Solved CBSE Sample Papers for Class 10 Science Set 1
GENERAL INSTRUCTIONS
I. The Question Paper comprises of two sections, A and B. You are to attempt both the sections.
II. All questions are compulsory.
III. All questions of Section A and all questions of Section B are to be attempted separately.
IV. Question numbers I to 2 in Section A are one mark questions. These are to be answered in one word or in one sentence.
V. Question numbers 3 to 5 in Section A are two marks questions. These are to be answered in about 30 words each.
VI. Question numbers 6 to 15 in Section A are three marks questions. These are to be answered in about 50 words each.
VII. Question numbers 16 to 21 in Section A are five marks questions. These are to be answered in about 70 words each.
VIII. Question numbers 22 to 27 in Section B are questions based on practical skills and are five marks questions.
SECTION A
Question 1:
Write the balanced chemical equation with the state symbols of the following reaction: Solutions of Barium chloride and Sodium sulphate in water react to give insoluble Barium sulphate and the solution of Sodium chloride. 1
Answer:
BaCl2(aq) + Na2SO4(aq) → BaSO4(s) + 2 NaCl (aq)
Question 2:
What is an ecosystem ?
Answer:
Ecosystem is a unit of biosphere where interaction between biotic community and abiotic factors of an area or place takes place with each other.
Question 3:
An object is placed at a distance of 30 cm in front of a convex mirror of focal length 15 cm. Write four characteristics of the image formed by the mirror.
Answer:
Four characteristics of the image formed are:
(i) Image is erect.
(ii) Image is virtual.
(iii) Image is diminished in size.
(iv) The image is formed behind the mirror between P & F points of the mirror.
(v) The image is laterally inverted. (any four)
Question 4:
What is sustainable management? Why is reuse considered better in comparisonto recycle?
Answer:
Sustainable management means the judicious use of the natural resources by the present generations so that these resources are also available for the future generations.
In the recycling of materials some energy is used and harmful gases are produced which pollute the environment, while reuse of materials does not consume energy.
Question 5:
Write down important properties of ionic compounds.
Answer:
(i) Ionic compounds are generally hard and solids.
(ii) Ionic compounds have high melting and boiling points.
(iii) Ionic compounds are mostly soluble in water and insoluble in solvents like kerosene oil, alcohols, etc.
(iv) In their molten state or aqueous solution these are good conductors of electric current.
Question 6:
Write the chemical name of Plaster of Paris. Write the chemical equation of its preparation. Why should Plaster of Paris be stored in a dry place?
Answer:
Chemical name of Plaster of Paris is calcium sulphate hemihydrate and its formula is
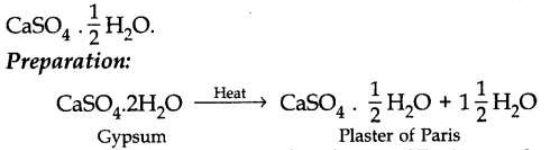
In the presence of moisture the Plaster of Paris powder changes to a hard solid mass called gypsum, so it has to be stored in a dry place.
Question 7:
Describe the structure and functioning of nephron.
Answer:
Structure of nephron. Nephron is the basic filtration unit in the kidney. It consists of a tubule which is connected with a collecting duct at one end and a cup shaped structure at the other end, called Bowman’s capsule. Every Bowman’s capsule contains a cluster of capillaries called glomerulus within the cup-shaped structure. The blood enters into glomerulus through afferent arteriole of renal artery and leaves it through efferent arteriole.
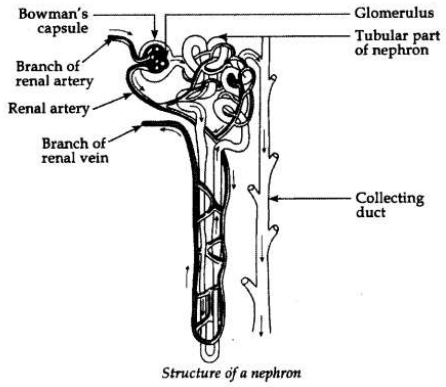
Basic functions of Nephrons:
(i) Filteration. Filteration of the blood takes place in Bowman’s capsule in the capillaries of the glomerulus. Then this filterate passes into the tubular part of the nephron. The filterate contains glucose, amino acids, urea and uric acid and a large amount of water.
(ii) Reabsorption. The filterate flows along the tubule and useful substances such as glucose, amino acids, salts and some water are re-absorbed into the blood by the capillaries surrounding the nephron tubule.
(iii) Urine. The filterate which remains after the re-absorption is called the urine, which is collected from nephron by the collecting duct to carry it to the urinary bladder and then to the urethra.
Question 8:
Compare and contrast nervous and hormonal mechanisms for control and
coordination in the animals.
Answer:
| Nervous mechanism | Hormonal mechanism |
| Nervous mechanism involves the movement of stimulus and response information in the form of chemical and electrical impulses through specialised neuron cells. | Hormones are special chemicals produced by specialised organs. Information reaches the target organs through the blood stream. |
| Nerve impulses are fast in their actions. | Hormones are slow in their actions. |
| Response is immediate in nervous control. | Response is usually slow. |
Question 9:
Two lamps, one rated 40W at 220V and the other 100 W at 220V, are connected in parallel to the electric supply at 220V.
(a) Draw a circuit diagram to show the connections.
(b) Calculate the current drawn from the electric supply.
(c) Calculate the total energy consumed by the two lamps together when they operate for one hour.
Answer: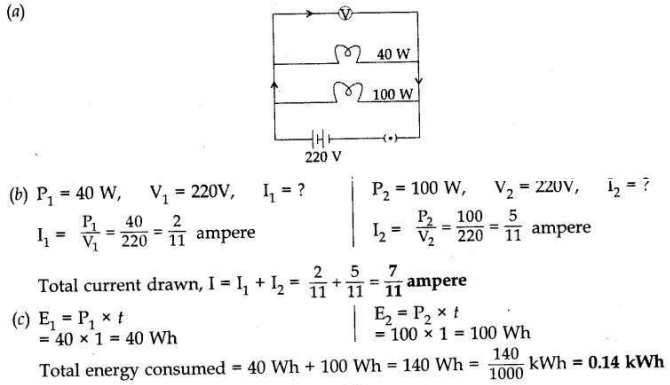
OR
Two resistors, with resistance 10 Ω and 15 Ω, are to be connected to a battery of e.m.f. 12V so as to obtain:
(i) minimum current
(ii) maximum current
(a) Describe the mode of connecting the resistances in each case.
(b) Calculate the strength of the total current in the circuit in each case.
Answer: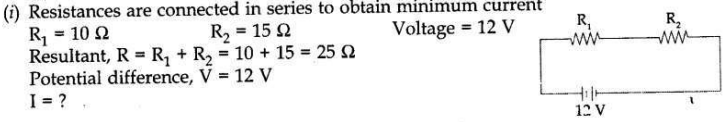

Question 10:
A coil of insulated copper wire is connected to a galvanometer. What will happen bar magnet is
(i) pushed into the coil,
(ii) withdrawn from inside the coil,
(iii) held stationary inside the coil?
Answer:
(i) When the north pole (N) of the magnet is pushed into the coil, the galvanometer is deflected towards the right.
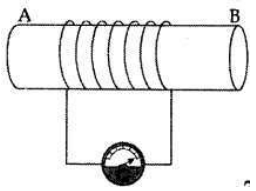
(ii) When the north pole (N) of the magnet is withdrawn from the coil, the galvanometer is deflected towards the left.
(iii) When the magnet is held stationary inside the coil, the deflection of the galvanometer is zero.
Question 11:
Complete the following chemical equations:
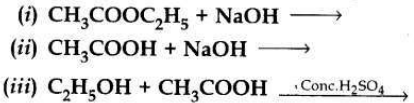
Answer:
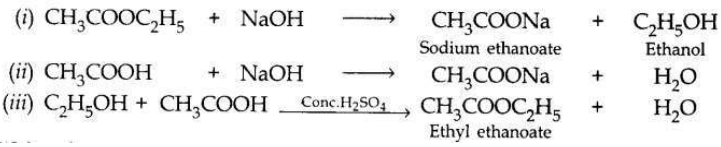
Question 12:
Write the names given to the vertical columns and horizontal rows in the Modern Periodic Table. How does the metallic character of elements vary on moving down a vertical column? How does the size of atomic radius vary on moving left to right in a horizontal row? Give reason in support of your answer in the above two cases.
Answer:
Vertical coloumns are called Groups.
The horizontal rows are called Periods.
• Metallic character increases on going down in a group.
Reason. Ability to lose electrons increases on moving down the group due to increase in distance between the nucleus and the valence electrons. There is decrease in the attraction between the nucleus and the valence electrons.
• Atomic radius decreases as we move from left to right in a period.
Reason. The nuclear charge increases on moving from left to right across a period resulting in increase in the attraction between the nucleus and the valence electrons.
OR
An element P (atomic number 20) reacts with an element Q (atomic number 17) to form a compound. Answer the following questions giving the reason. Write the position of P and Q in the Modern Periodic Table and the molecular formula of the compound formed when P reacts with Q.
Answer:
Element P: Electronic configuration: 2, 8, 8, 2
So, element P belongs to 4th period and group 2, as it has four shells and 2 valence electrons.
Element Q: Electronic configuration: 2, 8, 7
So element Q belongs to 3rd period and group 17, as it has three shells and seven valence electrons.
Molecular formula of compound will be PQ2. As valency of P is 2 and valency of Q is one.
Question 13:
State the basic requirement for sexual reproduction? Write the importance of such reproduction in nature. 3
Answer:
The basic requirement for sexual reproduction is sexual interaction of male and female parents to cause fusion of male gamete with female gamete to form zygote. Then from the zygote develops the new child.
Importance of such reproduction. Such sexual reproduction leads to more variation in the offsprings for their successful living in the changing environmental conditions. More variations also lead to evolution of new species after some generations. It leads to diversity in the population which helps in natural selection.
Question 14:
Explain with the help of an example each how the following provide evidences in favour of evolution:
(a) Homologous organs
(b) Analogous organs
(c) Fossils
Answer:
(a) Homologous orgAnswer: Those organs which have same basic structure among different types of living beings but perform different functions are called Homologous orgAnswer: For example, arms of human beings and forelegs of horse, lion, etc. are Homologous orgAnswer: Their same basic structure shows that these have been evolved from some common ancestors.
(b) Analogous orgAnswer: Those organs which have different basic structure but perform same functions in different types of living beings are known as Analogous orgAnswer: For example, wings of birds and wings of bat are analogous orgAnswer: Similarity in these organs is superficial and the design and structure of these organs is very different.
(c) Fossils. Fossils are the dead remain parts of the extinct form of living beings. They provide the missing link between the species, e.g., Fossils of dinosours with feathers/ fossils of prehistoric horse, etc.
Question 15:
Due to gradual weakening of ciliary muscles and diminishing flexibility of the eye lens a certain defect of vision arises. Write the name of this defect. Name the type of lens required by such persons to improve the vision. Explain the structure and function of such a lens.
Answer:
The defect of this vision is called Presbyopia.
Bifocal lens is used for the correction of this defect. The Upper part of this lens is concave and the lower part of the lens is Convex lens. The upper part of the lens is used to see the distant objects clearly and lower part of this lens is used to see the nearby objects clearly.
Question 16:
Write an activity to show the reaction of acids with metal carbonates and metal hydrogen carbonate salts.
Answer:
Take two test tubes. Mark them as ’A’ and ’B’. Take about 0.5 gm of sodium carbonate in the test tube ’A’ and 0.5 gm of sodium hydrogen carbonate in test tube ’B’. Add about 2 ml of dilute HCI acid to both of these test tubes. Pass the gas produced in each case through lime water as shown in the figure. Lime water in each case will turn milky due to the production of CO2 gas in each of the test tubes.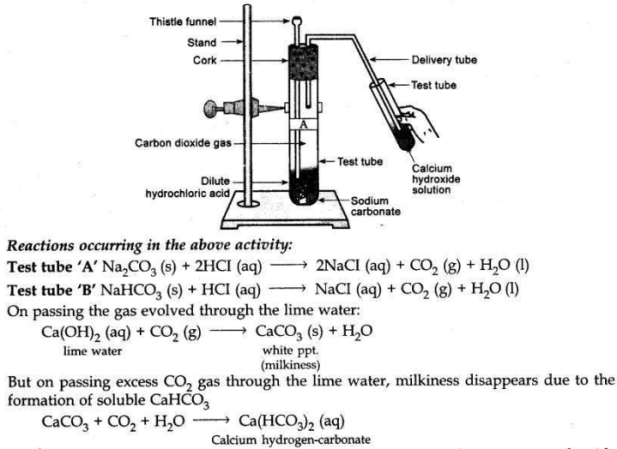
This activity shows that metal carbonates and metal hydrogen carbonate react with acids to form salt, CO2 and water.
Question 17:
Describe double circulation in human beings.
(a) Why is it necessary?
(b) How are arteries different from veins?
Answer:
(a) Double circulation of blood means, that the blood enters twice in the heart during its once circulation in other parts of the body. First time oxygenated blood from lungs enters the left atrium and second time de-oxygenated blood enters the right atrium through two main vena cava. Double circulation of blood is necessary to separate the oxygenated blood from the de-oxygenated blood to provide more energy for efficient working of body org
(b)
| Arteries | Veins |
| 1. Arteries are thick walled. | 1. Veins are thin walled. |
| 2. Arteries carry blood from the heart to different organs of the body. | 2. Veins carry the blood from body organs to the heart. |
| 3. Arteries do not have valves. | 3, Veins have valves in them. |
| 4. Arteries are deeply placed. | 4. Veins are superficially placed. |
Question 18:
Give the construction, working, advantages and disadvantages of a solar cooker.
Answer:
Solar Cooker is a device used to trap solar energy and utilize it to cook food. It consists of a box painted black from inside to absorb heat of the Sun (black colour is the best absorber of heat). A thick glass lid is placed over the box to trap heat energy of the Sun. A plane mirror reflector is also attached to the box so that a strong beam of sunlight falls over the cooker’s top.
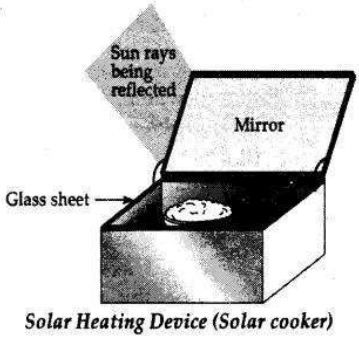
The reflector of the solar cooker sends strong beams of sunlight over the top of the cooker. Sunlight consists of about l/3rd of infra-red rays which have a heating effect. These infra-red rays are of shorter wavelength as these are produced by a very hot source of heat. The glass lid over the cooker allows these infra-red rays of short wavelength into the cooker but does not allow the infra-red rays which are emitted by the black surface of the cooker to escape as these are of longer wavelength. Thus, heat energy of the Sun gets trapped in the black box of the solar cooker. This heat cooks the food material kept in the black box.
Advantages of solar cooker:
(i) It is used to cook food and saves precious fossil fuel.
(ii) It does not cause any pollution.
(iii) No smoke is produced during the working of a solar cooker.
(iv) Nutrients of food material, which is to be cooked in the solar cooker, do not get destroyed.
(v) Four food items can be cooked at the same time.
Disadvantages of solar cooker.
(i) It cannot be used to cook food during the night.
(ii) It cannot be used to cook food on a cloudy day.
(iii) The direction of the reflector has to be changed after a small interval of time with the change of position of the Sun.
Question 19:
Soaps and detergents are both types of salts. State the difference between the two. Write the mechanism of cleansing action of soaps. Why do soaps not form lather (foam) with hard water? Mention any two problems that arise due to the use of detergents instead of soaps.
Answer:
• Soaps are the sodium salts or potassium salts of long chain carboxylic acids, while the detergents are the ammonium or sulphonate salts of long chain carboxylic acids.
• Mechanism of cleansing action of soaps. Soaps are molecules in which the two ends have differing properties, one is hydrophilic, that is, it dissolves in water, while the other end is hydrophobic, that is, it dissolves in hydrocarbons. When soap is at the surface of water, the hydrophobic ’tail’ of soap will not be soluble in water and the soap will align along the surface of water with the ionic end in water and the hydrocarbon ’tail’ protruding out of water.
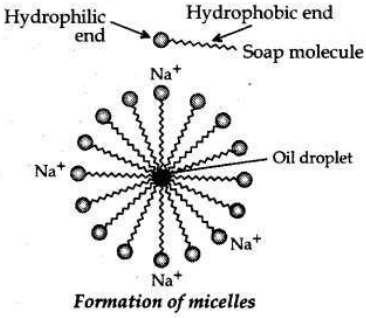
Inside water, these molecules have a unique orientation that keeps the hydrocarbon portion out of the water. This is achieved by forming clusters of molecules in which the hydrophobic tails are in the interior of the cluster and the ionic ends are on the surface of the cluster. This formation is called a micelle. Soap in the form of a micelle is able to clean, since the oily dirt will be collected in the centre of the micelle.
The micelles stay in solution as a colloid and will not come together to precipitate because of ion-ion repulsion. Thus, the dirt suspended in the micelles is also easily rinsed away.
• With hard water soap molecules form insoluble salts with Ca2+ or Mg2+ ions present in the hard water. So soaps do not form lather with hard water.
• Two problems of using detergents:
Detergents are non-biodegradable and thus these cause pollution of water bodies. Detergents are strong cleansing agents but these cause damage to the skins of the person using them.
Question 20:
(a) Name the organ that produces sperms as well as secretes a hormone in human males. Name the hormone it secretes and write its functions.
(b) Name the parts of the human female reproductive system where fertilization occurs.
(c) Explain how the developing embryo gets nourishment inside the mother’s body.
Answer:
(a) This organ is testes. The hormone secreted by testis is Testosterone. This hormone controls the changes which occur in human males during puberty. It leads to formation of sperms and development of secondary sexual characters.
(b) This part is Fallopian tube.
(c) Developing embryo gets the nourishment inside the mother’s body from Placenta which is developed on the wall of the uterus of the mother. It provides a large surface area for glucose and oxygen to pass from the mother’s blood to the developing embryo.
Question 21:
(a) To construct a ray diagram we use two rays which are so chosen that it is easy to
know their directions after reflection from the mirror. List two such rays and state the path of these rays after reflection in case of concave mirrors. Use these two rays and draw ray diagram to locate the image of an object placed between pole and focus of a concave mirror.
(b) A concave mirror produces three times magnified image on a screen. If the object is placed 20 cm in front of the mirror, how far is the screen from the object?
Answer:
(a) These two rays are:
(i) Ray of light parallel to principal axis, passes through point F of concave mirror after its reflection.
(ii) Ray of light passing through point C of the mirror, is reflected back on the same path after reflection from the mirror. Image is erect, virtual and magnified.
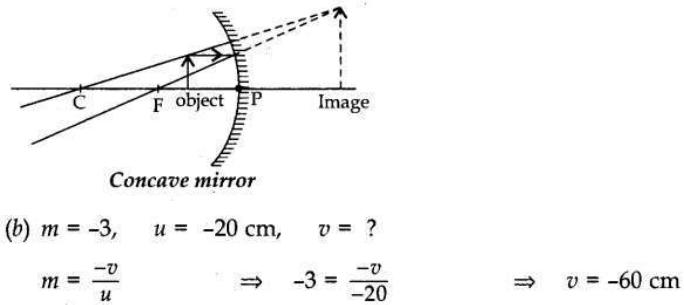
Distance between the screen and the object distance = [-60 – (-20)] = -40 cm So, the screen is at a distance of 40 cm from the object.
Minus sign is only as per sign conventions.
OR
(a) Draw a ray diagram to explain the term angle of deviation.
(b) Why do the component colours of incident white light split into a spectrum while passing through a glass prism, explain.
(c) Draw a labelled ray diagram to show the formation of a rainbow.
Answer:
(a) Angle of deviation is the angle between extended incident ray and extendent emergent ray through a prism.
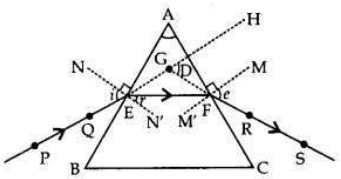
(b) PQ = Incident Ray, RS = Emergent Ray zD = Angle of Deviation The incident white light splits into a seven coloured spectrum as speed of different coloured light waves is different when these pass through the prism.
(C)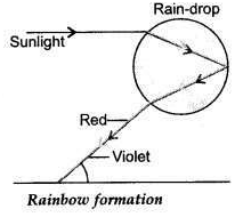
SECTION B
Question 22:
What type of material is formed when aqueous solutions of sodium sulphate and barium chloride are mixed. Give the balanced chemical equation involved. Name the type of reaction it is?
Answer:
• A white precipitate of BaSO4 will be formed.
• Na2SO4 (aq) + BaCl2 (aq) → BaSO4 (s) + 2NaCI (aq)
• It is a double displacement reaction.
Question 23:
Give any four precautions taken by a student to perform an experiment to determine the resultant resistance of two resistors when connected in series.
Answer:
(i) Clean the ends of the connecting wires by sand paper.
(ii) The connections should be tight.
(iii) Close the key (K) only when readings are to be taken.
(iv) Zero errors and the ranges of the ammeter and voltmeter should be noted.
Question 24:
Why is epidermal peel generally taken from lower surface of the leaf ?
Answer:
In dicot plants stomata are found in lower surface only, hence peel is taken from the lower surface. As stomata are present on both the surfaces of leaf in monocot plants so the peel can be taken from either surface.
Question 25:
A gas is liberated immediately with a brisk effervescence, when you add acetic acid to sodium hydrogen carbonate powder in a test tube. Name the gas and describe the test that confirms the identify of the gas. Answer:
The liberated gas is CO2 gas. The test that confirms the identity of the gas is its test with lime water. CO2 gas turns the lime water milky.
Ca(OH)2(aq) + Co2 → CaCo3(s) + H2o
Lime water White ppt
Question 26:
Name the type of asexual reproduction in which two individuals are formed from a single parent and the parental identity is lost. Write the first step from where such a type of reproduction begins. Draw first two stages of this reproduction.
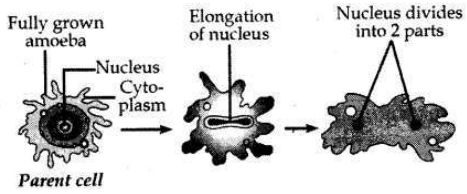
Answer:
The type of asexual reproduction in which two individuals are formed from a single parent and parental identity is lost is called Binary fission.
The first step of this reproduction is elongation of cells and its nucleus.
Question 27:
A student places a candle flame at a distance of about 60 cm from a convex lens of focal length 10 cm and focuses the image of the flame on a screen. After that he gradually moves the flame towards the lens and each time focuses the image on the screen.
(a) In which direction-toward or away from the lens, does he move the screen to focus the image?
(b) How does the size of the image change?
(c) How does the intensity of the image change as the flame moves towards the lens?
(d) Approximately for what distance between the flame and the lens, the image formed on the screen is inverted and of the same size?
Answer:
(a) He will move the screen away from the lens to focus the image.
(b) Size of the image goes on increasing.
(c) Intensity of image goes on decreasing.
(d) About 20 cms.