AP Chemistry 3.10 Solubility - Exam Style questions - FRQs- New Syllabus
Question
| Properties of Salicylic Acid \( \left( \mathrm{HC}_{7}\mathrm{H}_{5}\mathrm{O}_{3} \right) \) | |
|---|---|
| Melting point | \( 159^\circ \mathrm{C} \) |
| Solubility in \( \mathrm{H}_{2}\mathrm{O} \) at \( 25^\circ \mathrm{C} \) | \( 2.2\ \mathrm{g/L} \) |
| Specific heat capacity | \( 1.17\ \mathrm{J/(g \cdot ^\circ C)} \) |
| Heat of fusion | \( 27.1\ \mathrm{kJ/mol} \) |
Most-appropriate topic codes (AP Chemistry):
• Topic \( 3.10 \) — Solubility (Part \( \mathrm{(b)} \))
• Topic \( 6.5 \) — Energy of Phase Changes (Part \( \mathrm{(c)} \))
• Topic \( 3.1 \) — Intermolecular and Interparticle Forces (Part \( \mathrm{(d)} \))
• Topic \( 8.5 \) — Acid-Base Titrations (Parts \( \mathrm{(e)} \), \( \mathrm{(f)} \), \( \mathrm{(h)} \))
• Topic \( 8.7 \) — pH and \( pK_{a} \) (Part \( \mathrm{(g)} \))
▶️ Answer/Explanation
(a)
For the correct calculated value:
\( 0.300\ \mathrm{g\ C}_{8}\mathrm{H}_{8}\mathrm{O}_{3} \times \dfrac{1\ \mathrm{mol\ C}_{8}\mathrm{H}_{8}\mathrm{O}_{3}}{152.15\ \mathrm{g}} \times \dfrac{1\ \mathrm{mol\ HC}_{7}\mathrm{H}_{5}\mathrm{O}_{3}}{1\ \mathrm{mol\ C}_{8}\mathrm{H}_{8}\mathrm{O}_{3}} \times \dfrac{138.12\ \mathrm{g}}{1\ \mathrm{mol\ HC}_{7}\mathrm{H}_{5}\mathrm{O}_{3}} = 0.272\ \mathrm{g\ HC}_{7}\mathrm{H}_{5}\mathrm{O}_{3} \)
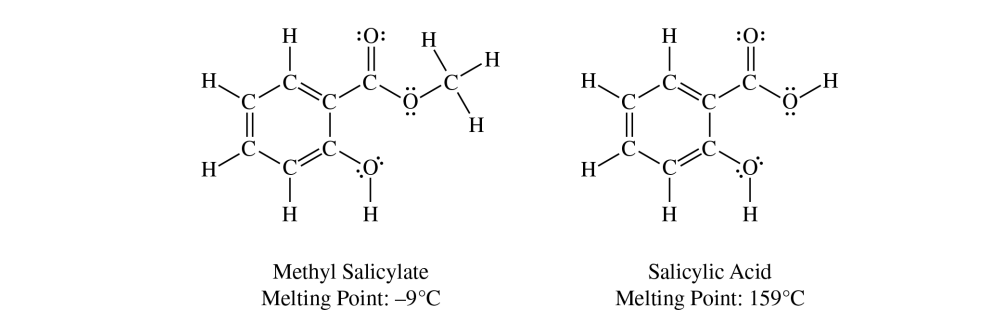
The mole ratio is \( 1:1 \), so after converting methyl salicylate to moles, the same number of moles of salicylic acid is produced.
Therefore, the maximum mass is \( \boxed{0.272\ \mathrm{g}} \).
(b)
For the correct answer and a valid justification:
Yes (consistent). Because the acid is soluble in water, some crystals may dissolve during rinsing, causing the mass of the collected precipitate to be lower than expected. This would lead to a percent yield less than \( 100\% \).
Since the reported yield is \( 87\% \), losing some product during the rinse step is a reasonable explanation for the lower recovered mass.
(c)
For the correct calculated value of either \( q \):
Accept one of the following:
• \( q_{\mathrm{heat}} = mc\Delta T = (0.105\ \mathrm{g})(1.17\ \mathrm{J/(g \cdot ^\circ C)})(159^\circ \mathrm{C} – 25^\circ \mathrm{C}) = 16.5\ \mathrm{J} \)
• \( q_{\mathrm{melt}} = 0.105\ \mathrm{g} \times \dfrac{1\ \mathrm{mol}}{138.12\ \mathrm{g}} \times \dfrac{27{,}100\ \mathrm{J}}{1\ \mathrm{mol}} = 20.6\ \mathrm{J} \)
For the correct calculated value of the other \( q \) and the total heat:
\( q_{\mathrm{total}} = q_{\mathrm{heat}} + q_{\mathrm{melt}} = 16.5\ \mathrm{J} + 20.6\ \mathrm{J} = 37.1\ \mathrm{J} \)
So the total heat absorbed is \( \boxed{37.1\ \mathrm{J}} \).
(d)
For a correct explanation:
Molecules of salicylic acid have more hydrogen-bonding sites than molecules of methyl salicylate have, which leads to stronger intermolecular forces and a higher melting point for salicylic acid.
Salicylic acid has both a carboxylic acid group and a phenolic \( \mathrm{O-H} \) group, so the crystal lattice can be held together more strongly by intermolecular attractions.
(e)
For the correct answer:
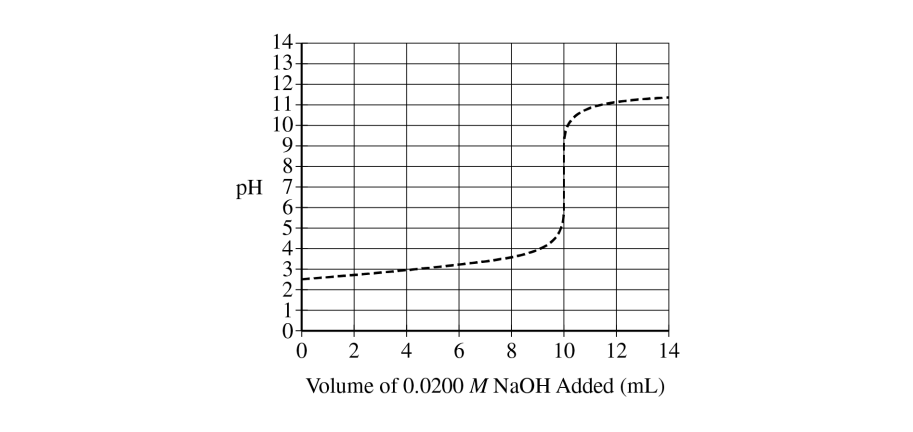
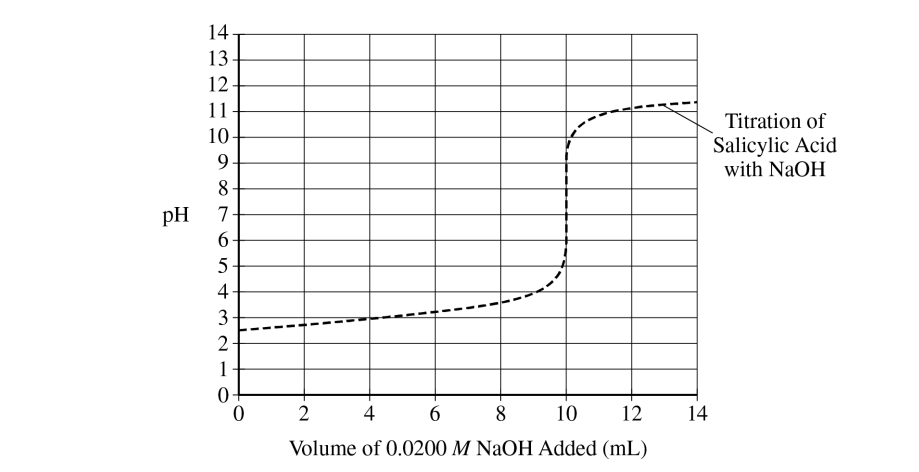
The \( pK_{a} \) is approximately \( 3 \).
The \( pK_{a} \) is read at the half-equivalence point. Since equivalence occurs near \( 10\ \mathrm{mL} \), the half-equivalence point is near \( 5\ \mathrm{mL} \), where the graph shows a pH of about \( 3 \).
(f)
For the correct answer and a valid justification, consistent with part \( \mathrm{(e)} \):
Accept one of the following:
• The conjugate base, \( \mathrm{C}_{7}\mathrm{H}_{5}\mathrm{O}_{3}^{-} \). When \( \mathrm{pH} = 4 \), the titration is beyond the half-equivalence point, where \( [\mathrm{HC}_{7}\mathrm{H}_{5}\mathrm{O}_{3}] = [\mathrm{C}_{7}\mathrm{H}_{5}\mathrm{O}_{3}^{-}] \). Thus, \( [\mathrm{C}_{7}\mathrm{H}_{5}\mathrm{O}_{3}^{-}] \) must be greater than \( [\mathrm{HC}_{7}\mathrm{H}_{5}\mathrm{O}_{3}] \).
• The conjugate base, \( \mathrm{C}_{7}\mathrm{H}_{5}\mathrm{O}_{3}^{-} \). Because the pH of the solution is greater than the \( pK_{a} \) of the acid, the majority of the molecules will be deprotonated.
In buffer logic, when \( \mathrm{pH} > pK_{a} \), the deprotonated form is present in higher concentration.
(g)
For the correct calculated value:
\( pK_{a} = -\log(6.3 \times 10^{-5}) = 4.20 \)
Therefore, the value is \( \boxed{4.20} \).
(h)
For a curve that shows a correct starting and half-equivalence point, consistent with part \( \mathrm{(g)} \):
The curve starts at \( \mathrm{pH} \approx 3.11 \) and passes through the \( pK_{a} \) calculated in part \( \mathrm{(g)} \) at \( 5\ \mathrm{mL} \).
See example response below.
For a curve that shows the correct equivalence point:
The curve inflects vertically at \( 10\ \mathrm{mL} \), showing the same volume of base needed to reach the equivalence point.
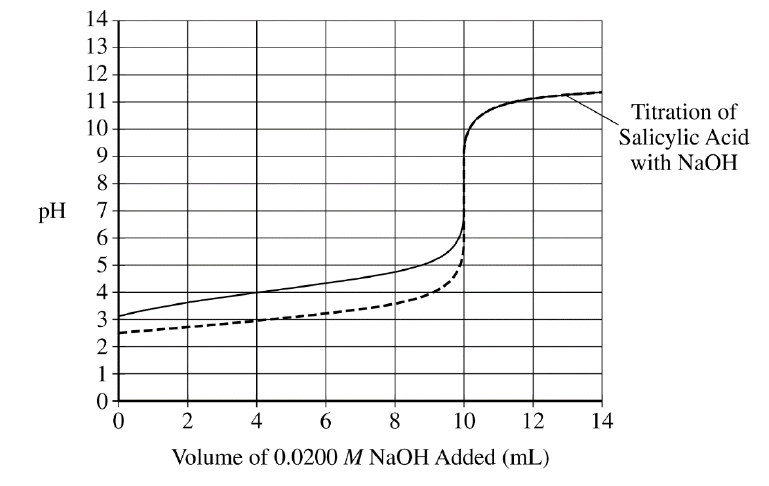
Because both acids have the same initial volume and concentration, they require the same volume of \( \mathrm{NaOH} \) to reach equivalence. The benzoic acid curve simply starts a bit higher and passes through \( \mathrm{pH} = 4.20 \) at the half-equivalence point.
