AP Chemistry 2.7 VSEPR and Bond Hybridization- MCQs - Exam Style Questions
Question
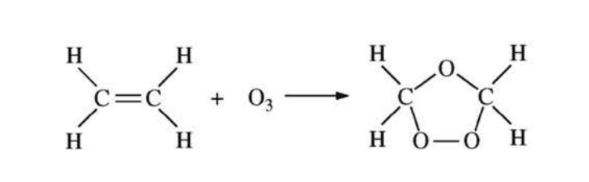
| Before | After | |
|---|---|---|
| (A) | \(sp\) | \(sp^2\) |
| (B) | \(sp\) | \(sp^3\) |
| (C) | \(sp^2\) | \(sp\) |
| (D) | \(sp^2\) | \(sp^3\) |
▶️ Answer/Explanation
1. Analyze the Reactant (Before):
The reactant molecule is ethene (\(C_2H_4\)). Each carbon atom has one double bond (to the other C) and two single bonds (to H atoms). The number of electron domains is \(3\) (one C=C bond counts as one domain, plus two C-H bonds). Three electron domains correspond to \(sp^2\) hybridization (trigonal planar geometry).
2. Analyze the Product (After):
In the product molecule (ethane-1,2-diol), the C=C double bond has been broken. Each carbon atom now has four single bonds (one to C, two to H, and one to O). The number of electron domains is \(4\). Four electron domains correspond to \(sp^3\) hybridization (tetrahedral geometry).
3. Conclusion:
The hybridization of the carbon atoms changes from \(sp^2\) before the reaction to \(sp^3\) after the reaction.
✅ Answer: (D)
Questions
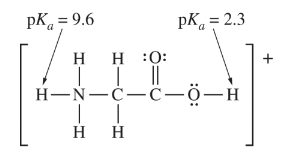
The structural formula of the glycinium cation is shown above. Arrows indicate the\( pK_{ a}\) values for the labile protons in the molecule.
Which of the following is true about the geometry of the glycinium cation?
(A) The leftmost C atom and all the atoms directly bonded to it lie in the same plane.
(B) Both C atoms and both O atoms lie in the same plane.
(C) The N-C-C bond angle is 180°.
(D) The geometry around the N atom is planar.
▶️Answer/Explanation
Ans: A
The correct answer is (A) The leftmost C atom and all the atoms directly bonded to it lie in the same plane.
Glycinium cation, represented as \( \text{NH}_3^+ \cdot \text{CH}_2 \cdot \text{COOH}^-\), has a planar geometry where the leftmost carbon atom (C) and all the atoms bonded directly to it are in the same plane. This is due to the sp² hybridization of the carbon atom, which forms three sigma bonds with the adjacent atoms, resulting in a trigonal planar geometry.
Question
Which of the following Lewis diagrams represents a molecule that is polar?
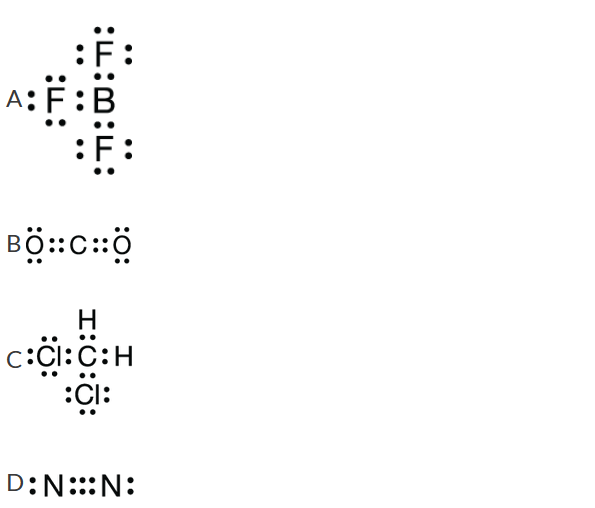
▶️Answer/Explanation
Ans:C
In the \(CH_2Cl_2\) molecule, the H and Cl atoms are in a tetrahedral arrangement around the central C atom. The two electronegative chlorine atoms create a partial negative charge on their side of the carbon atom, leaving a partial positive charge on the opposite (hydrogen) side of the carbon atom. This separation of charge constitutes a dipole, and the molecule is polar.
