AP Chemistry 1.7 Periodic Trends- Exam Style questions - FRQs- New Syllabus
Question
• Balance • \( 200.00\ \mathrm{mL} \) volumetric flask • Pipet
• \( 50.0\ \mathrm{mL} \) graduated cylinder
| Step | Step Description |
|---|---|
| 1. | Use weighing paper to measure the determined mass of solid \( \mathrm{AgNO_3} \) on a balance. |
| 2. | |
| 3. | |
| 4. | |
| 5. |
| Half-Reaction | \( E^\circ \) |
|---|---|
| \( \mathrm{Ag}^{+}(aq) + e^- \rightarrow \mathrm{Ag}(s) \) | \( 0.80\ \mathrm{V} \) |
| \( \mathrm{Al}^{3+}(aq) + 3e^- \rightarrow \mathrm{Al}(s) \) | \( -1.66\ \mathrm{V} \) |
Most-appropriate topic codes (AP Chemistry):
• Topic \( 1.7 \) — Periodic Trends (Part \( \mathrm{(b)} \))
• Topic \( 3.7 \) — Solutions and Mixtures (Part \( \mathrm{(c)} \))
• Topic \( 4.9 \) — Oxidation-Reduction (Redox) Reactions (Part \( \mathrm{(d)} \), Part \( \mathrm{(e)} \))
• Topic \( 9.9 \) — Cell Potential and Free Energy (Parts \( \mathrm{(f)} \), \( \mathrm{(g)} \))
▶️ Answer/Explanation
(a)
For a correct electron configuration:
Accept one of the following:
• \( 1s^2\,2s^2\,2p^6\,3s^2\,3p^1 \)
• \( [\mathrm{Ne}]\,3s^2\,3p^1 \)
Aluminum has atomic number \( 13 \), so its \( 13 \) electrons fill orbitals in order up to \( 3p^1 \).
(b)
For a correct explanation:
The highest occupied electron shell \( (n = 3) \) of \( \mathrm{Al} \) is at a greater average distance from the nucleus than the highest occupied electron shell \( (n = 2) \) of \( \mathrm{Al^{3+}} \).
When aluminum forms \( \mathrm{Al^{3+}} \), it loses its three valence electrons, so the entire \( n = 3 \) shell is removed. That makes the ion smaller.
(c)
For the correct steps to dissolve the solute in water \( (\text{steps may be consolidated}) \):
\( 2 \). Partially fill the volumetric flask with some distilled water.
\( 3 \). Add the weighed \( \mathrm{AgNO_3}(s) \) to the volumetric flask.
\( 4 \). Swirl to dissolve the solid.
For the correct step to ensure quantitative dilution:
\( 5 \). After the solid is dissolved, fill the flask to the calibration \( (200.0\ \mathrm{mL}) \) mark and mix.
The volumetric flask is used because it gives the most accurate final solution volume.
(d)
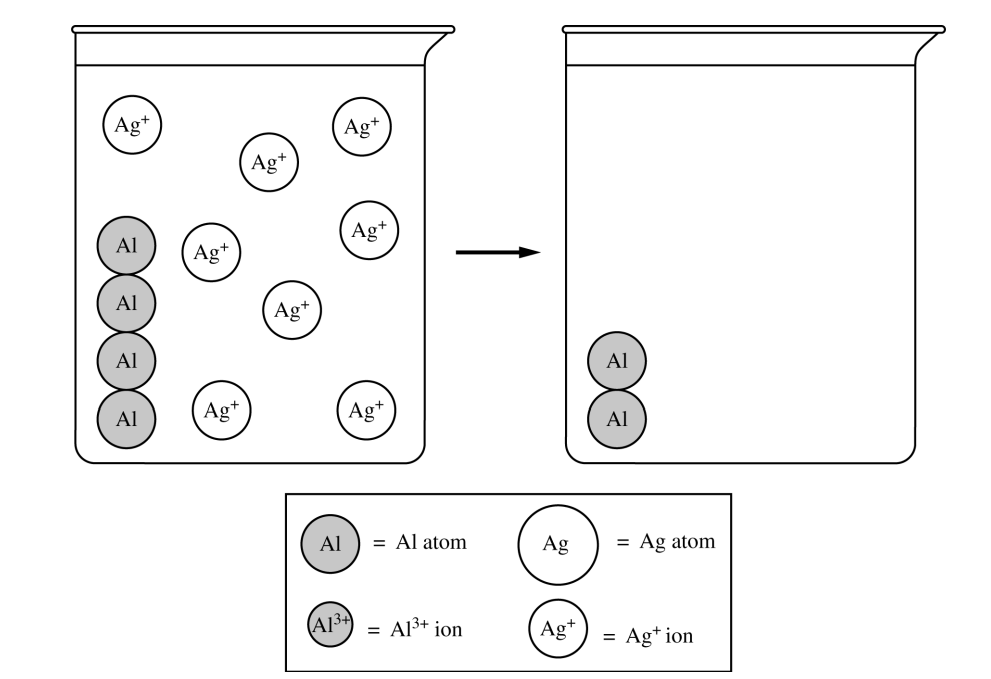
For a drawing that shows product formation and indicates the conservation of matter:
\( 4 \) \( \mathrm{Al} \) atoms and \( 8 \) \( \mathrm{Ag} \) particles in the beaker on right \( (\text{see sample drawing below}) \)
For a drawing that shows product formation and conservation of charge:
\( 2 \) \( \mathrm{Ag^{+}} \) ions and \( 2 \) \( \mathrm{Al^{3+}} \) ions in the beaker on the right \( (\text{see sample drawing below}) \)
For a drawing that shows product formation and correct phases of matter for all species:
\( 6 \) \( \mathrm{Ag} \) atoms that are solid and \( 2 \) \( \mathrm{Al^{3+}} \) ions that are aqueous in the beaker on the right
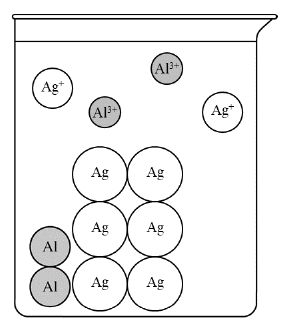
A quick count check: starting with \( 4 \) aluminum atoms and \( 8 \) silver ions, the reaction uses \( 2 \) aluminum atoms and \( 6 \) silver ions, leaving \( 2 \) aluminum atoms and \( 2 \) silver ions unreacted.
(e)
For the correct calculated value:
Accept one of the following:
• \( E^\circ = 0.80\ \mathrm{V} + 1.66\ \mathrm{V} = 2.46\ \mathrm{V} \)
• \( E^\circ_{\mathrm{cell}} = E^\circ_{\mathrm{red}} – E^\circ_{\mathrm{ox}} = 0.80\ \mathrm{V} – (-1.66\ \mathrm{V}) = 2.46\ \mathrm{V} \)
Silver ion is reduced, and aluminum is oxidized. The large positive cell potential shows the reaction is strongly favorable.
(f)
For the correct answer and a valid justification:
Negative. The reaction has a positive value of \( E^\circ \), indicating that it is thermodynamically favorable and would therefore have a negative value of \( \Delta G^\circ \). \( \left( \Delta G^\circ = -nFE^\circ \right) \)
Since \( E^\circ_{\mathrm{cell}} > 0 \), the sign of \( \Delta G^\circ \) must be negative.
(g)
For the correct answer and a valid justification:
Accept one of the following:
• Zero. The observation that the reaction stops progressing implies that \( E_{\mathrm{cell}} = 0 \), indicating that there is no longer a driving force for the reaction.
• Zero. The observation that reaction stops progressing implies that equilibrium is established, and \( \Delta G = 0 \) at equilibrium.
Once the system reaches equilibrium, forward and reverse processes balance, so there is no net free-energy change.
