AP Chemistry 3.4 Ideal Gas Law- MCQs - Exam Style Questions
Question
(B) \(HBr\)
(C) \(SO_2\)
(D) \(N_2\)
▶️ Answer/Explanation
1. Understand Ideal Gas Assumptions:
The ideal gas law assumes that gas particles have negligible volume and no intermolecular forces (IMFs).
2. Identify the Most Ideal Gas:
A real gas behaves most ideally when its IMFs are weakest. At the same temperature and pressure, we must find the molecule with the weakest IMFs.
3. Analyze IMFs of Options:
- (A) \(PH_3\): Polar (trigonal pyramidal shape), has dipole-dipole forces.
- (B) \(HBr\): Polar, has dipole-dipole forces.
- (C) \(SO_2\): Polar (bent shape), has dipole-dipole forces.
- (D) \(N_2\): Nonpolar, only has weak London dispersion forces (LDFs).
4. Conclusion:
Because \(N_2\) is the only nonpolar molecule in the list, it has the weakest intermolecular forces. Therefore, it will behave most like an ideal gas under the given conditions.
✅ Answer: (D)
Questions
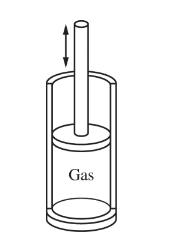
A mixture of two gases, 0.01 mol of\( C_{4}H_{10}\)(g) and 0.065 mol of O2(g), is pumped into a cylinder with a movable piston, as shown above. The mixture, originally at \(200^{\circ}\) C and 1.0 atm, is sparked and the reaction represented below occurs.
2 \(C_{ 4}H_{10}(g) + 13 O_{2}\)(g) \rightarrow 8\( CO_{2}(g) + 10 H_{2}\)O(g)
Which of the following is true after the product gases return to the original temperature and pressure, and why will the change occur? (Assume all gases behave ideally.)
(A) The piston will be higher than its original position, because the cylinder will contain a greater number of gas molecules.
(B) The position of the piston will be unchanged, because the total mass of the gases in the cylinder does not change.
(C) The position of the piston will be unchanged, because the temperature and pressure of the contents of the cylinder remain the same.
(D) The piston will be lower than its original position, because the product molecules are smaller than the reactant molecules.
▶️Answer/Explanation
Ans: A
Initial mixture: \(0.01 \, \text{mol} \, \text{C}_4\text{H}_{10}(g)\) and \(0.065 \, \text{mol} \, \text{O}_2(g)\)
Initial temperature: \(200^\circ \text{C} = 473 \, \text{K}\)
Initial pressure: \(1.0 \, \text{atm}\)
Balanced chemical reaction: \(2\text{C}_4\text{H}_{10}(g) + 13\text{O}_2(g) \rightarrow 8\text{CO}_2(g) + 10\text{H}_2\text{O}(g)\)
After the reaction, the product gases (\(8\text{CO}_2(g) + 10\text{H}_2\text{O}(g)\)) return to the original temperature (\(473 \, \text{K}\)) and pressure (\(1.0 \, \text{atm}\)).
According to the ideal gas law: \(PV = nRT\)
Since the temperature and pressure remain constant, the volume occupied by the gases (\(V\)) will be directly proportional to the total number of moles of gas (\(n\)).
Let’s calculate the total number of moles before and after the reaction:
Before the reaction:
Total moles \(=0.01 |, \text{mol} \, \text{C}_4\text{H}_{10} +0.065|, \text{mol} \, \text{O}_2=0.075 |, \text{mol}\)
After the reaction:
Total moles \(=8\,\text{mol} \,\text{CO}_2+10 \,\text{mol} \, \text{H}_2\text{O} =18 \, \text{mol}\)
Since the total number of moles increases after the reaction (\(18 \,\text{mol} >0.075 \,\text{mol}\)), the volume occupied by the gases will also increase at constant temperature and pressure.
Therefore, the correct option is (A) The piston will be higher than its original position because the cylinder will contain a greater number of gas molecules.
Question
A 1.0 L sample of a pure gas is found to have a lower pressure than that predicted by the ideal gas law. The best explanation for the observation is that the molecules of the gas
(A) have a combined volume that is too large to be considered negligible when compared to the volume of the container
(B) have a low molecular mass and therefore do not strike the container walls with as much force as expected
(C) are attracted to each other and do not exert as much force on the container walls as they would if they had no mutual attractions
(D) are attracted to the sides of the container and strike the container walls with more force than expected
▶️Answer/Explanation
Ans:C
The deviation from the ideal gas law can occur due to various factors, including the size of gas molecules, intermolecular forces, and the conditions under which the gas is being measured.
Option A suggests that the molecules have a combined volume that is too large to be negligible. In reality, gases are assumed to consist of point-like particles with negligible volume, according to the ideal gas law. When the size of gas molecules becomes significant compared to the volume of the container, the assumption of negligible volume breaks down, and the behavior of the gas deviates from ideal. Therefore, option A could be a valid explanation for the observed deviation from the ideal gas law.
Option B suggests that the low molecular mass of the gas results in fewer collisions with the container walls. However, the ideal gas law doesn’t depend on the molecular mass of the gas; it assumes that gas molecules have mass and move randomly, regardless of their mass.
Option C suggests that the gas molecules are attracted to each other, resulting in fewer collisions with the container walls. This phenomenon is characteristic of real gases, where intermolecular forces affect the behavior of the gas. When gas molecules experience attractive forces, they are less likely to collide with the container walls with the same force as in an ideal gas.
Option D suggests that the gas molecules are attracted to the sides of the container and strike the container walls with more force than expected. This scenario is less likely because attractive forces typically result in fewer collisions with the container walls, leading to lower pressure than expected by the ideal gas law.
Considering the options, option A (the combined volume of gas molecules is not negligible) or option C (the molecules are attracted to each other and do not exert as much force on the container walls) seem more plausible explanations for the observed deviation from the ideal gas law. However, given that real gases often experience both volume effects and intermolecular forces, option C is a more direct explanation for the lower pressure observed. So, the best explanation for the observed deviation is:
(C) The molecules are attracted to each other and do not exert as much force on the container walls as they would if they had no mutual attractions.
Question
A balloon filled with 0.25 mol of He(g) at 273 K and 1 atm is allowed to rise through the atmosphere. Which of the following explains what happens to the volume of the balloon as it rises from ground level to an altitude where the air temperature is 220 K and the air pressure is 0.1 atm?
(A) The volume will increase because the decrease in air pressure will have a greater effect than the decrease in temperature.
(B) The volume will remain unchanged because of the counteracting effects of the decrease in temperature and the decrease in air pressure.
(C) The volume will decrease because the decrease in temperature will have a greater effect than the decrease in air pressure.
(D) It cannot be determined whether the volume of the balloon will increase, decrease, or remain the same without knowing the initial volume of the balloon.
▶️Answer/Explanation
Ans:A
To determine what happens to the volume of the balloon as it rises from ground level to an altitude where the air temperature is 220 K and the air pressure is 0.1 atm, we can apply the ideal gas law equation:
\[ PV = nRT \]
Since the amount of helium gas (\( n \)) and the gas constant (\( R \)) remain constant, we can rewrite the ideal gas law as:
\[ \frac{P_1}{T_1} = \frac{P_2}{T_2} \]
Where:
\( P_1 \) and \( T_1 \) are the initial pressure and temperature, respectively,
\( P_2 \) and \( T_2 \) are the final pressure and temperature, respectively.
Substituting the given values, we get:
\[ \frac{1 \, \text{atm}}{273 \, \text{K}} = \frac{0.1 \, \text{atm}}{220 \, \text{K}} \]
Solving for the final volume (\( V_2 \)), we get:
\[ V_2 = \frac{P_1 \times V_1 \times T_2}{P_2 \times T_1} \]
Given that \( P_1 = 1 \, \text{atm} \), \( T_1 = 273 \, \text{K} \), \( P_2 = 0.1 \, \text{atm} \), and \( T_2 = 220 \, \text{K} \), we can calculate the final volume (\( V_2 \)).
When we plug in the values, we get:
\[ V_2 = \frac{1 \times V_1 \times 220}{0.1 \times 273} \]
\[ V_2 = \frac{2200}{27.3} \times V_1 \]
\[ V_2 \approx 80.52 \times V_1 \]
This means the final volume (\( V_2 \)) will be approximately \(80.52\) times the initial volume (\( V_1 \)). Since \(80.52\) is much greater than \(1\), it indicates that the volume of the balloon will increase significantly as it rises to the altitude with lower temperature and pressure.
Therefore, the correct answer is:
(A) The volume will increase because the decrease in air pressure will have a greater effect than the decrease in temperature.
