IBDP Physics- E.5 Fusion and stars- IB Style Questions For HL Paper 2 -FA 2025
Question
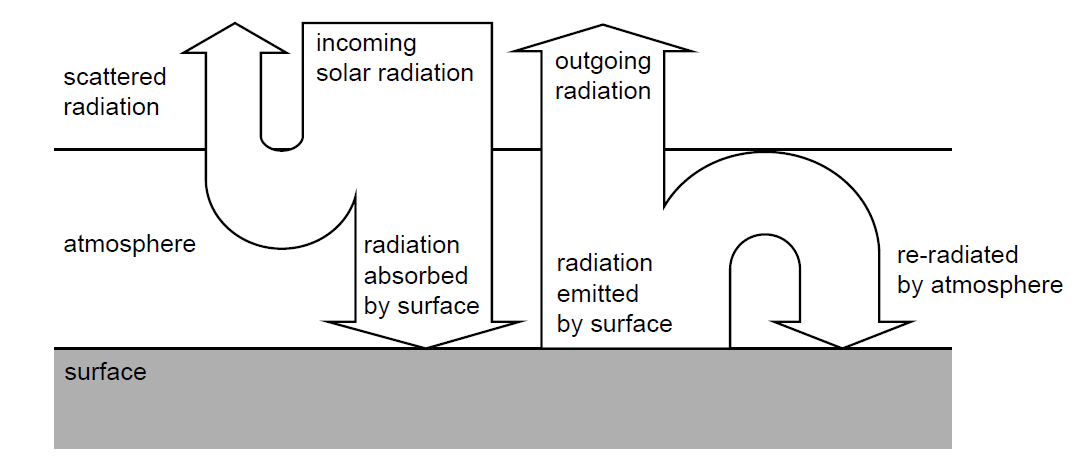
Average albedo of Earth \(= 0.30\)
Mean global surface temperature \(= 288\,K\)
Mean Earth–Sun distance \(= 1.5\times10^{11}\,m\)
(ii) Show that the average global intensity of radiation absorbed by the Earth’s surface is approximately \(240\,W\,m^{-2}\).
(iii) Calculate the average intensity of radiation re-emitted by the atmosphere toward the surface. Assume the surface emissivity is \(0.90\).
(i) Given that the mass of a helium nucleus is \(4.001506\,u\), calculate the energy released in this reaction in MeV.
(ii) Explain how fusion reactions help maintain a stable radius of the Sun.
(iii) Outline how the presence of helium in the Sun can be confirmed using observational evidence.
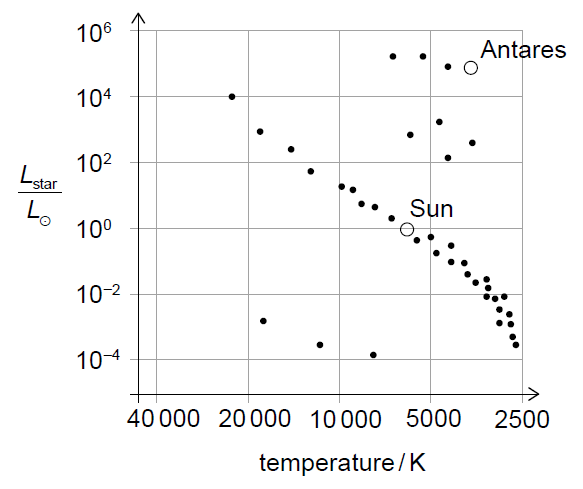
(ii) Discuss how nuclear fusion processes in Antares differ from those occurring in the Sun.
Most-appropriate topic codes (IB Physics):
• Topic B.1: Thermal energy transfers (black-body radiation) — part (b)(iii)
• Topic E.5: Fusion and stars (HR diagram, stellar evolution) — parts (d), (e)
▶️ Answer/Explanation
(a)
The solar constant is the power per unit area of solar radiation incident on a surface perpendicular to the Sun’s rays at the average Earth–Sun distance, measured at the top of the Earth’s atmosphere.
(b)
(i) Greenhouse gases absorb infrared radiation emitted by the Earth’s surface because the radiation matches energy transitions in the gas molecules. These molecules then re-emit the radiation in all directions, including back toward the surface.
(ii) Average incoming intensity \(= \frac{S}{4} = \frac{1360}{4} = 340\,W\,m^{-2}\).
Absorbed intensity \(= (1 – 0.30)\times340 \approx 238\,W\,m^{-2} \approx 240\,W\,m^{-2}\).
(iii) Emission from surface: \(\epsilon\sigma T^4 = 0.90 \times 5.67\times10^{-8} \times 288^4 \approx 351\,W\,m^{-2}\).
Energy balance gives re-radiated intensity: \(351 – 238 \approx 113\,W\,m^{-2}\).
(c)
Total solar power \(P = S \times 4\pi d^2 = 1360 \times 4\pi(1.5\times10^{11})^2 \approx 3.8\times10^{26}\,W \approx 4\times10^{26}\,W\).
(d)
(i) Mass defect: \(\Delta m = [4(1.007276) + 2(0.000549)] – 4.001506 = 0.028696\,u\).
Energy released: \(E = 0.028696 \times 931.5 \approx 26.7\,MeV\).
(ii) Energy released by fusion produces outward radiation pressure that balances gravitational contraction, maintaining hydrostatic equilibrium.
(iii) The presence of helium is confirmed through analysis of the Sun’s absorption spectrum, where characteristic helium spectral lines are observed.
(e)
(i) Antares is a red supergiant.
(ii) The Sun primarily fuses hydrogen into helium via the p–p chain, whereas Antares, due to its much larger mass and core temperature, undergoes fusion of heavier elements such as helium into carbon and beyond.
