iGCSE Physics (0625) 5.2.5 Safety precautions -Exam Style Questions Paper 1 - New Syllabus
Question
- student 1: Move the radioactive source with her bare hands.
- student 2: Stay as close as possible to the radioactive source during the experiment.
- student 3: Minimise the time of the experiment.
▶️ Answer/Explanation
Detailed solution:
To reduce health risks from ionising radiation, one must minimize exposure.
Student 1 is incorrect because handling sources with bare hands increases the risk of contamination and high local dosage; tools like tongs should be used.
Student 2 is incorrect because radiation intensity follows the inverse square law, meaning the dose increases significantly as distance decreases.
Student 3 is correct because the total radiation dose received is directly proportional to the time of exposure (Dose∝time).
Therefore, minimizing the duration of the experiment effectively reduces the total absorbed dose.
Only Student 3 provides a valid safety suggestion, making option D the correct choice.
Question
▶️ Answer/Explanation
Detailed solution:
High-frequency electromagnetic waves carry enough energy to ionize atoms and damage biological cells. According to the syllabus, ultraviolet ($UV$) radiation causes damage to surface cells and eyes, potentially leading to skin cancer. $X$-rays and $\gamma$-rays (gamma rays) possess even higher frequencies and can cause deeper mutations or significant cell death. In contrast, visible light (such as green and red) and radio waves do not have sufficient energy per photon to cause this type of ionising cell damage. Therefore, both gamma rays and ultraviolet are correct answers.
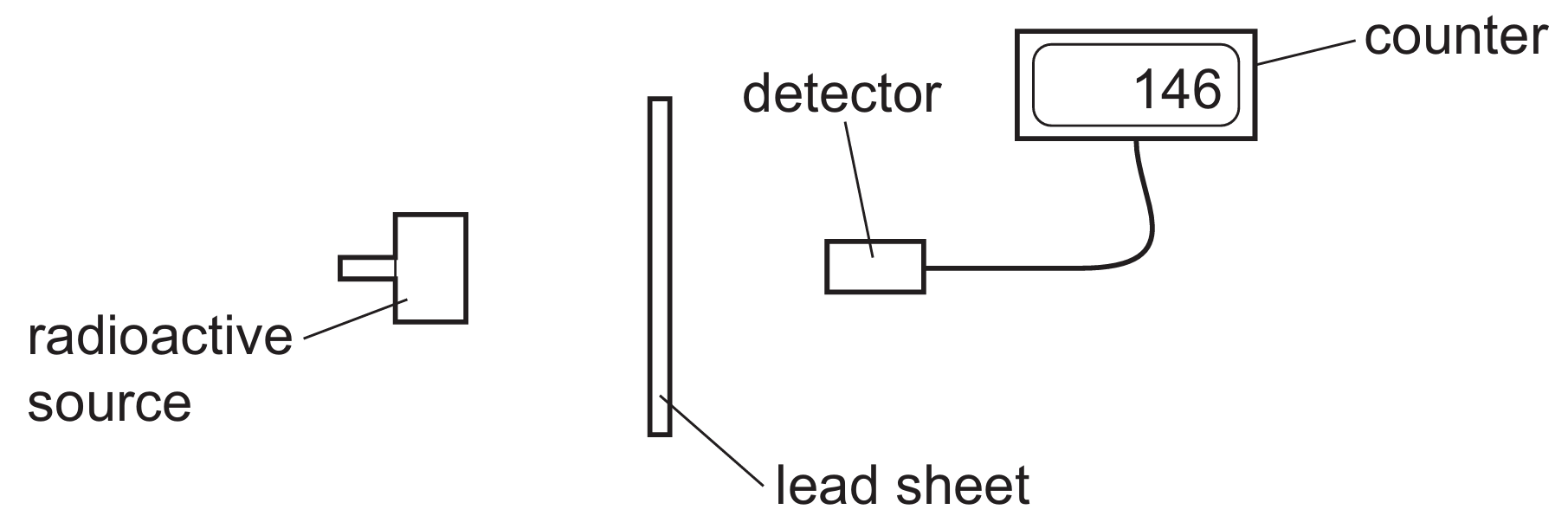
Question
▶️ Answer/Explanation
Detailed solution:
Lead is a very dense material with a high atomic number, making it highly effective at attenuating ionising radiation.
Radioactive sources may emit $\alpha$-particles, $\beta$-particles, or $\gamma$-radiation, all of which transfer energy to the material they encounter.
While $\alpha$ and $\beta$ are easily stopped, $\gamma$-rays are highly penetrating and require thick layers of dense material like lead to be significantly reduced.
The lead atoms interact with the incoming radiation, causing the radiation to lose its energy and be absorbed rather than passing through.
Radiation cannot be “neutralised,” “repelled,” or “dissolved”; it can only be blocked or shielded through absorption by a medium.
Therefore, lead-lined boxes ensure the safety of personnel by capturing the emissions within the container walls.
