iGCSE Physics (0625) 2.1.2 Particle model Paper 3 -Exam Style Questions- New Syllabus
Question
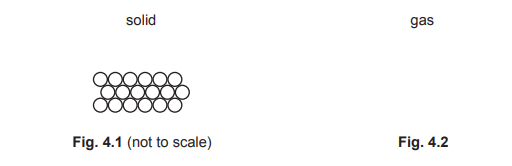
Most-appropriate topic codes (Cambridge IGCSE Physics 0625):
• Topic 2.1.1 — States of matter (Part (a)(i))
• Topic 2.1.2 — Particle model (Parts (a)(ii), (a)(iii))
• Topic 2.2.3 — Melting, boiling and evaporation (Parts (b)(i), (b)(ii))
▶️ Answer/Explanation
(a)(i)
vibrate
In a solid, particles are held closely together in a fixed, regular arrangement by strong intermolecular forces. They do not have enough energy to move from their positions, so their motion is limited to vibrating about fixed points.
(a)(ii)
random (arrangement)
clear separation of particles
Particles in a gas are widely spaced with no fixed pattern. The diagram should show particles distributed randomly across the available space with significant gaps between them, reflecting the negligible forces of attraction in the gaseous state.
(a)(iii)
any two from:
random
fast/high speed / high KE
colliding
Gas particles possess high kinetic energy, allowing them to overcome attractive forces. They move rapidly in straight lines in random directions, frequently colliding with each other and the walls of their container.
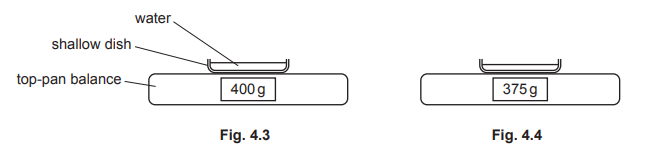
(b)(i)
evaporation
The mass of water decreases because some liquid water changes state to water vapour and escapes from the open dish into the surrounding air.
(b)(ii)
any two from:
(happens at the) surface
the more energetic particles escape
(liquid particles) → gas / vapour (particles)
Evaporation occurs at the liquid surface. Particles in the liquid have a range of kinetic energies; those near the surface with the highest energy overcome the attractive forces of neighbouring particles and escape to become vapour, reducing the average kinetic energy and mass of the remaining liquid.
