AP Chemistry 4.6 Introduction to Titration - MCQs - Exam Style Questions
Question
A 20. mL sample of 0.50 M \(HC_{2}H_{3}O_{2}\)(aq) is titrated with 0.50 M NaOH(aq). Which of the following best represents the species that react and the species produced in the reaction?
(A) \(H^{+}(aq) + OH^{−}(aq) → H_{2}O(l)\)
(B) \(H^{+}(aq) + C_{2}H_{3}O_{2}^{−}(aq) + Na^{+}(aq) + OH^{−}(aq)\) →\( H_{2}O(l) + NaC_{2}H_{3}O_{2}\)(aq)
(C) \(HC_{2}H_{3}O_{2}(aq)\) + OH^{−}(aq) → \(C_{2}H_{3}O_{2}^{−}(aq) + H_{2}O(l)\)
(D) \(HC_{2}H_{3}O_{2}(aq) + NaOH(aq) \)→ \(H_{2}O(l) + Na+(aq) + C_{2}H_{3}O_{2}^{−}\)(aq)
▶️Answer/Explanation
Ans:D
Question
A 25.5 mL aliquot of HCl (aq) of unknown concentration was titrated with 0.113 M NaOH (aq). It took 51.2 mL
of the base to reach the endpoint of the titration. What was the concentration (M) of the acid?
A) 0.454 B) 0.113 C) 1.02 D) 0.114 E) 0.227
▶️Answer/Explanation
Ans: E
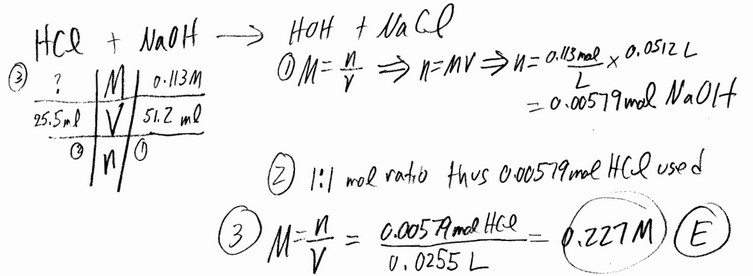
Questions (a)-(b) refer to the figures below. The figures show portions of a buret used in a titration of an acid solution of known concentration with a saturated solution of Ba(OH)2. Figures 1 and 2 show the level of the Ba(OH)2 solution at the start and at the endpoint of the titration, respectively. Phenolphthalein was used as the indicator for the titration.
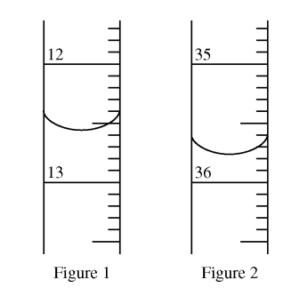
Question(a)
What is the evidence that the endpoint of the titration has been reached?
(A) The color of the solution in the buret changes from pink to colorless.
(B) The color of the solution in the buret changes from blue to red.
(C) The color of the contents of the flask below the buret changes from colorless to pink.
(D) The color of the contents of the flask below the buret changes from blue to red.
(E) The contents of the flask below the buret change from clear to cloudy.
▶️Answer/Explanation
Ans:C
Question(b)
The volume of saturated Ba(OH)2 used to neutralize the acid was closest to
(A) 6.60 mL
(B) 22.80 mL
(C) 23.02 mL
(D) 23.20 mL
(E) 29.80 mL
▶️Answer/Explanation
Ans:D
