AP Chemistry 3.10 Solubility- MCQs - Exam Style Questions
Question
(B) \(CCl_{4}\)
(C) \(NH_{3}\)
(D) \(PH_{3}\)
▶️ Answer/Explanation
1. Use the Principle of Solubility:
The principle of “like dissolves like” states that polar solvents, like water ($H_2O$), will dissolve polar solutes. Water is highly polar and forms extensive hydrogen bonds.
2. Analyze the Options:
- (A) \(CH_{4}\) (Methane): This molecule is tetrahedral and the C-H bonds are nonpolar. It is a nonpolar molecule and will not dissolve in water.
- (B) \(CCl_{4}\) (Carbon tetrachloride): Although C-Cl bonds are polar, the molecule has a symmetrical tetrahedral shape, causing the dipoles to cancel out. It is a nonpolar molecule and will not dissolve in water.
- (C) \(NH_{3}\) (Ammonia): This molecule is trigonal pyramidal and is highly polar. Furthermore, it contains H-N bonds, allowing it to form strong hydrogen bonds with water molecules. It is very soluble in water.
- (D) \(PH_{3}\) (Phosphine): This molecule is trigonal pyramidal, but the electronegativity difference between P and H is very small, making the P-H bonds nearly nonpolar. It does not form hydrogen bonds and is only slightly soluble in water.
3. Conclude:
\(NH_3\) is the only molecule that is both highly polar and capable of hydrogen bonding with water, making it the most soluble.
✅ Answer: (C)
Questions
\(CaF_{2}(s)\rightleftharpoons Ca^{2+}(aq)+2F^{-}(aq) K_{sp}\)=\(4.0\times 10^{-11}\)
The concentration of \(F^{ − }\)(aq) in drinking water that is considered to be ideal for promoting dental health is \(4.0 × 10^{−5}\) M. Based on the information above, the maximum concentration of Ca2+(aq) that can be present in drinking water without lowering the concentration of \(F^{−}\)(aq) below the ideal level is closest to
(A) 0.25 M
(B) \(0.025 \)M
(C) \(1.6 × 10^{ −6}\) M
(D)\(1.6 × 10 ^{−15}\) M
▶️Answer/Explanation
Ans: B
To find the maximum concentration of \(Ca^{2+}(aq)\) that can be present in drinking water without lowering the concentration of \(F^{-}(aq)\) below the ideal level, we need to use the solubility product constant (\(K_{sp}\)) and the given concentration of \(F^{-}(aq)\).
The solubility product constant (\(K_{sp}\)) expression for the dissolution of \(CaF_{2}(s)\) is:
\[ K_{sp} = [Ca^{2+}][F^{-}]^2 \]
Given:
\(K_{sp} = 4.0 \times 10^{-11}\)
\([F^{-}] = 4.0 \times 10^{-5}\) M (ideal concentration)
We can rearrange the expression to solve for \([Ca^{2+}]\):
\[ [Ca^{2+}] = \frac{K_{sp}}{[F^{-}]^2} \]
\[ [Ca^{2+}] = \frac{4.0 \times 10^{-11}}{(4.0 \times 10^{-5})^2} \]
\[ [Ca^{2+}] = \frac{4.0 \times 10^{-11}}{1.6 \times 10^{-9}} \]
\[ [Ca^{2+}] \approx 2.5 \times 10^{-2} \, \text{M} \]
So, the maximum concentration of \(Ca^{2+}(aq)\) that can be present in drinking water without lowering the concentration of \(F^{-}(aq)\) below the ideal level is closest to \(0.025 \, \text{M}\).
Questions
Based on the structural formulas, which of the following identifies the compound that is more soluble in water and best helps to explain why?
(A) Ethane, because the electron clouds of its molecules are more polarizable than those of propanol.
(B) Ethane, because its molecules can fit into the spaces between water molecules more easily than those of propanol can.
(C) Propanol, because its molecules have a greater mass than the molecules of ethane have.
(D) Propanol, because its molecules can form hydrogen bonds with water molecules but those of ethane cannot
▶️Answer/Explanation
Ans: D
Based on the structural formulas provided, the correct answer is (D) Propanol, because its molecules can form hydrogen bonds with water molecules but those of ethane cannot.
Here’s the reasoning:
Ethane is a non-polar hydrocarbon molecule with no polar bonds or lone pairs of electrons. It cannot form hydrogen bonds with water molecules, which are polar and can participate in hydrogen bonding.
On the other hand, propanol is an alcohol with a polar O-H bond. The oxygen atom in propanol has lone pairs of electrons, and the hydrogen atom attached to the oxygen can participate in hydrogen bonding
with water molecules.
The ability to form hydrogen bonds with water is a key factor that increases the solubility of a compound in water. Propanol molecules can form favorable intermolecular interactions with water molecules
through hydrogen bonding, leading to increased solubility compared to ethane, which cannot form such interactions.
Therefore, propanol is expected to be more soluble in water than ethane due to its ability to form hydrogen bonds with water molecules.
The other options are incorrect because:
(A) The polarizability of electron clouds does not significantly affect the solubility in water for these compounds.
(B) The molecular size or ability to fit into water spaces is not the primary factor determining solubility in this case.
(C) The molecular mass alone does not directly relate to water solubility for these compounds.
Questions
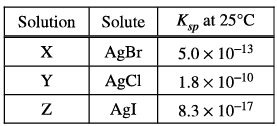
Three saturated solutions (X, Y, and Z) are prepared at \(25^{\circ }C\). Based on the information in the table above, which of the following lists the solutions in order of increasing [\(Ag^{+}\)] ?
(A) X < Z < Y
(B) Y < X < Z
(C) Z < Y < X
(D) Z < X < Y
▶️Answer/Explanation
Ans: D
Given information about the solubility products (\( K_{\text{sp}} \)) for the three different silver halide salts:
Solution X contains \( \text{AgBr} \) with a solubility product of \( 5.0 \times 10^{-13} \).
Solution Y contains \( \text{AgCl} \) with a solubility product of \( 1.8 \times 10^{-10} \).
Solution Z contains \( \text{AgI} \) with a solubility product of \( 8.3 \times 10^{-17} \).
The solubility product expression for each salt is as follows:
\( \text{AgBr} \rightleftharpoons \text{Ag}^+ + \text{Br}^-: K_{\text{sp}} = [\text{Ag}^+][\text{Br}^-] = 5.0 \times 10^{-13} \)
\( \text{AgCl} \rightleftharpoons \text{Ag}^+ + \text{Cl}^-: K_{\text{sp}} = [\text{Ag}^+][\text{Cl}^-] = 1.8 \times 10^{-10} \)
\( \text{AgI} \rightleftharpoons \text{Ag}^+ + \text{I}^-: K_{\text{sp}} = [\text{Ag}^+][\text{I}^-] = 8.3 \times 10^{-17} \)
We want to compare the concentrations of \( \text{Ag}^+ \) ions in each solution. Since the solubility product expression involves the product of \( \text{Ag}^+ \) concentration with the anion concentration, we can infer the following:
Solution Z (\( \text{AgI} \)) has the lowest solubility product, which means it has the lowest concentration of \( \text{Ag}^+ \) ions.
Solution X (\( \text{AgBr} \)) has an intermediate solubility product.
Solution Y (\( \text{AgCl} \)) has the highest solubility product, indicating the highest concentration of \( \text{Ag}^+ \) ions.
Therefore, the correct order of increasing \( \text{Ag}^+ \) concentration is:
\[ \text{Z} < \text{X} < \text{Y} \]
Hence, the answer is option (D): Z < X < Y.
