AP Chemistry 8.1 Introduction to Acids and Bases - Exam Style questions - FRQs- New Syllabus
Question
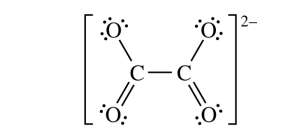
(ii) Calculate the molar solubility of \( \mathrm{Ag_2C_2O_4} \) in neutral distilled water.
(iii) The molar solubility of \( \mathrm{Ag_2C_2O_4} \) increases when it is dissolved in \( 0.5\ \mathrm{M\ HClO_4}(aq) \) instead of neutral distilled water. Write a balanced, net-ionic equation for the process between species in solution that contributes to the increased solubility of \( \mathrm{Ag_2C_2O_4}(aq) \) in \( \mathrm{HClO_4}(aq) \).
Most-appropriate topic codes (AP Chemistry):
• Topic \( 7.11 \) — Introduction to Solubility Equilibria (Parts \( \mathrm{(b)(i)} \), \( \mathrm{(b)(ii)} \))
• Topic \( 8.1 \) — Introduction to Acids and Bases (Part \( \mathrm{(b)(iii)} \))
▶️ Answer/Explanation
(a)
For the correct answer:
\( sp^2 \)
Each carbon is bonded in a trigonal planar arrangement with three electron domains around it, so \( sp^2 \) hybridization is expected.
(b)(i)
For the correct answer:
\( K_{sp} = [\mathrm{Ag^+}]^2[\mathrm{C_2O_4^{2-}}] \)
The exponent \( 2 \) on \( [\mathrm{Ag^+}] \) comes from the coefficient \( 2 \) in the dissolution equation \( \mathrm{Ag_2C_2O_4}(s) \rightleftharpoons 2\,\mathrm{Ag^+}(aq) + \mathrm{C_2O_4^{2-}}(aq) \).
(b)(ii)
For the correct calculated value:
Let the molar solubility be \( s \).
Then
\( [\mathrm{Ag^+}] = 2s \)
\( [\mathrm{C_2O_4^{2-}}] = s \)
So,
\( 5.40 \times 10^{-12} = (2s)^2(s) \)
\( 5.40 \times 10^{-12} = 4s^3 \)
\( s = 1.11 \times 10^{-4}\ \mathrm{M} \)
Therefore, the molar solubility is \( \boxed{1.11 \times 10^{-4}\ \mathrm{M}} \).
(b)(iii)
For a correct equation (state symbols not required):
Accept one of the following:
• \( \mathrm{C_2O_4^{2-}}(aq) + \mathrm{H_3O^+}(aq) \rightarrow \mathrm{HC_2O_4^-}(aq) + \mathrm{H_2O}(l) \)
• \( \mathrm{C_2O_4^{2-}}(aq) + \mathrm{H^+}(aq) \rightarrow \mathrm{HC_2O_4^-}(aq) \)
• \( \mathrm{C_2O_4^{2-}}(aq) + 2\,\mathrm{H_3O^+}(aq) \rightarrow \mathrm{H_2C_2O_4}(aq) + 2\,\mathrm{H_2O}(l) \)
• \( \mathrm{C_2O_4^{2-}}(aq) + 2\,\mathrm{H^+}(aq) \rightarrow \mathrm{H_2C_2O_4}(aq) \)
Acid removes \( \mathrm{C_2O_4^{2-}} \) from solution by protonating it. That lowers the oxalate ion concentration and shifts the dissolution equilibrium to the right, increasing the solubility of \( \mathrm{Ag_2C_2O_4} \).
