iGCSE Physics (0625) 2.2.2 Specific heat capacity Paper 3 -Exam Style Questions- New Syllabus
Question
(a) A teacher shows the class a demonstration about heat energy and states of matter.
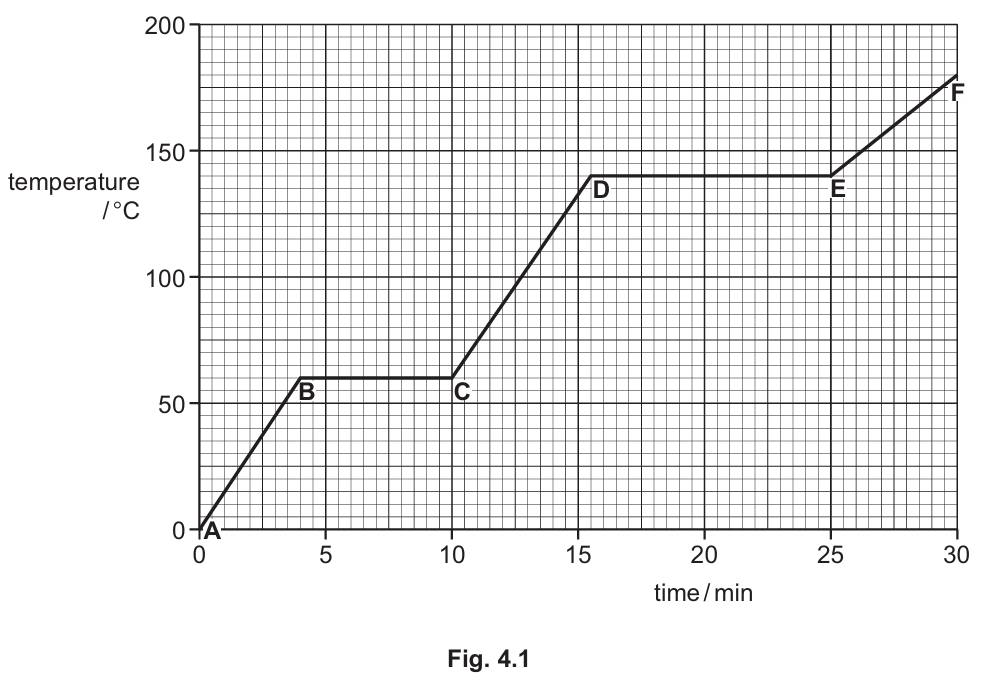
The teacher heats a substance. A heater supplies thermal energy at a steady rate. The students measure the temperature of the substance every minute for $30\text{ minutes}$.
Fig. $4.1$ shows the graph of their results.
At point $A$, the substance is solid.
Most-appropriate topic codes (Cambridge IGCSE Physics $0625$):
• Topic $2.2.3$ — Melting, boiling and evaporation (Parts $\text{(a)(ii)}$, $\text{(a)(iii)}$, $\text{(a)(iv)}$, $\text{(b)}$)
• Topic $2.2.2$ — Specific heat capacity (Part $\text{(a)(i)}$)
• Topic $2.1.2$ — Particle model (Part $\text{(b)(i)}$)
▶️ Answer/Explanation
(a)(i)
For the correct answer:
Temperature increasing OR internal energy increasing.
In section $AB$, the graph shows the temperature of the solid increasing from $0~^\circ\text{C}$ to $60~^\circ\text{C}$. As thermal energy is supplied at a steady rate, the kinetic energy of the particles within the solid increases. This continuous input of heat results in a direct increase in both the temperature and the internal energy of the substance before it reaches its melting point.
(a)(ii)
For the correct answer:
Melting OR changing from solid to liquid.
During section $BC$, the temperature remains constant at $60~^\circ\text{C}$ despite the continuous input of thermal energy. This flat region on the heating curve indicates a change of state where the solid is turning into a liquid. The supplied energy is being used to overcome the intermolecular forces of attraction holding the solid particles together, which is the process of melting.
(a)(iii)
For the correct answer:
$140~^\circ\text{C}$
The second horizontal line on the temperature-time graph occurs at section $DE$. At this stage, the substance undergoes a second phase change from a liquid to a gas, meaning it is boiling. By reading the value on the y-axis corresponding to the horizontal line $DE$, we can determine that the boiling point of the substance is exactly $140~^\circ\text{C}$.
(a)(iv)
For the correct answer:
Gas
After the substance has completely boiled at $140~^\circ\text{C}$ (end of section $DE$), the temperature begins to rise again in section $EF$. Since the substance has already transitioned from a solid to a liquid, and then from a liquid to a gas, any further steady thermal energy input increases the temperature of the gas. Therefore, in section $EF$, the substance exists entirely in the gaseous state.
(b)(i)
For the correct answer:
Any $3$ from: liquid (particles) $\rightarrow$ gas / vapour (particles); happens at a surface; absorbs thermal energy / heat; kinetic energy of particle increases; more energetic particles escape.
Evaporation occurs when a substance changes from a liquid to a gas at temperatures below its boiling point. Particles near the surface of the liquid absorb thermal energy from their surroundings, increasing their kinetic energy. The most energetic particles overcome the attractive forces of neighboring particles and escape into the air. Unlike boiling, this process happens exclusively at the liquid’s surface.
(b)(ii)
For the correct answer:
(Remaining liquid) cools.
As the most energetic particles escape the surface of the puddle during evaporation, the average kinetic energy of the particles left behind decreases. Since temperature is a macroscopic measure of the average kinetic energy of the particles, this loss of energy results in a temperature drop. Consequently, the remaining liquid in the puddle cools down.
