AP Chemistry 7.11 Introduction to Solubility Equilibria Study Notes - New Syllabus Effective fall 2024
AP Chemistry 7.11 Introduction to Solubility Equilibria Study Notes- New syllabus
AP Chemistry 7.11 Introduction to Solubility Equilibria Study Notes – AP Chemistry – per latest AP Chemistry Syllabus.
LEARNING OBJECTIVE
Calculate the solubility of a salt based on the value of Ksp for the salt.
Key Concepts:
- The Dissolution of a Salt and the Solubility Product Constant
- Calculating Solubility from Ksp
- Relating Solubility Rules and Ksp
- Using Molar Solubility to Calculate \( \mathrm{K_{sp}} \)
The Dissolution of a Salt and the Solubility Product Constant
The dissolution of an ionic compound is a reversible process that reaches an equilibrium between undissolved solid and its dissolved ions in solution. The extent of this equilibrium is represented by the solubility-product constant \( \mathrm{K_{sp}} \).
For a general salt \( \mathrm{A_{m}B_{n(s)}} \):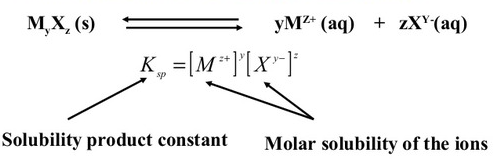
\( \mathrm{A_{m}B_{n(s)} ⇄ mA^{n+}(aq) + nB^{m-}(aq)} \)
The equilibrium expression for the solubility product is:
\( \mathrm{K_{sp} = [A^{n+}]^m [B^{m-}]^n} \)
- Solids are not included in \( \mathrm{K_{sp}} \) because their concentrations are constant.
- \( \mathrm{K_{sp}} \) provides a quantitative measure of a salt’s solubility — larger \( \mathrm{K_{sp}} \) → more soluble salt.
Example :
Reaction: \( \mathrm{PbCl_2(s) ⇄ Pb^{2+}(aq) + 2Cl^-(aq)} \)
Write the expression for \( \mathrm{K_{sp}} \).
▶️ Answer / Explanation
Step 1: Use stoichiometric coefficients as exponents.
Step 2: \( \mathrm{K_{sp} = [Pb^{2+}][Cl^-]^2} \)
Step 3: Note that \( \mathrm{[PbCl_2(s)]} \) is omitted because it’s constant.
Final Answer: \( \mathrm{K_{sp} = [Pb^{2+}][Cl^-]^2} \)
Calculating Solubility from Ksp
The solubility of an ionic compound in mol/L can be calculated from its \( \mathrm{K_{sp}} \) value by relating ion concentrations to the molar solubility (s).
General Formula:
For \( \mathrm{A_{m}B_{n(s)} ⇄ mA^{n+} + nB^{m-}} \):
\( \mathrm{K_{sp} = (m s)^m (n s)^n = s^{m+n} (m^m n^n)} \)
- \( \mathrm{s} \): molar solubility (mol/L)
- Use algebra to solve for \( \mathrm{s} \) when \( \mathrm{K_{sp}} \) is known.
Example :
Reaction: \( \mathrm{Ag_2CrO_4(s) ⇄ 2Ag^+(aq) + CrO_4^{2−}(aq)} \)
Given: \( \mathrm{K_{sp} = 1.1 \times 10^{-12}} \)
Find the molar solubility (\( \mathrm{s} \)) of \( \mathrm{Ag_2CrO_4} \).
▶️ Answer / Explanation
Step 1: Let \( \mathrm{s} \) = molar solubility.
\( \mathrm{[Ag^+] = 2s, [CrO_4^{2−}] = s} \)
Step 2: Substitute into \( \mathrm{K_{sp}} \):
\( \mathrm{K_{sp} = (2s)^2 (s) = 4s^3} \)
Step 3: Solve for \( \mathrm{s} \): \( \mathrm{s = \sqrt[3]{\dfrac{K_{sp}}{4}} = \sqrt[3]{\dfrac{1.1 \times 10^{-12}}{4}} = 6.5 \times 10^{-5}\ mol/L} \)
Final Answer: \( \mathrm{s = 6.5 \times 10^{-5}\ mol/L} \)
Relating Solubility Rules and Ksp
The empirical solubility rules can be explained quantitatively using \( \mathrm{K_{sp}} \): salts with large \( \mathrm{K_{sp}} \) values (>1) are highly soluble, whereas those with very small \( \mathrm{K_{sp}} \) values are sparingly soluble or practically insoluble.

Key Relationship:
- \( \mathrm{K_{sp} \gg 1} \): soluble salt — dissolves extensively.
- \( \mathrm{K_{sp} \ll 1} \): insoluble salt — dissolves very slightly.
- Solubility rules (e.g., “all nitrates are soluble”) reflect very high \( \mathrm{K_{sp}} \) values.
Example :
Data:
- \( \mathrm{K_{sp}(AgCl) = 1.8 \times 10^{-10}} \)
- \( \mathrm{K_{sp}(AgBr) = 5.0 \times 10^{-13}} \)
- \( \mathrm{K_{sp}(AgI) = 8.3 \times 10^{-17}} \)
Rank the halide salts by solubility.
▶️ Answer / Explanation
Step 1: For 1:1 salts, solubility is directly proportional to \( \mathrm{\sqrt{K_{sp}}} \).
Step 2: \( \mathrm{AgCl > AgBr > AgI} \)
Step 3: AgCl dissolves most readily; AgI is least soluble.
Using Molar Solubility to Calculate \( \mathrm{K_{sp}} \)
The molar solubility of a salt (\( \mathrm{s} \), in mol/L) represents the number of moles of solute that dissolve per liter of solution to reach equilibrium. Once \( \mathrm{s} \) is known, it can be used to calculate the solubility product constant \( \mathrm{K_{sp}} \) using the stoichiometry of the dissolution equilibrium.
General Equation for Ionic Dissolution:
\( \mathrm{A_{m}B_{n(s)} ⇄ mA^{n+}(aq) + nB^{m-}(aq)} \)
Expression for \( \mathrm{K_{sp}} \):
\( \mathrm{K_{sp} = [A^{n+}]^m [B^{m-}]^n} \)
If the molar solubility is \( \mathrm{s} \):
- \( \mathrm{[A^{n+}] = m \times s} \)
- \( \mathrm{[B^{m-}] = n \times s} \)
Substitute into the equilibrium expression:
\( \mathrm{K_{sp} = (m s)^m (n s)^n = s^{(m+n)} (m^m n^n)} \)
Key Idea:
- The stoichiometric coefficients become exponents in the \( \mathrm{K_{sp}} \) expression.
- Solubility \( \mathrm{s} \) can be used directly to find \( \mathrm{K_{sp}} \), or vice versa.
- Units of \( \mathrm{K_{sp}} \) depend on the total number of ions produced.
Example :
Reaction: \( \mathrm{CaF_2(s) ⇄ Ca^{2+}(aq) + 2F^-(aq)} \)
Given: \( \mathrm{s = 2.1 \times 10^{-4}\ mol/L} \)
Calculate \( \mathrm{K_{sp}} \).
▶️ Answer / Explanation
Step 1: Write the equilibrium concentrations:
\( \mathrm{[Ca^{2+}] = s = 2.1 \times 10^{-4}} \)
\( \mathrm{[F^-] = 2s = 4.2 \times 10^{-4}} \)
Step 2: Substitute into the \( \mathrm{K_{sp}} \) expression:
\( \mathrm{K_{sp} = [Ca^{2+}][F^-]^2 = (s)(2s)^2 = 4s^3} \)
Step 3: Plug in \( \mathrm{s = 2.1 \times 10^{-4}} \):
\( \mathrm{K_{sp} = 4(2.1 \times 10^{-4})^3 = 4(9.26 \times 10^{-12}) = 3.7 \times 10^{-11}} \)
Final Answer: \( \mathrm{K_{sp} = 3.7 \times 10^{-11}} \)
Example :
Reaction: \( \mathrm{Al(OH)_3(s) ⇄ Al^{3+}(aq) + 3OH^-(aq)} \)
Given: \( \mathrm{s = 1.3 \times 10^{-9}\ mol/L} \)
Find \( \mathrm{K_{sp}} \).
▶️ Answer / Explanation
Step 1: Write the ion concentrations:
\( \mathrm{[Al^{3+}] = s} \)
\( \mathrm{[OH^-] = 3s} \)
Step 2: Write the \( \mathrm{K_{sp}} \) expression:
\( \mathrm{K_{sp} = [Al^{3+}][OH^-]^3 = (s)(3s)^3 = 27s^4} \)
Step 3: Substitute \( \mathrm{s = 1.3 \times 10^{-9}} \):
\( \mathrm{K_{sp} = 27(1.3 \times 10^{-9})^4 = 27(2.8561 \times 10^{-36}) = 7.7 \times 10^{-35}} \)
Final Answer: \( \mathrm{K_{sp} = 7.7 \times 10^{-35}} \)
