Respiration in Plants : Notes and Study Materials -pdf
Respiration in Plants Class 11 Notes Biology Chapter 14
Topics and Subtopics in for Class 11 Biology Chapter 14 Respiration in Plants:
| Section Name | Topic Name |
| 14 | Respiration in Plants |
| 14.1 | Do Plants Breathe? |
| 14.2 | Glycolysis |
| 14.3 | Fermentation |
| 14.4 | Aerobic Respiration |
| 14.5 | The Respiratory Balance Sheet |
| 14.6 | Amphibolic Pathway |
| 14.7 | Respiratory Quotient |
| 14.8 | Summary |
All living organisms require a continuous supply of energy for their survival. Energy is used to carry out various functions such as uptake of materials, absorption, growth, development, movement and even breathing. About 50% of the energy produced by the cell is utilized by these cellular activities and rest of it is changed into heat and get lost. Now, the question arises from where does this energy comes to carry out all these processes of life.
Topic 1 Respiration : The Basics
We eat food in order to obtain energy. The food we eat in the form of macro molecules is oxidized to fulfill energy requirement of body for caring out all the basic life processes.
As we have already studied in last chapter, that only green plants and cyanobacteria can prepare their own food by the process called photosynthesis. They use trapped energy in order to obtain their food by converting light energy into chemical energy, which thereby gets stored into the bonds of carbohydrates such as glucose, sucrose, starch, etc.
But all cells, tissues and organs in plants do not photosynthesise instead, photosynthesis takes place in only some parts of plants, i.e., only cells that contain chloroplasts (mosdy areas located in superficial layers).
Hence, all other organs, tissues and cells in green plant that are non-green are need food for oxidation. Hence, food has to be translocated from the green parts to the non-green parts for oxidation processes.
Need of Photosynthesis
Animals on the other hand are heterotrophic in nature, i.e., they either obtain their food directly from the plants (herbivores) or indirectly, dependent on herbivores for their food (carnivores).
Saprophytes are dependent on dead and decaying matter for their food (e.g., Fungi). Thus, it can be concluded that all the food that is respired for life processes ultimately comes from photosynthesis.
Cellular Respiration
Cellular respiration or the mechanism of breakdown of food materials within-the cell to release energy and trapping the same energy for synthesis of ATP.
Respiration is the process of breaking of the C-C bonds of complex compounds through oxidation within the cells, leading to release of considerable amount of energy.
It is to be noted that site of breaking down of complex molecules to yield energy is cytoplasm and mitochondria (also only in eukaryotes) which is different from the site of photosynthesis, which is chloroplast in plants.
Respiratory Substrates
The compounds that are oxidised during the process of respiration are called respiratory substrates. Carbohydrates are used as major respiratory substrates are oxidised in high amounts, to release energy, but under some conditions in some plants, proteins, fats and organic acids are also used as respiratory substrates.
Differences between Respiration and Combustion
ATP : Energy Currency of the Cell
During the process of oxidation of food within a cell, all the energy contained in the respiratory substrates is not released free into the cell, or in a single step. Instead it gets released in a series of step-wise reactions controlled by enzymes and is trapped as chemical energy in the form of ATP.
Hence, the energy released in respiration by the process of oxidation is not used directly but is used in synthesising ATP (which is utilised whenever energy needs to be utilised).
Thus, it is said that ATP acts as the energy currency of the cell. The energy trapped in ATP is utilised in many energy requiring processes of the organisms, and the carbon skeleton produced during respiration is used as precursors for the biosynthesis of other molecules in a cell.
Do Plants Breathe : Exchange of Gases in Plants
For the process of respiration, plant takes O2 and releases CO2. Plants have stomata and lenticles for gaseous exchange instead of specialised organs that are present in animals for exchange of gases.
Following are the reasons which shows, how plants can get along without respiratory organs
(i) Every part of the plant has the ability to take care of its own needs of gas exchange and also very little transport of gases occur from one part of the plant to another.
(ii) It is only during the process of photosynthesis that large volumes of gases are exchanged and each leaf of the plant has ability to take care of its own needs during these periods.
Thus, when cells photosynthesis, availability of O2 is not a problem in these cells due to a continuous release of O2 that takes place within the cell.
(iii) Gases may easily diffuse in large, bulky plants as distance for diffusion is not so great because living cells in a plant are located quite close to the surface of the plant.
In case of stems, which are thick and woody in nature, the organisation of living cells is in the form of thin layers, which are found inside and beneath the bark. Like leaves which have stomata for gaseous exchange, these stems also have openings called lenticels. Internal cells are dead and provide only mechanical support to the plant.
This depicts that most cells of plant have atleast a part of their surface in contact with the air. The loose packing of parenchyma cells in leaves, stems and roots and provides an interconnected network of air spaces helps in facilitating this process.
Types of Respiration
We know that during the process of respiration, utilisation of O2 takes place with the release of CO2, water and energy as products.
According to the dependence of cells on oxygen, cellular respiration may be classified into two types as given below
1 Aerobic Respiration
This is the type of respiration in which organism utilise oxygen for the complete oxidation of organic food into CO2 and water. It occurs inside the mitochondria.
Aerobic respiration yields more energy as the respiratory substrate gets completely oxidised in the presence of O2.
2. Anaerobic Respiration
This is the type of respiration in which organic food is oxidised incompletely without utilising energy as oxidant. It occurs in cytoplasm and often releases small amount of energy.
It is believed that the first cells on this planet lived in an oxygen free environment, i.e., they were anaerobes. Even among present day living organisms, several are adapted to anaerobic conditions.
Some of them are facultative anaerobes (organisms that have capability of switching from aerobic to anaerobic conditions according to the availability of oxygen) while others are obligate anaerobes (organisms that are killed by normal atmospheric concentration of oxygen of 21%).
Thus, in any case, all living organisms retain the enzymatic machinery for partial oxidation of glucose in the absence of oxygen. And this breakdown of glucose to pyruvic acid is called glycolysis.
Topic 2 Respiration: The Mechanism
Cellular respiration occurs inside the cell and proceeds with the help of enzymes. The first step in respiration (taking glucose as substrate) is the glycolysis (glucose oxidised to pyruvic acid). After which the pyruvic acid may enter the Krebs’ cycle (aerobic respiration) or undergo fermentation (anaerobic respiration).
Glycolysis
Glycolysis (Gr. Glycor-sugar; lysis-splitting), is a step-wise process by which one molecule of glucose (6C) breaks down into two molecules of pyruvic acid (3C).
The scheme of glycolysis was given by Gustav Embden, Otto Meyerhof and J Parnas and is often referred as the EMP pathway. It is a common pathway in both aerobic and anaerobic modes of respiration. But in case of anaerobic organisms, it is the only process of respiration.
Glycolysis occurs in the cytoplasm of the cell. During the process glucose gets partially oxidised. In plants this glucose is derived from sucrose (end product of photosynthesis) or from storage carbohydrates.
During the course of process in plant this sucrose is first converted into glucose and fructose by the action of invertase enzyme after this, these two monosaccharides enter the glycolytic pathway.
Steps Involved in Glycolysis
In glycolysis, a chain of 10 reactions often reactions occur under the control of different enzymes.
It involves the following steps
Step I Phosphorylation of glucose occur under the action of an enzyme hexokinase and Mg2+ that gives rise to glucose-6-phosphate by the utilisation of ATP.
Step II Isomerisation of this phosphorylated glucose-6-phsophate takes place to form fructose-6-phosphate with the help of an enzyme phosphohexose isomerase (Reversible Reaction).
Step III This fructose-6-phosphate is again phosphorylated by ATP in order to form fructose 1, 6-bisphosphate in the presence of an enzyme phosphofructokinase and Mg2+.
The steps of phosphorylation of glucose to fructose 1, 6-bisphosphate (i.e., from step 1 to 3) activates the sugar thus, preventing it from getting out of the cell.
Step IV Splitting of fructose 1, 6-bisphosphate takes place into two triose phosphate molecules, i.e., dihydroxyacetone 3-phosphate and 3-phosphoglyceraldehyde (i.e., PGAL). This reaction is catalysed by an enzyme aldolase.
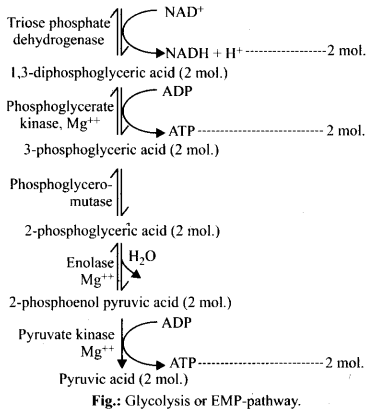
Step V Each molecule of PGAL removes two redox equivalents in the form of hydrogen atom and transfer them to a molecule of NAD+ (This NAD+ forms NADH + H+) and accepts inorganic phosphate (Pi) from phosphoric acid. This reaction in turn leads to the conversion to PGAL (which gets oxidised) to 1, 3-bisphosphoglycerate (BPGA) (Reversible reaction).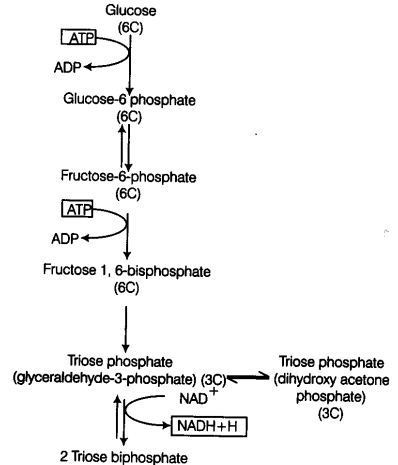
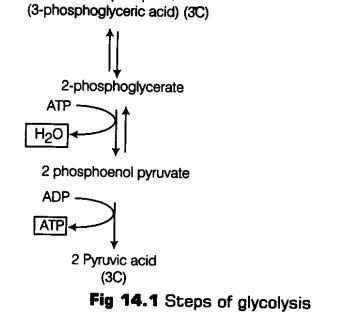
Step VI 1, 3-bisphosphoglycerate is converted to 3-phosphoglycerate with the formation of ATP.
This reaction is catalysed by an enzyme phosphoglycerate kinase.
It is also known as energy yielding process. The formatibn of ATP directly from metabolites constitutes substrate level phosphorylation (Reversible reaction).
Step VII In the next step, 3-phosphoglycerate is subsequently isomerised to form 2-phosphoglycerate, catalysed by enzyme phosphoglyceromutase (Reversible reaction).
Step VIII In the presence of enzyme enolase and Mg2+, with the loss of a water molecule, 2-phosphoglycerate is converted to Phosphoenol Pyruvate (PEP) (Reversible reaction).
Step IX High energy phosphate group of Phosphoenol Pyruvate (PEP) is transferred to a molecule of ADP, by the action of enzyme pyruvate kinase in the presence of Mg2+ and K+ This in turn produces two molecules of pyruvic acid (pyruvate) and a molecule of ATP by substrate level phosphorylation. The pyruvic acid thus, produced is the key product of glycolysis.
Metabolic Fate of Glycolysis
The overall reaction of glycolysis can be depicted as
* Glucose + 2Pi + 2ADP + 2NAD+ —> 2 Pyruvate + 2ATP + 2NADH + 2H+
* Two molecules of NADH on oxidation produce 6 molecules of ATP. Therefore, a net gain of 8ATP molecules occurs during glycolysis.
* The fete of glycolysis depends upon the availability of oxygen in the cell. In the presence of oxygen, pyruvic acid will enter the mitochondrion and undergo complete oxidation of glucose to CO 2 andH20 in aerobic respiration (Krebs’ cycle).
* On the other hand in the absence of oxygen, the pyruvic acid will undergo anaerobic respiration (lactic acid fermentation or alcoholic fermentation).
Note:
* Kostytcher (1902) coined the term anaerobic respiration.
* Glycolysis has two phases, i.e., preparatory (glucose is broken down to glyceraldehyde-3-phosphate) and pay off phase (the GAL-3-PD4 is changed into pyruvate producing NADH and ATP).
Fermentation
Various microorganisms, bacteria, animals and plants are known to catabolise pyruvic acid into various organic compounds depending upon the specific enzymes they possess.
Some of these types are as follows
(i) During alcoholic fermentation, in fungi (e.g., yeast), and some higher plants, the incomplete oxidation of glucose is achieved under anaerobic condition by a series of reactions in which pyruvic acid is converted to CO2 and ethanol.
It is done under two steps
(а) Pyruvic acid is first decarboxylated to acetaldehyde in the presence of enzyme pyruvic acid decarboxylase.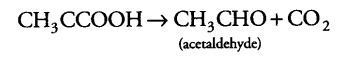
(b) This acetaldehyde is further reduced to ethyl alcohol or ethanol in the presence of enzyme, i.e., alcohol dehydrogenase.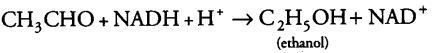
(ii) During lactic acid fermentation, organisms like some bacteria produces lactic acid as an end product from pyruvic acid.
During the reduction, the pyruvic acid produced in glycolysis is reduced by NADH2 to form lactic acid, CO2 is not produced and NADH2 is oxidised to NAD+.
This reaction is catalysed by an lactic acid dehydrogenase, FMN proteins and Zn2+ ions.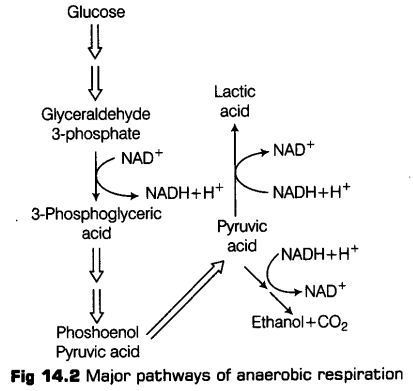
Likewise, in case of animal cells also (such as muscles) during exercise, when there is inadequate amount of oxygen for cellular respiration, pyruvic acid is reduced to lactic acid by lactate dehydrogenase.
Thus, in both the processes reoxidation of reducing (NADH + H+) agent takes place.
Energy Yield in Fermentation
In both alcoholic and lactic acid fermentation, the energy released is very less, i.e., not more than 7% of the energy is released from glucose and not all of it is trapped as high energy bonds of ATP.
Also, the fermentation processes are proved to be hazardous in nature because either acid or alcohol is produced on oxidation. Apart from this, yeasts may also poison themselves to death if the concentration of alcohol reaches about 13%.
Drawback of this process is that organisms cannot carryout complete oxidation of glucose and are also unable to extract out the energy stored to synthesise a larger number of ATP molecules required for cellular metabolism.
Differences between Glycolysis and Fermentation
Aerobic Respiration
Aerobic respiration is the next step (after glycolysis) that leads to complete oxidation of organic substances. It occurs in the presence of oxygen. The oxygen acts as a final acceptor of electron and protons are removed from the substrate. For aerobic respiration to take place within the mitochondria, the final product of glycolysis, i.e., pyruvic acid is transported into from the cytoplasm mitochondria and thus, the second phase of respiration is initiated.
The process of aerobic respiration involves two crucial events
(i) The complete oxidation of pyruvate occurs by the step-wise removal of all the hydrogen atoms, thereby, leaving three molecules of CO2. This occurs in the matrix of mitochondria.
(ii) The electrons removed as part of the hydrogen atoms are then passed on to molecular 02 with the simultaneous synthesis of ATP. This on the contrary takes place on the inner membrane of the mitochondria.
Oxidative Decarboxylation of Pyruvic Acid
In mitochondria, pyruvic acid (formed by the glycolytic catabolism of carbohydrates in cytosol) undergoes oxidative decarboxylation (i.e., removal of CO2 in aerobic conditions) forming a key compound, i.e., acetyl Co-A by the action of pyruvic acid dehydrogenase (in mitochondrial matrix) through a series of reactions.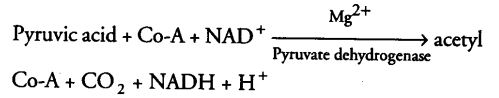
Thus, acetyl Co-A acts as a connecting link between glycolysis and citric acid cycle.
During this process, two molecules of NADH are produced from the metabolism of two molecules of pyruvic acid (produced from one glucose molecule during glycolysis).
Tricarboxylic Acid (TCA) Cycle
The acetyl Co-A then enters a cyclic pathway, Krebs’ cycle (or tricarboxylic acid cycle, TCA) in mitochondrial matrix. Various coenzymes including NAD+ and Co-A also participates in the reaction catalysed by pyruvic acid dehydrogenase.
It was first elucidated by Sir Hans Kreb, a British Biochemist in 1940.
The whole cycle explains how pyruvate is broken down to CO2 and water.
Following are the steps of Krebs ’ cycle
(i) Condensation The Krebs’ cycle starts with the condensation of acetyl group with oxaloacetic acid and water to yield citric acid, a 6C compound. This is the first stable product of the cycle.
This step is catalysed by an enzyme citrate synthetase. Co-A is liberated during this reaction.
(ii) Citric acid then undergoes reorganisation in two steps in order to form in the presence of an enzyme acinotase. intermediate
(iii) Oxidative decarboxylation Isocitrate is followed by two successive steps of oxidative decarboxylation, that leads to the formation of a-ketoglutaric acid, (a 5C compound in the presence of an enzyme isocitrate dehydrogenase and Mn1 ) and then succinyl Co-A, catalysed by a-complex. .
The succinyl Co-A then splits into a 4C compound succinic acid and Co-A with the addition of water. During this conversion, a molecule of GTP (guanosine triphosphate) is synthesised catalysed by an enzyme succinyl Co-A synthetase (this occurs when co-enzyme A transfers its high energy to a phosphate group that joins GDP forming GTP).
(i) GTP is also an energy carrier like ATP. Thus, this is the only high energy phosphate produced in the Krebs’ cycle.
(ii) In plants cells, this reaction also produces ATP from ADP.
In the remaining steps of Krebs’ cycle, succinyl Co-A is oxidised to oxaloacetic acid, a 4C compound following the formation of fumaric acid and malic acid catalysed by enzymes succinate dehydrogenase and fumacase respectively.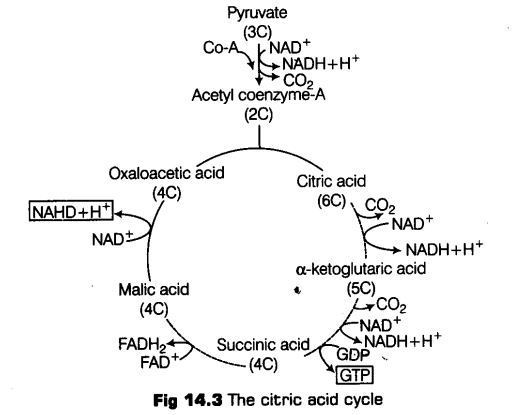
Output of Krebs’ Cycle or Citric Acid Cycle
During this cycle of reactions, 3 molecules of NAD+ are reduced to NADH + H+, and one molecule of FAD+ is reduced to FADH2. And also one molecule of ATP is reduced directly from GTP (by substrate level phosphorylation).
For continuous oxidation of acetyl Co-A, continued replenishment of oxaloacetic acid is necessary. In addition to this regeneration of NAD+ and FAD+ from NADH and FADH2 respectively are also required.
The summary equation for this phase of respiration is as follows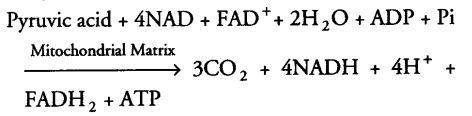
Till now, glucose has been broken down to release CO2 and 8 molecules of NADH+H+, two FADH2 are synthesised and just two molecules of ATP.
Importance of Citric Acid Cycle
The citric acid cycle is important in thefollowing ways
(i) This is the major pathway for the formation for ATP molecules.
(ii) Many intermediate compounds of this cycle are used in the synthesis of other biomolecules.
Differences between glycolysis and Krebs’cycle
CBSE Class 11 Biology Chapter-14 Important Questions
1 Marks Questions
1.Define fermentation and aerobic respiration.
Ans. Fermentation is partial breakdown of glucose.
Aerobic respiration glucose is completely degraded into CO2 and H2O.
2.What are the different types of respiration occurs in plants?
Ans. Aerobic respiration and Anaerobic respiration.
3.Name the energy currency of the cells.
Ans. ATP.
4.What are the other two names for kreb’s cycle?
Ans. Citric acid cycle (CAC), Tricarboxylic acid cycle (TCA)
5.In which organelle does kreb’s cycle occur in living cells?
Ans. Mitochondria.
6.Mention the conditions under which
(i) RQ is 1 (ii) R.Q is less than 1
Ans. (i) If carbohydrates are used as substrate and are fully oxidized the R.Q will be 1.
(ii) If fats are used in respiration, the R.Q well be less than 1.
7.What is respiration?
Ans. A process of physiochemical change by which environmental oxygen is taken into, to oxidize the stored food, for release of CO2, water and energy. The energy released is used for doing various life activities, whereas CO2 is used by the plants.
8.Give two types of cellular respiration.
Ans. (a) Aerobic (b) Anaerobic
9.How many carbon atoms are present in the molecule of each of :
Ans. (i) 6 carbon in glucose (ii) 3 carbon in pyruvate.
(i)Glucose and (ii) Pyruvate?
10.Nain the molecule which is terminal acceptor of electron.
Ans. Oxygen.
11.How many ATP mo1ecuIe am produced from a molecule of glucose on i complete oxidation in eukaryotes.
Ans. 36 ATP.
12.Where does ETC found in eukaryotic cell?
Ans. Mitochondrial membrane.
13.Name the enzyme which convert sugar into glucose and fructose.
Ans. Invertase.
14.How many molecules of ATP are produced by the oxidation of one molecule of FADH2?
Ans. 2 ATP molecules.
15.Why do the person with sufficient while fibres get fatigued ma short period ?
Ans. due to formation of Lactic acid.
16.Write the name of end product of glycolysis.
Ans. Pyruvic acid
17.Name the first product formed in Kerb’s cycle.
Ans. Citric acid.
2 Marks Questions
1.Define RQ. What is its value for fats?
Ans. Respiratory Quotient (RQ) : The ratio of the volume of CO2 evolved to the volume of O2 consumed in respiration is termed as the respiratory quotient or respiratory ratio.
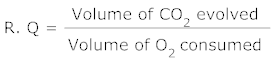
Its value for fats is less than one.
2.What is the importance of F0-F1 particles in ATP production during aerobic Respiration?
Ans. F1 head piece contains the site for ATP synthesis from ADP and phosphate.
F0 forms the channel through which protons cross the inner membrane.
3.What is oxidative decarboxylation? What happens to pyruvate immediately after this reaction?
Ans. Oxidative decarboxylation – It is the process in which carbon is removed from a compound as carbon-dioxide and the compound is oxidized.
Pyruvate is oxidatively decarboxylated into 2C acetate unit, which joins coenzyme A (COA) to form acetyl CO – A.
4.What is respiration?
Ans. A process of physicochemical change by which environmental oxygen is taken into, to oxidize the stored food, for release of CO2, water and energy. The energy released is used for doing various life activities, where as CO2 is used by the plaints.
5.Why less energy is produced during anaerobic respiration?
Ans. i) Incomplete breakdown of respiratory substrate takes place.
ii) Some of the products of anaerobic respiration can be oxidized further to release energy which shows that anaerobic respiration does not liberate the whole of energy contained on the respiratory substrate.
iii) O2 is not utilized for securing electrons & protons.
iv) NADH2 does not produce ATP as electron transport is absent.
6. What is the function of phosphofructokinase in glycolysis?
Ans. It catalyses the formation of fructose -1, 6- biphosphate from fructose-6- phosphate and adenosine –tri- phosphate (ATP) Fructose -1,6- biophosphate is splited into 2 molecules of triose phosphate – 3 phosphoglyceraldhyde and dihydroxyacetone phosphate.
7.Give difference between Breathing and Respiration?
Ans.
| Breathing | Respiration | |
| a. | It is a biophysical process. | It is a biochemical process |
| b. | Oxygen is taken in and CO2 is given out. | Water, carbon dioxide and energy is released by the oxidation of carbohydrates. |
8.Define aerobic respiration?
Ans. The process of release of energy through intake of molecular oxygen and release of CO2 is known as aerobic respiration.
9.What is compensation point?
Ans. At low concentration of CO2 and non-limiting light intensity, photosynthetic rate of a given plant will be equal to the total amount of respiration. Atmospheric concentration of CO2 at which photosynthesis just compensates for respiration is referred to as the CO2 compensation point.
10. Mention two steps of glycol sis in which ATP is utilized.
Ans. (i) ATP molecules are formed by direct transfer of Pi to ADP.
(ii) By oxidation of NADH.
3 Marks Questions
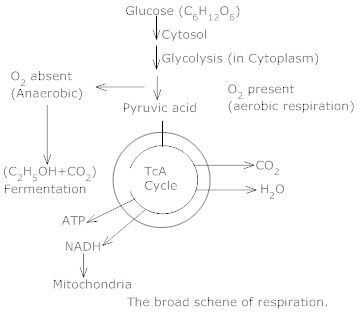
1.Describe the mechanism of Respiration.
Ans. Mechanism of respiration – Glucose molecule is broken down into an intermediate molecule, Pyruvic acid.
a) Breakdown of pyruvic acid in anaerobic respiration – In this process in absence of oxygen the pyruvic acid is incompletely reduced to ethyl alcohol.
Glucose → Ethyl alcohol + CO2 + 2ATP
b) Breakdown of pyruvic acid in aerobic respiration – In this process the pyruvic acid is completely oxidized into CO2 and H2O is the presence of oxygen. This process occurs in the mitochondria of the cell and is khown as kreb’s cycle.
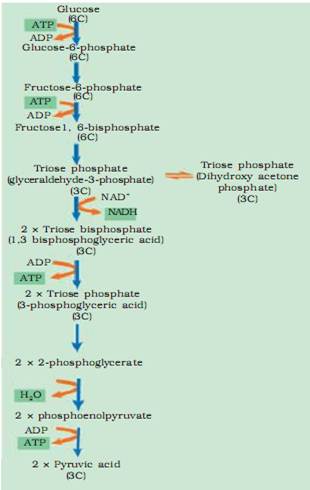
2.What are the various steps involved in glycolysis?
Ans. Steps of Glycolysis –
3.Explain Respiratory Balance sheet.
Ans. a) As equetial, orderly pathway functioning, with one substrate forming next one with glycolysis, TCA cycle and ETS pathway following one after another.
b) NADH synthesized in glycolysis. It is transferred into mitochondria and undergoes oxidative phosphorylation.
c) None of intermediates in pathway are used to form any other compound.
d) Only glucose is being respired; no other alternative substrates enter in pathway at any of intermediary stages.
4.What is the significance of stepwise release of energy in respiration?
Ans. Advantages of step wise oxidation during respirations-
a) It facilitates the utilization of a relatively higher proportion of that energy in ATP synthesis.
b) Activities of enzymes for the different steps may be enhanced or inhibited by specific compounds. This provides a means of controlling the rate of the pathway and the energy output according to the need of the cell.
c) The same pathway may be utilized for forming intermediates used in the synthesis of other bimolecular like amino acids.
5.Write the significance of citric acid cycle.
Ans. i) It explains the process of breaking of pyruvate into CO2 and water.
It is major pathway of generation of ATP.
(ii) More energy is released (30 ATP) in this process as compared to glycolysis.
(iii) Many intermediates compounds are formed. They are used in the synthesis of other bimolecules like amino-acids, nucleotides, Chlorophyll, cytochromes and fats.
6.Explain fermentation.
Ans. It occurs in some organisms like some bacteria that produce lactic acid from pyruvic acid.
In animal cells, such as muscles during exercise, when O2 is inadequate for cellular exercise, the pyruvic acid is reduced to lactic acid by lactate dehydrogrogenase. Reducing agent is NADH + H+ that is reoxidised to NAD+ in both processes.
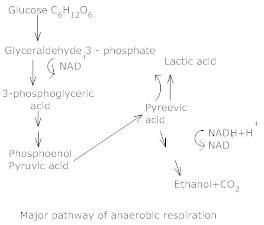
In both lactic acid and alcohol fermentation not much energy is relased; less than seven per cent of the energy in glucose is released and not all it is trapped as high energy bonds of ATP. The processes are hazardous either the acid or alcohol is produced. Yeasts poison themselves to death when the concentration of alcohol reaches approximately 13%.
5 Marks Questions
1.Describe the process and role of citric acid cycle in living organisms.
Ans. It is called “tricarboxylic acid cycle”. Following steps are present for completing this cycle-
(i)In this step, CO2 is removed from pyruvic acid and resulting 2- carbon unit with the sulphur containing compound coenzyme A forming Acetyl CoA. During this process the hydrogen released is accepted by NAD and NADH2 is produced.
Pyruvic acid _ CoA + NAD → Acetyl CoA + NADH2 + CO2
(ii) Acetyl coenzyme A reacts with a 4 – carbon compound oxaloacetic acid to form citric acid.
(iii) The citrate remains in equilibrium with cisaconitic acid and isocitric acid in the presence of the enzyme aconitase.
Citric acid → Isocitric acid.
(iv) Isocitrate is dehydrogenated in the presence of isocitrate dehydrogenase enzyme to form oxalosuccinate. The hydrogen released is accepted by NAD to form NADH2.
Isocitric acid + NAD → Oxalosuccinate + NADH2.
(v) A molecule of CO2 is lost from oxalosuccinate and a 5 – carbon compound  –ketoglutaric acid is formed in the presence of decarboxylate enzyme.
–ketoglutaric acid is formed in the presence of decarboxylate enzyme.
(vi)  – ketoglutarate loses a molecule of CO2 and 4 – carbon compound succinyl CoA is formed.
– ketoglutarate loses a molecule of CO2 and 4 – carbon compound succinyl CoA is formed.
 – keloglutarate + CoA + NAD → succinyl + CoA + NADH2 + CO2
– keloglutarate + CoA + NAD → succinyl + CoA + NADH2 + CO2
(vii) Succinyl CoA forms succinate, and ATP is found by linking ADP and inorganic phosphate (Pi)
Succinate CoA + ADP + Pi → Succinylate + CoA + ATP
b Succinate is oxidized into fumarate in the presence of succinate dehydrogenase enzyme. The hydrogen liberated is accepted by FAD and FADH2 is formed.
Succinate + FAD → Fumerate + FADH2
(ix) In this step the fumarate is converted into malate in the presence of enzyme fumarate hydrase (fumarase)
Fumarate → Malate.
(x) Malate is changed into oxaloacetate in the presence of the enzyme malate denydrogenase. NAD is reduced to NADH2 by the liberated hydrogen.
Thus oxaloacetic acid produced is ready to combine with the fresh acetyl CoA obtained from pyruvic acid for completing one cycle.
Net yield kreb’s cycle :- 1 Pyruvic acid + 1ADP + 4NAD + 1FAD à3CO2 + 1FADH2 + 4NADH2 + 1ATP
Thus total yield of energy
1ATP = 1 ATP
3×4NADH2 = 12ATP
2×1FADH2 = 2ATP
Total = 15 ATP
Thus 2 Pyruvic acid in ghycolysis yield, 15×2=30ATP.
2..Explain ETS.
Ans. Mechanism of Electron transport system – Glucose molecule is completely oxidized by the end of the citric acid cycle. The energy is not released unless NADH and FADH are oxidized through the ETS. The oxidation means ‘removal of electrons from it’. Metabolic pathway through which the electron passes from one carrier to another is called “Electron transport system” It is operative in the inner mitochondria membrane. Electrons from NADH produced in mitochondrial matrix are oxidized by NADH dehydrogenase (complex I) and electrons are then transferred to ubiqinone located within the inner membrane also receives reducing equivalents via FADH; that is generated during oxidation of succinate, through activity of enzyme named succinate dehydrogenase (complex II). Reduced ubiquinone is then oxidized with the transfer of electrons to cytochrome complex (complex III). Cytothrome is small protein attached to outer surface of inner membrane and acts as a mobile carries for transfer of electrons between complex III and complex IV. (complex IV) is cytochromes ‘c’ oxidize complex having cytochromes ‘a’ and a3.
When electron pass from one carries to another via complex I to IV in ETS, they are coupled to ATP synthase (complex V) for production ATP from ADP and inorganic phosphate. Oxidation of one molecule of NADH, gives rise to 3 molecules of ATP, while that of FADH, produces 2 molecules of ATP. Electrons are curried by cytochromes and recombine with their protons before the final stage when hydrogen atom is accepted by oxygen to form water. O2 acts as final hydrogen acceptor. Whole process by which oxygen allows the production of ATP by phosphorylation of ADP is called ‘oxidative phosphorylation’.
3.Give the various steps involved in Glycolysis.
Ans. The steps are as follows-
1) Glucose is phosphorylated in the presence of ATP, catalyzed by the enzyme hexokinase.
2) Glucose – 6 – phosphate is changed into its isomer fructose – 6 – phosphate catalyzed by phosphohexose isomers.
3) Fructose – 6 – phosphate is phosphorylated in the presence of ATP to form Fructose 1, 6 biphosphate.
4) Fructose 1, 6 biposphate is split into two molecules of triose phosphate one of 3 – phosphoglyceral dehyde and one of dihydroxyacetose phosphate, which are interconvertible. This reaction is catalysed by phosphofructokinase.
5) 3 – phosphoglyceraldehyde is oxieised to 1,3 biphosphoglycerate, with the reduction of NAD to NADH.
6) Phosphoglycerate kinase catalyses the formation of 3-phosphoglycerate to 1,3 biposphoglycerate and 1 molecule of ATP is produced directly (substrate phosphation).
7) 3-phosphoglycerate is converted into 2-phosphoglycerate and then into phosphoenlpysuvate (PEP)
8) PEP is converted into pyrucate long with the formation of one molecule of ATP directly. The enzyme pyruvate kinase catalyses this step.
The end products of glycolysis are 2 molecules of pyruvc acid + 2 NADH + 2ATP.
NCERT TEXTBOOK QUESTIONS FROM SOLVED
1. Give the schematic representation of an overall view of Krebs’ cycle.
Solution: 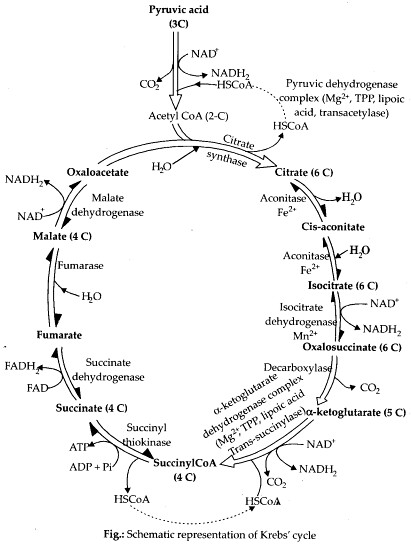
2. Differentiate between
(a) Respiration and Combustion
(b) Glycolysis and Krebs’cycle
(c) Aerobic respiration and Fermentation
Solution: (a) Differences between respiration and combustion are as follows :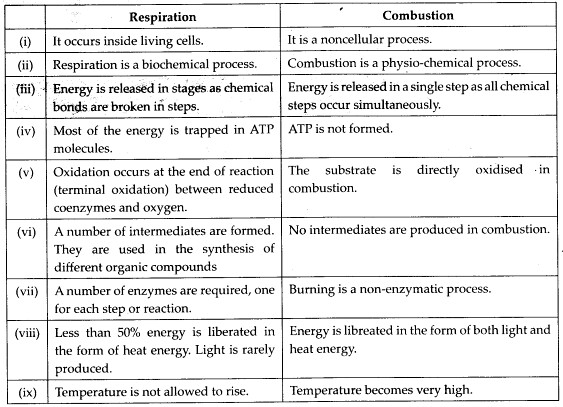
(b) Differences between glycolysis and Krebs’ cycle are as follows:

(C)Differences between aerobic respiration and fermentation are as follows: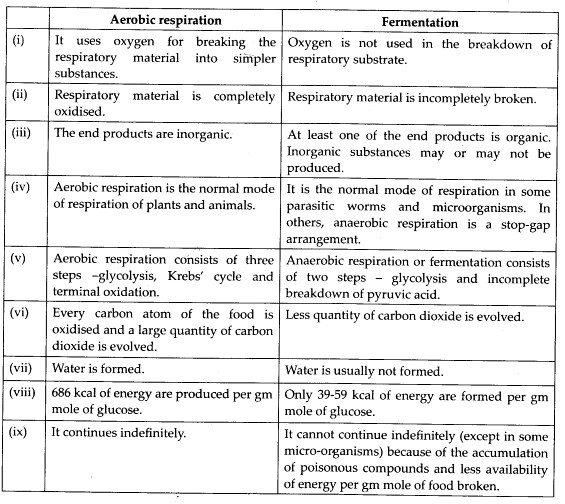
3. What are respiratory substrates? Name the most common respiratory substrate.
Solution: Respiratory substrates are those organic substances which are oxidised during respiration to liberate energy inside the living cells. The common respiratory substrates are carbohydrates, proteins, fats and organic acids. The most common respiratory substrate is glucose. It is a hexose monosaccharide.
4. Give the schematic representation of glycolysis.
Solution: 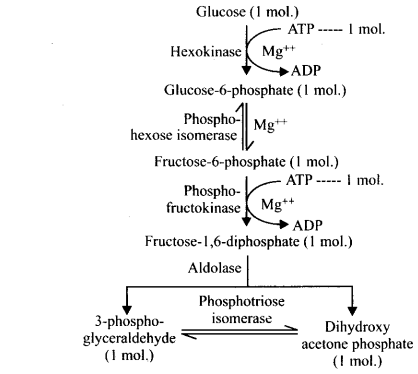
5. Explain ETS.
Solution: An electron transport chain or system (ETS) is a series of coenzymes and cytochromes that take part in the passage of electrons from
a chemical to its ultimate acceptor. Reduced coenzymes participate in electron transport chain. Electron transport takes place on cristae of mitochondria [oxysomes ( F0 -F1 , particles) found on the inner surface of the membrane of mitochondria]. NADH formed in glycolysis and citric acid cycle are oxidised by NADH dehydrogenase (complex I) and the electrons are transferred to ubiquinone. Ubiquinone also receives reducing equivalents via FADH2 through the activity of succinate dehydrogenase (complex II). The reduced ubiquinone is then oxidised by transfer of electrons of cytochrome c via cytochrome Fc, complex (complex III). Cytochrome c acts as a mobile carrier between complex III and complex IV. Complex IV refers to cytochrome c oxidase complex containing cytochromes a and a3and two copper centres. When the electrons are shunted over the carriers via complex I to IV in the electron transport chain, they are coupled to ATP synthetase (complex V) for the formation of ATP from ADP and Pi. Oxygen functions as the terminal acceptor of electrons and is reduced to water along with the hydrogen atoms. Reduced coenzymes (coenzyme I, II and FAD) do not combine directly with the molecular O2. Only their hydrogen or electrons are transferred through various substances and finally reach O2. The substances useful for the transfer of electron are called electron carriers. Only electrons are transferred through cytochromes (Cyt F1 Cyt c,,C2, a, a3) and finally reach molecular O2. Both cytochrome a and a3 form a system called cytochrome oxidase. Copper is also present in Cyt a3 in addition to iron. The molecular oxygen that has accepted electrons now receives the protons that were liberated into the surrounding medium to give rise to a molecule of water. The liberated energy is utilised for the synthesis of ATP from ADP and Pi.
6. What are the main steps in aerobic respiration? Where does it take place?
Solution: Aerobic respiration is an enzymatically controlled release of energy in a stepwise catabolic process of complete oxidation of organic food into carbon dioxide and water with oxygen acting as terminal oxidant. It
occurs by two methods, common pathway and pentose phosphate pathway. Common pathway is known so because its first step, called glycolysis, is common to both aerobic and anaerobic modes of respiration. The common pathway of aerobic respiration consists of three steps – glycolysis, Krebs’ cycle and terminal oxidation. Aerobic respiration takes place within mitochondria. The final product of glycolysis, pyruvate is transported from the cytoplasm into the mitochondria.
7. What are the assumptions made during the calculation of net gain of ATP?
Solution: It is possible to make calculations of the net gain of ATP for every glucose molecule oxidised; but in reality this can remain only a theoretical exercise. These calculations can be made only on certain assumptions that:
- There is a sequential, orderly pathway functioning, with one substrate forming the next and with glycolysis, TCA cycle and ETS pathway following one after another.
transferred into the mitochondria and undergoes oxidative phosphorylation. - None of the intermediates in the pathway are utilised to synthesise any other compound.
- Only glucose is being respired – no other alternative substrates are entering in the pathway at any of the intermediary stages.
But these kind of assumptions are not really valid in a living system; all pathway work simultaneously and do not take place one after another; substrates enter the pathways and are withdrawn from it as and when necessary; ATP is utilised as and when needed; enzymatic rates are controlled by multiple means. Hence, there can be a net gain of 36 ATP molecules during aerobic respiration of one molecule of glucose.
8. Distinguish between the following:
(a) Aerobic respiration and Anaerobic respira¬tion.
(b) Glycolysis and Fermentation.
(c) Glycolysis and Citric acid cycle.
Solution: (a) Differences between aerobic and anaerobic respiration are as follows:
(b) Differences between glycolysis and fermentation are as follows: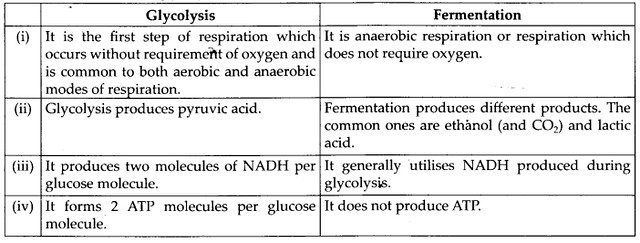
9. Discuss “The respiratory pathway is an amphibolic pathway”.
Solution: Amphibolic pathway is the one which is used for both breakdown (catabolism) and build-up (anabolism) reactions. Respiratory pathway is mainly a catabolic process which serves to run the living system by providing energy. The pathway produces a number of intermediates. Many of them are raw materials for building up both primary and secondary metabolites. Acetyl CoA is helpful not only in Krebs’ cycle but is also raw material for synthesis of fatty acids, steroids, terpenes, aromatic compounds and carotenoids, a-ketoglutarate is organic acid which forms glutamate (an important amino acid) on amination. OAA (Oxaloacetic acid) on amination produces asparate. Both aspartate and glutamate are components of proteins. Pyrimidines and alkaloids are other products. Succinyl CoA forms cytochromes and chlorophyll.
Hence, fatty acids would be broken down to acetyl CoA before entering the respiratory pathway when it is used as a substrate. But when the organism needs to synthesise fatty acids, acetyl CoA would be withdrawn from the respiratory pathway for it. Hence, the respiratory pathway comes into the picture both during breakdown and synthesis of fatty acids. Similarly, during breakdown and synthesis of proteins too, respiratory intermediates form the link. Breaking down processes within the living organism is catabolism, and synthesis is anabolism. Because the respiratory pathway is involved in both anabolism and catabolism, it would hence be better to consider the respiratory pathway as an amphibolic pathway rather than as a catabolic one.
10. Define RQ. What is its value for fats?
Solution: Respiratory quotient (RQ) is the ratio of the volume of carbon dioxide produced to the volume of oxygen consumed in respiration over a period of time. Its value can be one, zero, more than 1 or less than one.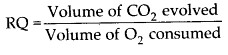
Volume of C02 evolved Volume of 02 consumed
RQ is less than one when the respiratory substrate is either fat or protein.
C57 H104O6 + 80 O2-» 57 CO2+ 52H2O
RQ = 57CO2/80O2 = 0.71
RQ is about 0.7 for most of the common fats.
11. What is oxidative phosphorylation?
Solution: Oxidative phosphorylation is the synthesis of energy rich ATP molecules with the help of energy liberated during oxidation of reduced co-enzymes (NADH, FADH2) produced in respiration. The enzyme required for this synthesis is called ATP synthase. It is considered to be the fifth complex of electron transport chain. ATP synthase is located in FT or head piece of F0 -F1 or elementary particles. The particles are present in the inner mitochondrial membrane. ATP synthase becomes active in ATP formation only where there is a proton gradient having higher concentration of H+ or protons on the F0 side as compared to F x side (chemiosmotic hypothesis of Peter Mitchell).
Increased proton concentration is produced in the outer chamber or outer surface of inner mitochondrial membrane by the pushing of proton with the help of energy liberated by passage of electrons from one carrier to another. Transport of the electrons from NADH over ETC helps in pushing three pairs of protons to the outer chamber while two pairs of protons are sent outwardly during electron flow from FADH2. The flow of protons through the F0 channel induces F1 particle to function as ATP-synthase. The energy of the proton gradient is used in attaching a phosphate radical to ADP by high energy bond. This produces ATP. Oxidation of one molecule of NADH2 produces 3 ATP molecules while a similar oxidation of FADH2 forms 2 ATP molecules.
12. What is the significance of step-wise release of energy in respiration?
Solution: The utility of step-wise release of energy in respiration are given as follows :
(i) There is a step-wise release of chemical bond energy which is very easily trapped in forming ATP molecules.
(ii) Cellular temperature is not allowed to rise.
(iii) Wastage of energy is reduced.
(iv) There are several intermediates which can be used in production of a number of biochemicals.
(v) Through their metabolic intermediates different substances can undergo respiratory catabolism.
(vi) Each step of respiration is controlled by its own enzyme. The activity of different enzymes can be enhanced or inhibited by specific compounds.
This helps in controlling the rate of respiration and the amount of energy liberated by it.