iGCSE Physics (0625) 2.1.2 Particle model -Exam Style Questions Paper 1 - New Syllabus
Question
▶️ Answer/Explanation
Detailed solution:
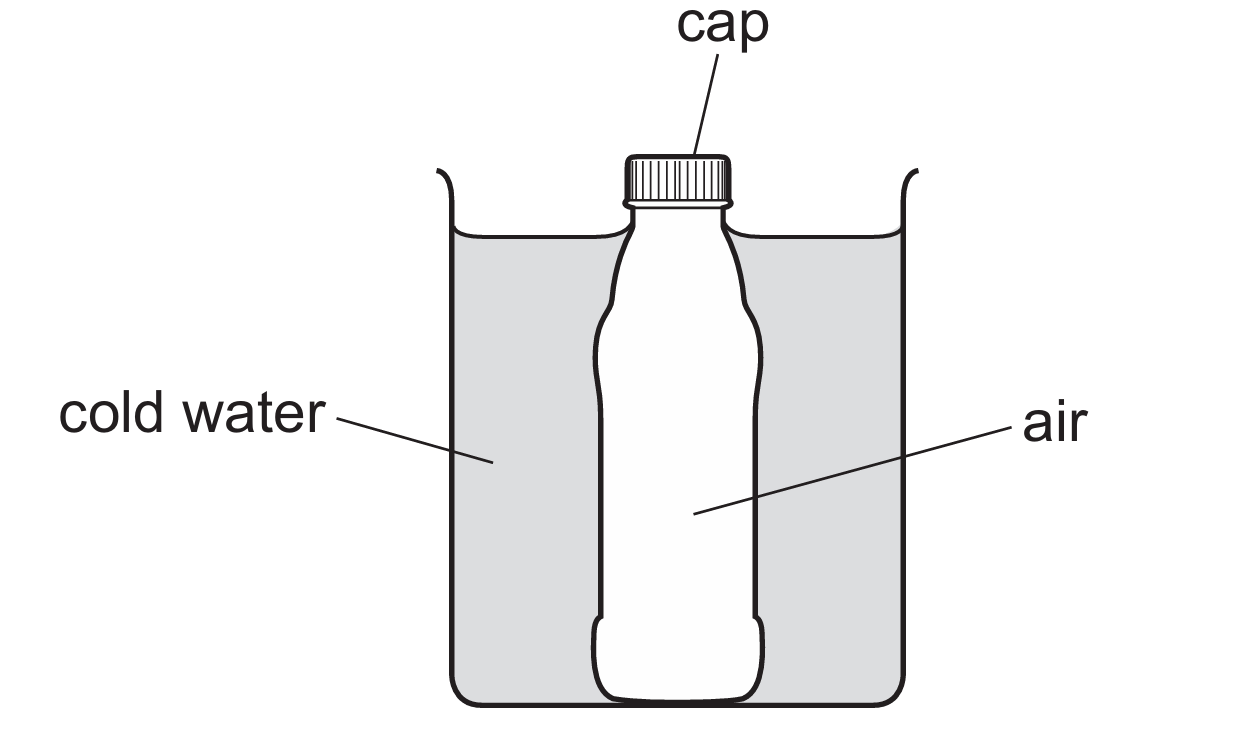
Since the bottle is sealed, the mass of air is constant, and because the bottle’s contraction is ignored, the volume $V$ remains the same.
As the air cools, the temperature $T$ decreases, causing the average kinetic energy of the particles to decrease.
According to the kinetic theory, slower particles collide with the walls less frequently and with less force, reducing the pressure $P$.
Since pressure is force per unit area ($P = \frac{F}{A}$), the force exerted on the cap also decreases as the pressure drops.
Therefore, while pressure, energy, and force all change with temperature, the volume of the air is fixed by the rigid container.
Thus, the only quantity that remains constant throughout the cooling process is the volume of air in the bottle.
Question

▶️ Answer/Explanation
Detailed solution:
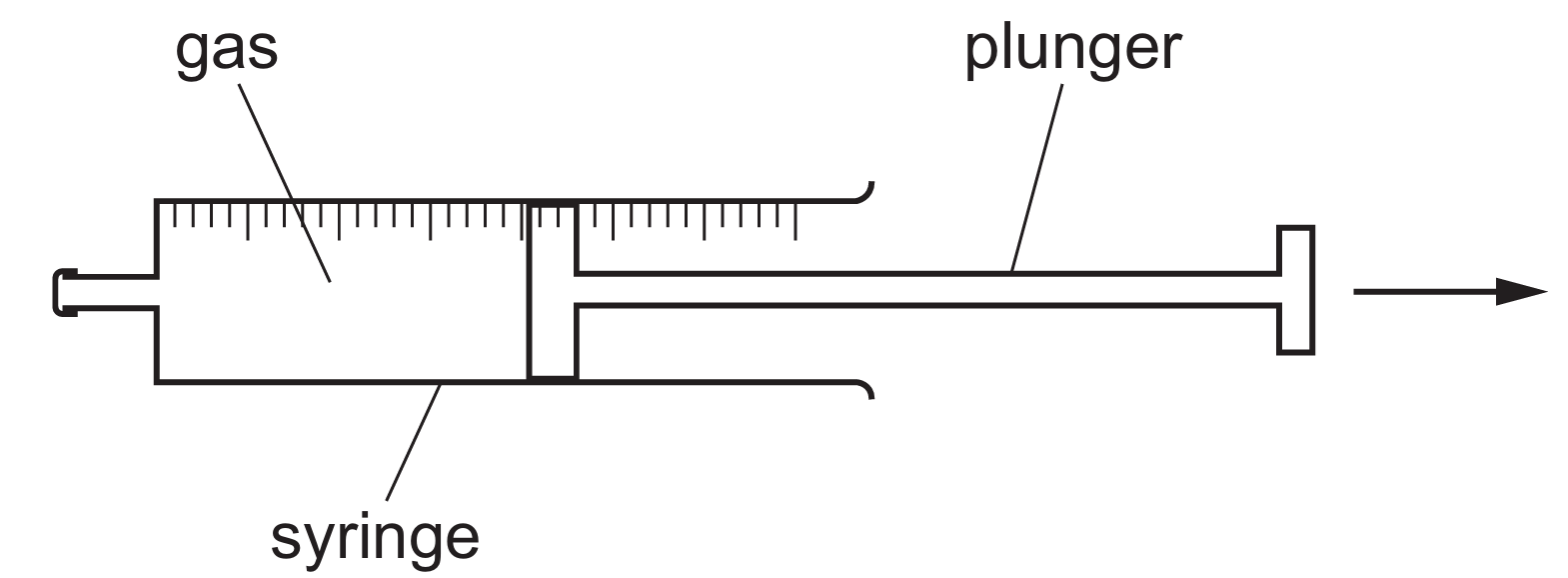
When the plunger is moved out, the volume $V$ of the gas increases while the temperature $T$ remains constant.
According to Boyle’s Law, $pV = \text{constant}$, so an increase in volume leads to a decrease in pressure $p$.
In terms of the kinetic particle model, the gas particles are now spread out over a larger space.
As a result, the particles hit the walls of the syringe less often, meaning the frequency of collisions decreases.
Since pressure is defined as the force per unit area, fewer collisions per second result in a lower pressure.
Therefore, Row A correctly identifies that the pressure decreases because particles collide with the walls less frequently.
Question
▶️ Answer/Explanation
Detailed solution:
In a liquid, particles are packed closely together with very small gaps between them.
When water boils into steam (a gas), the particles gain enough energy to overcome attractive forces.
The particles in a gas move randomly and are spaced much further apart than in a liquid.
Since the number of particles and the size of individual particles remain constant during a state change,
the massive increase in volume from $1.0 cm^3$ to $1600 cm^3$ is due solely to the increased separation.
Therefore, the average distance between particles in steam is significantly greater than in liquid water.
