IB Biology HL (HIGHER level)- 2024 – Practice Questions- All Topics
Topic 2.1 Molecules to metabolism
Topic 2 Weightage : 10%
All Questions for Topic 2.1 –Molecular Biology, Organic Compounds, Organic Subunits -glucose, ribose and fatty and amino acids, Organic Polymers -sugars and lipids, Falsifying Vitalism, Metabolism, Anabolism and Catabolism, Periodic Table, Types of Bonding, Trace Elements
Question
What characteristic shows that this steroid molecule is a lipid?
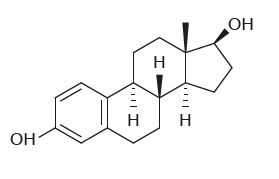
A. It is made of carbon rings.
B. It has a very low proportion of oxygen to carbon.
C. It contains OH groups as do fatty acids.
D. It is made only of nitrogen, oxygen and hydrogen.
▶️Answer/Explanation
B
Given figure is of “Methyl estradiol”, which is not a lipid but actually a estrogen medication which is used in the treatment of menopausal symptoms. Due to its oxygen carbon proportion it looks like lipids.
Question
Glucose is absorbed through protein channels in the plasma membrane of epithelium cells in the small intestine. Which characteristics of glucose prevent its diffusion through the phospholipid bilayer?
A. It is non-polar and therefore hydrophobic.
B. Its hydrogen bonds link with amino acids in the protein channel.
C. It is polar and therefore hydrophilic.
D. Its covalent bonds interact with the phospholipids.
▶️Answer/Explanation
C
The hydrophobic nature of the phospholipid bilayer in the small intestine prevents polar glucose from being absorbed into the bloodstream. The cell membrane is composed of a lipid bilayer which is hydrophobic, meaning it repels water. Glucose has a hydrophilic nature, meaning it is attracted to water. Therefore, cell membranes act as barriers to most molecules. For water molecules and a few other small molecules, such as oxygen and carbon dioxide, the lipid bilayer is permeable.
Question
What are the most frequently occurring elements in living organisms?
A. calcium, phosphorus, iron and sodium
B. calcium, sodium, nitrogen and phosphorus
C. carbon, phosphorus, oxygen and nitrogen
D. nitrogen, carbon, oxygen and hydrogen
▶️Answer/Explanation
D
The four most abundant elements in living matter are carbon, nitrogen, oxygen, and hydrogen. These elements have low atomic numbers and are thus light elements capable of forming strong bonds with other atoms to produce molecules. Unlike carbon, nitrogen forms up to three bonds, oxygen forms up to two, and hydrogen forms one. These elements are also found in organic molecules which take a variety of chemical forms.
Question
Which process is an example of catabolism?
Translation of mRNA
Replication of DNA
Hydrolysis of protein
Synthesis of a disaccharide
▶️Answer/Explanation
Ans: C
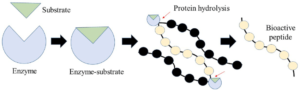
Hydrolysis is a catabolic reaction because it breaks down large molecules into smaller ones. In the case of proteins, hydrolysis breaks down the peptide bonds between amino acids, which are the building blocks of proteins. This process is catalyzed by enzymes called hydrolases.
Question
The diagram shows the product of a polymerization reaction.
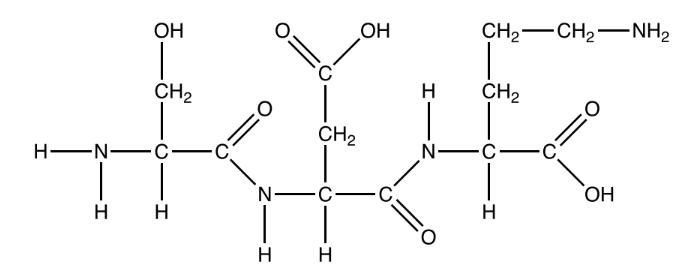
What is formed in this polymerization reaction?
A. A dipeptide formed by the hydrolysis of two nucleotides
B. A tripeptide formed by the hydrolysis of three amino acids
C. A dipeptide formed by the condensation of two amino acids
D. A tripeptide formed by the condensation of three amino acids
▶️Answer/Explanation
Ans:D
