AP Chemistry 5.7 Introduction to Reaction Mechanisms - Exam Style questions - FRQs- New Syllabus
Question
Step \( 1 \): \( \mathrm{NO_2}(g) + \mathrm{NO_2}(g) \rightarrow \mathrm{NO}(g) + \mathrm{NO_3}(g) \) slow
Step \( 2 \): \( \mathrm{NO_3}(g) \rightarrow \mathrm{NO}(g) + \mathrm{O_2}(g) \) fast
Step \( 1 \): \( \mathrm{NO_2}(g) + \mathrm{NO_2}(g) \rightleftharpoons \mathrm{N_2O_4}(g) \) fast equilibrium
Step \( 2 \): \( \mathrm{N_2O_4}(g) \rightarrow 2\,\mathrm{NO}(g) + \mathrm{O_2}(g) \) slow
Most-appropriate topic codes (AP Chemistry):
• Topic \( 5.3 \) — Concentration Changes Over Time (Part \( \mathrm{(a)} \))
• Topic \( 5.7 \) — Introduction to Reaction Mechanisms (Part \( \mathrm{(c)} \))
• Topic \( 5.8 \) — Reaction Mechanism and Rate Law (Part \( \mathrm{(c)(i)} \), Part \( \mathrm{(c)(ii)} \))
▶️ Answer/Explanation
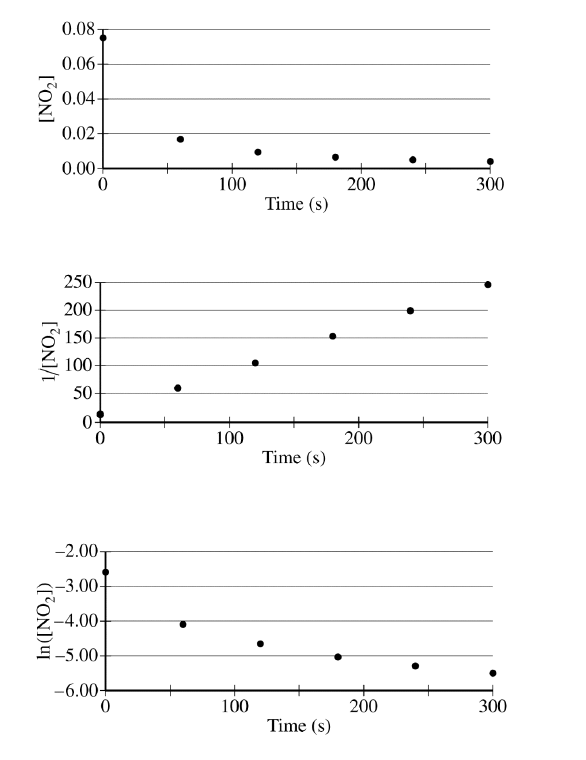
(a)
The graph of \( \dfrac{1}{[\mathrm{NO_2}]} \) versus time is linear, which indicates that the reaction is second order.
For comparison, a zero-order reaction would give a linear plot of \( [\mathrm{NO_2}] \) versus time, and a first-order reaction would give a linear plot of \( \ln[\mathrm{NO_2}] \) versus time. Since neither of those is linear here, the second-order model is the correct one.
(b)
\( \text{rate} = k[\mathrm{NO_2}]^2 \)
Because the reaction is second order overall and involves only \( \mathrm{NO_2} \) as the reactant in the experimentally determined rate law, the concentration of \( \mathrm{NO_2} \) must be squared.
(c)(i)
Yes, mechanism I is consistent with the rate law in part \( \mathrm{(b)} \).
Step \( 1 \) is slow, so it is the rate-determining step. Since step \( 1 \) is an elementary reaction,
\( \text{rate} = k[\mathrm{NO_2}][\mathrm{NO_2}] = k[\mathrm{NO_2}]^2 \)
This matches the experimentally determined rate law from part \( \mathrm{(b)} \).
(c)(ii)
Yes, mechanism II is also consistent with the rate law in part \( \mathrm{(b)} \).
Step \( 2 \) is slow, so it is the rate-determining step. Therefore, the rate law from mechanism II begins as
\( \text{rate} = k[\mathrm{N_2O_4}] \)
However, \( \mathrm{N_2O_4} \) is an intermediate, so it cannot appear in the final overall rate law. Use the fast equilibrium in step \( 1 \):
\( \mathrm{NO_2}(g) + \mathrm{NO_2}(g) \rightleftharpoons \mathrm{N_2O_4}(g) \)
\( K_{eq} = \dfrac{[\mathrm{N_2O_4}]}{[\mathrm{NO_2}]^2} \)
Rearranging gives
\( [\mathrm{N_2O_4}] = K_{eq}[\mathrm{NO_2}]^2 \)
Substitute into the rate law for the slow step:
\( \text{rate} = k[\mathrm{N_2O_4}] = k\!\left(K_{eq}[\mathrm{NO_2}]^2\right) \)
\( \text{rate} = (kK_{eq})[\mathrm{NO_2}]^2 \)
Since \( kK_{eq} \) is still just a constant, this simplifies to the same form:
\( \text{rate} = k'[\mathrm{NO_2}]^2 \)
So mechanism II is also consistent with the rate law in part \( \mathrm{(b)} \).
