AP Chemistry 8.7 pH and pKa - MCQs - Exam Style Questions
Question
\(HCHO_{2}(aq)+H_{2}O(l)\rightleftharpoons H_{3}O^{+}(aq)+CHO_{2}^{-}(aq)\)
\(HCHO_2\)(aq), a weak acid (K = \(2 × 10^{−4})\) a , dissociates in water according to the equation above. Which of the following provides the best estimate of the pH of 0.5 M \(HCHO_2\)(aq) and identifies the species at the highest concentration (excluding\( H_2O)\) in the solution?

▶️Answer/Explanation
Ans:D
The dissociation of a weak acid in water can be represented as follows:
\[ HA(aq) \rightleftharpoons H^{+}(aq) + A^{-}(aq) \]
The equilibrium constant expression for this reaction is:
\[ K_a = \frac{[H^{+}][A^{-}]}{[HA]} \]
Given that \( K_a = 2 \times 10^{-4} \) and the initial concentration of \( HCHO_2 \) is 0.5 M, we can set up the following table to represent the concentrations of the species at equilibrium:
\[
\begin{array}{lccc}
& \text{Initial Concentration} & \text{Change in Concentration} & \text{Equilibrium Concentration} \\
HCHO_2 & 0.5 & -x & 0.5 – x \\
H^{+} & 0 & +x & x \\
CHO_2^{-} & 0 & +x & x \\
\end{array}
\]
Substituting these into the \( K_a \) expression gives:
\[ 2 \times 10^{-4} = \frac{x^2}{0.5 – x} \]
Assuming that \( x << 0.5 \), we can approximate \( 0.5 – x \approx 0.5 \), which simplifies the equation to:
\[ x = \sqrt{2 \times 10^{-4} \times 0.5} \approx 0.01 \]
The \( H^{+} \) ion concentration is equal to \( x \), so the pH of the solution is \( -\log[H^{+}] = -\log(0.01) = 2 \).
Since \( HCHO_2 \) is a weak acid, it does not fully dissociate in water. Therefore, at equilibrium, the concentration of \( HCHO_2 \) will be higher than the concentrations of \( H^{+} \) and \( CHO_2^{-} \). Thus, the species at the highest concentration (excluding \( H_2O \)) in the solution is \( HCHO_2 \).
Question
The pH of a solution made by combining 150.0 mL of 0.10 M KOH(aq) with 50.0 mL of 0.20 M\( HBr(aq)\) is closest to which of the following?
(A) 2
(B) 4
(C) 7
(D) 12
▶️Answer/Explanation
Ans:D
To find the pH of the resulting solution, we need to first determine the moles of \( OH^{-} \) and \( H^{+} \) ions produced by the reaction between \( KOH \) and \( HBr \).
From the given concentrations and volumes, we can calculate the moles of each species:
For \( KOH \):
\[
\text{Moles of } OH^{-} = \text{Molarity} \times \text{Volume} = 0.10 \, \text{M} \times 0.150 \, \text{L} = 0.015 \, \text{mol}
\]
For \( HBr \):
\[
\text{Moles of } H^{+} = \text{Molarity} \times \text{Volume} = 0.20 \, \text{M} \times 0.050 \, \text{L} = 0.010 \, \text{mol}
\]
Since \( KOH \) and \( HBr \) react in a 1:1 ratio according to the balanced equation:
\[ KOH(aq) + HBr(aq) \rightarrow KBr(aq) + H_2O(l) \]
Therefore, the moles of \( OH^{-} \) in excess are \( 0.015 \, \text{mol} – 0.010 \, \text{mol} = 0.005 \, \text{mol} \).
We then convert the excess moles of \( OH^{-} \) to \( \text{pOH} \):
\[ \text{pOH} = -\log(\text{OH}^-) = -\log(0.005 \, \text{M}) \]
\[ \text{pOH} \approx 2.3 \]
Finally, we can find the pH using the relation:
\[ \text{pH} + \text{pOH} = 14 \]
\[ \text{pH} = 14 – \text{pOH} = 14 – 2.3 \]
\[ \text{pH} \approx 11.7 \]
Since the closest option is 12, the correct answer is: (D) 12
Question
\(HN0_{2}(aq)\rightleftharpoons H^{+}N0_{2}^{-}(aq)\) \(K_{a}=4.0\times 10^{-4}\)
On the basis of the information above, what is the approximate percent ionization of \(HNO_{2}\) in a 1.0 M\( HNO_{2}\)(aq) solution?
(A) 0.00040%
(B) 0.020%
(C) 0.040%
(D) 2.0%
▶️Answer/Explanation
Ans:D
To find the percent ionization of \(HNO_2\) in a 1.0 M \(HNO_2(aq)\) solution, we need to use the given equilibrium constant (\(K_a\)) and the initial concentration of \(HNO_2\).
The equilibrium constant \(K_a\) is given as \(4.0 \times 10^{-4}\).
For a weak acid like \(HNO_2\), the percent ionization is low, so we can assume that the concentration of the ionized species (\(H^+\) and \(NO_2^-\)) is much smaller than the initial concentration of \(HNO_2\). This assumption allows us to use the approximation:
\([H^+] = [NO_2^-] = x\) (let’s call this the ionization concentration)
\([HNO_2] \approx 1.0\) M (the initial concentration)
Substituting these values into the equilibrium constant expression:
\[ K_a = \frac{[H^+][NO_2^-]}{[HNO_2]} \]
\[ 4.0 \times 10^{-4} = \frac{x^2}{1.0} \]
Solving for \(x\):
\[ x^2 = 4.0 \times 10^{-4} \]
\[ x = \sqrt{4.0 \times 10^{-4}} = 0.02 \text{ M} \]
This means that the ionization concentration \([H^+] = [NO_2^-] = 0.02\) M.
To find the percent ionization, we divide the ionization concentration by the initial concentration and multiply by 100:
\[ \text{Percent ionization} = \left( \frac{0.02 \text{ M}}{1.0 \text{ M}} \right) \times 100\% = 2\% \]
Therefore, the correct answer is (D) 2.0%.
Question
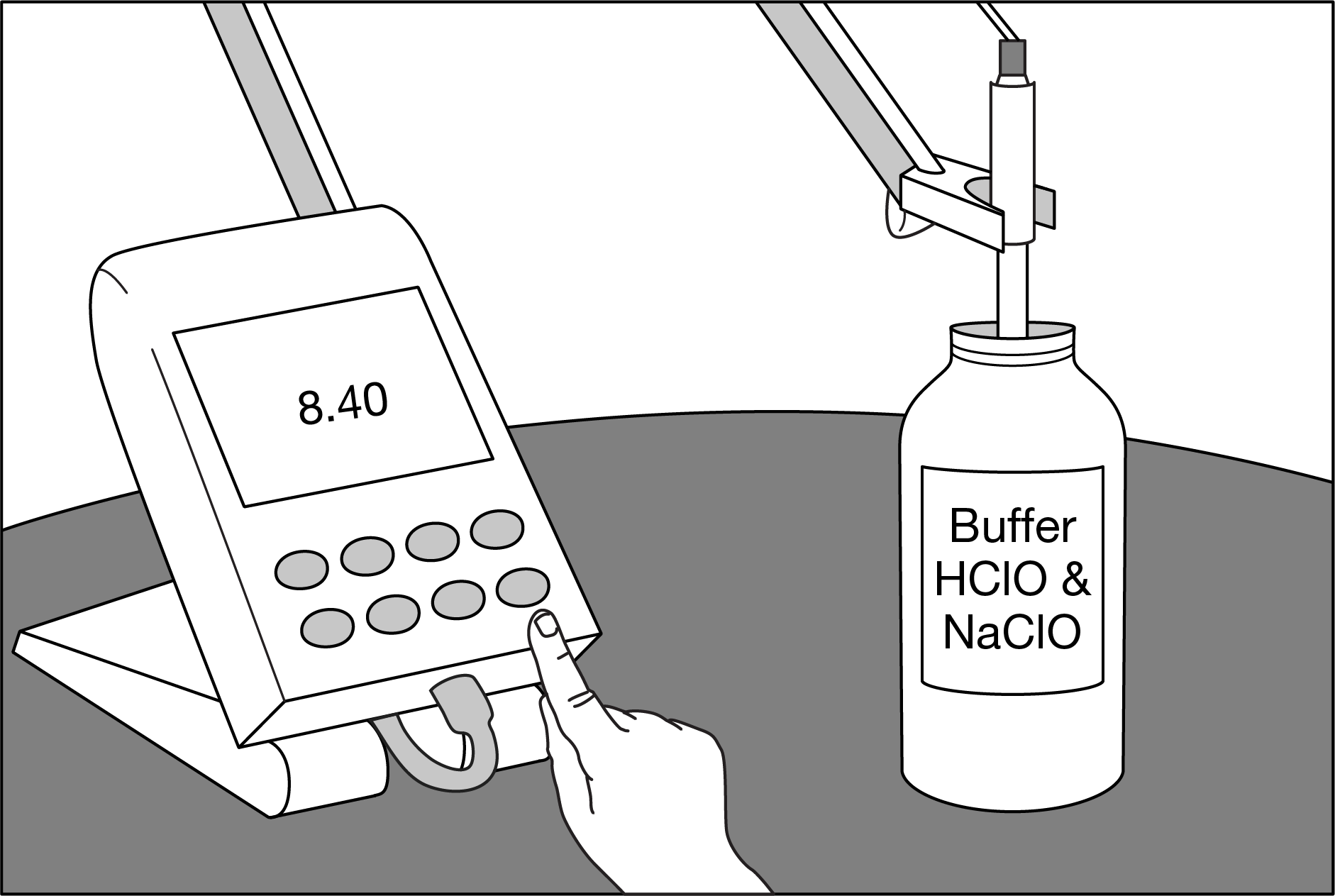
A student measures the pH of a 0.0100M buffer solution made with \(HClO\) and \(NaClO\), as shown above. The \(pK_a\) of \(HClO\) is 7.40 at 25°C. Based on this information, which of the following best compares the relative concentrations of \(ClO^−\) and \(HClO\) in the buffer solution?
A \([ClO^−]=[HClO]\)
B \( [ClO^−]>[HClO]\)
C \([ClO^−]<[HClO]\)
D It is not possible to compare the concentrations without knowing the \(pK_b\) of \(ClO^−\).
▶️Answer/Explanation
Ans:B
\( [ClO^−]>[HClO]\). When the \(pH>pK_a\), the base form has a higher concentration than the acid form.
