iGCSE Physics (0625) 5.1.1 the atom Paper 3 -Exam Style Questions- New Syllabus
Question
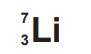
Most-appropriate topic codes (Cambridge IGCSE Physics 0625):
• Topic 5.1.1 — The atom (Parts (a), (b))
• Topic 5.2.4 — Half-life (Part (c))
▶️ Answer/Explanation
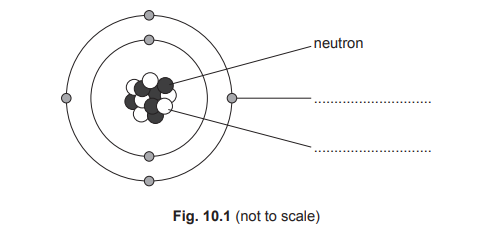
(a)
For the correct answer:
electron
proton
The diagram shows a central nucleus composed of protons and neutrons, with particles orbiting in shells around it. The particle labelled in the outer orbit is an electron, which carries a negative charge. The particle labelled within the nucleus is a proton, which carries a positive charge.
(b)
For the correct answer (any three from):
3 protons (in nucleus)
4 neutrons (in nucleus)
3 electrons outside nucleus
nucleus labelled
electron orbits seen
The nuclide notation $^{7}_{3}\text{Li}$ indicates that the nucleus contains 3 protons (the atomic number) and a total of 7 nucleons, meaning there are $7 – 3 = 4$ neutrons. Since atoms are neutral, there must be 3 electrons orbiting the nucleus in distinct shells.
(c)
For the correct answer:
$(5700 \times 3 =)$ 17 100 (years)
(from 120 mg to 15 mg takes) 3 half-lives
The mass of the isotope halves with each half-life: $120\text{ mg} \rightarrow 60\text{ mg} \rightarrow 30\text{ mg} \rightarrow 15\text{ mg}$. This sequence requires three half-lives. The total time taken is therefore $3 \times 5700\text{ years} = 17100\text{ years}$.
