AP Chemistry 9.3 Gibbs Free Energy and Thermodynamic Favorability Study Notes - New Syllabus Effective fall 2024
AP Chemistry 9.3 Gibbs Free Energy and Thermodynamic Favorability Study Notes- New syllabus
AP Chemistry 9.3 Gibbs Free Energy and Thermodynamic Favorability Study Notes – AP Chemistry – per latest AP Chemistry Syllabus.
LEARNING OBJECTIVE
Explain whether a physical or chemical process is thermodynamically favored based on an evaluation of ΔGo.
Key Concepts:
Gibbs Free Energy and the Standard State
Thermodynamic Favorability and the Sign of \( \mathrm{\Delta G^\circ} \)
- Calculating \( \mathrm{\Delta G^\circ} \) from Standard Free Energies of Formation
- Role of Enthalpy and Entropy in Thermodynamic Favorability
- Calculating ( \mathrm{\Delta G^\circ} ) from ( \mathrm{\Delta H^\circ} ) and ( \mathrm{\Delta S^\circ} )
- Predicting Temperature Dependence of Favorability
Gibbs Free Energy and the Standard State
The Gibbs free energy change (\( \mathrm{\Delta G} \)) represents the maximum amount of energy available to do useful work during a chemical or physical process at constant temperature and pressure. When all substances involved are in their standard states, the Gibbs free energy change is written as \( \mathrm{\Delta G^\circ} \).
The standard Gibbs free energy change is the change in free energy when all reactants and products are in their standard states at 1.0 bar (or 1.0 atm) and 298 K (25 °C):
\( \mathrm{\Delta G^\circ = \Delta H^\circ – T\Delta S^\circ} \)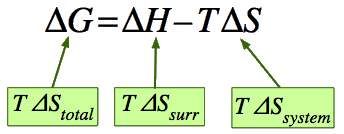
Where:
- \( \mathrm{\Delta H^\circ} \): standard enthalpy change (kJ/mol)
- \( \mathrm{\Delta S^\circ} \): standard entropy change (J/mol·K)
- \( \mathrm{T} \): absolute temperature (K)
Key Conditions for Standard State:
- Gases: 1.0 bar (or 1.0 atm) pressure
- Solutes: 1.0 M concentration
- Pure solids or liquids: most stable form at 1 bar and 298 K
- Temperature: typically 298 K (25 °C)
\( \mathrm{\Delta G^\circ} \) provides a thermodynamic reference point to compare reactions under identical, standardized conditions.
| Symbol | Quantity | Standard Condition |
|---|---|---|
| \( \mathrm{\Delta G^\circ} \) | Standard Gibbs free energy change | 1 bar, 1 M, 298 K |
| \( \mathrm{\Delta H^\circ} \) | Standard enthalpy change | 1 bar, 1 M, 298 K |
| \( \mathrm{\Delta S^\circ} \) | Standard entropy change | 1 bar, 1 M, 298 K |
Example :
At 298 K, the standard enthalpy change for the formation of liquid water is \( \mathrm{\Delta H^\circ = -285.8\ kJ/mol} \) and the standard entropy change is \( \mathrm{\Delta S^\circ = -163.3\ J/(mol·K)} \). Calculate the standard Gibbs free energy change \( \mathrm{(\Delta G^\circ)} \) for the reaction: \( \mathrm{H_2(g) + \tfrac{1}{2}O_2(g) \rightarrow H_2O(l)} \)
▶️ Answer/Explanation
Step 1: Convert entropy to kJ/mol·K for consistency: \( \mathrm{\Delta S^\circ = -163.3\ J/(mol·K) = -0.1633\ kJ/(mol·K)} \)
Step 2: Apply \( \mathrm{\Delta G^\circ = \Delta H^\circ – T\Delta S^\circ} \):
\( \mathrm{\Delta G^\circ = (-285.8) – (298)(-0.1633)} \)
\( \mathrm{\Delta G^\circ = -285.8 + 48.7 = -237.1\ kJ/mol} \)
Step 3: Interpretation: Since \( \mathrm{\Delta G^\circ < 0} \), the formation of water from hydrogen and oxygen is thermodynamically favorable under standard conditions.
Final Answer: \( \mathrm{\Delta G^\circ = -237.1\ kJ/mol} \)
Thermodynamic Favorability and the Sign of \( \mathrm{\Delta G^\circ} \)
The standard Gibbs free energy change (\( \mathrm{\Delta G^\circ} \)) is the key indicator of whether a chemical or physical process is thermodynamically favored under standard conditions. It combines enthalpy and entropy changes to determine the direction of spontaneity at constant temperature and pressure.
A process is said to be thermodynamically favored when the change in Gibbs free energy is negative under standard conditions:
\( \mathrm{\Delta G^\circ < 0} \Rightarrow \text{Process is thermodynamically favored} \)
Historically, the term “spontaneous” was used, but “thermodynamically favored” is preferred to avoid confusion with “fast” or “immediate.” A process can be thermodynamically favored yet occur very slowly (for example, the conversion of diamond to graphite).
Key Relationships:
\( \mathrm{\Delta G^\circ = \Delta H^\circ – T\Delta S^\circ} \)
- If \( \mathrm{\Delta G^\circ < 0} \): process is thermodynamically favored.
- If \( \mathrm{\Delta G^\circ > 0} \): process is non-favored (requires input of energy).
- If \( \mathrm{\Delta G^\circ = 0} \): system is at equilibrium.
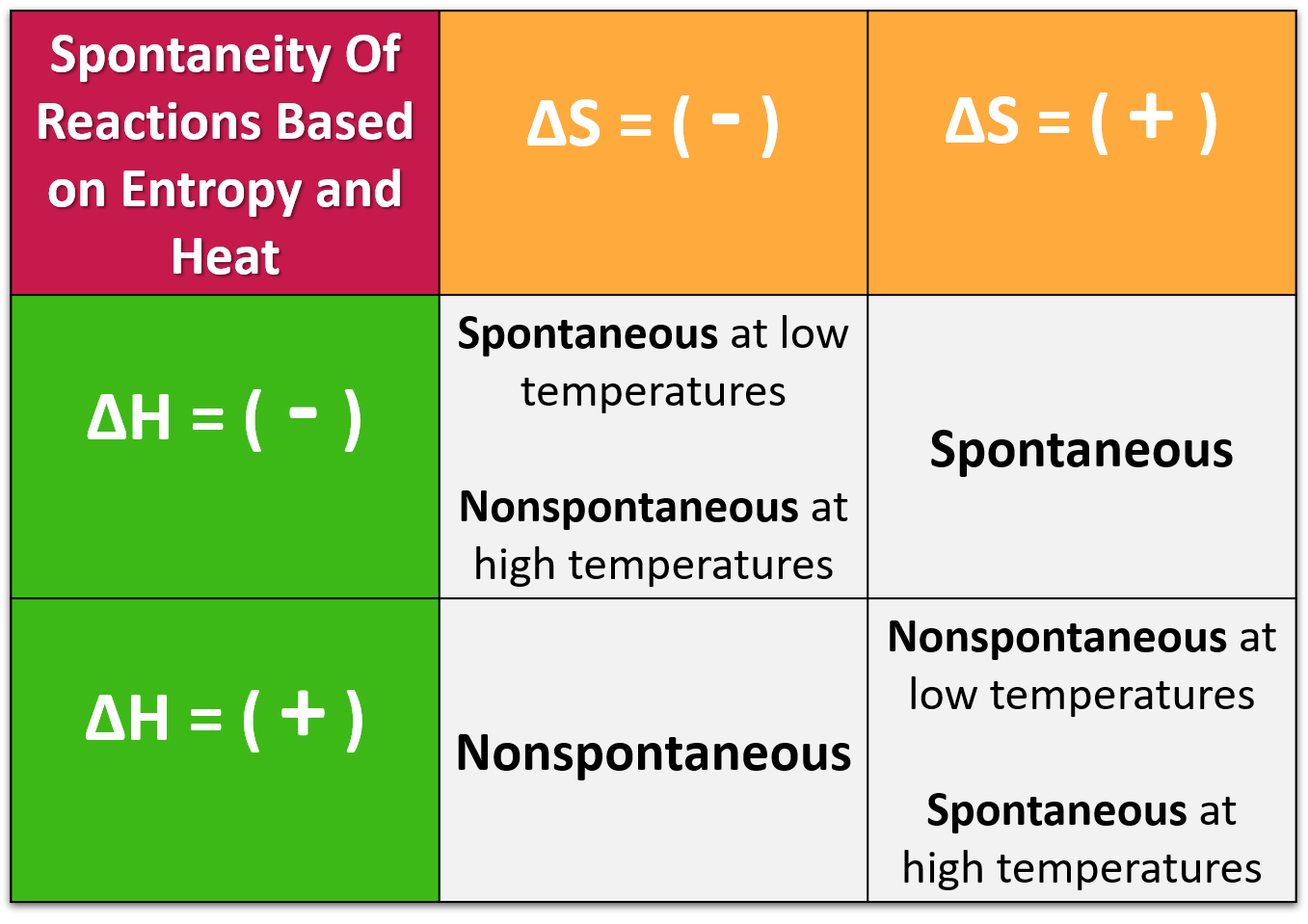
Effect of Temperature:
The sign of \( \mathrm{\Delta G} \) depends on both enthalpy and entropy changes. The four possible cases are summarized below:
| \( \mathrm{\Delta H} \) | \( \mathrm{\Delta S} \) | Temperature Dependence | Thermodynamic Favorability |
|---|---|---|---|
| − | + | All temperatures | Always favored (ΔG < 0) |
| + | − | All temperatures | Never favored (ΔG > 0) |
| − | − | Favored at low T | Temperature-dependent |
| + | + | Favored at high T | Temperature-dependent |
The sign and magnitude of \( \mathrm{\Delta G^\circ} \) reveal whether a reaction can occur spontaneously under standard conditions and how temperature affects that favorability.
Example:
For the decomposition of calcium carbonate: \( \mathrm{CaCO_3(s) \rightarrow CaO(s) + CO_2(g)} \) Given \( \mathrm{\Delta H^\circ = +178.3\ kJ/mol} \) and \( \mathrm{\Delta S^\circ = +160.5\ J/(mol·K)} \). Determine the temperature above which this reaction becomes thermodynamically favored.
▶️ Answer/Explanation
Step 1: For thermodynamic favorability, \( \mathrm{\Delta G^\circ = 0} \) (transition point between non-favored and favored).
Step 2: Use \( \mathrm{\Delta G^\circ = \Delta H^\circ – T\Delta S^\circ} \):
\( \mathrm{0 = \Delta H^\circ – T\Delta S^\circ} \Rightarrow T = \dfrac{\Delta H^\circ}{\Delta S^\circ} \)
Step 3: Convert entropy to kJ/mol·K: \( \mathrm{\Delta S^\circ = 160.5\ J/(mol·K) = 0.1605\ kJ/(mol·K)} \)
Step 4: Substitute values:
\( \mathrm{T = \dfrac{178.3}{0.1605} = 1111\ K} \)
Step 5: Interpretation:
- Below 1111 K → \( \mathrm{\Delta G^\circ > 0} \): decomposition is non-favored.
- Above 1111 K → \( \mathrm{\Delta G^\circ < 0} \): decomposition becomes thermodynamically favored.
Final Answer: The reaction becomes thermodynamically favored at temperatures above 1111 K.
Calculating \( \mathrm{\Delta G^\circ} \) from Standard Free Energies of Formation
The standard Gibbs free energy change for a chemical or physical process can be calculated from the standard Gibbs free energies of formation (\( \mathrm{\Delta G_f^\circ} \)) of the reactants and products. This allows prediction of thermodynamic favorability without direct experimental data.
The standard Gibbs free energy of formation (\( \mathrm{\Delta G_f^\circ} \)) is the free energy change that accompanies the formation of 1 mole of a compound from its elements in their standard states.
Formula for Reaction Free Energy:
\( \mathrm{\Delta G_{reaction}^\circ = \sum \Delta G_f^\circ (products) – \sum \Delta G_f^\circ (reactants)} \)
Where:
- \( \mathrm{\sum \Delta G_f^\circ (products)} \): sum of free energies of formation of all products (multiplied by their coefficients)
- \( \mathrm{\sum \Delta G_f^\circ (reactants)} \): sum of free energies of formation of all reactants (multiplied by their coefficients)
Conventions:
- \( \mathrm{\Delta G_f^\circ} \) for any element in its standard state (e.g., \( \mathrm{O_2(g), N_2(g), C(s)} \)) is zero.
- Negative \( \mathrm{\Delta G_f^\circ} \) indicates a compound that is more stable than its constituent elements.
A negative \( \mathrm{\Delta G_{reaction}^\circ} \) means the reaction is thermodynamically favored under standard conditions, while a positive value means it is not favored.
| Term | Symbol | Typical Unit | Meaning |
|---|---|---|---|
| Standard Gibbs free energy of formation | \( \mathrm{\Delta G_f^\circ} \) | kJ/mol | Free energy change to form 1 mol of a compound from elements |
| Standard Gibbs free energy of reaction | \( \mathrm{\Delta G_{reaction}^\circ} \) | kJ/mol | Sum of products − sum of reactants |
Example:
Calculate the standard Gibbs free energy change \( \mathrm{(\Delta G^\circ_{reaction})} \) at 298 K for the reaction:
\( \mathrm{2NO(g) + O_2(g) \rightarrow 2NO_2(g)} \)
Given data:
- \( \mathrm{\Delta G_f^\circ(NO(g)) = +86.6\ kJ/mol} \)
- \( \mathrm{\Delta G_f^\circ(NO_2(g)) = +51.3\ kJ/mol} \)
- \( \mathrm{\Delta G_f^\circ(O_2(g)) = 0\ kJ/mol} \)
▶️ Answer/Explanation
Step 1: Apply the formula: \( \mathrm{\Delta G^\circ_{reaction} = \sum \Delta G_f^\circ(products) – \sum \Delta G_f^\circ(reactants)} \)
Step 2: Substitute known values:
\( \mathrm{\Delta G^\circ_{reaction} = [2(+51.3)] – [2(+86.6) + 1(0)]} \)
Step 3: Simplify:
\( \mathrm{\Delta G^\circ_{reaction} = (102.6) – (173.2)} \)
\( \mathrm{\Delta G^\circ_{reaction} = -70.6\ kJ/mol} \)
Step 4: Interpretation:
- The negative sign indicates that the formation of \( \mathrm{NO_2(g)} \) from \( \mathrm{NO(g)} \) and \( \mathrm{O_2(g)} \) is thermodynamically favored under standard conditions.
- This aligns with observation: nitrogen oxides readily form at high temperatures and are stable once cooled.
Final Answer: \( \mathrm{\Delta G^\circ_{reaction} = -70.6\ kJ/mol} \) → thermodynamically favored.
Role of Enthalpy and Entropy in Thermodynamic Favorability
For many processes, both enthalpy (\( \mathrm{\Delta H^\circ} \)) and entropy (\( \mathrm{\Delta S^\circ} \)) changes influence whether a reaction is thermodynamically favored. A process may release heat (favorable enthalpy) but decrease disorder (unfavorable entropy), or vice versa. Determining overall favorability requires considering both quantities together.
A process is thermodynamically favored if it results in a decrease in Gibbs free energy (\( \mathrm{\Delta G^\circ < 0} \)), where:
\( \mathrm{\Delta G^\circ = \Delta H^\circ – T\Delta S^\circ} \)
Key Ideas:
- Enthalpy change (\( \mathrm{\Delta H^\circ} \)) reflects heat flow: exothermic processes (\( \mathrm{\Delta H^\circ < 0} \)) are usually favorable.
- Entropy change (\( \mathrm{\Delta S^\circ} \)) reflects disorder: increases in disorder (\( \mathrm{\Delta S^\circ > 0} \)) favor spontaneity.
- Temperature (\( \mathrm{T} \)) controls the relative influence of enthalpy and entropy on Gibbs free energy.
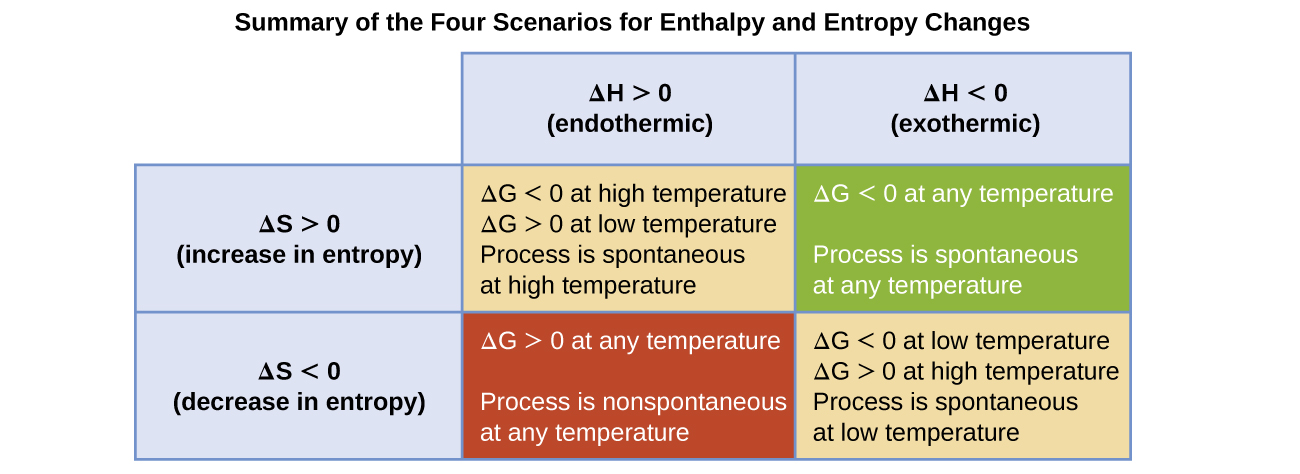
Some processes require evaluating both \( \mathrm{\Delta H^\circ} \) and \( \mathrm{\Delta S^\circ} \) to determine whether they are thermodynamically favored at a given temperature.
Example :
The freezing of water at 0 °C involves the process: \( \mathrm{H_2O(l) \rightarrow H_2O(s)} \). Given \( \mathrm{\Delta H^\circ = -6.01\ kJ/mol} \) and \( \mathrm{\Delta S^\circ = -22.0\ J/(mol·K)} \). Determine whether the process is thermodynamically favored at (a) 0 °C and (b) 25 °C.
▶️ Answer/Explanation
Step 1: Convert units for entropy: \( \mathrm{-22.0\ J/(mol·K) = -0.0220\ kJ/(mol·K)} \)
Step 2: Use \( \mathrm{\Delta G = \Delta H – T\Delta S} \)
(a) At 0 °C = 273 K: \( \mathrm{\Delta G = (-6.01) – (273)(-0.0220) = -6.01 + 6.01 = 0.00\ kJ/mol} \)
→ At 0 °C, \( \mathrm{\Delta G = 0} \): the process is at equilibrium (melting and freezing coexist).
(b) At 25 °C = 298 K: \( \mathrm{\Delta G = (-6.01) – (298)(-0.0220) = -6.01 + 6.56 = +0.55\ kJ/mol} \)
→ \( \mathrm{\Delta G > 0} \): freezing is not thermodynamically favored at 25 °C (melting dominates).
Final Answer: • At 0 °C → equilibrium • At 25 °C → not favored (water melts).
Calculating ( \mathrm{\Delta G^\circ} ) from ( \mathrm{\Delta H^\circ} ) and ( \mathrm{\Delta S^\circ} )
When the standard enthalpy and entropy changes for a process are known, the standard Gibbs free energy change can be calculated directly using the equation:
\( \mathrm{\Delta G^\circ = \Delta H^\circ – T\Delta S^\circ} \)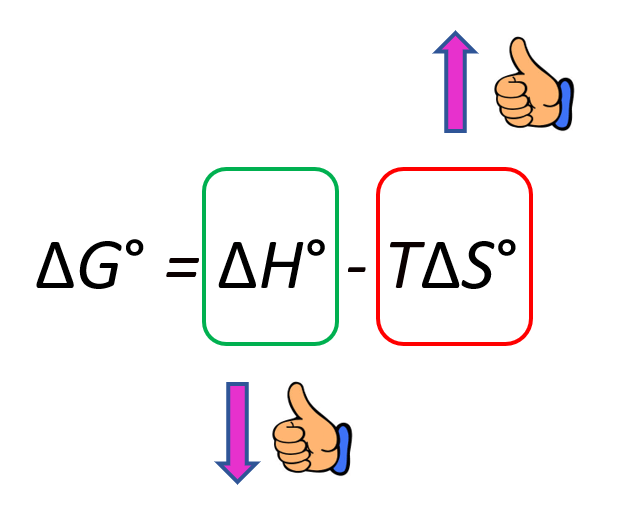
Key Points:
- \( \mathrm{\Delta H^\circ} \) and \( \mathrm{\Delta S^\circ} \) must be expressed in consistent units (usually kJ/mol and kJ/mol·K).
- Temperature plays a critical role — a process unfavorable at one temperature may become favorable at another.
- \( \mathrm{\Delta G^\circ} \) quantifies the balance between enthalpic and entropic effects.
By substituting known values of \( \mathrm{\Delta H^\circ} \) and \( \mathrm{\Delta S^\circ} \), chemists can determine whether a process is thermodynamically favorable at any temperature.
Example:
The decomposition of hydrogen peroxide: \( \mathrm{2H_2O_2(l) \rightarrow 2H_2O(l) + O_2(g)} \) has \( \mathrm{\Delta H^\circ = -196.0\ kJ/mol} \) and \( \mathrm{\Delta S^\circ = +126.1\ J/(mol·K)} \). Calculate \( \mathrm{\Delta G^\circ} \) at 298 K and determine whether the process is thermodynamically favored.
▶️ Answer/Explanation
Step 1: Convert \( \mathrm{\Delta S^\circ} \) to kJ/mol·K: \( \mathrm{+126.1\ J/(mol·K) = +0.1261\ kJ/(mol·K)} \)
Step 2: Apply \( \mathrm{\Delta G^\circ = \Delta H^\circ – T\Delta S^\circ} \):
\( \mathrm{\Delta G^\circ = (-196.0) – (298)(+0.1261)} \)
\( \mathrm{\Delta G^\circ = -196.0 – 37.6 = -233.6\ kJ/mol} \)
Step 3: Interpretation: \( \mathrm{\Delta G^\circ < 0} \) → process is thermodynamically favored.
Final Answer: \( \mathrm{\Delta G^\circ = -233.6\ kJ/mol} \), thermodynamically favored at 298 K.
Predicting Temperature Dependence of Favorability
The signs of \( \mathrm{\Delta H^\circ} \) and \( \mathrm{\Delta S^\circ} \) allow prediction of how temperature affects thermodynamic favorability. The equation \( \mathrm{\Delta G^\circ = \Delta H^\circ – T\Delta S^\circ} \) shows that temperature can either enhance or oppose spontaneity depending on whether the process is enthalpy- or entropy-driven.
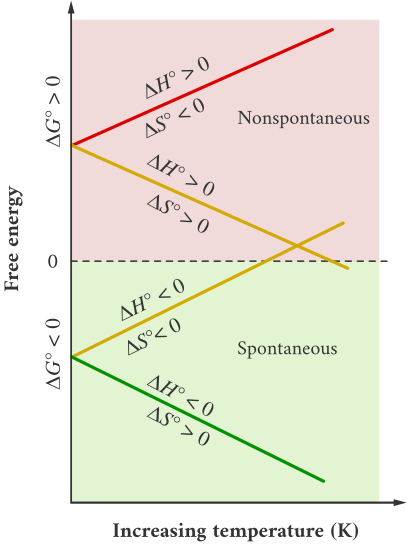
Temperature Dependence Table:
| \( \mathrm{\Delta H^\circ} \) | \( \mathrm{\Delta S^\circ} \) | Thermodynamic Favorability (\( \mathrm{\Delta G^\circ < 0} \)) |
|---|---|---|
| − | + | Favored at all \( \mathrm{T} \) |
| + | − | Never favored |
| − | − | Favored at low \( \mathrm{T} \) |
| + | + | Favored at high \( \mathrm{T} \) |
Key Note:
- If both \( \mathrm{\Delta H^\circ} \) and \( \mathrm{\Delta S^\circ} \) favor spontaneity, the reaction is always favored.
- If both oppose spontaneity, it’s never favored.
- If one favors and the other opposes, temperature determines the outcome.
Example:
Consider the evaporation of liquid water: \( \mathrm{H_2O(l) \rightarrow H_2O(g)} \) Given \( \mathrm{\Delta H^\circ = +44.0\ kJ/mol} \), \( \mathrm{\Delta S^\circ = +118.8\ J/(mol·K)} \). Determine the temperature above which this process is thermodynamically favored.
▶️ Answer/Explanation
Step 1: Set \( \mathrm{\Delta G^\circ = 0} \) at equilibrium: \( \mathrm{T = \dfrac{\Delta H^\circ}{\Delta S^\circ}} \)
Step 2: Convert \( \mathrm{\Delta S^\circ} \) to kJ/mol·K: \( \mathrm{+118.8\ J/(mol·K) = +0.1188\ kJ/(mol·K)} \)
Step 3: Calculate \( \mathrm{T} \): \( \mathrm{T = \dfrac{44.0}{0.1188} = 370.5\ K} \)
Step 4: Interpretation:
- Below 370 K (97 °C) → \( \mathrm{\Delta G^\circ > 0} \): condensation favored.
- Above 370 K → \( \mathrm{\Delta G^\circ < 0} \): evaporation favored.
Final Answer: Evaporation becomes thermodynamically favored above 370 K (≈97 °C), close to the boiling point of water.
