iGCSE Physics (0625) 2.2.2 Specific heat capacity -Exam Style Questions Paper 1 - New Syllabus
Question
▶️ Answer/Explanation
Detailed solution:
Temperature is a macroscopic measure of the average kinetic energy of the particles in a substance.
When a gas cools, its temperature $\theta$ decreases, which directly corresponds to a reduction in the average kinetic energy $E_{k}$ of its constituent molecules.
The internal energy of an ideal gas is primarily determined by the sum of the kinetic energies of all its particles.
Therefore, a decrease in internal energy during cooling is caused by the particles moving more slowly on average.
Option C and D describe changes that would typically increase energy or occur during expansion, while B is irrelevant to thermal cooling.
Thus, the decrease in internal energy is due to a decrease in the average kinetic energy of the gas particles.
Question
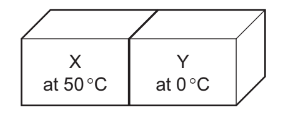
What has happened to the internal energy of each block?
▶️ Answer/Explanation
Detailed solution:
Block X cools from \(50^{\circ}\mathrm{C}\) to \(25^{\circ}\mathrm{C}\), so its particles lose kinetic energy and its internal energy decreases.
Block Y warms from \(0^{\circ}\mathrm{C}\) to \(25^{\circ}\mathrm{C}\), so its particles gain kinetic energy and its internal energy increases.
Energy is transferred thermally from the hotter block to the colder block until thermal equilibrium is reached.
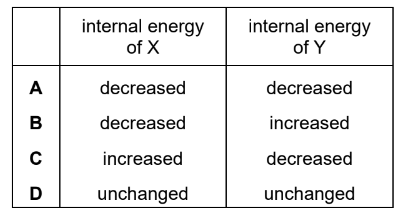
Therefore, the internal energy of X decreases and the internal energy of Y increases.
This matches Option B.
What is the change in temperature, expressed in kelvin?
▶️ Answer/Explanation
Detailed solution:
To find the change in temperature, we first calculate the difference in degrees Celsius: $40^\circ\text{C} – 10^\circ\text{C} = 30^\circ\text{C}$.
It is a fundamental rule in physics that a change of $1$ degree Celsius is exactly equal to a change of $1$ kelvin.
Even if you convert both temperatures to kelvin first ($10^\circ\text{C} = 283\text{ K}$ and $40^\circ\text{C} = 313\text{ K}$), the difference remains the same.
Subtracting these values gives: $313\text{ K} – 283\text{ K} = 30\text{ K}$.
Therefore, the change in temperature expressed in kelvin is $30\text{ K}$, which matches Option A.
