS OF CHEMICALS BEHAVIOUR
Oxides of non metals which give acids when dissolved in water are called acidic oxides eg.
BASIC OXIDES
- Ionic in nature. Oxides of alkali and alkaline earth metals eg Na2O, CaO, BaO.
In water they give basic solutions
- Covalent oxides – Oxides of transition metals are covalent in nature eg CuO, FeO. Insoluble in water.
OXYGEN
OCCURRENCE
PREPARATION
- By action of heat on oxygen rich compounds
- From oxides
- From peroxides and other oxides
- From certain compounds
- By the action of some chemical reagent on compound rich in O2.
- By electrolysis of water either acidified with H2SO4 using platinum electrodes or by making it alkaline with NaOH or Ba(OH)2 using nickel electrodes.
- By decomposition of steam by chlorine
MANUFACTURE
PHYSICAL PROPERTIES
CHEMICAL PROPERTIES
USES
- For breathing
- In welding and cutting – oxy-hydrogen or oxy-acetylene torch is used
- In iron and steel industry – to increase the content of blast in the Bessemer and open hearth process
- As a fuel in rockets
TESTS
- With NO it gives reddish brown fumes of NO2
- Absorbed by alkaline pyrogallol
- A smouldering wood splinter bursts into flames in a jar of O2
STRUCTURE
SULPHUR
OCCURENCE
- Gypsum CaSO4.2H2O
- S-Celesite SrSO4
- Galena PbS
- Zinc blende ZnS
- Copper pyrites Cu2SFe2S3
- Iron pyrites FeS2
PROPERTIES
- Crystalline – Rhombic, monoclinic
- Amorphous – Plastic, milk of sulphur, colloidal sulphur
SULPHURIC ACID (H2SO4)
MANUFACTURE
- Production of SO2 : By burning S or iron pyrites.
- Production of catalyst – Oxides of nitrogen
- Reaction in lead chamber
- Conditions – Temperature 50ºC, excess of steam, lead chamber since lead is not attacked H2SO4.
- Purification – The acid obtained contains the impurities of PbSO4, AS2O3, NO and NO2 which are removed as follows
- Concentration – The sulphuric acid is concentrated by evaporation
- Production of SO2 – It is produced by burning sulphur and iron pyrites and purified by treating with steam to remove dust particles. The arsenic is removed by ferric hydroxide, water vapour removed by conc H2SO4. The gases are filtered through coke filters and purity is tested by Tyndal box.
- Conversion of SO2 to SO3 – It is done in contact or catalyst chamber after being preheated to 450ºC.
- SO3 is absorbed by conc H2SO4 and then water is added to produce the acid of desired concentration
PROPERTIES
- Physical Properties – Its specific gravity is 1.8 and it is 98% by weight normality is 36N.
- Action of heat
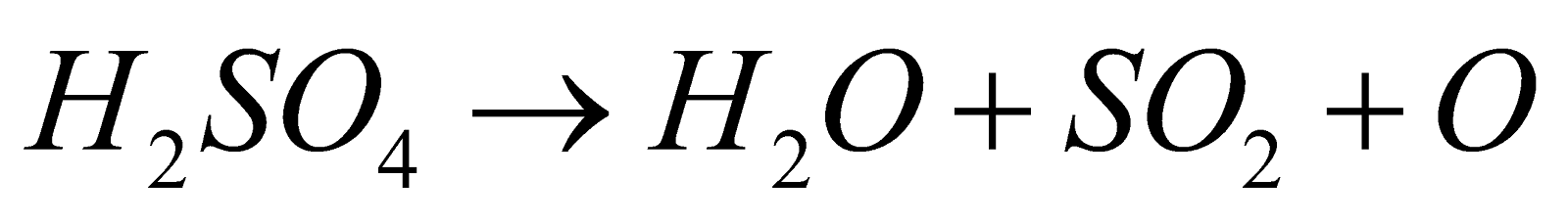
- Displacement reactions – It displaces volatile acids from their salts.
- Dehydrating agent – It is strong dehydrating in nature
- Reaction with PCl5
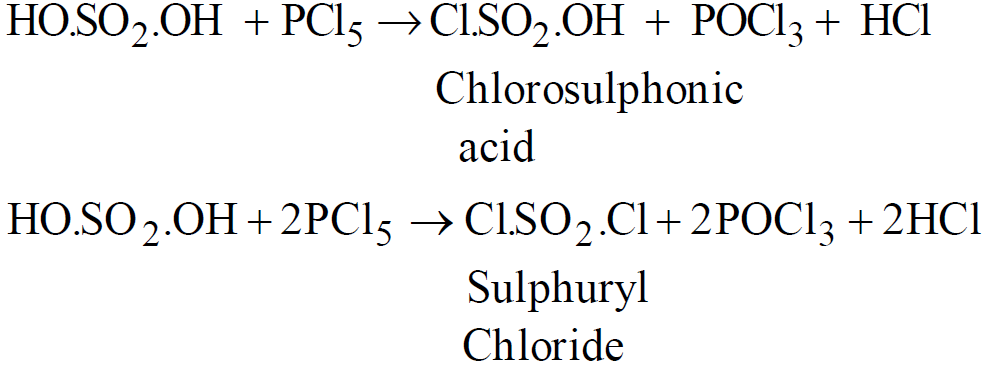
- Sulfonation of aromatic compounds
- Precipitation – Barium and lead are precipitated as sulphates
USES
STRUCTURE
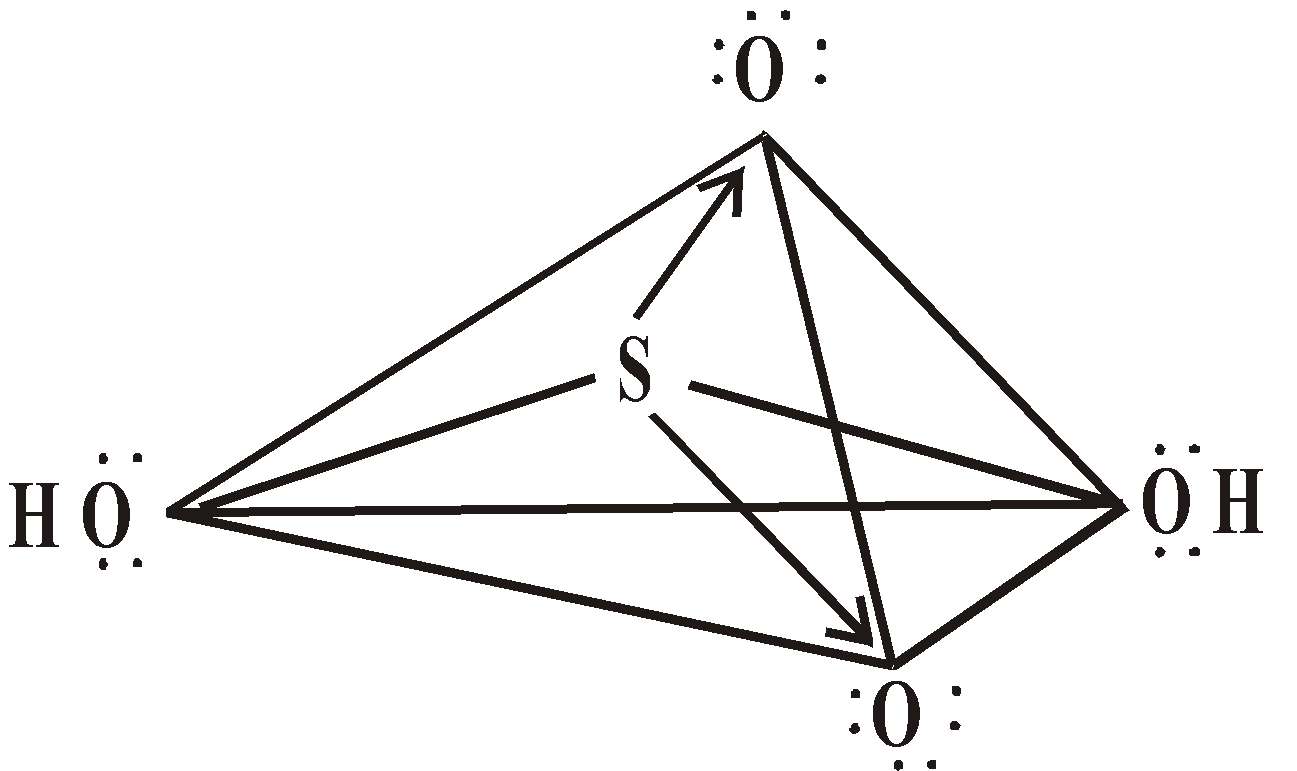 or
or 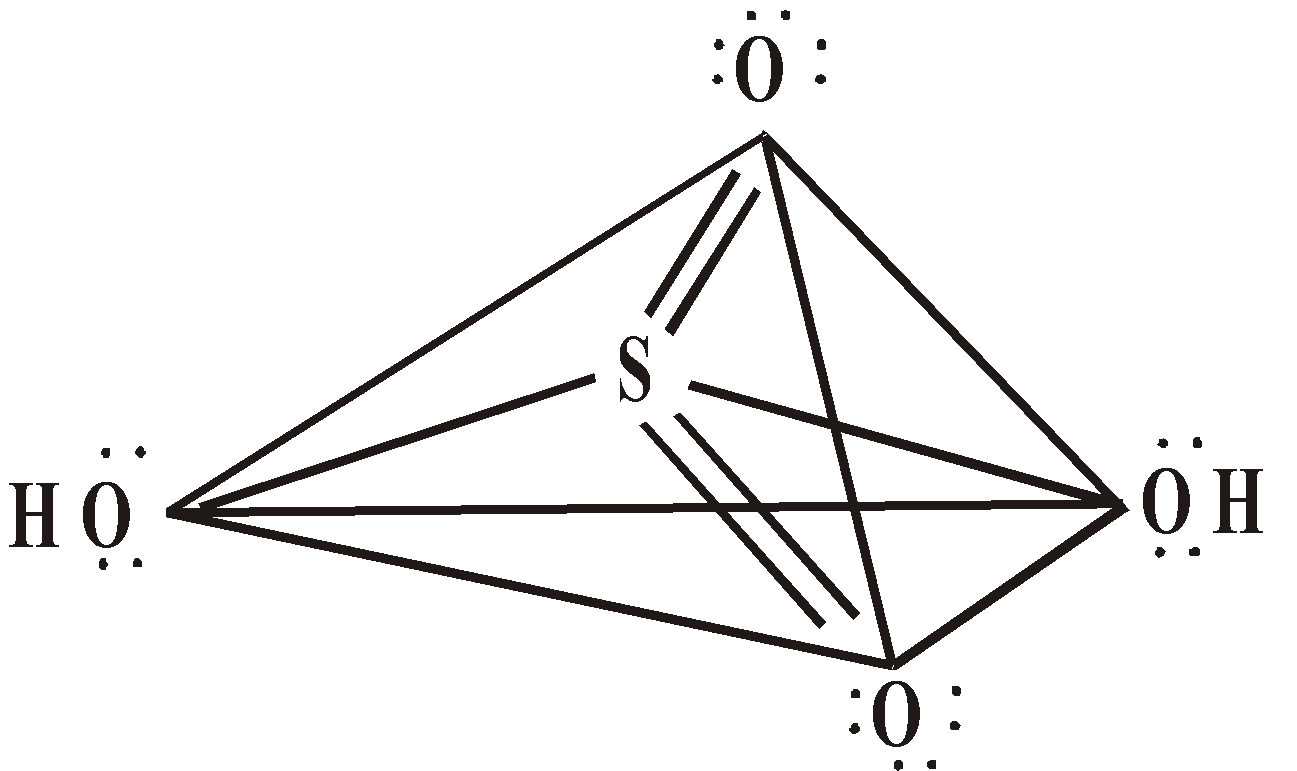
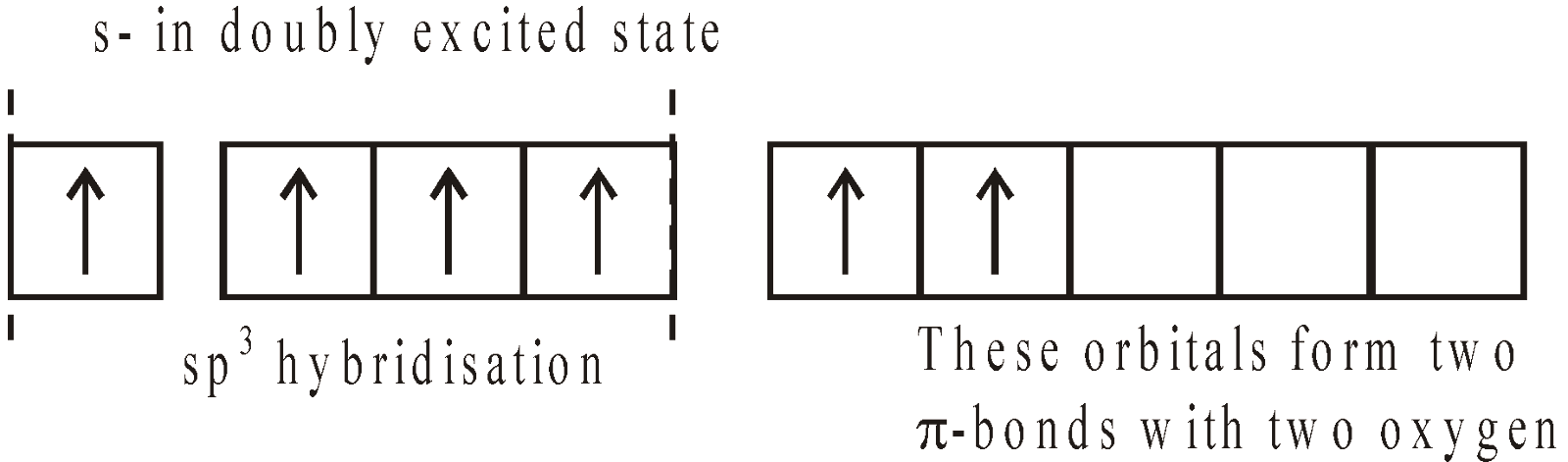
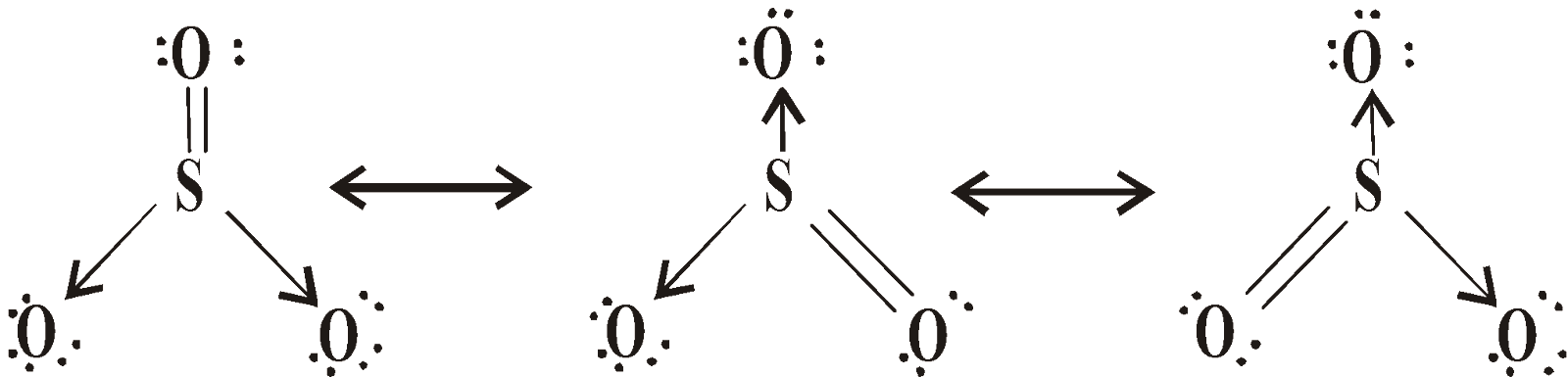
SULPHUR DIOXIDE (SO2)
PREPARATION
- By heating sulphur in air
- Roasting iron pyrites in excess of air
- Lab method : By heating Cu with conc H2SO4
PROPERTIES
- As reducing agent
- As oxidising agent
- Burning of potassium – Potassium burns in SO2 giving sulphate and thiosulphate
- Bleaching action – Its bleaching action is due to reduction
STRUCTURE
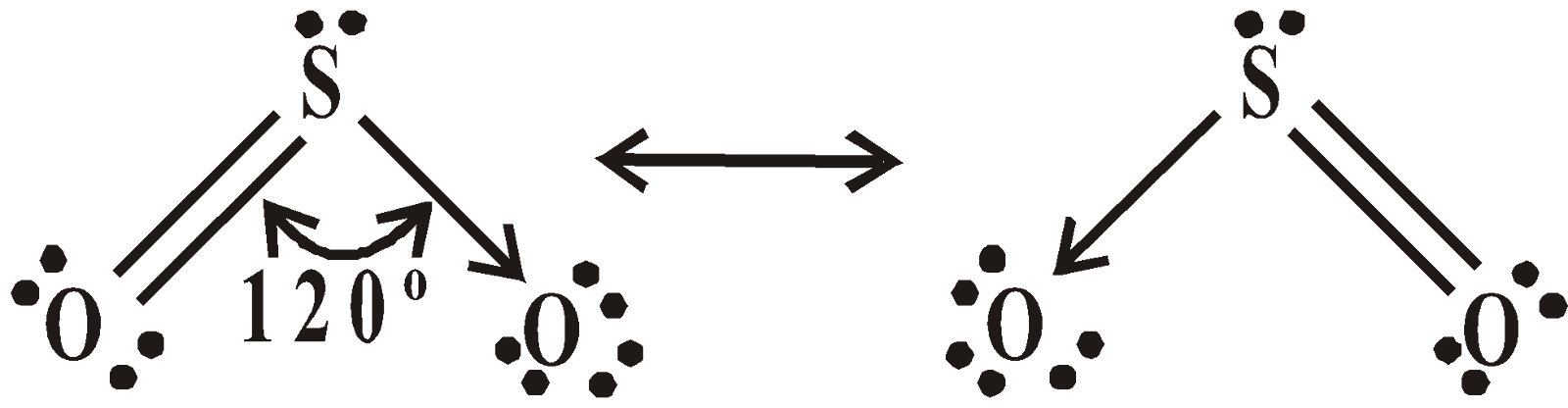 or
or 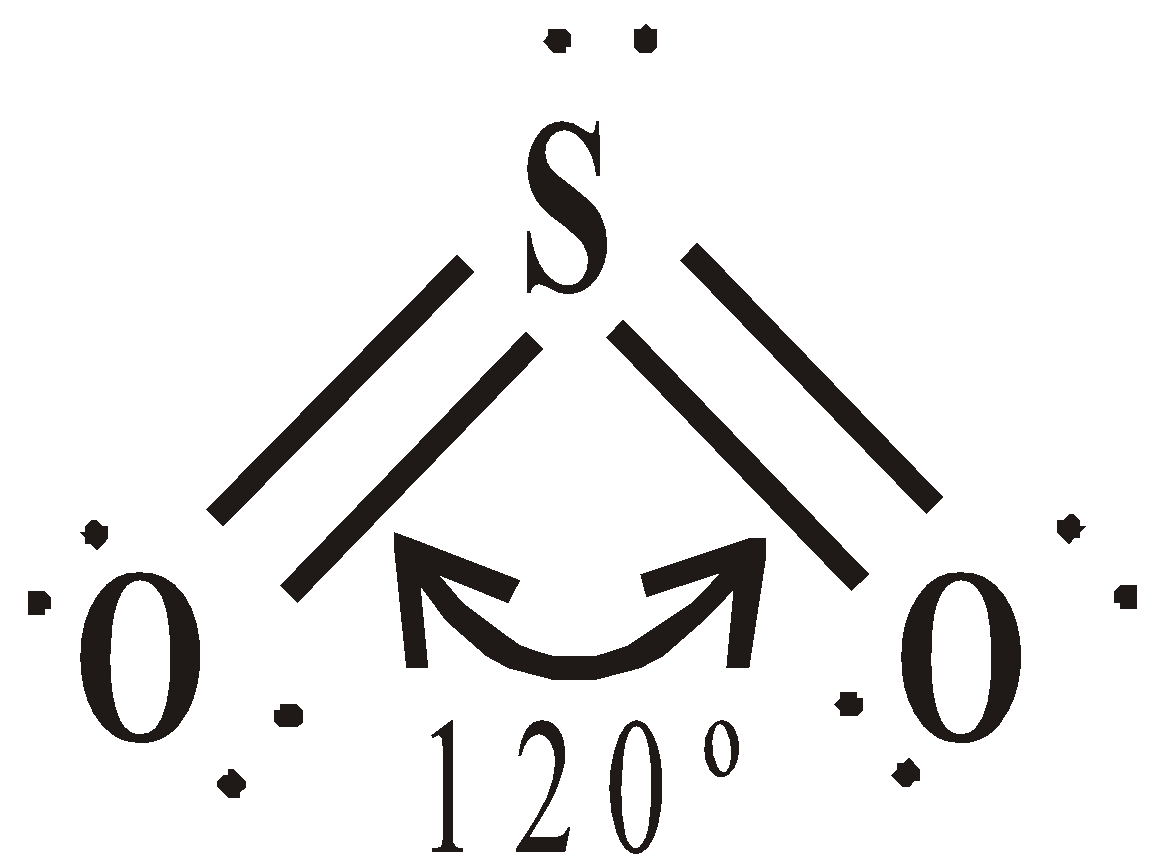
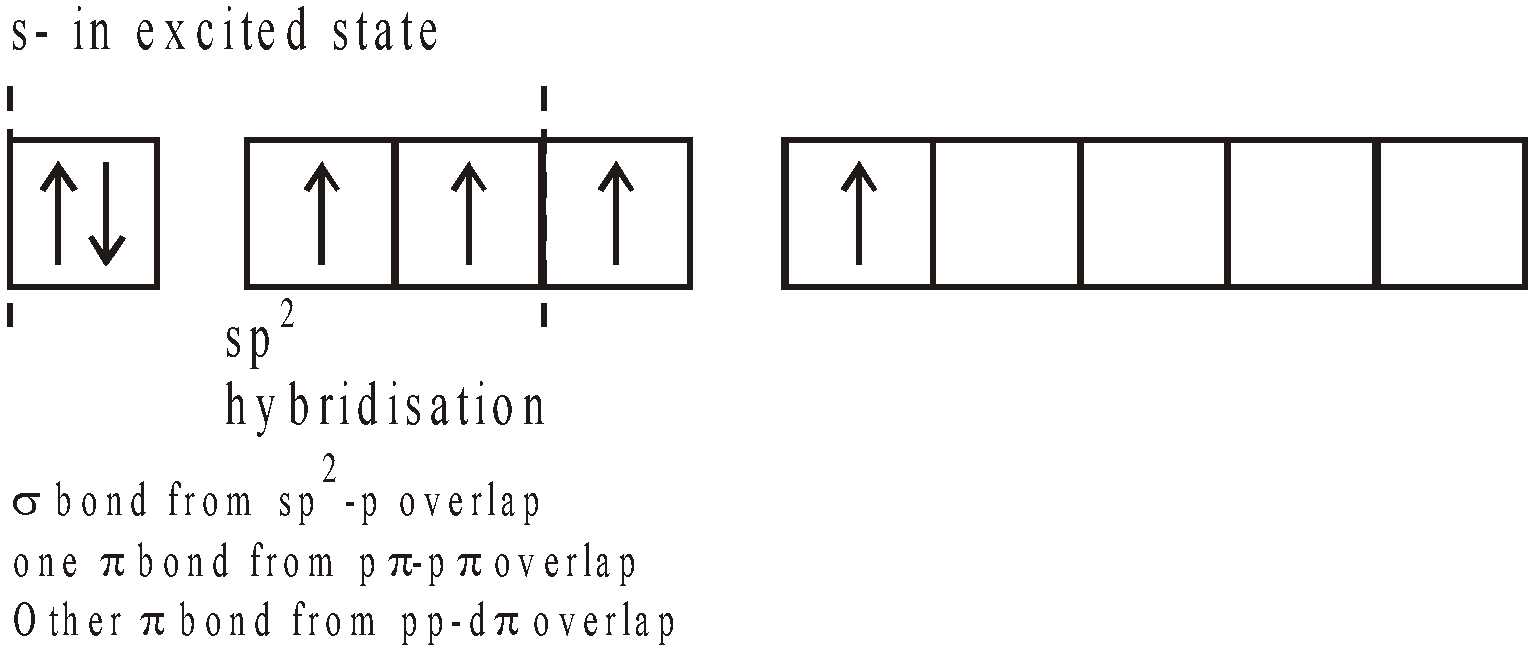
SULPHUR TRIOXIDE (SO3)
PREPARATION
- By heating conc H2SO4 with P2O5
- Manufacture : It is done by contact process
PROPERTIES
- Action of heat

- Water

- With
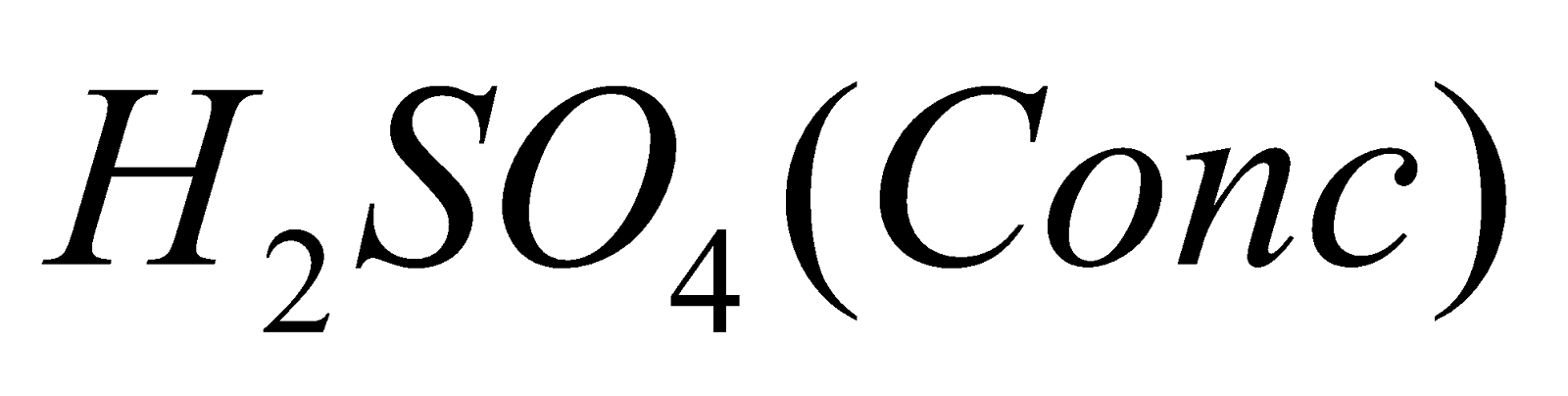
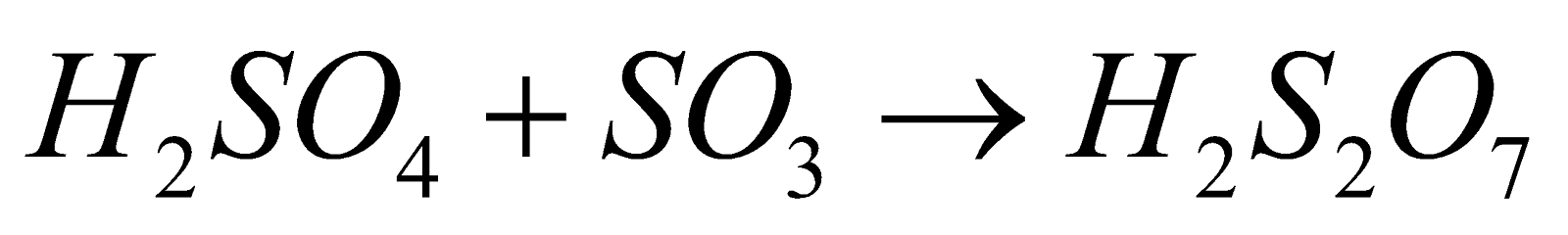 (oleum)
(oleum) - Acidic nature
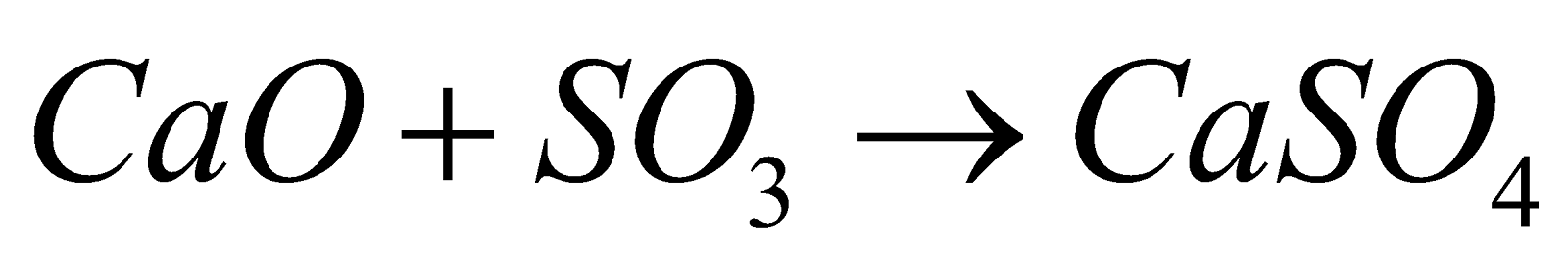
- With HCl
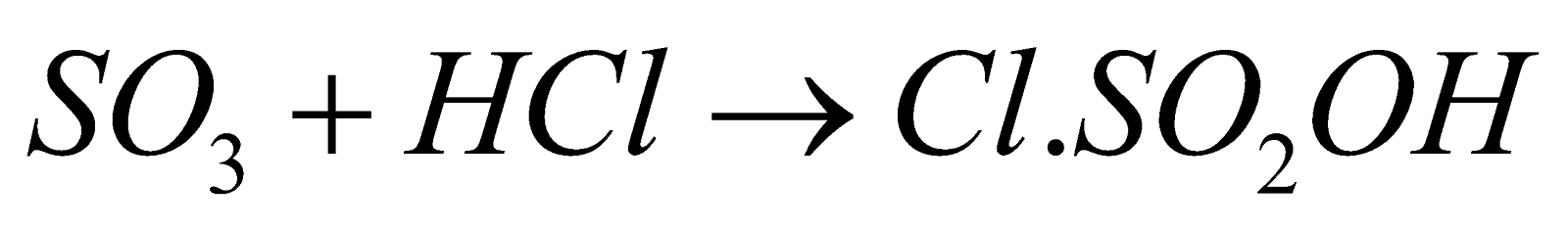 (Chloro Sulphonic acid)
(Chloro Sulphonic acid) - Oxidising agent – It is powerful oxidising agent
STRUCTURE
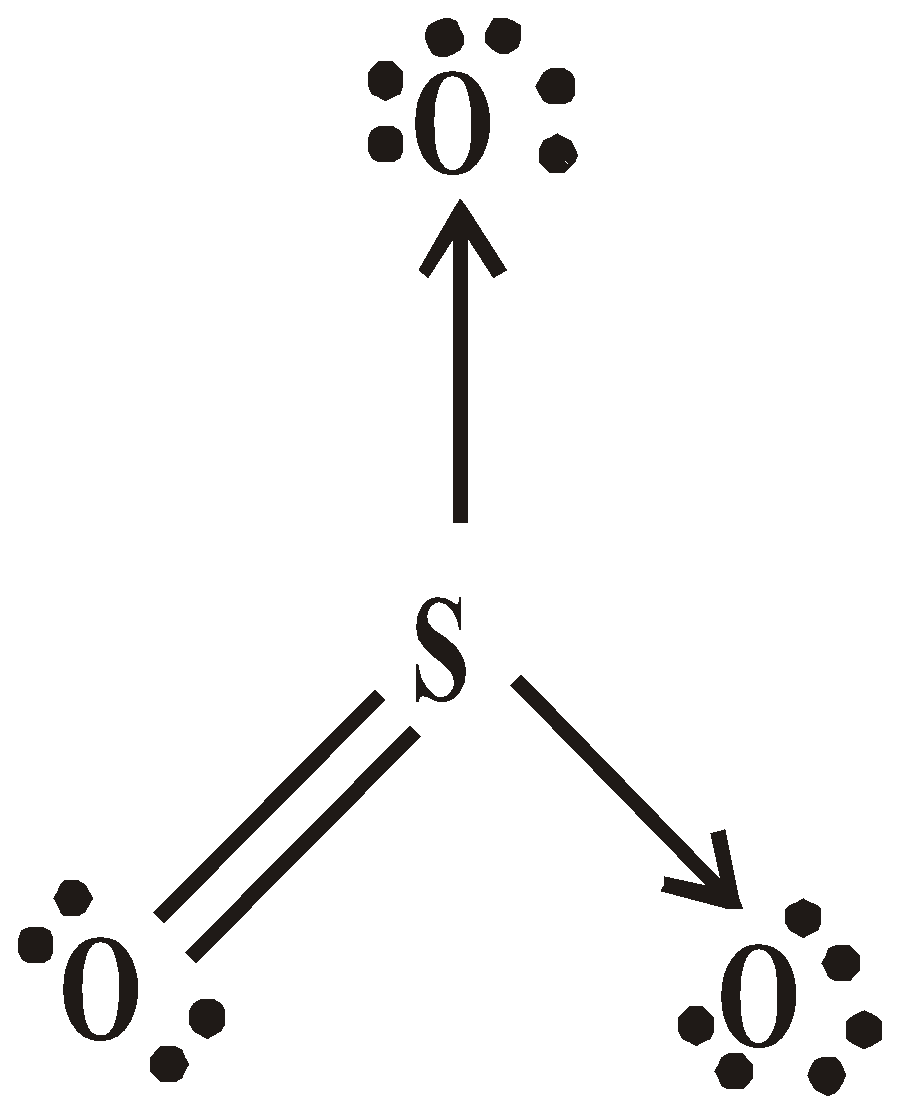
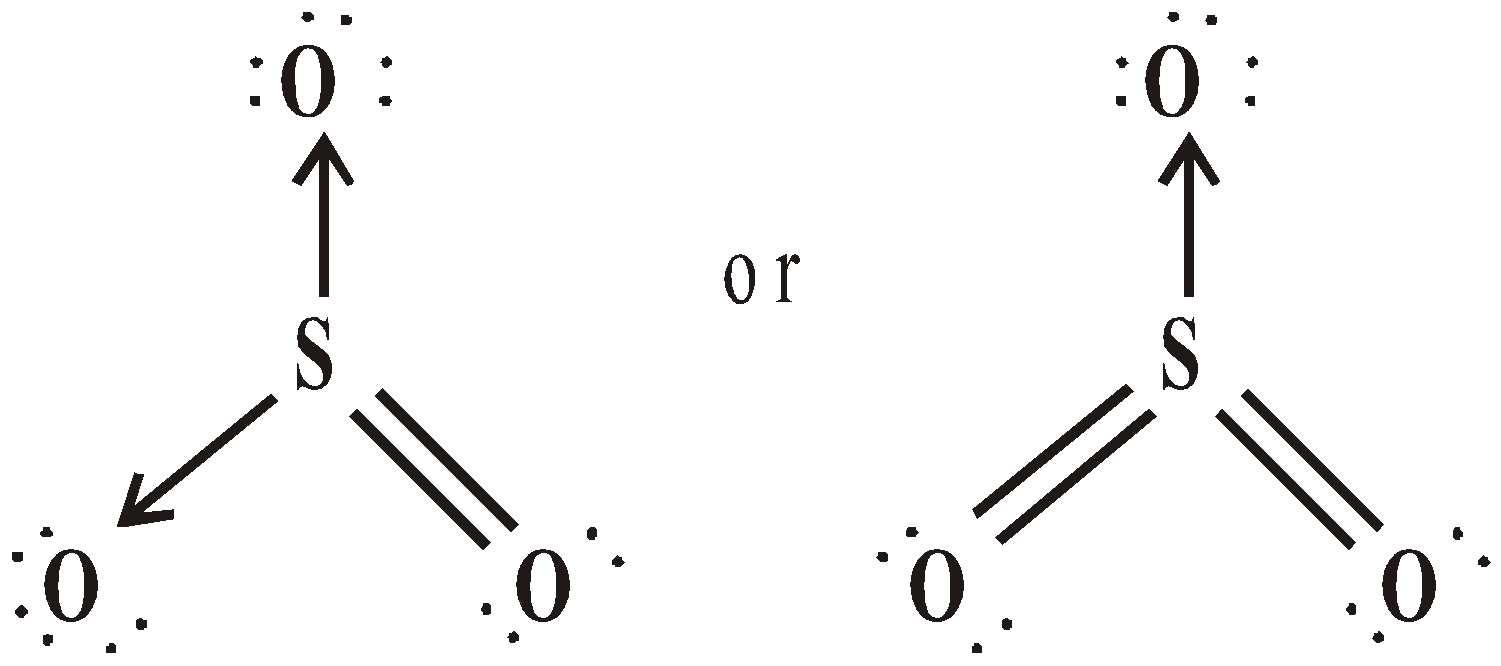
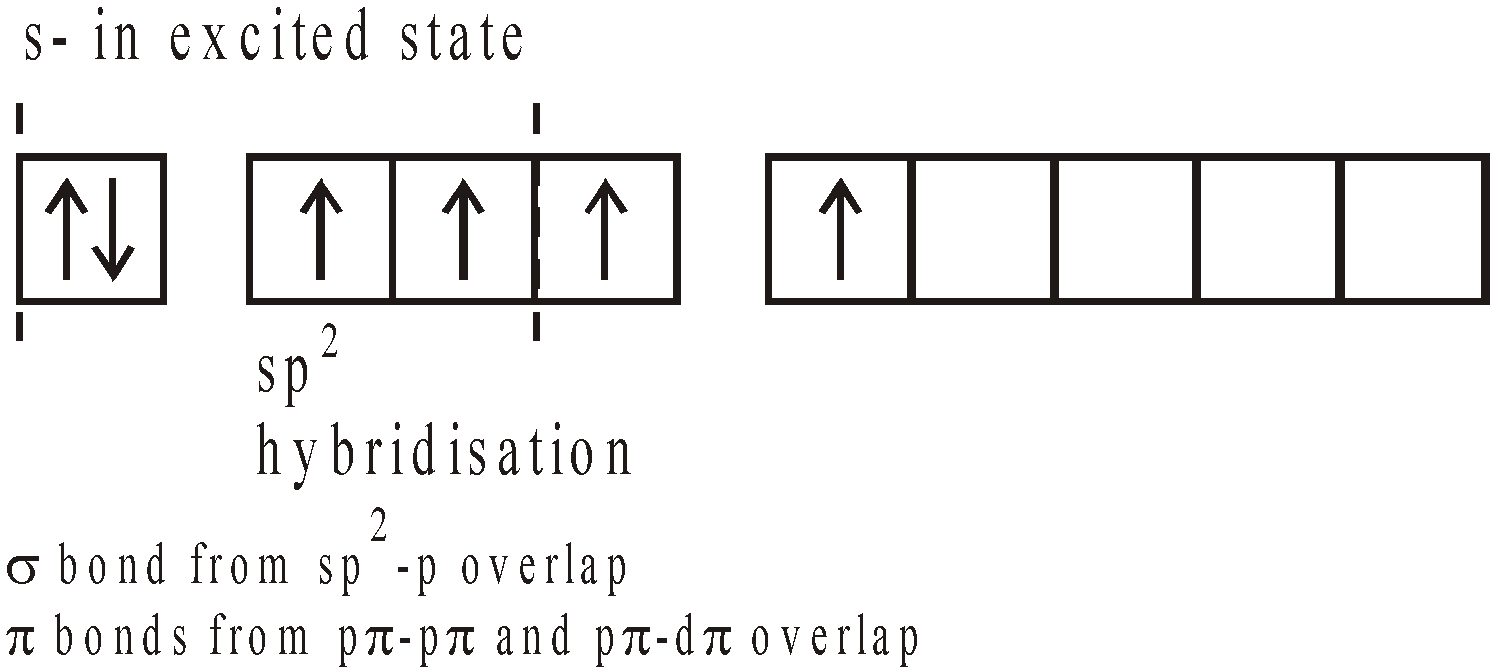
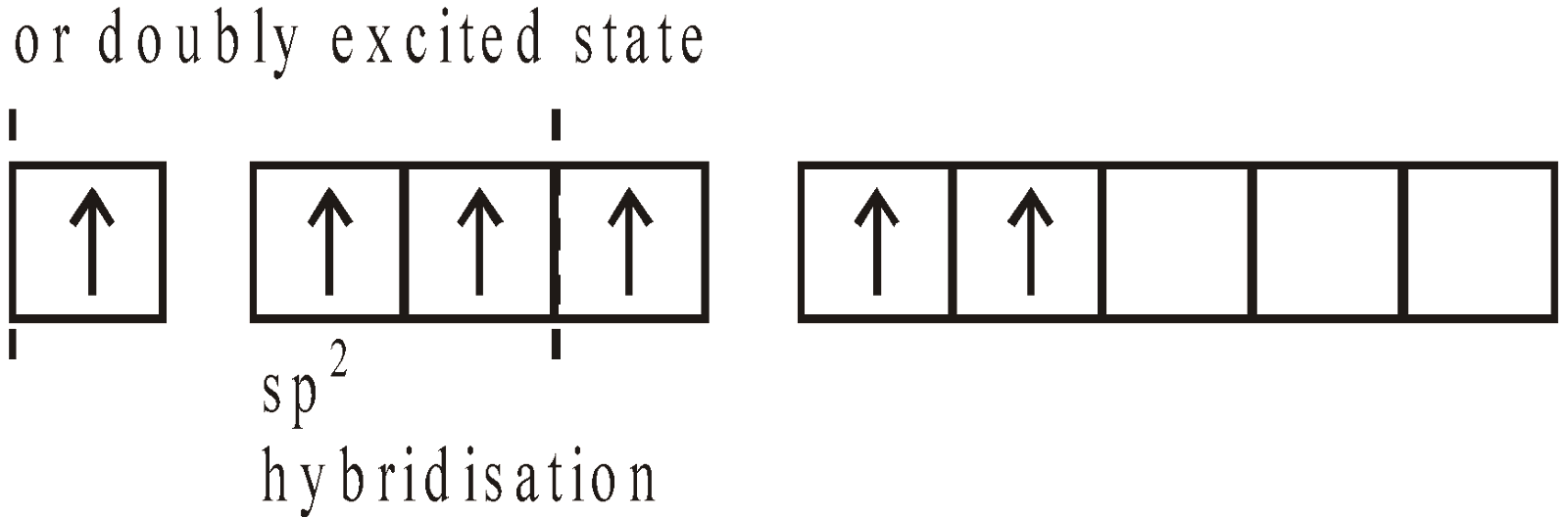
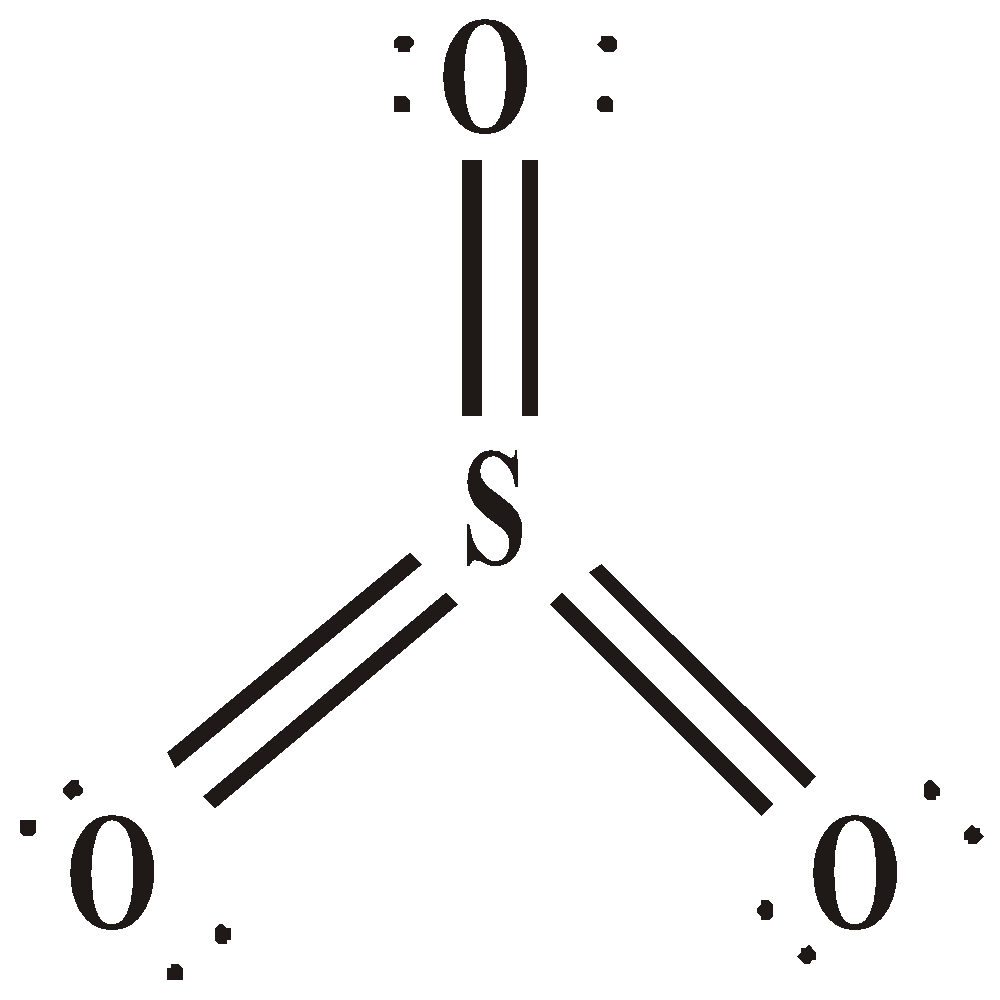
USES
HYDROGEN SULPHIDE (H2S)
PREPARATION
PROPERTIES
- Colourless poisonous gas with odour of rotten eggs fairly soluble in water
- Combustibility
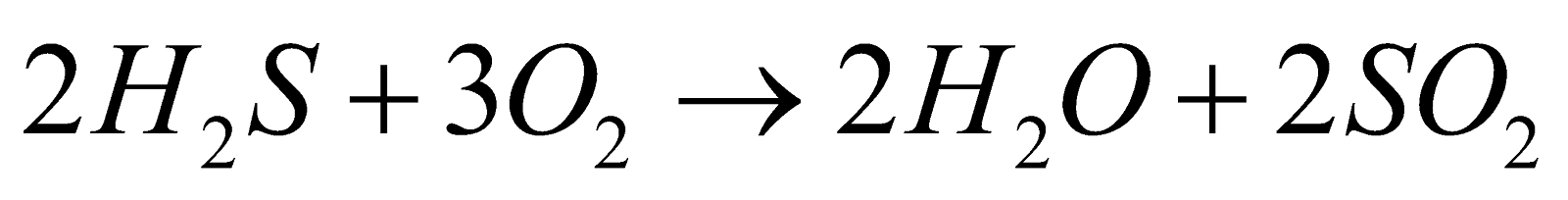
- Reducing nature
USES
SODIUM THIOSULPHATE (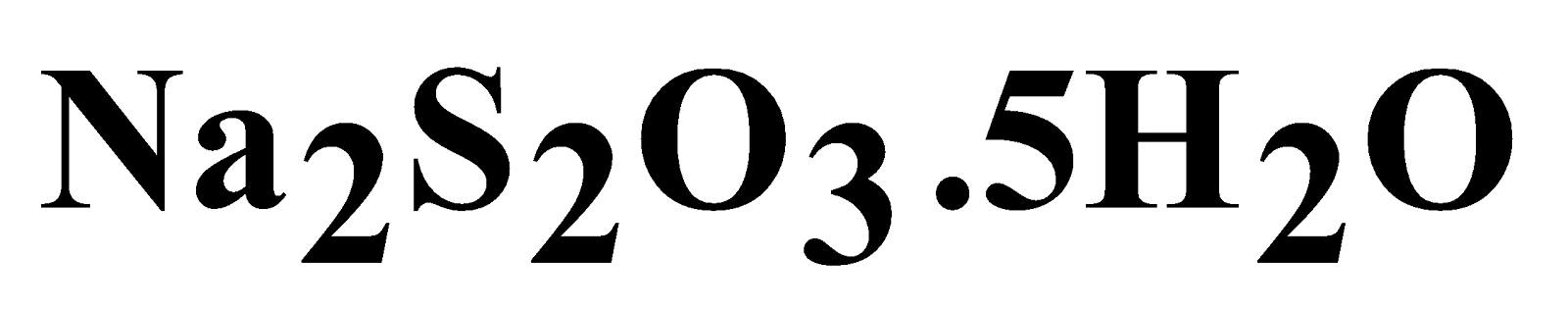 )
)
PREPARATION
- By boiling with flowers of sulphur in absence of air
- By heating sodium hydrogen sulphide and sodium hydrogen sulphite together
PROPERTIES
- Complexing – It dissolves silver halides forming complex
- It absorbs halogens
- Reaction with AgNO3
- Action of dil. H2SO4
USES
- It is used as an antichlor compound.
- Fixer in photography to remove AgBr left.
- In the metallurgy for extraction of Ag and Au.
