About this unit
Group 15 elements: General introduction, electronic configuration, occurrence, oxidation states, trends in physical and chemical properties; preparation and properties of ammonia and nitric acid, oxides of nitrogen (structure only); Phosphorous- allotropic forms; compounds of phosphorous: preparation and properties of phosphine, halides (PCI3 , PCI5 ) and oxoacids (elementary idea only).
Group 16 elements: General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemical properties; dioxygen: preparation, properties and uses; classification of oxides; ozone. Sulphur – allotropic forms; compounds of sulphur: preparation, preparation, properties and uses of sulphur dioxide; sulphuric acid: industrial process of manufacture, properties and uses, oxoacids of sulphur (structures only).
Group 17 elements: General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemical properties; compounds of halogens: preparation, properties and uses of chlorine and hydrochloric acid, interhalogen compounds oxoacids of halogens (structures only).
Group 18 elements: General introduction, electronic configuration, occurrence, trends in physical and chemical properties, uses.
| p-Block Elements – Nitrogen Family |
| p-Block Elements – Oxygen Family |
| p-Block Elements – Halogens |
p-BLOCK ELEMENTS – NOBLE GASES
INTRODUCTION
GENERAL CHARACTERISTICS
ELECTRONIC CONFIGURATION
Element | Symbol | At No. | Valence shell electronic confg. |
Helium | He | 2 | 1s2 |
Neon | Ne | 10 | [He] 2s22p6 |
Argon | Ar | 18 | [Ne] 3s23p6 |
Krypton | Kr | 36 | [Ar] 3d104s24p6 |
Xenon | Xe | 54 | [Kr] 4d10, 5s25p6 |
Radon | Rn | 86 | [Xe] 4f14, 3d10, 6s26p6 |
PHYSICAL STATE
PRESENCE
ABUNDANCE
ATOMICITY
MELTING AND BOILING POINTS
ATOMIC RADII
CRITICAL TEMPERATURE/CRITICAL PRESSURE
Element | He | Ne | Ar | Kr | Ye | Rn |
Tc(K) | 5.1 | 44.3 | 150.6 | 211.0 | 256.4 | 373.5 |
Pc(Atm) | 2.26 | 26.86 | 47.99 | 54.3 | 58.2 | 62.4 |
DENSITY
HEAT OF VAPORISATION
SOLUBILITY IN WATER
LIQUEFACTION
IONISATION ENERGY
ELECTRON AFFINITY
POLARIZABILITY
ADSORPTION BY CHARCOAL
DISCOVERY
- Argon – It was isolated by Rayleigh and confirmed by Ramsay, from air (free from O2, moisture and CO2). Due to its inertness the gas was named as argon (meaning lazy).
- Helium – It was observed in the spectrum of the sun hence name helium, from helios, which means sun, by Lockyer and Frankland. Ramsay obtained it from gases occluded in uranium minerals.
- Neon, Krypton and Xenon – These were obtained by fractional distillation of liquid air under reduced pressure. These were named as neon means “new” krypton means “hidden” and Xenon means “stranger or foreigner”.
- Radon – Spectroscopically was noticed by Dorn and isolated by disintegration of radium by Rutherford and Soddy.
ISOLATION OF RARE GASES
RAMSAY AND RAYLEIGH’S FIRST METHOD
RAMSAY AND RAYLEIGH’S SECOND METHOD
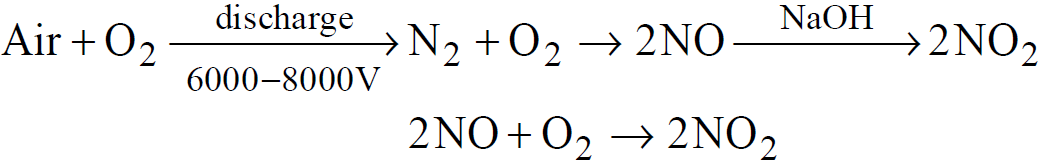
FISCHER AND RINGE’S METHOD
SEPARATION OF RARE GASES BY DEWAR’S METHOD
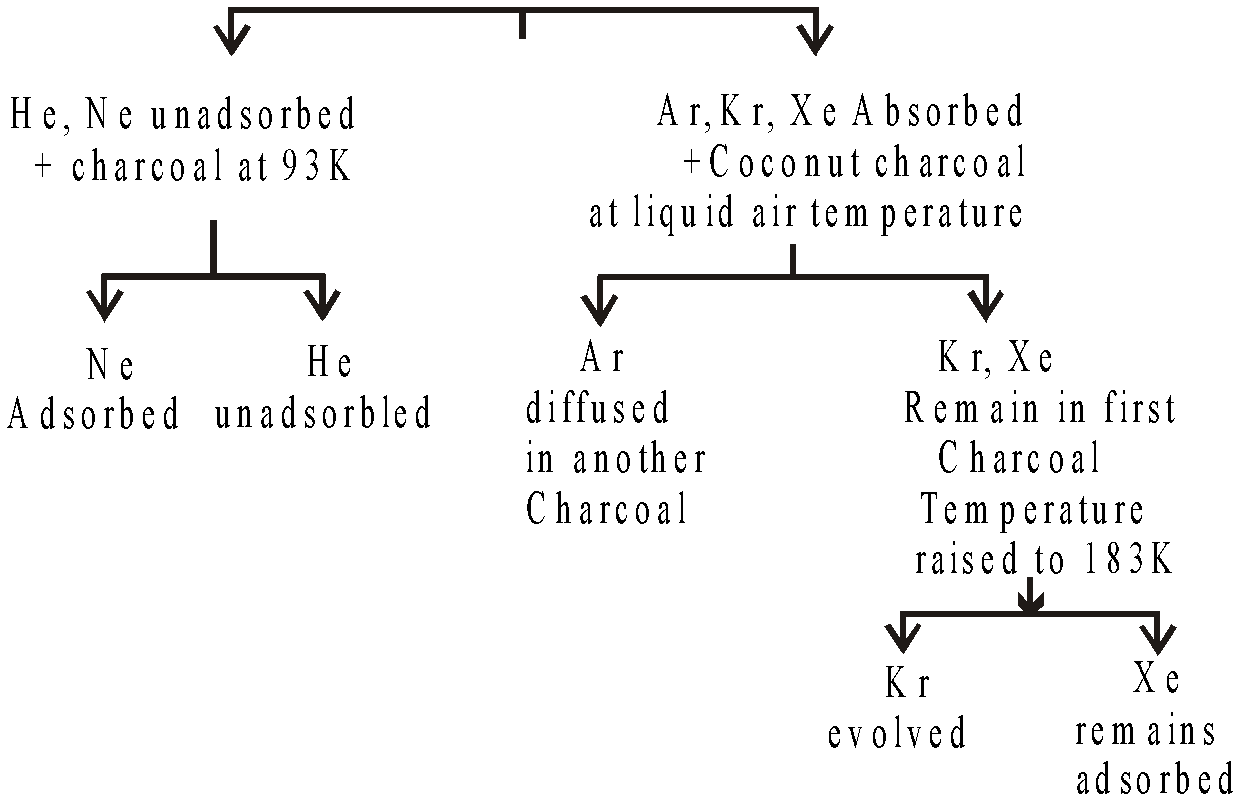
Element | He | Ne | N2 | Ar | O2 | Kr | Xe |
B.pt (K) | 4 | 27 | 77 | 87 | 90 | 121 | 124 |
HELIUM FROM OTHER SOURCES
PROPERTIES OF NOBLE GASES
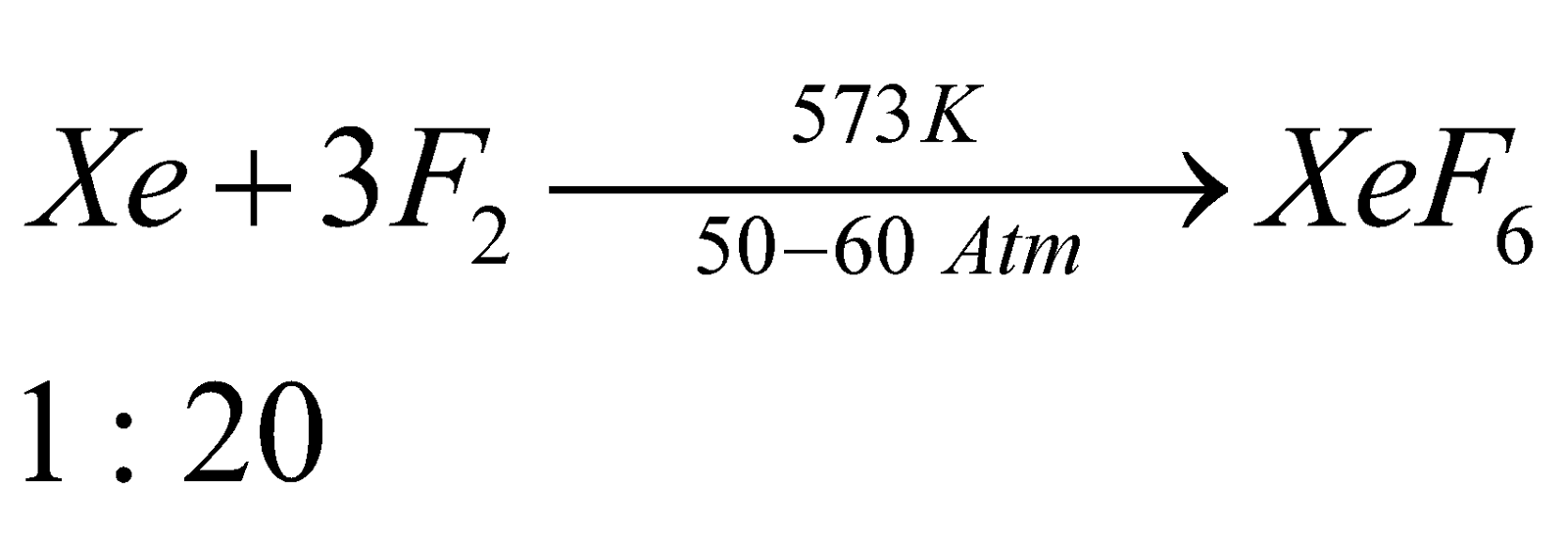
STRUCTURES OF XENON FLUORIDES
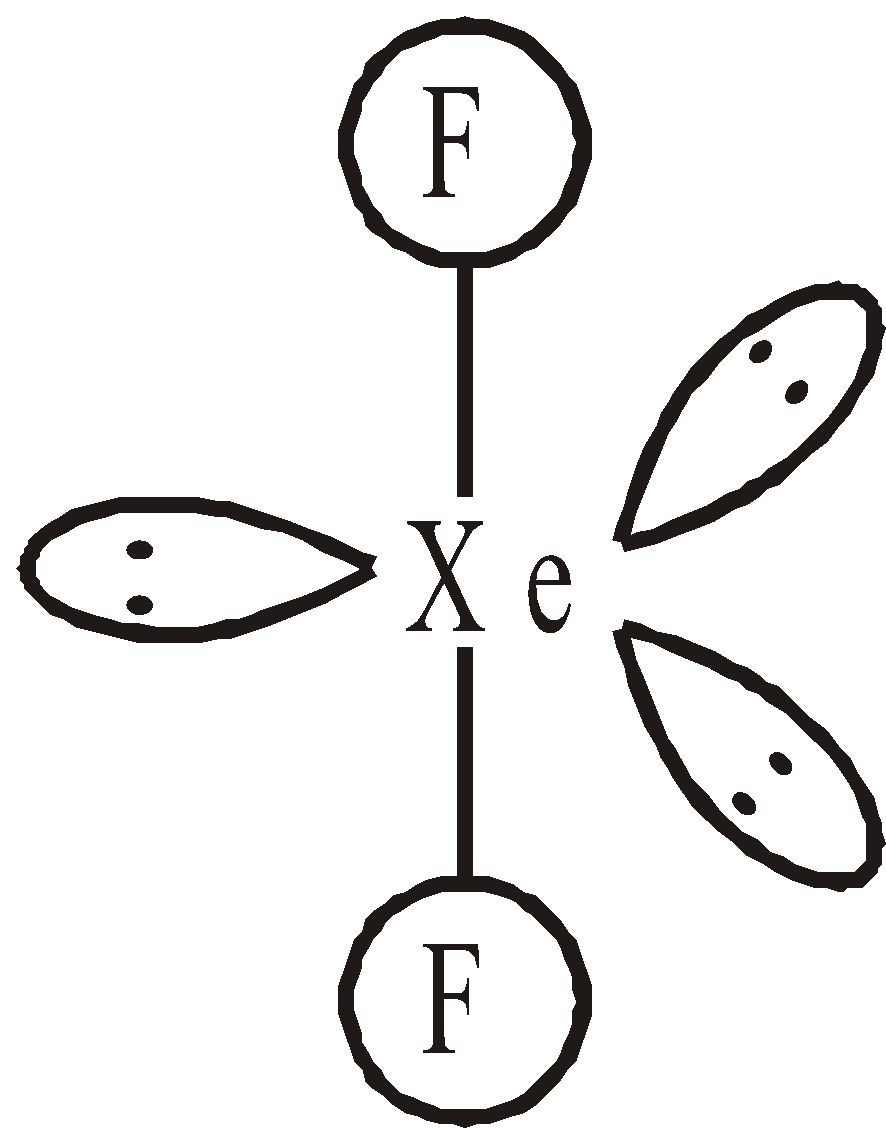 Linear
Linear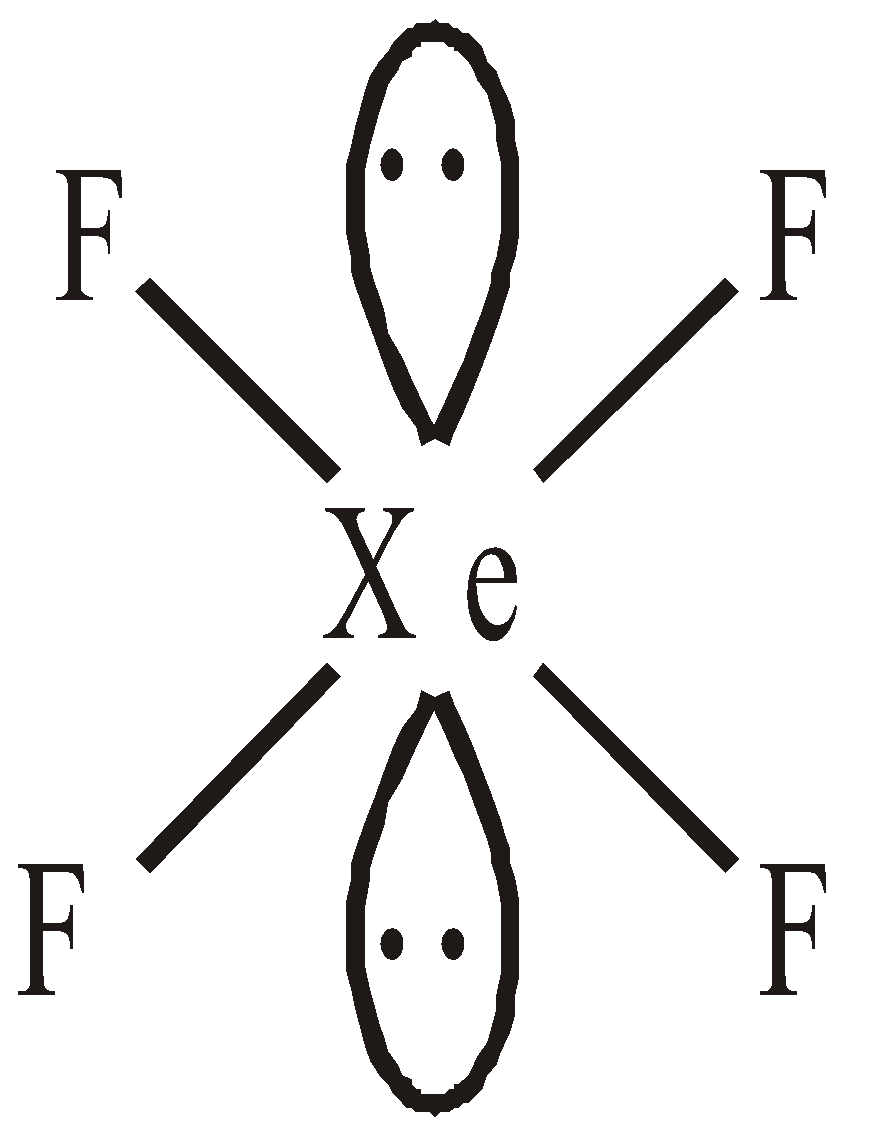 square planar
square planar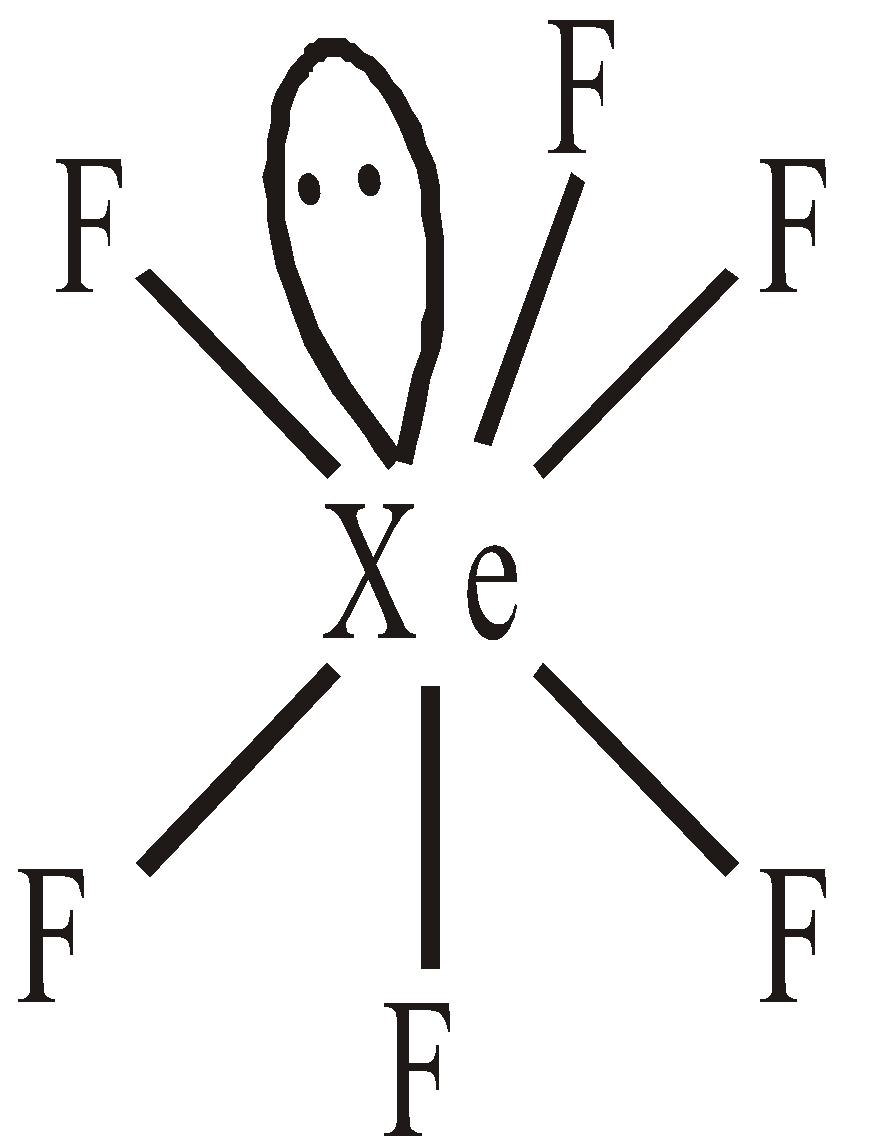 Pentagonal pyramidal or distorted octahedral
Pentagonal pyramidal or distorted octahedralSTRUCTURES OF OXYFLUORIDES AND OXIDES OF XENON
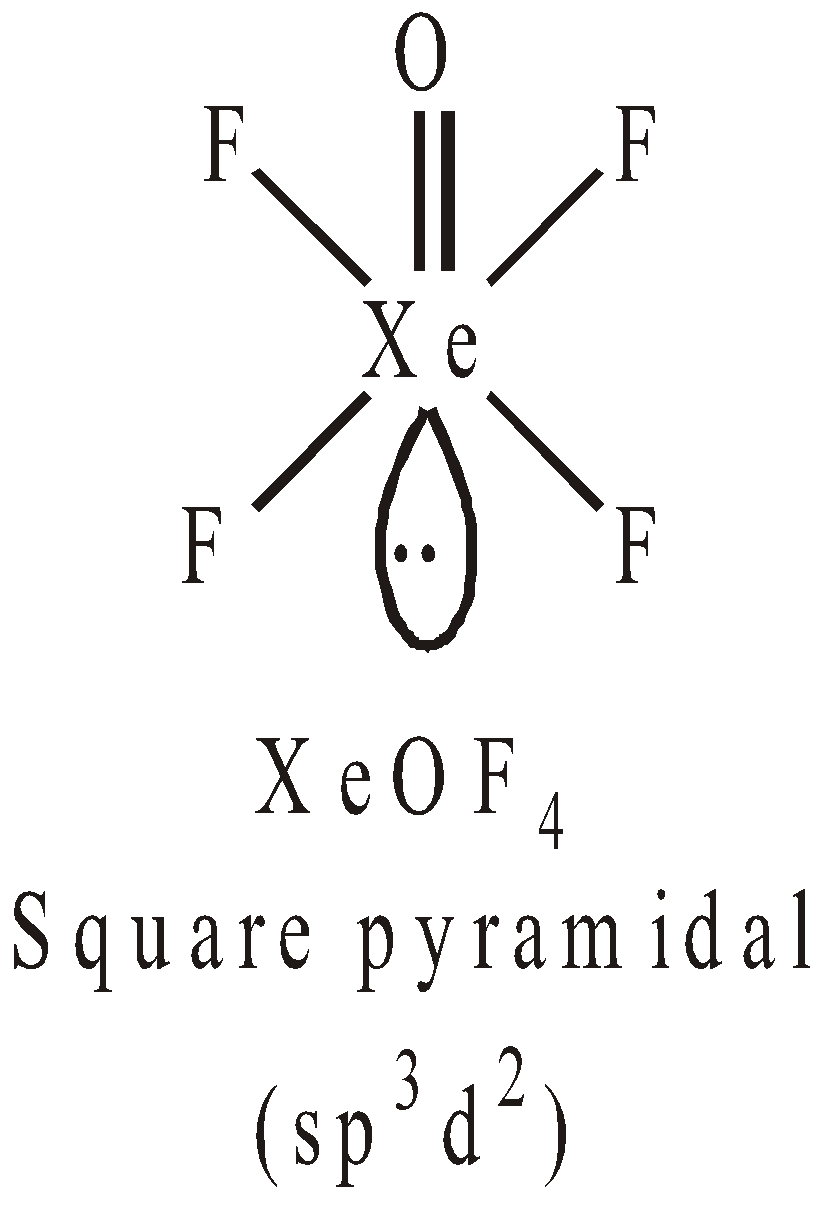
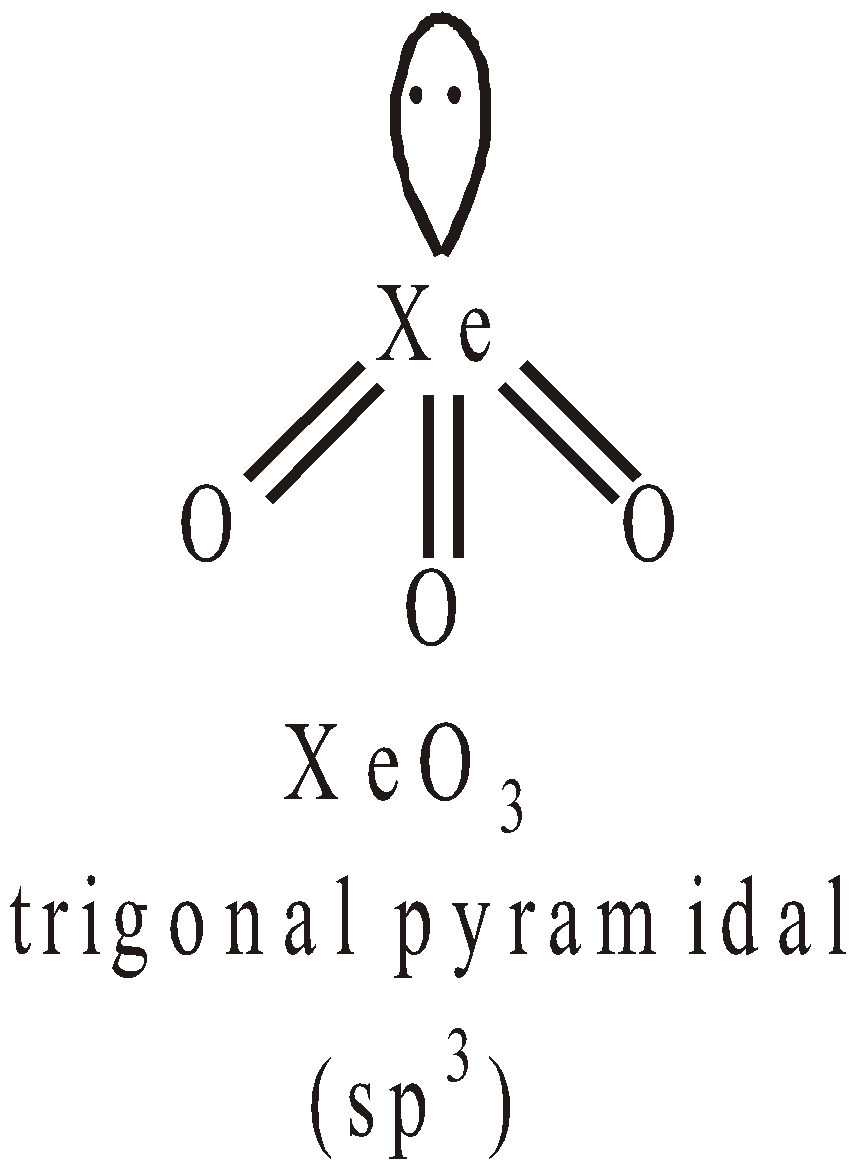
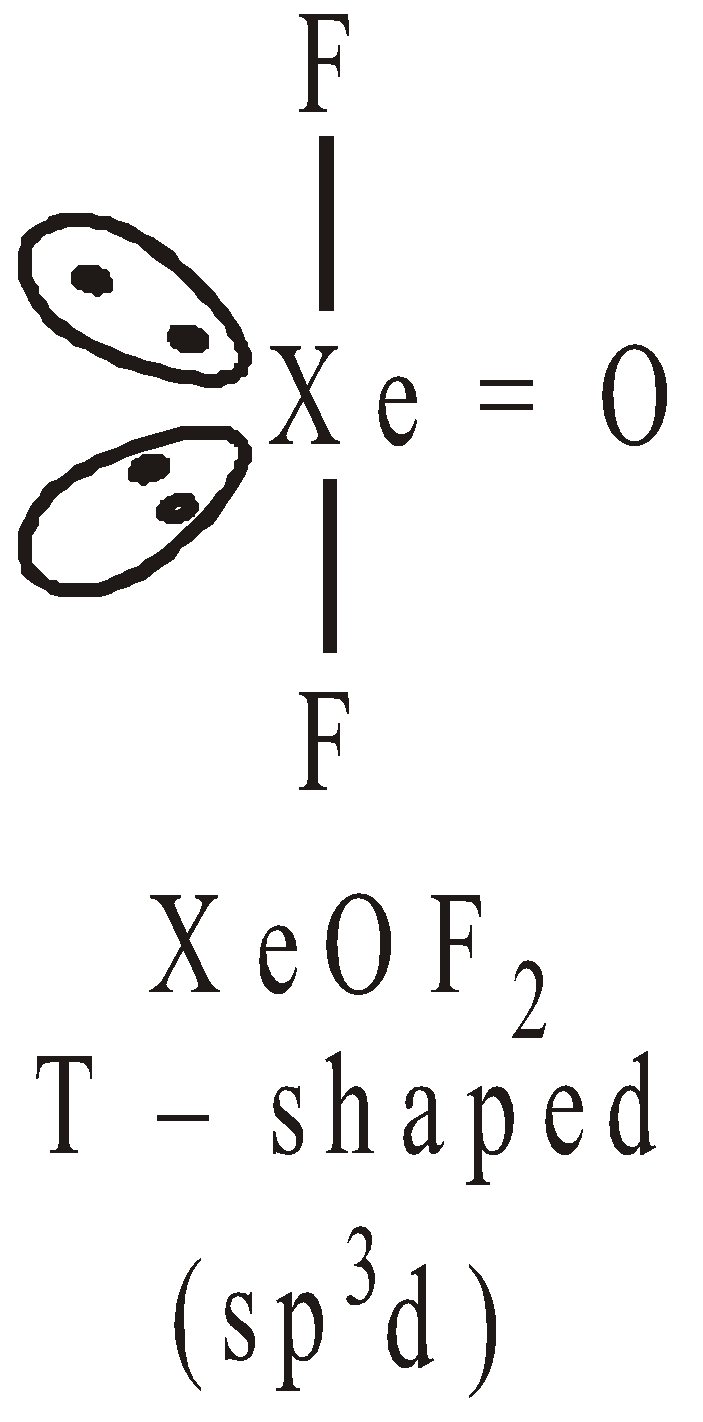
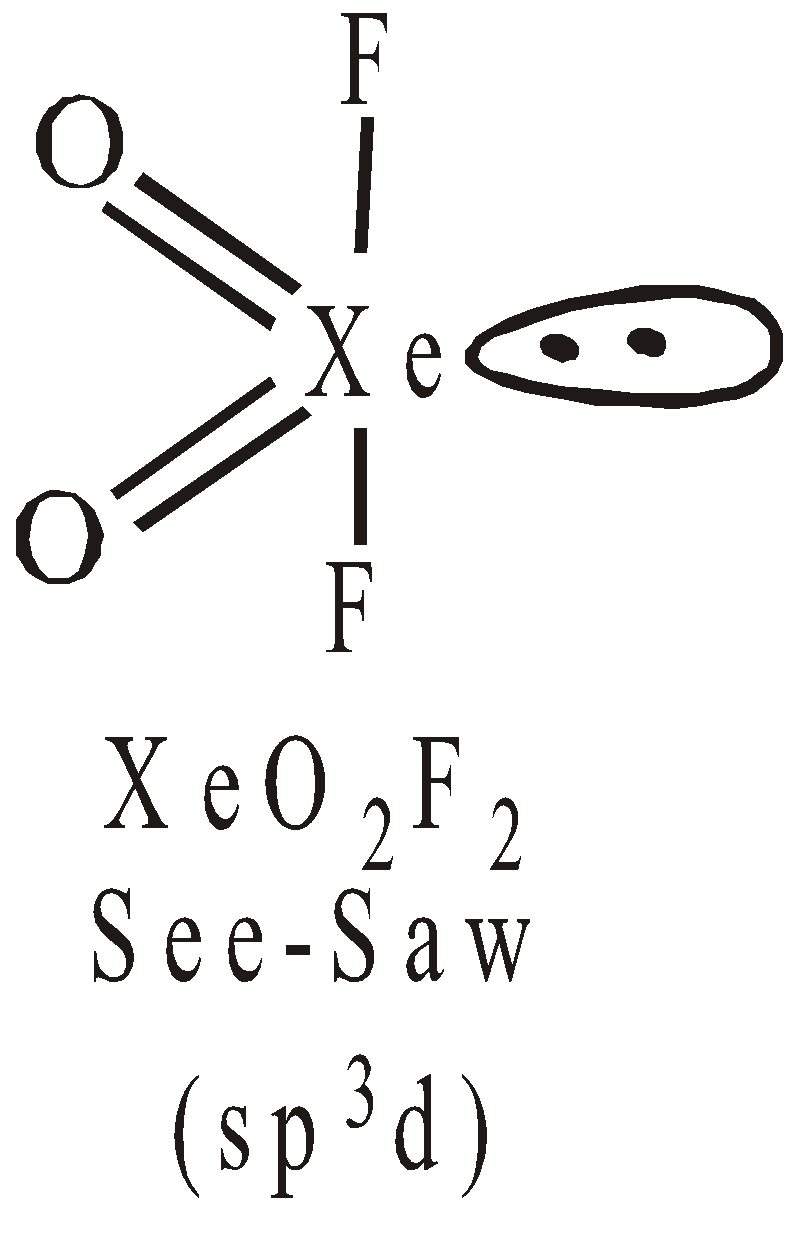
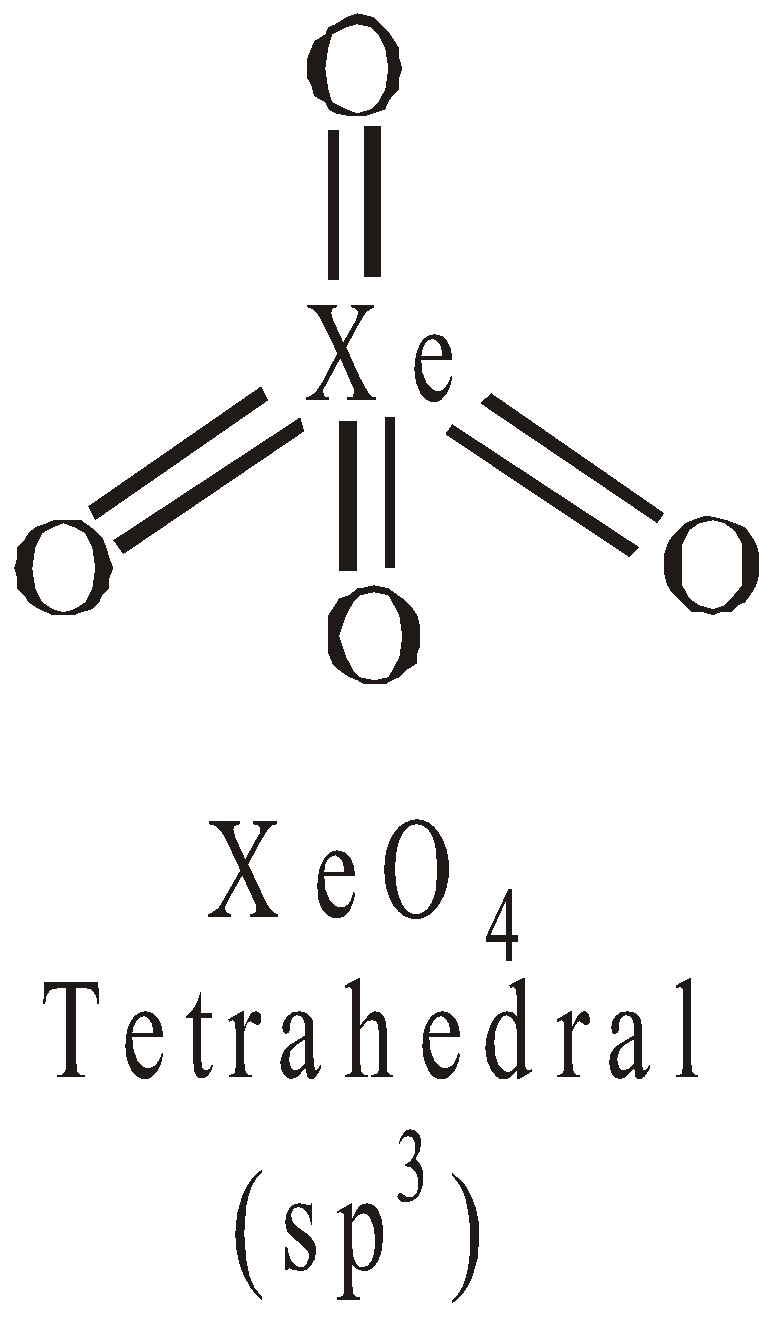
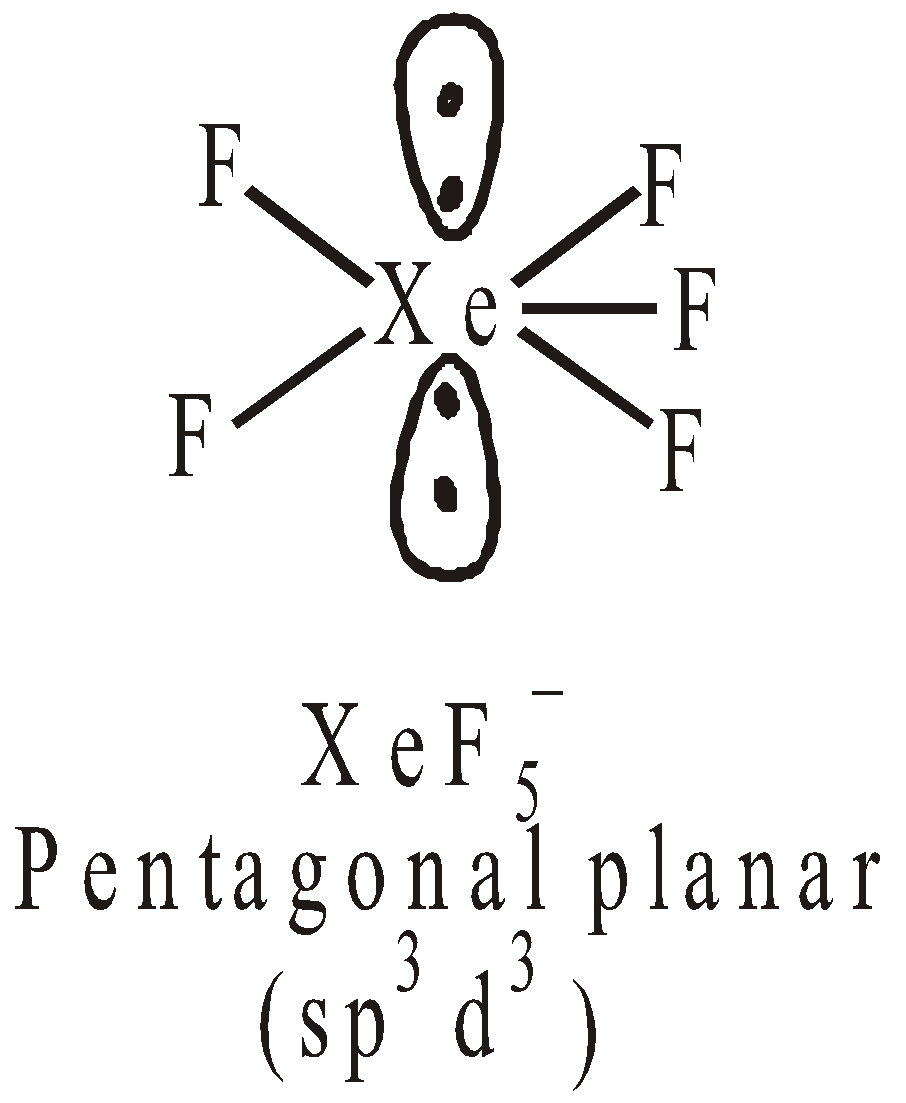
USES
- Helium – It is non-inflammable. It has low density and its lifting power is 92% that of hydrogen and used to lift weather balloons and airships. It is used as breathing mixture (or oxygen diluent) for divers. Mixture of O2 and He is used in the treatment of asthma. It is also used for inflating the tyres of aeroplanes.
- Neon – When an electric current is passed through a sample of the gas, it has a characteristic orange – red glow. Neon lighting is used for advertising. The noble gases argon (purple), Xenon (blue green) and Krypton (pale violet) are also used in “neon” lighting.
- Argon – It is used primarily to create an inert atmosphere in light bulbs, welding and fluorescent bulbs. It is also used in geiger counters, as it becomes ionised in the presence of radiation. The ratio of 40K to 40Ar can be used to date the age of rocks since argon is obtained by radioactive decay of an isotope of potassium.
- Krypton – The light emitted by Krypton in an electric discharge tube is used for runway and approach lights in airports.
- Xenon – It is used in electrical flash bulbs for high speed photography.
- Radon – In radiotherapy of cancer.
CLATHRATES
TYPES OF CLATHRATES
- Gas hydrates – Solid water having entrapped Ar, Kr, or Xe
- Quinol Clathrates – Quinol having entrapped Ar, Kr, and Xe
USES OF CLATHRATES
- Separation of noble gases – Since Ne does not form a clathrate with Quinol it is separated from Ar, Kr and Xe. The latter form a clathrate with quinol.
- Xe- 133 clathrate is a source of ɣ- radiations
- Kr – 85 clathrate is a source of β – radiations
- As an anaesthetic – Xe clathrate is used for this
- For transporting isotopes of noble gases.
