AP Chemistry 3.12 Properties of Photons Study Notes - New Syllabus Effective fall 2024
AP Chemistry 3.12 Properties of Photons Study Notes- New syllabus
AP Chemistry 3.12 Properties of Photons Study Notes – AP Chemistry – per latest AP Chemistry Syllabus.
LEARNING OBJECTIVE
Explain the properties of an absorbed or emitted photon in relationship to an electronic transition in an atom or molecule.
Key Concepts:
- Photon Absorption and Emission in Atoms and Molecules
- Relationships Among Wavelength, Frequency, and Photon Energy
Photon Absorption and Emission in Atoms and Molecules
When electromagnetic radiation interacts with matter, atoms or molecules can either absorb or emit photons. The energy of the absorbed or emitted photon corresponds exactly to the difference between two quantized energy levels of the atom or molecule.
1. Mechanism of Photon Absorption: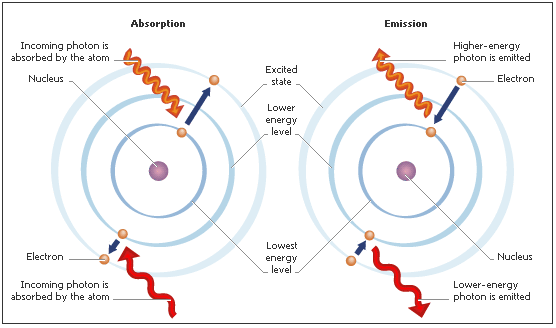
- When a photon of frequency \( \mathrm{\nu} \) strikes an atom or molecule, it can be absorbed only if its energy exactly matches the difference between two allowed energy levels.
- This causes the system to transition from a lower-energy state (ground or vibrational/electronic state) to a higher-energy excited state.
- Excess photon energy that does not match an energy gap will not be absorbed — hence, absorption occurs only at specific frequencies (quantization).
2. Mechanism of Photon Emission:
- When an excited atom or molecule returns to a lower-energy state, it emits a photon whose energy equals the difference between the two energy levels.
- This process is responsible for emission spectra — discrete lines corresponding to the released photon energies.
- In molecules, emissions may occur from excited electronic, vibrational, or rotational levels, leading to characteristic spectral patterns.
3. Sign Convention:
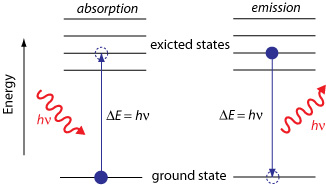
- Photon absorption: \( \mathrm{\Delta E > 0} \) → energy of the atom/molecule increases.
- Photon emission: \( \mathrm{\Delta E < 0} \) → energy of the atom/molecule decreases as it releases a photon.
4. Applications and Spectral Observations:
![]()
- Atomic Absorption/Emission Spectra: Each element emits or absorbs radiation at characteristic wavelengths (e.g., hydrogen emission lines: Balmer series). These are used for identifying elements in stars or flames.
- Molecular Spectra: Molecules show vibrational and rotational fine structure superimposed on electronic transitions, observed in infrared and UV–visible spectroscopy.
- Energy–Color Relationship: The energy of visible photons determines perceived color. For example, blue photons (\(\lambda \approx 450\, \mathrm{nm}\)) have higher energy than red photons (\(\lambda \approx 700\, \mathrm{nm}\)).
Example :
Explain why only certain wavelengths of light are absorbed by gaseous hydrogen atoms.
▶️ Answer/Explanation
Step 1: The electrons in hydrogen atoms occupy discrete energy levels given by the Bohr model.
Step 2: When a photon of specific energy \( \mathrm{E = h\nu} \) matches the difference between two allowed energy levels, it is absorbed, exciting the electron to a higher orbit.
Step 3: Photons with energies that do not correspond to any allowed transition will not be absorbed.
Final Answer: Hydrogen absorbs only specific wavelengths because its electrons can move only between quantized energy levels — leading to a line absorption spectrum rather than a continuous one.
Relationships Among Wavelength, Frequency, and Photon Energy
Electromagnetic radiation behaves as both a wave and a particle. Its wave nature relates the speed, frequency, and wavelength, while its particle nature relates the energy of individual photons to their frequency. These relationships form the mathematical foundation for understanding how light interacts with matter.
1. Wave Relationship — Speed, Wavelength, and Frequency
The speed of an electromagnetic wave (\( \mathrm{c} \)) is related to its wavelength (\( \mathrm{\lambda} \)) and frequency (\( \mathrm{\nu} \)) by the equation:
\( \mathrm{c = \lambda \nu} \)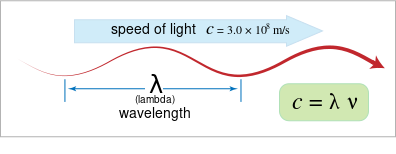
- \( \mathrm{c} \): Speed of light in vacuum = \( \mathrm{3.00 \times 10^8\, m/s} \)
- \( \mathrm{\lambda} \): Wavelength (m) — the distance between successive wave crests
- \( \mathrm{\nu} \): Frequency (s⁻¹ or Hz) — the number of wave cycles per second
Key Relationship: Wavelength and frequency are inversely related — as one increases, the other decreases.
\( \mathrm{\lambda \propto \dfrac{1}{\nu}} \)
Therefore, short-wavelength radiation (like gamma rays) has very high frequency, while long-wavelength radiation (like radio waves) has low frequency.
2. Photon Energy Relationship — Planck’s Equation
The energy of a photon depends directly on its frequency and inversely on its wavelength, as described by Planck’s equation: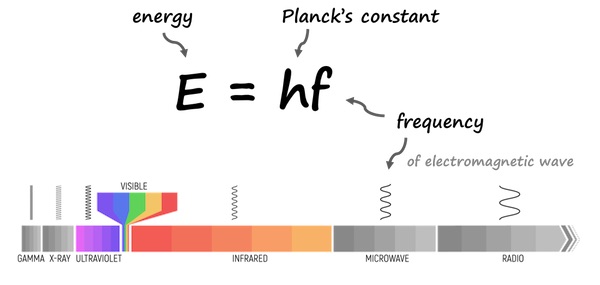
\( \mathrm{E = h \nu = \dfrac{hc}{\lambda}} \)
- \( \mathrm{E} \): Energy of a single photon (J)
- \( \mathrm{h} \): Planck’s constant = \( \mathrm{6.626 \times 10^{-34}\, J·s} \)
- \( \mathrm{\nu} \): Frequency of radiation (s⁻¹)
- \( \mathrm{\lambda} \): Wavelength (m)
Key Relationship:
- Higher frequency → higher photon energy.
- Shorter wavelength → higher photon energy.
Photon energy is quantized — it comes in discrete packets, not continuous amounts.
3. Combining Both Relationships:
By combining \( \mathrm{E = h \nu} \) and \( \mathrm{c = \lambda \nu} \):
\( \mathrm{E = \dfrac{hc}{\lambda}} \)
This equation directly connects a photon’s energy to its wavelength — shorter wavelengths correspond to higher-energy photons.
Example :
Calculate the energy of a photon with wavelength \( \mathrm{500\, nm} \) (green light).
▶️ Answer/Explanation
Step 1: Convert wavelength to meters. \( \mathrm{500\, nm = 500 \times 10^{-9}\, m = 5.00 \times 10^{-7}\, m} \)
Step 2: Use \( \mathrm{E = \dfrac{hc}{\lambda}} \).
\( \mathrm{E = \dfrac{(6.626 \times 10^{-34})(3.00 \times 10^8)}{5.00 \times 10^{-7}}} \)
\( \mathrm{E = 3.98 \times 10^{-19}\, J} \) per photon.
Step 3: Convert to energy per mole: \( \mathrm{E_{mol} = 3.98 \times 10^{-19} \times 6.022 \times 10^{23} = 2.40 \times 10^5\, J/mol = 240\, kJ/mol.} \)
Final Answer: A photon of 500 nm green light has energy \( \mathrm{3.98 \times 10^{-19}\, J} \) or \( \mathrm{240\, kJ/mol.} \)
Example :
Compare the energy of a 300 nm UV photon with a 600 nm red photon.
▶️ Answer/Explanation
Step 1: Since \( \mathrm{E \propto \dfrac{1}{\lambda}} \), shorter wavelength → higher energy.
Step 2: Ratio of energies = \( \mathrm{\dfrac{E_{UV}}{E_{red}} = \dfrac{\lambda_{red}}{\lambda_{UV}} = \dfrac{600}{300} = 2.} \)
Final Answer: A UV photon (300 nm) has twice the energy of a red photon (600 nm).
