iGCSE Physics (0625) 5.1.2 Nucleus -Exam Style Questions Paper 1 - New Syllabus
Question
What does the nucleus of one atom of this isotope contain?
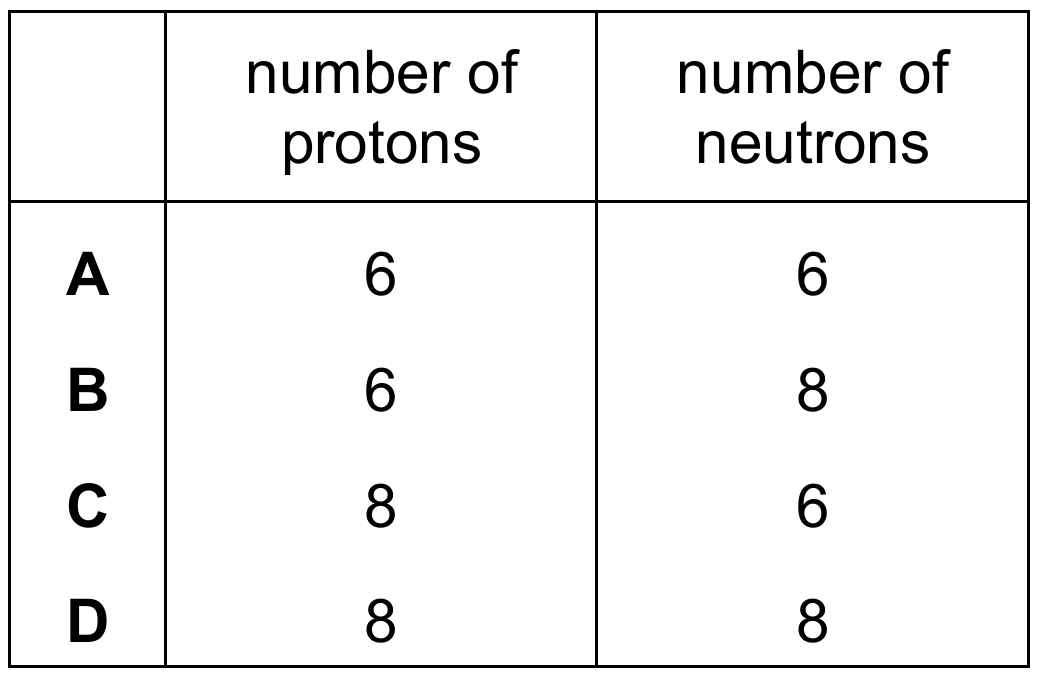
▶️ Answer/Explanation
Detailed solution:
In the nuclide notation ${}_{Z}^{A}\text{X}$, the lower number $Z$ represents the proton number (atomic number).
For ${}_{6}^{14}\text{C}$, the number of protons is $Z = 6$.
The upper number $A$ represents the nucleon number (mass number), which is the sum of protons and neutrons ($A = Z + N$).
To find the number of neutrons $N$, we subtract the proton number from the nucleon number: $N = A – Z$.
Calculating for this isotope: $14 – 6 = 8$ neutrons.
Thus, the nucleus contains $6$ protons and $8$ neutrons, matching the values provided in Row B.
Question
What is the number of protons in a krypton nucleus?
▶️ Answer/Explanation
Detailed solution:
The atomic number, often represented by the symbol $Z$, is defined as the number of protons found in the nucleus of an atom.
In this question, the isotope of krypton is explicitly stated to have an atomic number of $36$.
The mass number (or nucleon number, $A = 78$) represents the total sum of protons and neutrons, but does not change the identity or proton count of the element.
Since the atomic number is $36$, the number of protons must also be $36$.
This matches Option A.
Question
What is the number of neutrons in this nuclide?
▶️ Answer/Explanation
Detailed solution:
In nuclide notation ${}_{Z}^{A}\text{X}$, $A$ represents the nucleon number (total protons and neutrons) and $Z$ represents the proton number.
For the given nuclide ${}_{7}^{15}\text{N}$, the nucleon number is $A = 15$ and the proton number is $Z = 7$.
The number of neutrons $N$ is calculated using the formula $N = A – Z$.
Substituting the values, we get $N = 15 – 7 = 8$.
Therefore, there are $8$ neutrons in this nitrogen nuclide, making option B the correct choice.
