AP Chemistry 5.4 Elementary Reactions Study Notes - New Syllabus Effective fall 2024
AP Chemistry 5.4 Elementary Reactions Study Notes – New syllabus
AP Chemistry 5.4 Elementary Reactions Study Notes – AP Chemistry – per latest AP Chemistry Syllabus.
LEARNING OBJECTIVE
Represent an elementary reaction as a rate law expression using stoichiometry.
Key Concepts:
- Rate Laws and Stoichiometry in Elementary Reactions
- Probability of Multi-Particle Collisions
Rate Laws and Stoichiometry in Elementary Reactions
An elementary reaction is a single-step reaction that occurs exactly as written, involving a direct collision between reactant species. For such reactions, the rate law can be written directly from the stoichiometric coefficients because they represent the actual molecular event.
In an elementary reaction, the molecularity (number of particles colliding) determines the form of the rate law.
General Rule:
| Type of Elementary Step | Reaction Equation | Rate Law | Molecularity |
|---|---|---|---|
| Unimolecular | \( \mathrm{A \rightarrow Products} \) | \( \mathrm{Rate = k[A]} \) | 1 |
| Bimolecular | \( \mathrm{A + B \rightarrow Products} \) | \( \mathrm{Rate = k[A][B]} \) | 2 |
| Termolecular | \( \mathrm{A + B + C \rightarrow Products} \) | \( \mathrm{Rate = k[A][B][C]} \) | 3 (rare) |
Key Points:
- Each elementary step represents a single collision event.
- The rate law is written directly from the reactants appearing in that step.
- Complex reactions consist of multiple elementary steps, but only the rate-determining step dictates the overall rate law.
Example
Consider the elementary reaction:
\( \mathrm{2NO + O_2 \rightarrow 2NO_2} \)
If this reaction occurs in a single elementary step, write the rate law and determine the molecularity.
▶️ Answer / Explanation
Step 1: Identify the reactants and their stoichiometric coefficients.
Reactants: \( \mathrm{NO} \) (coefficient 2) and \( \mathrm{O_2} \) (coefficient 1).
Step 2: Write rate law directly from the equation.
\( \mathrm{Rate = k[NO]^2[O_2]} \)
Step 3: Identify molecularity.
Two \( \mathrm{NO} \) molecules and one \( \mathrm{O_2} \) molecule collide → termolecular.
Result: \( \mathrm{Rate = k[NO]^2[O_2]} \); the reaction is termolecular and therefore would be rare as a single-step process.
Probability of Multi-Particle Collisions
Elementary reactions involving simultaneous collisions of three or more particles are rare because the likelihood of three reactant molecules colliding with the correct orientation and sufficient energy at the same instant is extremely small.
Most complex reactions that appear to involve multiple reactants actually occur as a sequence of simpler, bimolecular steps (two-body collisions), rather than a single termolecular step.
Reasons Termolecular Reactions Are Rare: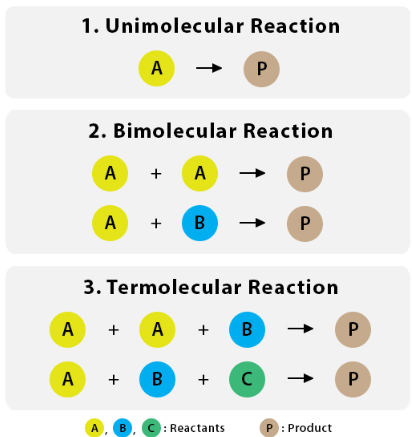
- The probability of three-body collisions occurring simultaneously is extremely low compared to two-body collisions.
- Even if three particles collide, they must have the correct orientation and sufficient energy to form products.
- Most apparent termolecular reactions occur through a series of elementary bimolecular steps.
Illustration — Two-Step Mechanism Instead of One:
Overall reaction:
\( \mathrm{2NO + O_2 \rightarrow 2NO_2} \)
Proposed mechanism:
- \( \mathrm{NO + O_2 \rightarrow NO_3} \) (slow)
- \( \mathrm{NO_3 + NO \rightarrow 2NO_2} \) (fast)
Explanation: Instead of a single collision among three molecules, the reaction proceeds through two simpler, sequential bimolecular collisions, which are far more probable.
Example
Why are elementary reactions involving three or more molecules colliding at once rarely observed?
▶️ Answer / Explanation
Step 1: Consider collision probability.
For a reaction to occur, particles must collide with correct energy and orientation.
Step 2: Compare likelihoods.
Two-particle collisions occur frequently; three-particle collisions at the same instant are extremely rare.
Step 3: Explain via reaction mechanism.
Instead of one termolecular event, the reaction proceeds via multiple bimolecular elementary steps, each much more probable.
Result: Elementary steps involving more than two molecules colliding simultaneously are statistically improbable, so complex reactions occur through simpler bimolecular processes.
