AP Chemistry 9.5 Free Energy and Equilibrium Study Notes - New Syllabus Effective fall 2024
AP Chemistry 9.5 Free Energy and Equilibrium Study Notes- New syllabus
AP Chemistry 9.5 Free Energy and Equilibrium Study Notes – AP Chemistry – per latest AP Chemistry Syllabus.
LEARNING OBJECTIVE
Explain whether a process is thermodynamically favored using the relationships between K, ΔGo, and T.
Key Concepts:
- Meaning of “Thermodynamically Favored”
- Relationship Between Gibbs Free Energy and the Equilibrium Constant
- Qualitative Connections Between \( \mathrm{K} \) and \( \mathrm{\Delta G^\circ} \)
- Interpreting Favorability Using \( \mathrm{\Delta G^\circ} \)
Meaning of “Thermodynamically Favored”
The phrase “thermodynamically favored” (also called spontaneous) describes a process for which the Gibbs free energy change is negative under standard conditions:
\( \mathrm{\Delta G^\circ < 0} \)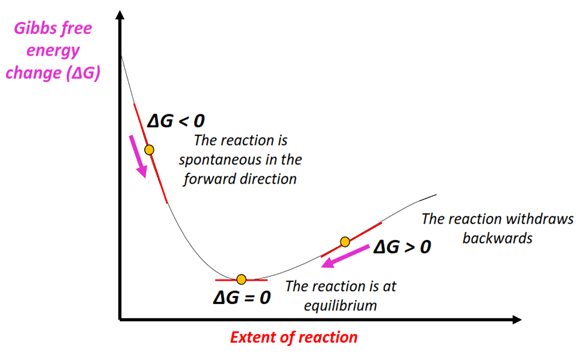
- When \( \mathrm{\Delta G^\circ < 0} \), the process is energetically favorable — meaning that the formation of products is preferred at equilibrium.
- At equilibrium, the position lies toward the products, so the equilibrium constant \( \mathrm{K > 1} \).
- The larger the value of \( \mathrm{K} \), the more product-favored the reaction.
Key Relationship
The relationship between Gibbs free energy and equilibrium constant is expressed as:
\( \mathrm{\Delta G^\circ = -RT \ln K} \)
- \( \mathrm{R} \): gas constant = 8.314 J·mol⁻¹·K⁻¹
- \( \mathrm{T} \): temperature (K)
- \( \mathrm{K} \): equilibrium constant
Key Concept Summary
- \( \mathrm{\Delta G^\circ < 0} \) → Products favored → \( \mathrm{K > 1} \).
- \( \mathrm{\Delta G^\circ > 0} \) → Reactants favored → \( \mathrm{K < 1} \).
- Thermodynamic favorability refers to equilibrium position, not reaction rate.
Example:
A reaction has \( \mathrm{\Delta G^\circ = -34.2\ kJ/mol} \) at 298 K. Calculate the equilibrium constant \( \mathrm{K} \), and state whether the reaction is thermodynamically favored.
▶️ Answer / Explanation
Step 1: Use the equation \( \mathrm{\Delta G^\circ = -RT \ln K} \).
\( \mathrm{-34200 = -(8.314)(298)\ln K} \)
Step 2: Simplify:
\( \mathrm{\ln K = \dfrac{34200}{(8.314)(298)} = 13.8} \)
Step 3: Calculate \( \mathrm{K} \):
\( \mathrm{K = e^{13.8} = 9.8 \times 10^5} \)
Step 4: Interpretation:
- \( \mathrm{K > 1} \): products favored at equilibrium.
- \( \mathrm{\Delta G^\circ < 0} \): thermodynamically favored reaction.
Final Answer: \( \mathrm{K = 9.8 \times 10^5} \); the reaction is thermodynamically favored and product-favored at equilibrium.
Relationship Between Gibbs Free Energy and the Equilibrium Constant
The relationship between the Gibbs free energy change (\( \mathrm{\Delta G^\circ} \)) and the equilibrium constant (\( \mathrm{K} \)) connects thermodynamics and equilibrium. It expresses how the position of equilibrium depends on the energy difference between products and reactants under standard conditions.
- At equilibrium, the forward and reverse reaction rates are equal, and the system’s free energy is minimized.
- \( \mathrm{K} \) provides a quantitative measure of how far the equilibrium lies toward products or reactants.
- \( \mathrm{\Delta G^\circ} \) tells us whether a reaction is spontaneous under standard conditions.
Key Equations
1. The equilibrium constant is related to free energy by:
\( \mathrm{K = e^{-\frac{\Delta G^\circ}{RT}}} \)
2. Alternatively, rearranging gives:
\( \mathrm{\Delta G^\circ = -RT \ln K} \)
- \( \mathrm{R = 8.314\ J\ mol^{-1}\ K^{-1}} \)
- \( \mathrm{T} \): absolute temperature in kelvin
- \( \mathrm{K} \): equilibrium constant (dimensionless)
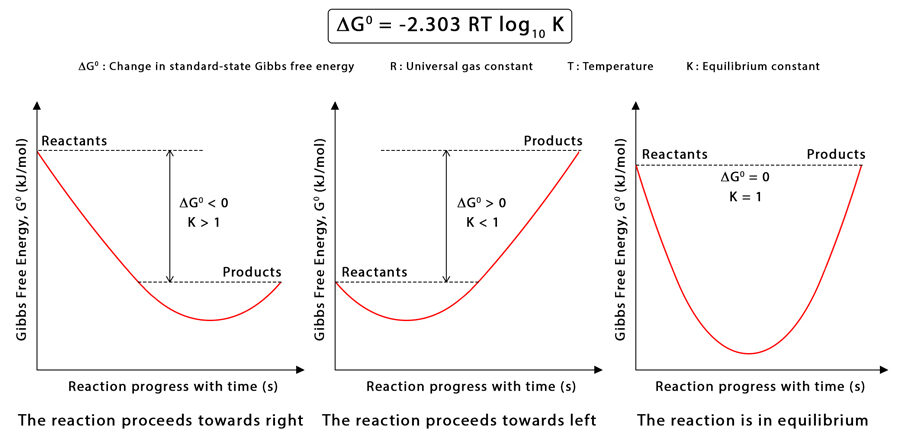
Derivation Overview
The relationship is derived from the condition for equilibrium:
\( \mathrm{\Delta G = \Delta G^\circ + RT \ln Q} \)
At equilibrium, \( \mathrm{\Delta G = 0} \) and \( \mathrm{Q = K} \). Substituting gives:
\( \mathrm{0 = \Delta G^\circ + RT \ln K} \Rightarrow \Delta G^\circ = -RT \ln K \)
Interpretation:
- If \( \mathrm{\Delta G^\circ < 0} \) → \( \mathrm{K > 1} \): Products are favored at equilibrium.
- If \( \mathrm{\Delta G^\circ > 0} \) → \( \mathrm{K < 1} \): Reactants are favored at equilibrium.
- If \( \mathrm{\Delta G^\circ = 0} \) → \( \mathrm{K = 1} \): System is equally balanced between reactants and products.
Key Concept Summary
- \( \mathrm{\Delta G^\circ} \) determines whether a reaction is spontaneous.
- \( \mathrm{K} \) quantifies the position of equilibrium.
- The exponential relationship means even small changes in \( \mathrm{\Delta G^\circ} \) can cause large changes in \( \mathrm{K} \).
Example
For a reaction at 298 K, the equilibrium constant is \( \mathrm{K = 2.5 \times 10^4} \). Calculate the standard Gibbs free energy change (\( \mathrm{\Delta G^\circ} \)) for the reaction, and interpret its meaning.
▶️ Answer / Explanation
Step 1: Use the equation \( \mathrm{\Delta G^\circ = -RT \ln K} \).
\( \mathrm{\Delta G^\circ = -(8.314)(298)\ln(2.5\times10^4)} \)
Step 2: Simplify:
\( \mathrm{\ln(2.5\times10^4) = 10.13} \)
\( \mathrm{\Delta G^\circ = -(8.314)(298)(10.13) = -25,100\ J/mol = -25.1\ kJ/mol} \)
Step 3: Interpretation:
- \( \mathrm{\Delta G^\circ = -25.1\ kJ/mol} \): negative value → spontaneous reaction.
- \( \mathrm{K > 1} \): products strongly favored at equilibrium.
Final Answer: \( \mathrm{\Delta G^\circ = -25.1\ kJ/mol} \); the reaction is thermodynamically favored and product-favored at equilibrium.
Qualitative Connections Between \( \mathrm{K} \) and \( \mathrm{\Delta G^\circ} \)
The relationship between the Gibbs free energy change (\( \mathrm{\Delta G^\circ} \)) and the equilibrium constant (\( \mathrm{K} \)) allows qualitative predictions about how favorable a reaction is. Even without exact calculation, we can estimate whether \( \mathrm{K} \) will be large, small, or approximately 1 based on the sign and magnitude of \( \mathrm{\Delta G^\circ} \).
- \( \mathrm{\Delta G^\circ} \) determines the thermodynamic favorability of a process.
- \( \mathrm{K} \) expresses how far the equilibrium lies toward products or reactants.
- The magnitude of \( \mathrm{\Delta G^\circ} \) indicates how strongly equilibrium favors one side.
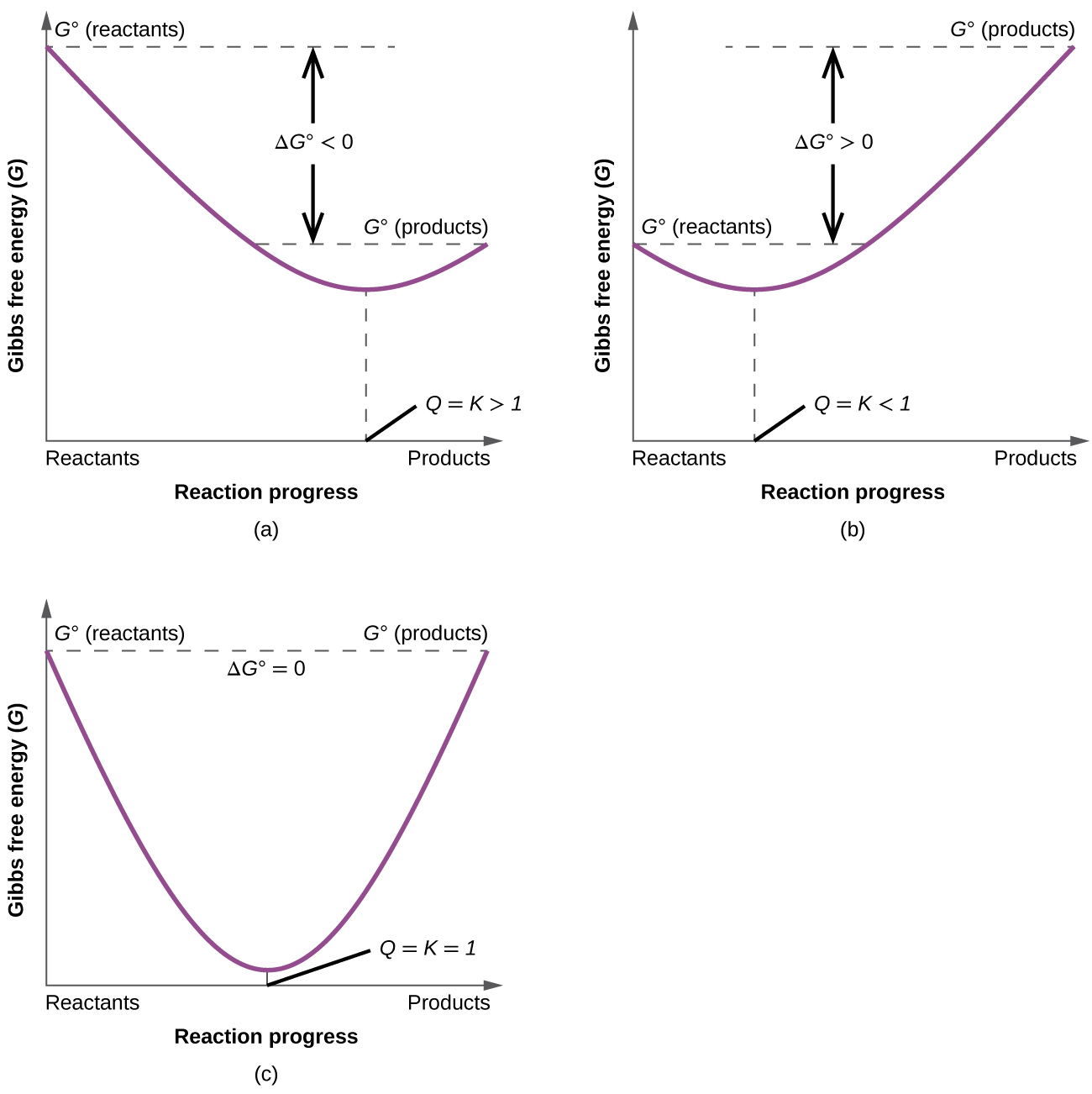
Key Equations
\( \mathrm{\Delta G^\circ = -RT \ln K} \)
\( \mathrm{K = e^{-\frac{\Delta G^\circ}{RT}}} \)
- \( \mathrm{R = 8.314\ J\ mol^{-1}\ K^{-1}} \)
- \( \mathrm{T} \): temperature in kelvin
Qualitative Interpretation
| Sign of \( \mathrm{\Delta G^\circ} \) | Magnitude of \( \mathrm{\Delta G^\circ} \) | Approximate Size of \( \mathrm{K} \) | Reaction Tendency |
|---|---|---|---|
| Large negative | ≫ \( \mathrm{RT} \) | ≫ 1 | Strongly product-favored |
| Slightly negative | ≈ \( \mathrm{RT} \) | > 1 but moderate | Moderately product-favored |
| ≈ 0 | ≈ 0 | ≈ 1 | Nearly balanced between reactants and products |
| Slightly positive | ≈ \( \mathrm{RT} \) | < 1 but moderate | Moderately reactant-favored |
| Large positive | ≫ \( \mathrm{RT} \) | ≪ 1 | Strongly reactant-favored |
Key Observations
- When \( \mathrm{\Delta G^\circ} \) is close to zero → \( \mathrm{K \approx 1} \), meaning both reactants and products are present in similar amounts at equilibrium.
- When \( |\mathrm{\Delta G^\circ}| \) is much greater than \( \mathrm{RT} \) (~2.5 kJ/mol at 298 K), \( \mathrm{K} \) deviates strongly from 1.
- Negative \( \mathrm{\Delta G^\circ} \) values produce large \( \mathrm{K} \); positive values produce small \( \mathrm{K} \).
Example:
Without detailed calculation, determine whether \( \mathrm{K} \) is large, small, or approximately 1 for each of the following reactions at 298 K:
- \( \mathrm{\Delta G^\circ = -50\ kJ/mol} \)
- \( \mathrm{\Delta G^\circ = +15\ kJ/mol} \)
- \( \mathrm{\Delta G^\circ = 0} \)
▶️ Answer / Explanation
Step 1: Recall that \( \mathrm{\Delta G^\circ = -RT \ln K} \).
Step 2: Compare each value of \( \mathrm{\Delta G^\circ} \) to \( \mathrm{RT \approx 2.5\ kJ/mol} \) at 298 K.
- Case (1): \( \mathrm{-50\ kJ/mol \ll 0} \) → much larger than \( \mathrm{RT} \) → \( \mathrm{K \gg 1} \) → strongly product-favored.
- Case (2): \( \mathrm{+15\ kJ/mol > 0} \) → larger than \( \mathrm{RT} \) → \( \mathrm{K \ll 1} \) → reactant-favored.
- Case (3): \( \mathrm{\Delta G^\circ = 0} \) → \( \mathrm{K = 1} \) → neither side favored.
Final Answer:
- (1) \( \mathrm{K \gg 1} \) → products dominate.
- (2) \( \mathrm{K \ll 1} \) → reactants dominate.
- (3) \( \mathrm{K = 1} \) → equilibrium balanced.
Interpreting Favorability Using \( \mathrm{\Delta G^\circ} \)
The sign of the standard Gibbs free energy change (\( \mathrm{\Delta G^\circ} \)) directly indicates whether a chemical or physical process is product-favored or reactant-favored at equilibrium under standard conditions.
- \( \mathrm{\Delta G^\circ} \) reflects the balance between enthalpy (\( \mathrm{\Delta H^\circ} \)) and entropy (\( \mathrm{\Delta S^\circ} \)).
- It determines the direction of spontaneous change and the equilibrium position.
- The corresponding equilibrium constant \( \mathrm{K} \) quantifies this favorability.
Key Equations
\( \mathrm{\Delta G^\circ = -RT \ln K} \)
\( \mathrm{K = e^{-\frac{\Delta G^\circ}{RT}}} \)
- \( \mathrm{R = 8.314\ J\ mol^{-1}\ K^{-1}} \)
- \( \mathrm{T} \): absolute temperature (K)
- \( \mathrm{K} \): equilibrium constant
Interpreting the Sign of \( \mathrm{\Delta G^\circ} \)
| Sign of \( \mathrm{\Delta G^\circ} \) | Relative Value of \( \mathrm{K} \) | Equilibrium Tendency | Thermodynamic Favorability |
|---|---|---|---|
| \( \mathrm{\Delta G^\circ < 0} \) | \( \mathrm{K > 1} \) | Products favored at equilibrium | Thermodynamically favored (spontaneous) |
| \( \mathrm{\Delta G^\circ = 0} \) | \( \mathrm{K = 1} \) | Reactants and products equally favored | System at equilibrium |
| \( \mathrm{\Delta G^\circ > 0} \) | \( \mathrm{K < 1} \) | Reactants favored at equilibrium | Not thermodynamically favored (nonspontaneous) |
Conceptual Summary
- \( \mathrm{\Delta G^\circ} \) and \( \mathrm{K} \) are inversely related — a negative \( \mathrm{\Delta G^\circ} \) corresponds to a large \( \mathrm{K} \), and vice versa.
- When \( \mathrm{K > 1} \), the forward reaction is spontaneous under standard conditions.
- When \( \mathrm{K < 1} \), the reverse reaction is spontaneous under standard conditions.
Example:
The reaction \( \mathrm{N_2(g) + 3H_2(g) \rightleftharpoons 2NH_3(g)} \) has \( \mathrm{\Delta G^\circ = -33.0\ kJ/mol} \) at 298 K. Determine whether the reaction is product- or reactant-favored and estimate \( \mathrm{K} \).
▶️ Answer / Explanation
Step 1: Use the equation \( \mathrm{K = e^{-\frac{\Delta G^\circ}{RT}}} \).
\( \mathrm{K = e^{-\frac{-33000}{(8.314)(298)}} = e^{13.3}} \)
Step 2: Simplify:
\( \mathrm{K = 6.0 \times 10^5} \)
Step 3: Interpretation:
- \( \mathrm{\Delta G^\circ < 0} \) → spontaneous → product-favored.
- \( \mathrm{K \gg 1} \) → equilibrium lies far to the right (toward \( \mathrm{NH_3} \)).
Final Answer: \( \mathrm{K = 6.0 \times 10^5} \); the reaction is strongly product-favored and thermodynamically spontaneous at 298 K.
