IB DP Chemistry Structure 2.3 The metallic model HL Paper 2- Exam Style Questions - New Syllabus
Question
Answer all questions. Answers must be written within the answer boxes provided.
Iron may be extracted from iron(II) sulfide, FeS.
(a) Outline why metals, like iron, can conduct electricity. [1]
(b) Justify why sulfur is classified as a non-metal by giving two of its chemical properties. [2]
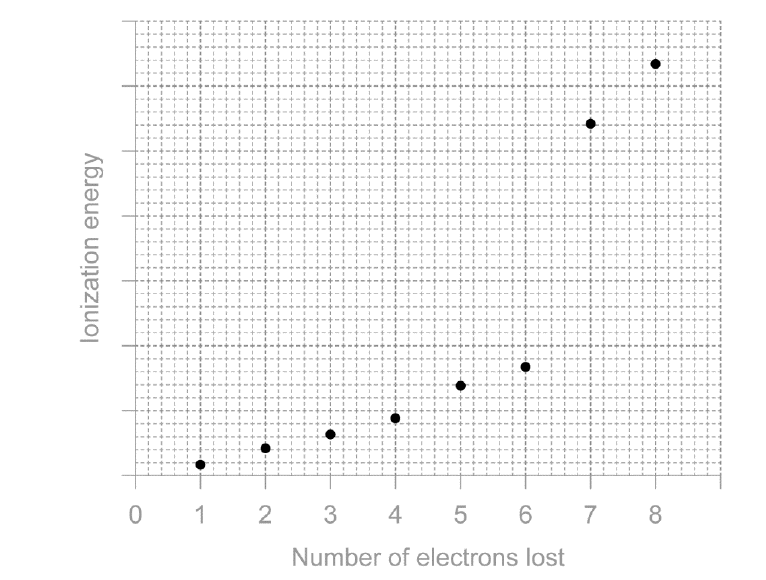
(c) Sketch the first eight successive ionisation energies of sulfur. [2]
(d) Iron (II) sulfide, FeS, is ionically bonded.
(i) Describe the bonding in this type of solid. [2]
(ii) State a technique that could be used to determine the crystal structure of the solid compound. [1]
(iii) State the full electron configuration of the sulfide ion. [1]
(iv) Outline, in terms of their electronic structures, why the ionic radius of the sulfide ion is greater than that of the oxide ion. [1]
(v) Suggest why chemists find it convenient to classify bonding into ionic, covalent and metallic. [1]
(e) The first step in the extraction of iron from iron (II) sulfide is to roast it in air to form iron (III) oxide and sulfur dioxide.
(i) Write the equation for this reaction. [1]
(ii) Deduce the change in the oxidation state of sulfur. [1]
(iii) Suggest why this process might raise environmental concerns. [1]
(f) Explain why the addition of small amounts of carbon to iron makes the metal harder. [2]
▶️ Answer/Explanation
Markscheme (concise points)
(a) [1]
Metals have delocalized/mobile valence electrons in a lattice of positive ions → electrons move to carry charge.
(b) [2]
Any two valid chemical properties of non-metals, e.g. forms acidic oxides; tends to gain electrons/act as oxidizing agent; forms covalent compounds; forms anions (rather than cations).
(c) [2]
Sketch shows two regions of small increases with a large jump from the 6th to 7th ionisation energy (removal from an inner shell). Axes correctly labelled.

(d)
(i) [2] Giant ionic lattice; strong electrostatic attraction between oppositely charged ions (Fe2+ and S2−).
(ii) [1] X-ray crystallography / X-ray diffraction.
(iii) [1] 1s2 2s2 2p6 3s2 3p6. (Do not use [Ne] shorthand.)
(iv) [1] S2− valence electrons are in n=3 (3s/3p) → further from nucleus/greater shielding than O2− (n=2) → larger radius.
(v) [1] Classification aids explanation, generalization, and prediction of properties (bonding models as a continuum).
(e)
(i) [1] 4FeS(s) + 7O2(g) → 2Fe2O3(s) + 4SO2(g). (Any correct balanced ratio earns credit.)
(ii) [1] Sulfur: −2 → +4, change = +6.
(iii) [1] SO2 emissions → acid rain and/or respiratory irritation → environmental concern.
(f) [2]
Carbon atoms (different size) distort the Fe lattice and impede dislocation/slip; layers cannot slide easily → harder alloy/steel.
Total for Question 1: 16
