About this unit
Group 15 elements: General introduction, electronic configuration, occurrence, oxidation states, trends in physical and chemical properties; preparation and properties of ammonia and nitric acid, oxides of nitrogen (structure only); Phosphorous- allotropic forms; compounds of phosphorous: preparation and properties of phosphine, halides (PCI3 , PCI5 ) and oxoacids (elementary idea only).
Group 16 elements: General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemical properties; dioxygen: preparation, properties and uses; classification of oxides; ozone. Sulphur – allotropic forms; compounds of sulphur: preparation, preparation, properties and uses of sulphur dioxide; sulphuric acid: industrial process of manufacture, properties and uses, oxoacids of sulphur (structures only).
Group 17 elements: General introduction, electronic configuration, oxidation states, occurrence, trends in physical and chemical properties; compounds of halogens: preparation, properties and uses of chlorine and hydrochloric acid, interhalogen compounds oxoacids of halogens (structures only).
Group 18 elements: General introduction, electronic configuration, occurrence, trends in physical and chemical properties, uses.
| p-Block Elements – Nitrogen Family |
| p-Block Elements – Oxygen Family |
| p-Block Elements – Halogens |
p-BLOCK ELEMENTS – NITROGEN FAMILY
GENERAL CHARACTERISTICS
ELECTRONIC CONFIGURATION
Element | At. No. | Electronic configuration | Valence shell |
Nitrogen | 7 | [He] 2s2 2p3 | 2s2 2p3 |
Phosphorous | 15 | [Ne] 3s2, 3p3 | 3s2 3p3 |
Arsenic | 33 | [Ar] 3d10, 4s2 4p3 | 4s2 4p3 |
Antimony | 51 | [Kr] 4d10, 5s2 5p3 | 5s2 5p3 |
Bismuth | 83 | [Xe] 4f14, 5d10 6s2 6p3 | 6s2 6p3 |
METALLIC CHARACTER
PHYSICAL STATE
ATOMICITY
MELTING AND BOILING POINTS
DENSITY
ATOMIC RADII
COVALENT RADII
ALLOTROPY
OXIDATION STATE
N | P | As | Sb | Bi |
–3 to +5 | –3, +3, +4, +5 | +3, +5 | +3, +5 | +3, +5 |
Nitrogen has a wide range of oxidation states
Oxidation state | Example |
+5 | N2O5, HNO3, |
+4 | NO2, N2O4 |
+3 | HNO2, , NF3 |
+2 | NO |
0 | N2 |
–1 | NH2OH, NH2F |
–2 | N2H4 |
–3 | NH3, , |
NEGATIVE OXIDATION STATES
INERT PAIR EFFECT
IONISATION ENERGY
ELECTRONEGATIVITY
CATENATION
REACTIVITY
MULTIPLE BOND FORMATION
COMPOUNDS OF GROUP 15 ELEMENTS
HYDRIDES
- Ease of formation
- Stability
- Basic character
- Solubility
- Bond angle (NH3 107.5º ; PH3 92º, AsH3 91, SbH3 90º)
- Strength of M – H bond
- Dipole moment
- Decomposition temperature
- Reducing character
- Covalent character
- Poisonous character
- Rate of combustion
- Ammonia
- Any ammonium salt + metal oxide or hydroxide
 NH3
NH3
- Phosphine
- Other hydrides
HALIDES
when X = Cl, M can be P, As and Sb
- Ease of hydrolysis BiCl3 > SbCl3 > AsCl3 > PCl3 > NCl3
- Trihalides except BiF3 are covalent in nature
- Trihalides have pyramidal structure.
PENTAHALIDES
- The hybridisation in pentahalides is sp3d (trigonal bipyramidal)
- Thermally less stable than trihalides
- Act as Lewis acids
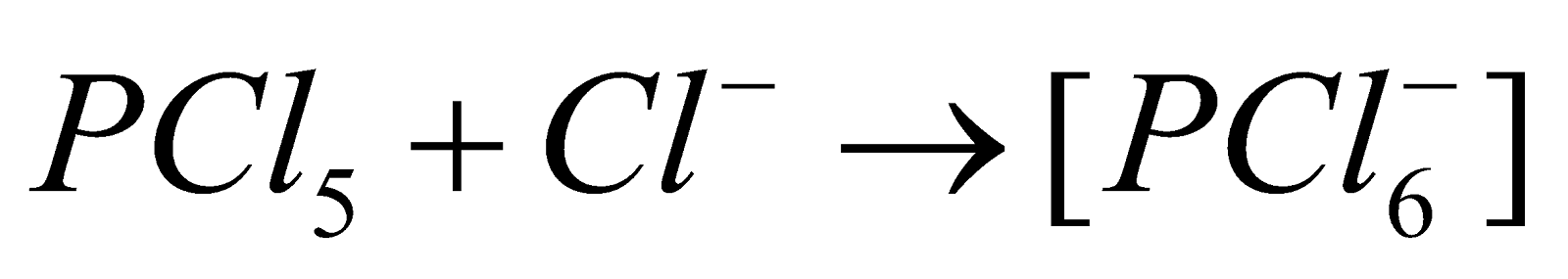
- On complete hydrolysis they produce acids
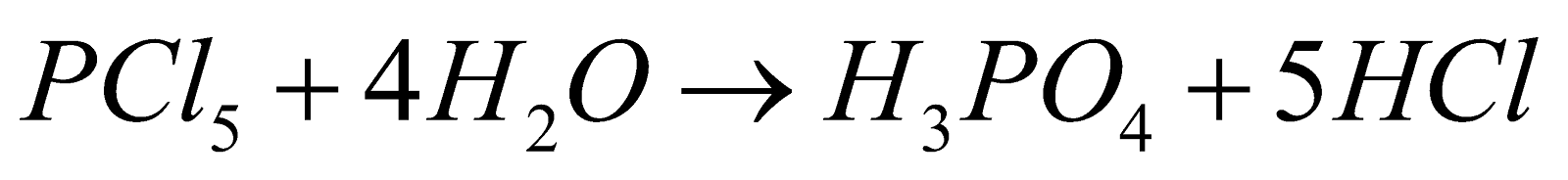
OXIDES
- Priestley’s method :
- Bertholet’s (common method) :
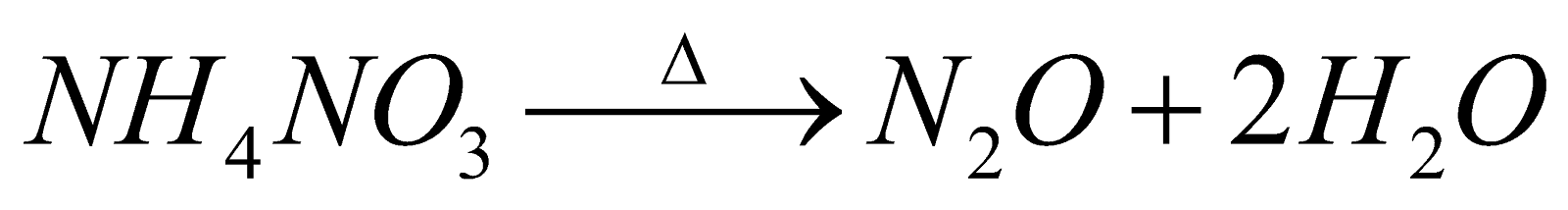

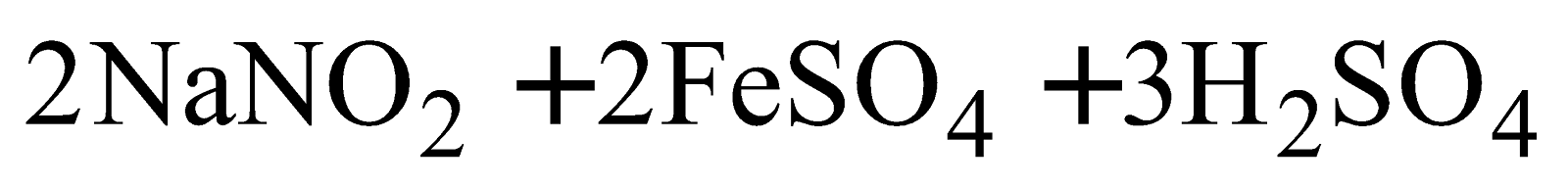
 (common method)
(common method)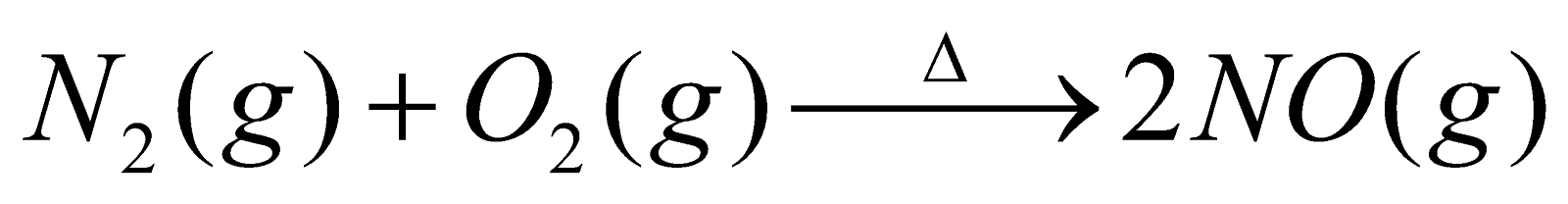 (commercial)
(commercial)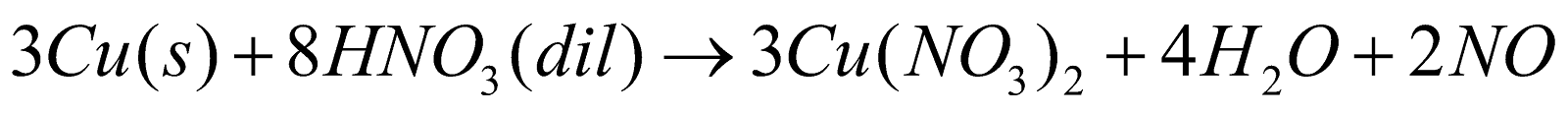 (Lab method)
(Lab method)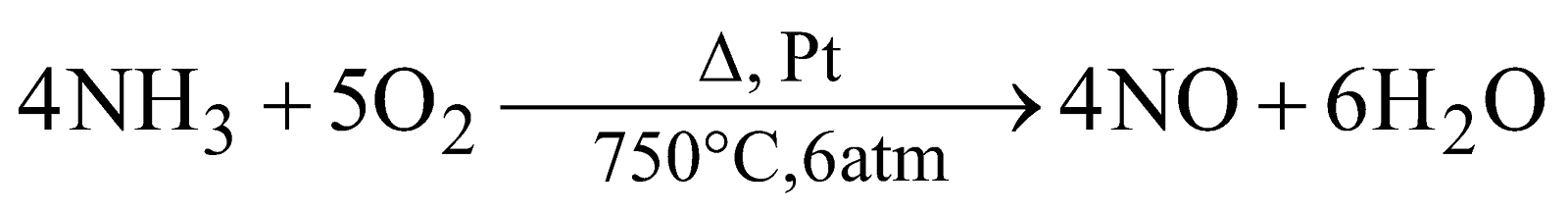 (Ostwald’s process)
(Ostwald’s process)
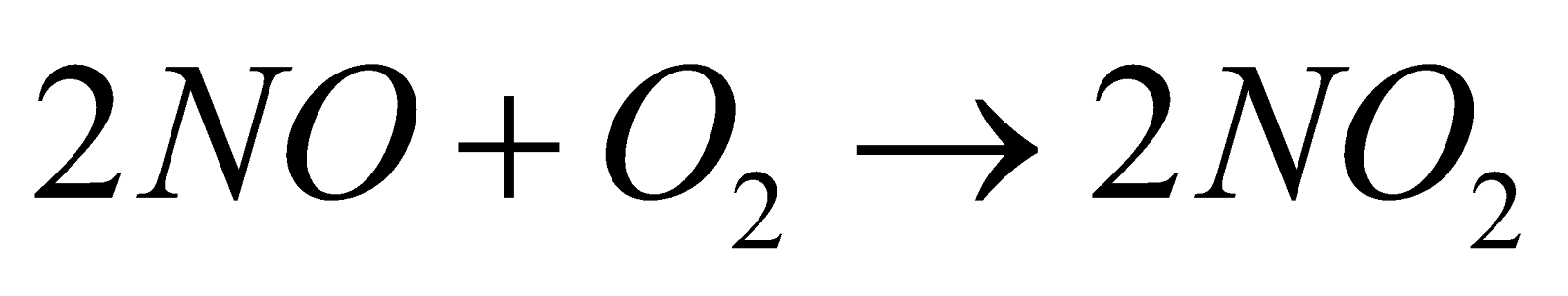
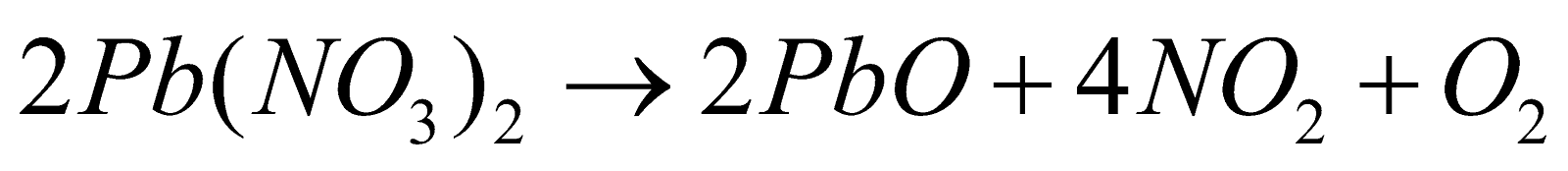 (Common method)
(Common method)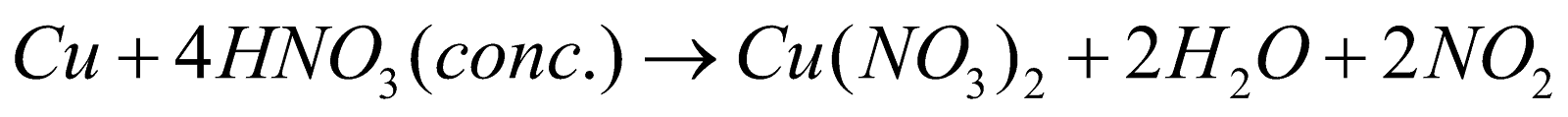
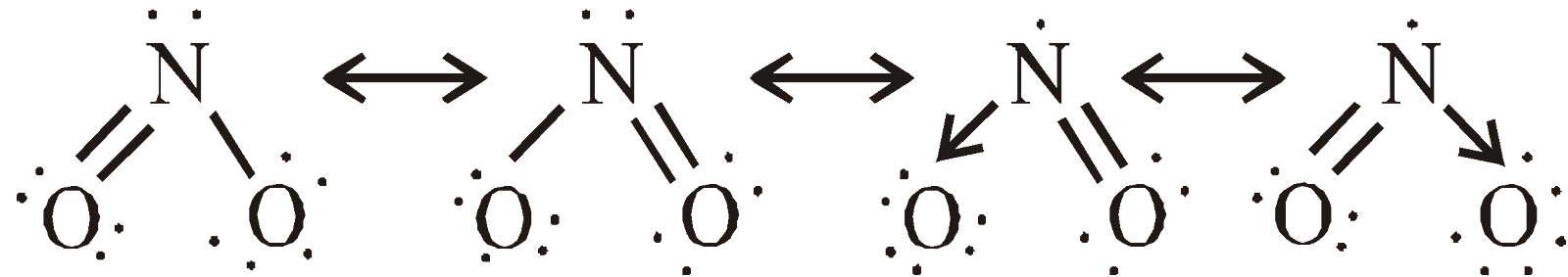
Highly toxic, paramagnetic, reddish brown gas with choking odour, acidic
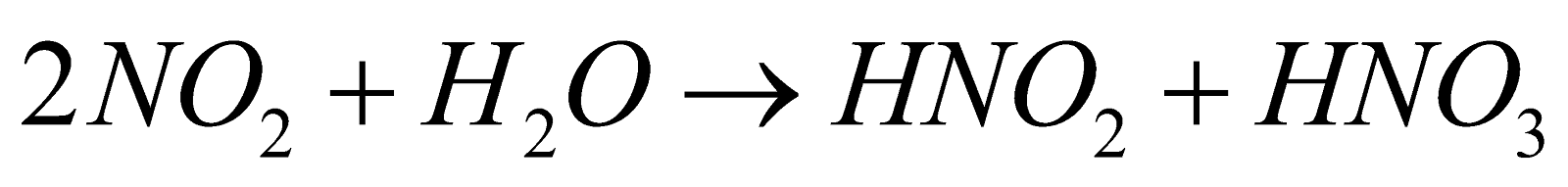 (Hence it is mixed anhydride of HNO2 and HNO3)
(Hence it is mixed anhydride of HNO2 and HNO3)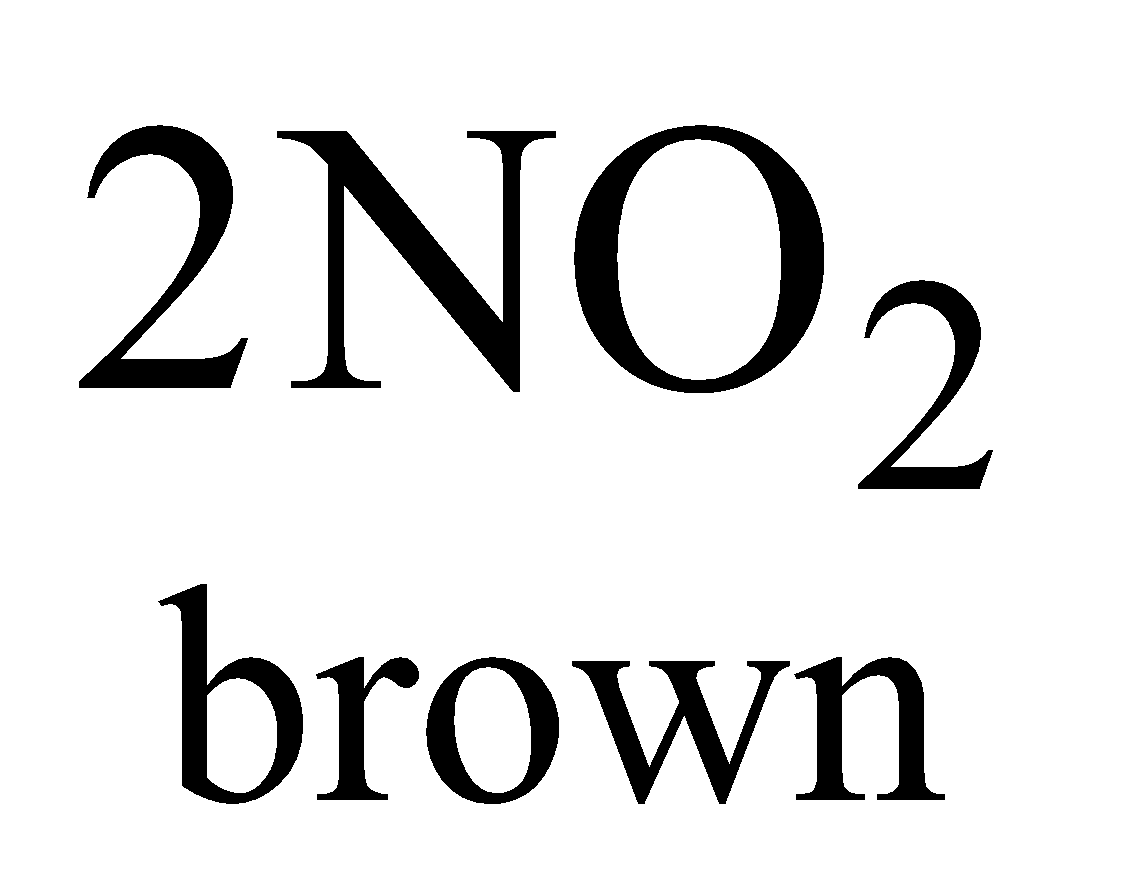
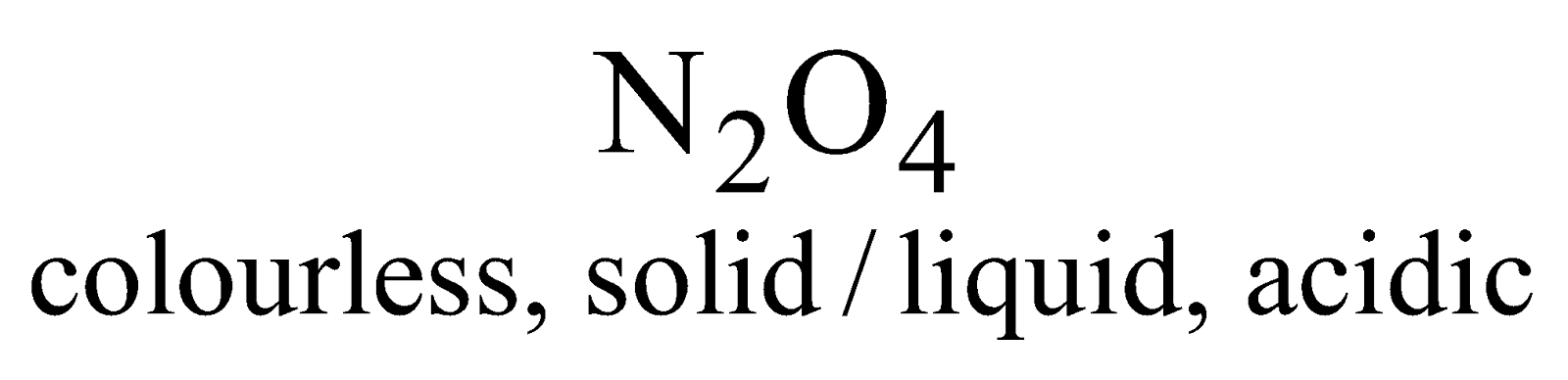
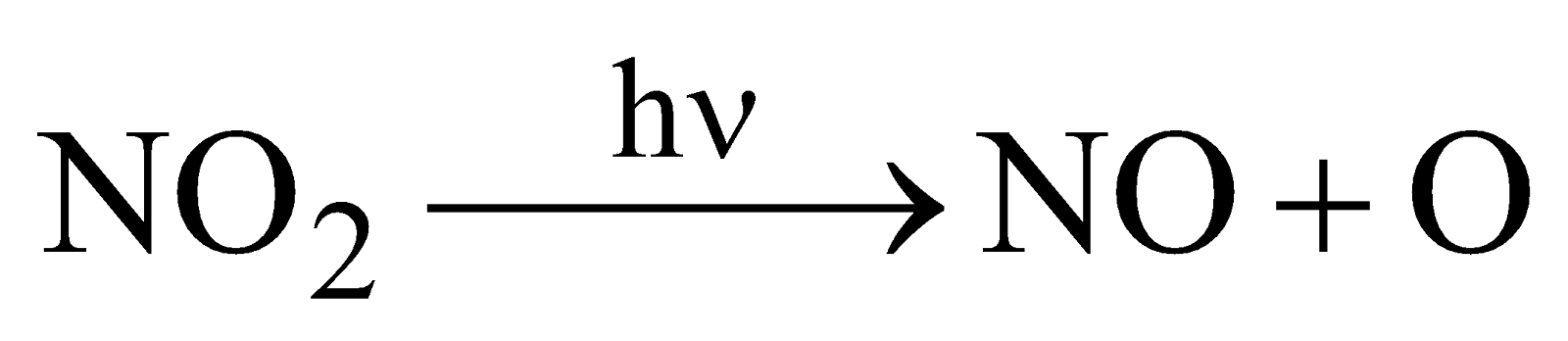
- It is combustible and supports the combustion of burning P, Mg or charcoal. Burning S or candle is extinguished.
- It is oxidising and reducing in nature
- For Manufacturing of HNO3
- As Catalyst in lead chamber process for Sulphuric acid
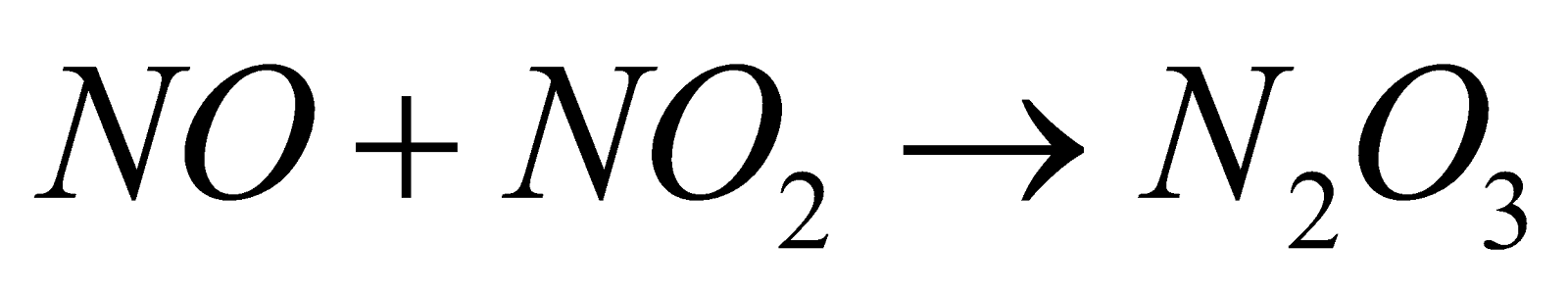
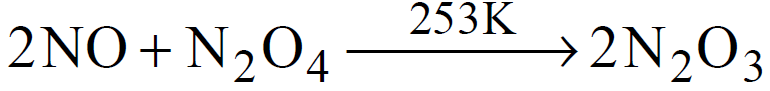 (common method)
(common method)
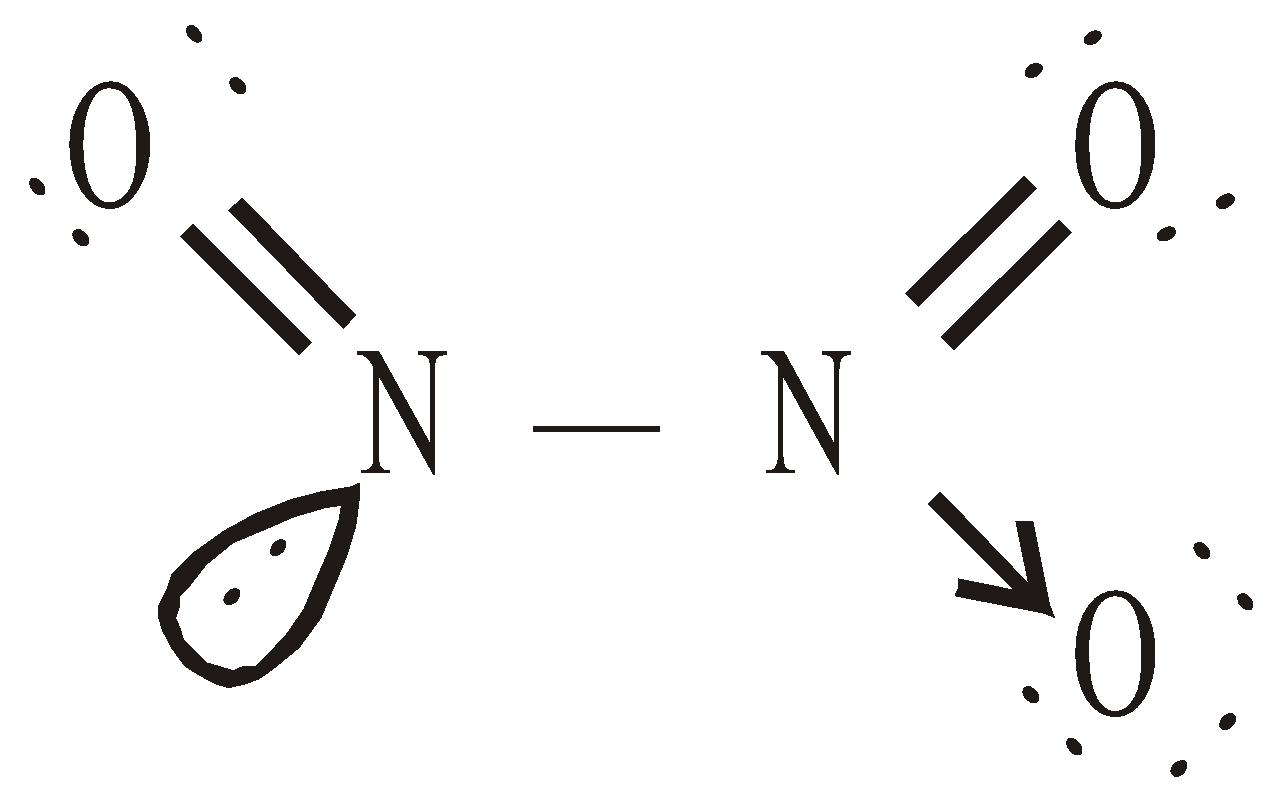
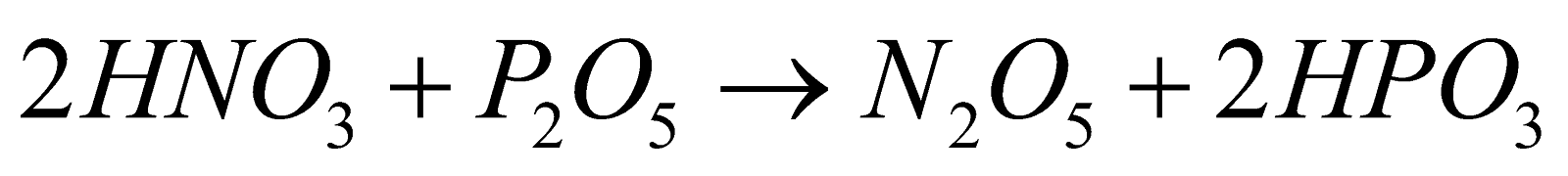 (common method)
(common method)

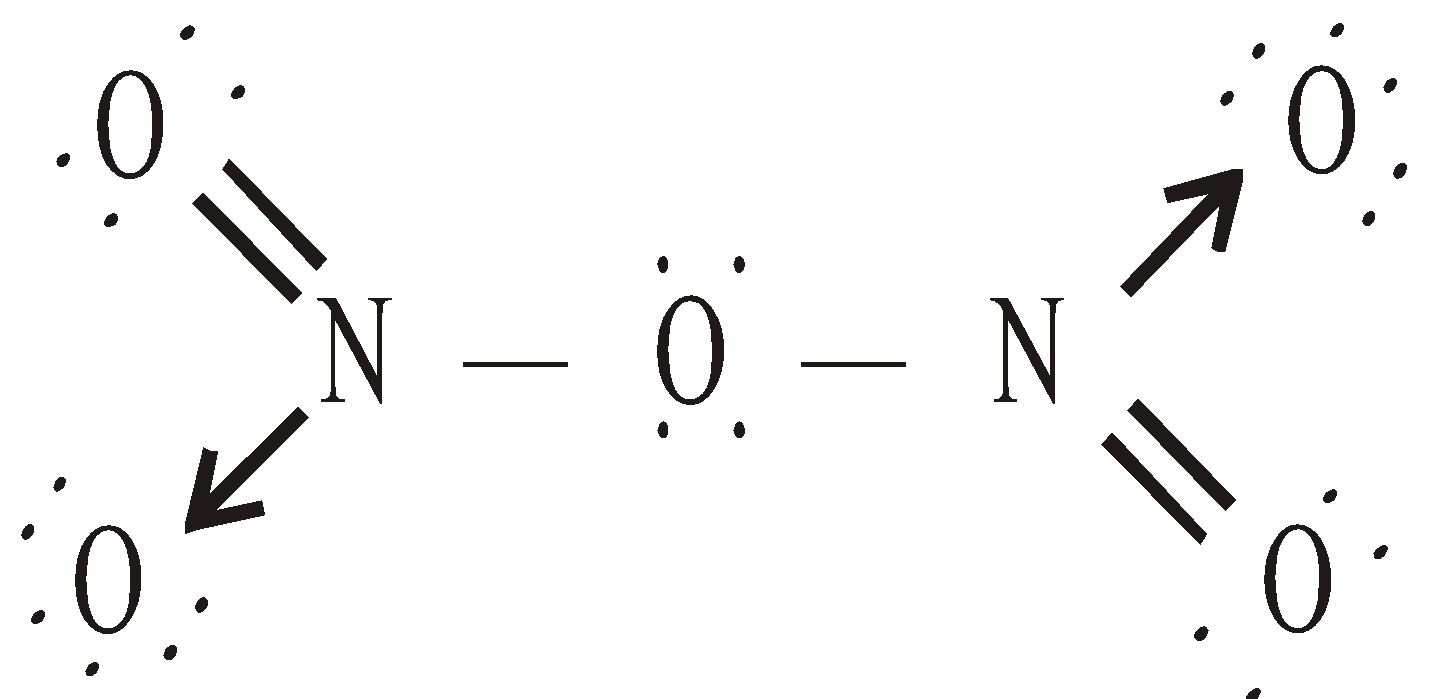
It’s ionic structure (by X-ray) is . It is also called nitronium nitrate.
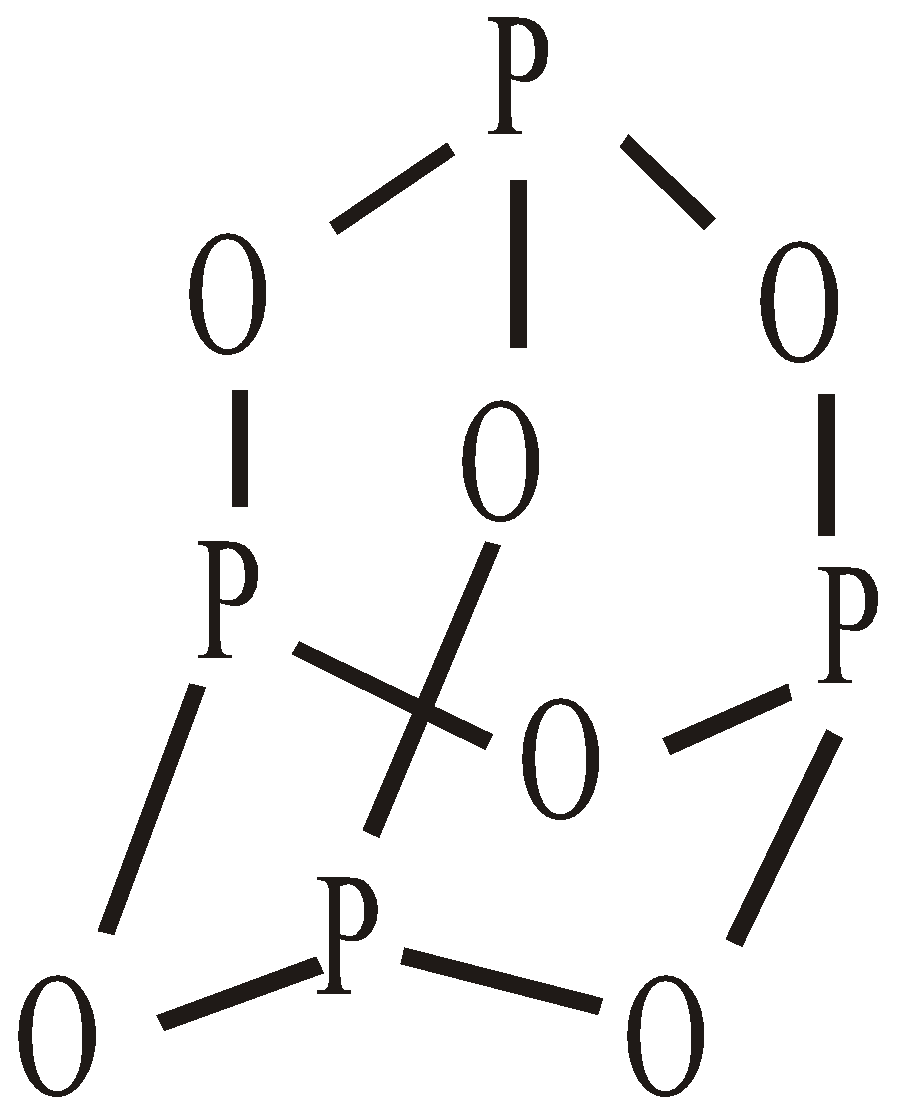
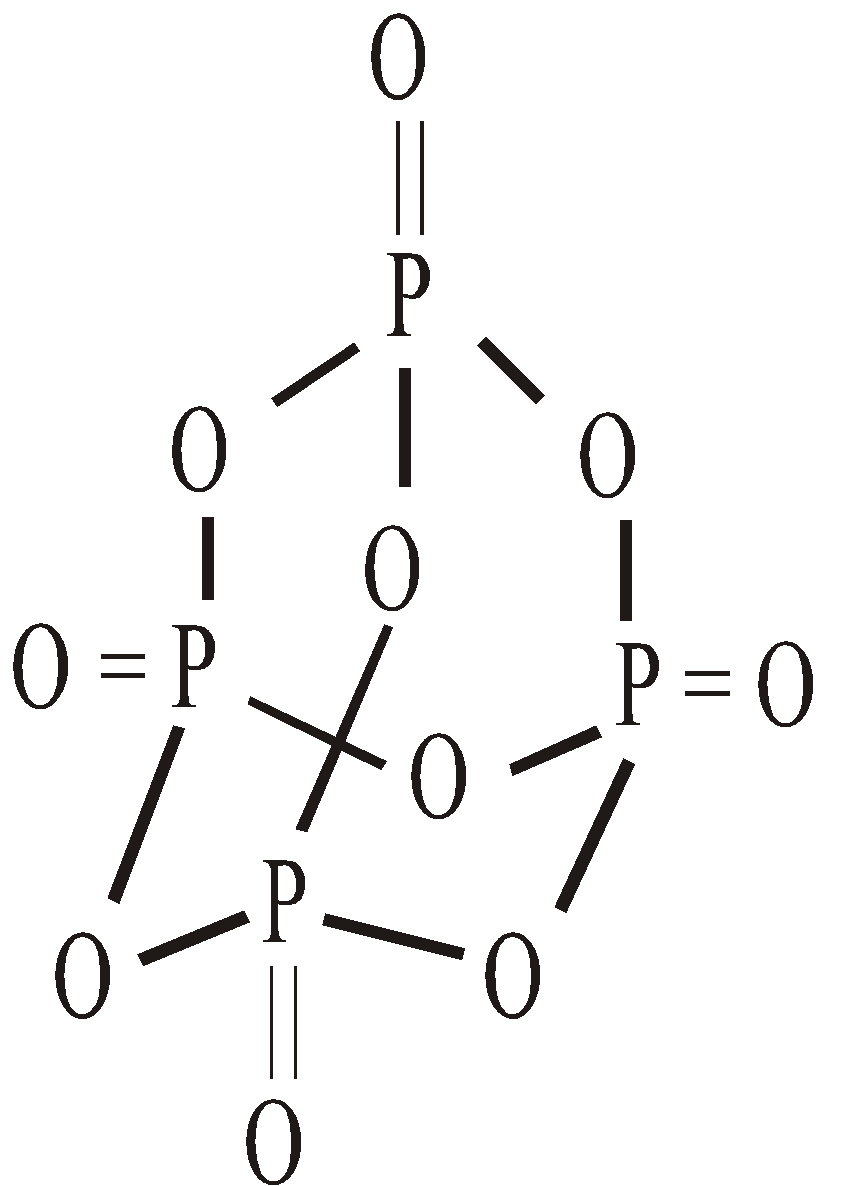
OXYACIDS OF N AND P
Oxidation number | Basicity | ||
Hyponitrous acid | H2N2O2 | +1 | |
Nitroxylic acid | H4N2O4 | +2 | |
Nitrous acid | HNO2 | +3 | |
Nitric acid | HNO3 | +5 | |
Peroxynitric acid | HNO4 | +5 | |
Hydronitrous acid | H2NO2 | +2 | |
Hypophosphorus acid | H3PO2 | +1 | 1 |
Phosphorus acid | H3PO3 | +3 | 2 |
Orthophosphoric acid | H3PO4 | +5 | 3 |
Pyrophosphoric acid | H4P2O7 | +5 | 4 |
Meta phosphoric acid | HPO3 | +5 | 1 |
Hypophosphoric acid | H4P2O6 | +4 | 4 |
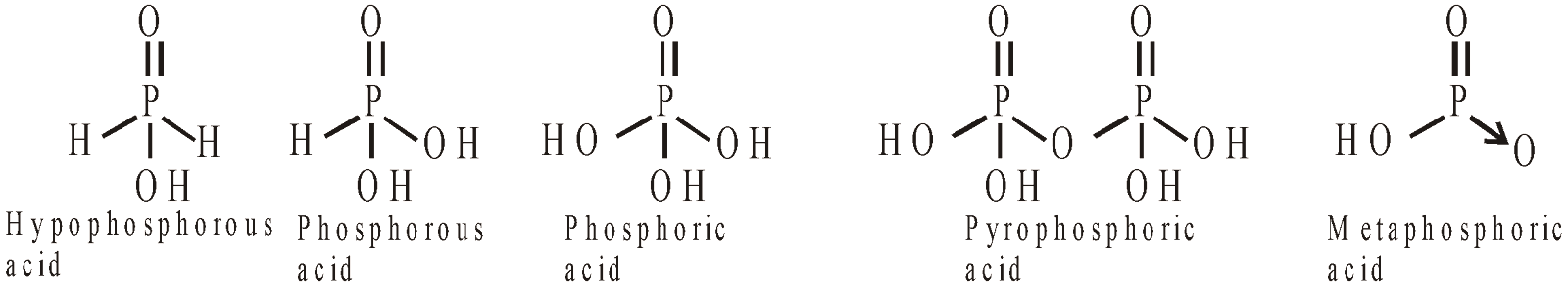
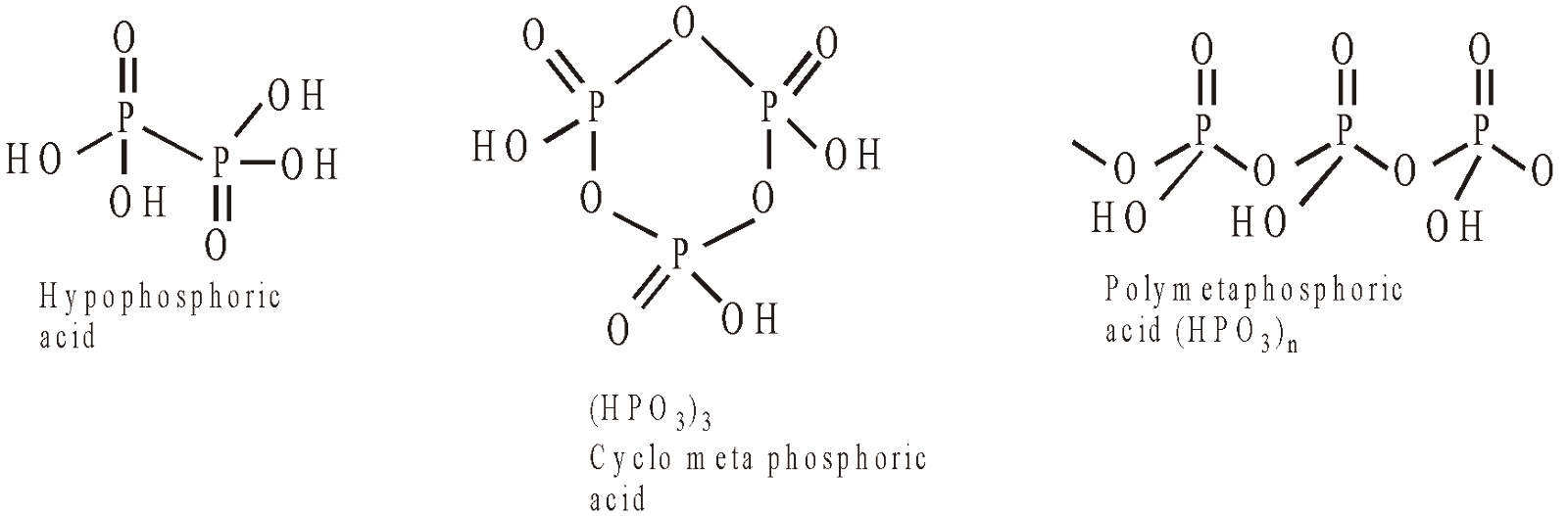
NITROGEN
PREPARATION
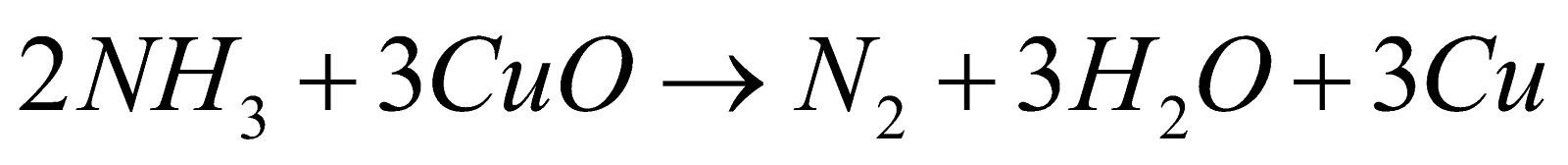
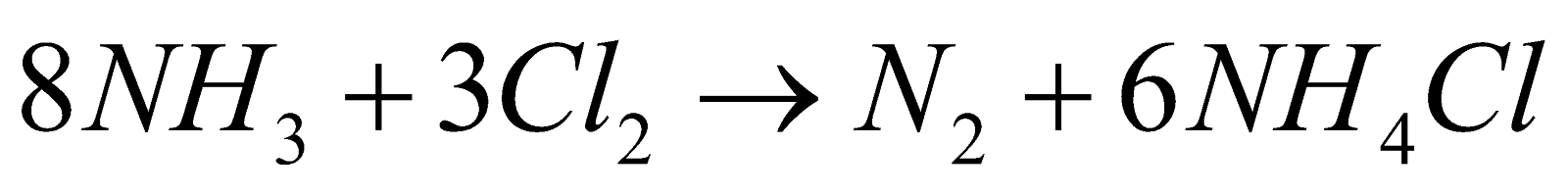
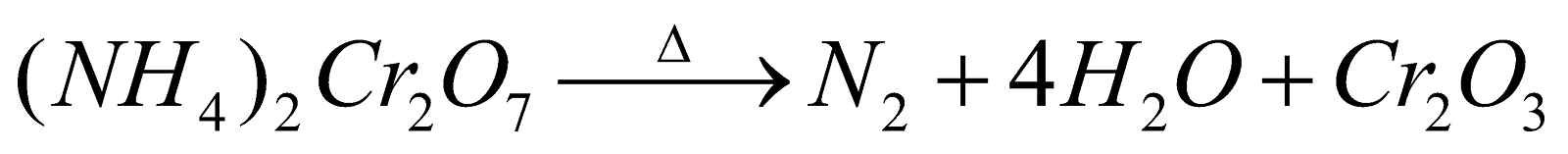 . It is violent reaction with flashes of light (volcano experiment)
. It is violent reaction with flashes of light (volcano experiment)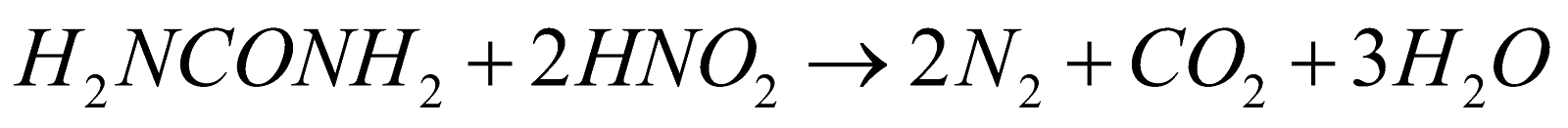
- Preparation of very pure N2 : By heating Sodium azide
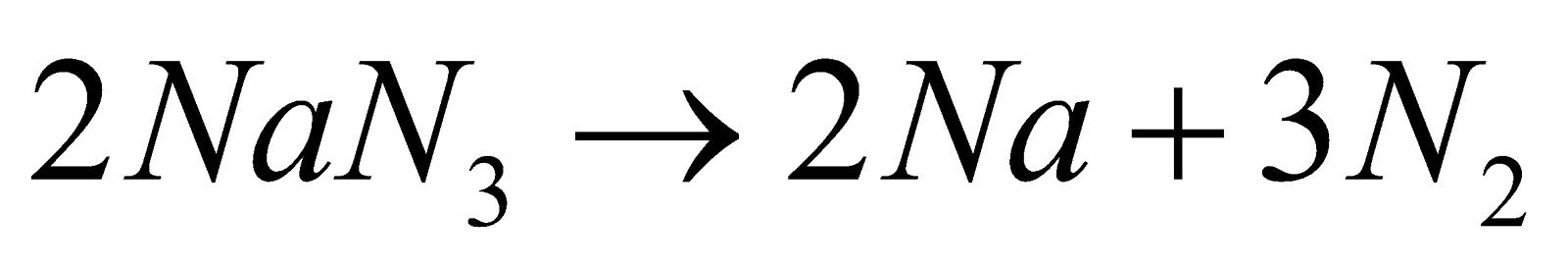
MANUFACTURING
PROPERTIES
- It is colourless, odourless, tasteless, slightly lighter than air, slightly soluble in water, non poisonous gas.
- It is incombustible and non supporter of combustion.
- It combines with metals and non metals to form number of compounds.
USES
AMMONIA (NH3)
PREPARATION
MANUFACTURING
- Haber’s process :
- Cyanamide process :
- Serpeck’s process : As by product during hydrolysis of AlN
PROPERTIES
- Colourless gas, characteristic pungent odour, brings tears into eyes, collected by downward displacement of air.
- Extremely soluble in water due to H-bonding. It is a strong lewis base.
- Ammonia is dried over any metal oxide but CaO is cheaper.
- When passed through alkaline solution of Nessler’s reagent a brown coloured complex known as Millon’s base is formed.
NITRIC ACID (HNO3)
PREPARATION
MANUFACTURING
- Birkeland – Eyde process : Air is passed through an electric arc (3000ºC) when N2 combines with O2 to form NO. It is cooled and allowed to combine with O2 to form NO2. The latter is passed in water in presence of excess of air to give HNO3.
- Ostwald’s Process : From ammonia
PHYSICAL PROPERTIES
CHEMICAL PROPERTIES
- As an acid
- As oxidising agent
ACTION OF NITRIC ACID ON METALS
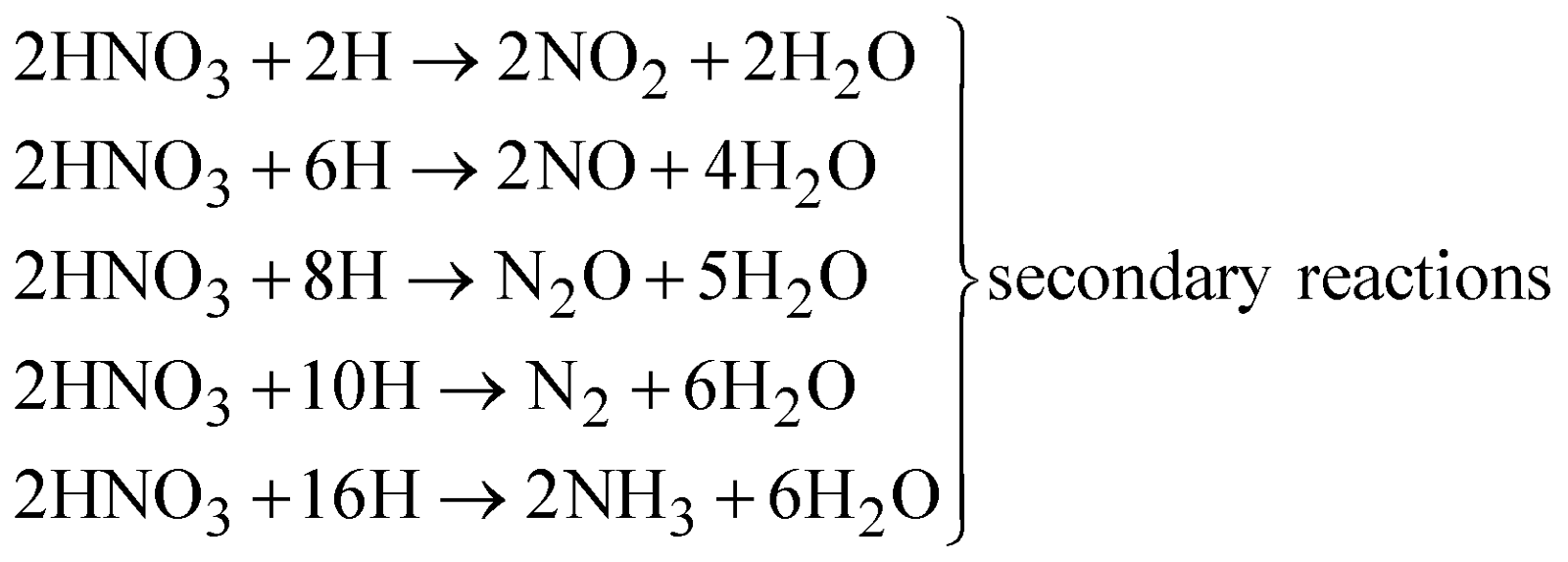
- Nature of the metal
- Concentration of the acid
- Temperature
- Presence of impurities
- Cold and very dil. acid evolves ammonia which reacts with HNO3 forming ammonium nitrate
- Cold and dil HNO3
- Cold and moderately conc.
- Cold and concentrated
- Cold and dil.
- Cold and moderately concentrated
- Cold and concentrated
- Hot and conc.
STRUCTURE OF NITRIC ACID
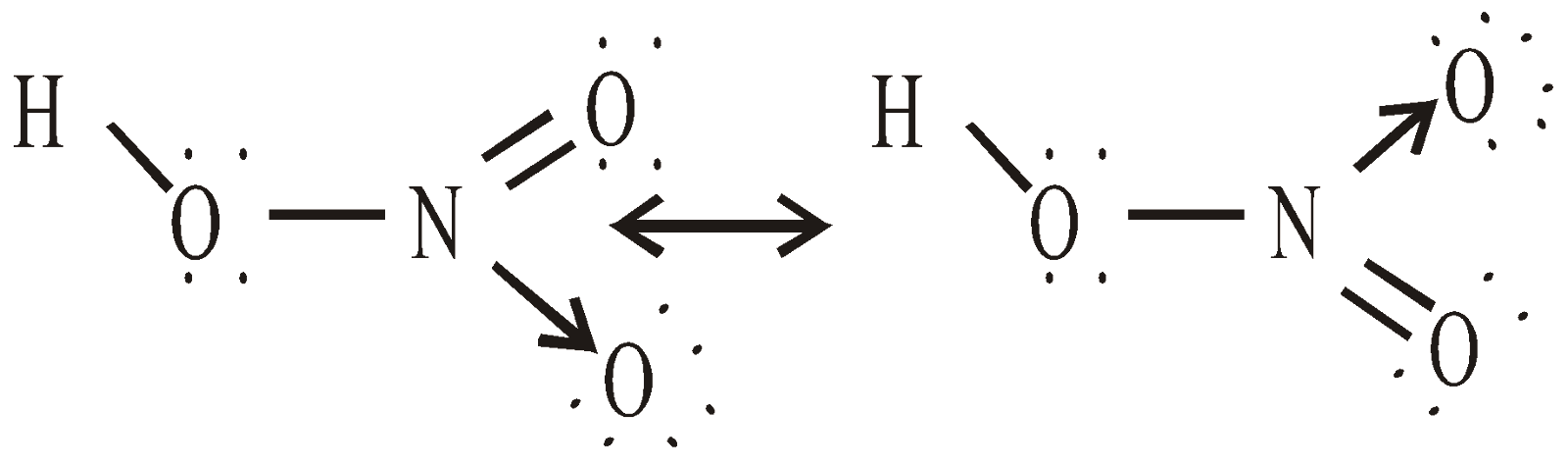
USES
NITROUS ACID (HNO2)
PREPARATION
PROPERTIES
- Decomposition
- Action of heat
- Oxidising nature
- Reducing nature
- Reaction with ammonia
- Formation of diazonium compounds
USES
STRUCTURE
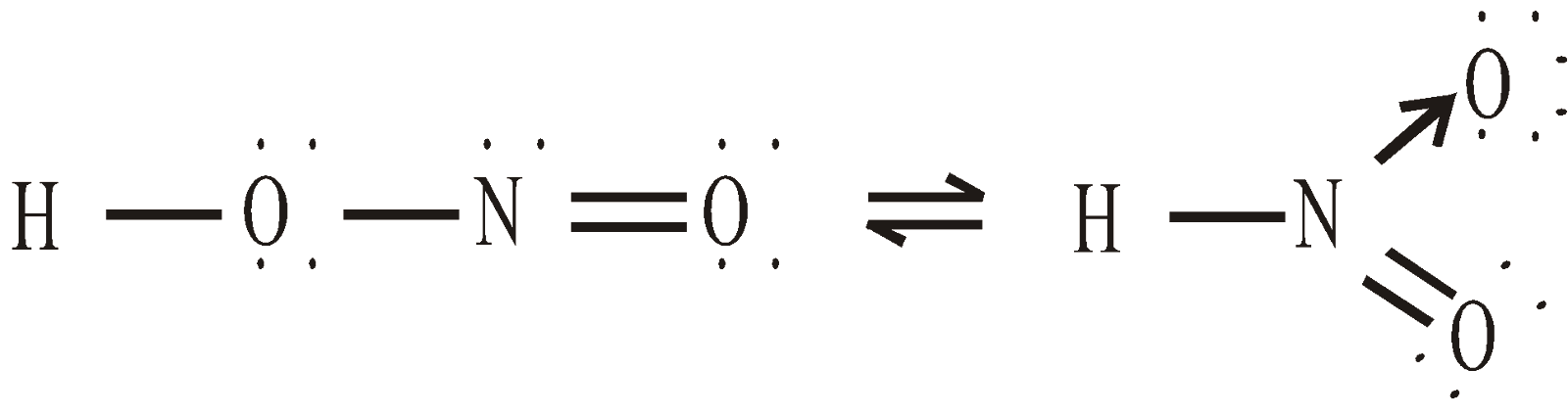
PHOSPHOROUS
OCCURRENCE
- Phosphorite Ca3(PO4)2
- Chlorapatite CaCl2.3Ca3(PO4)2
- Fluorapatite CaF2.3Ca3(PO4)2
MANUFACTURING
PROPERTIES
- Red phosphorous
By carefully heating yellow phosphorus in an inert atmosphere for about 8 days - Violet phosphorus
By crystallisation of white phosphorous from molten lead - Scarlet
- Black
By heating white P to 473K under 1000kg/sq. cm.
It is the most stable form, good conductor of electricity.
CHEMICAL PROPERTIES
- With non metals
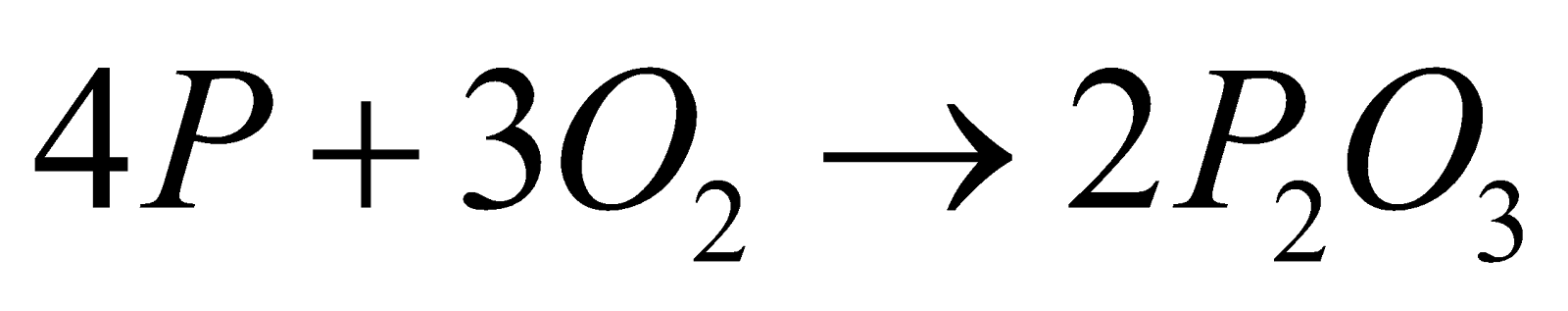 ,
,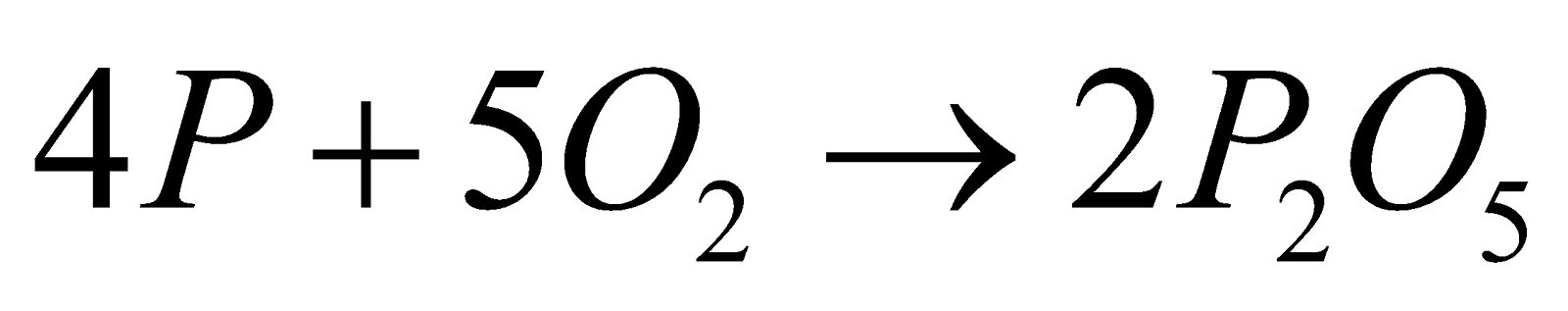 ,
,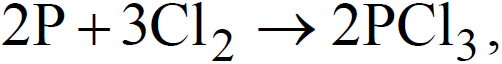
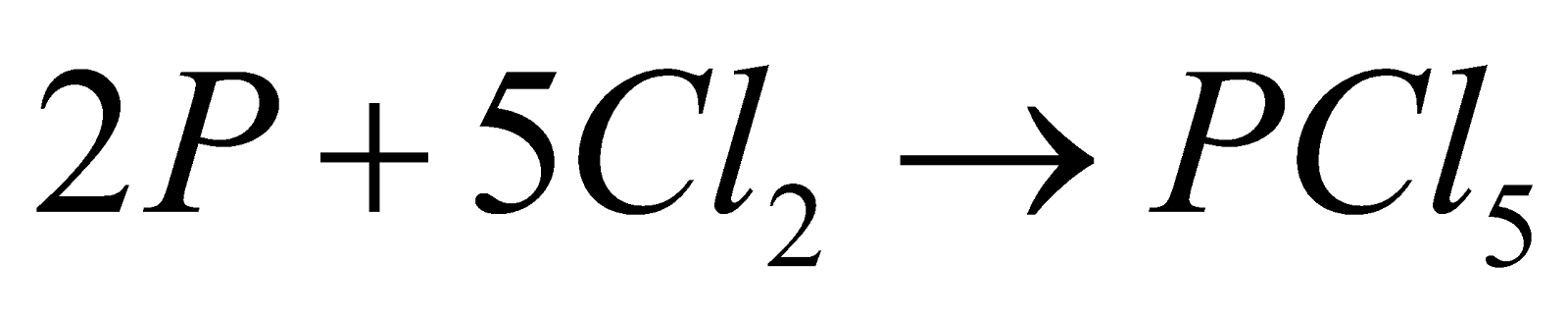
- With metals
- With compounds
USES
Side contains : Red P or P2S3 + Sand + Glue
PHOSPHINE (PH3)
PREPARATION
- Any phosphide + H2O
 PH3
PH3
- Decomposition of H3PO3 :
- Lab. method :
- Pure PH3 :
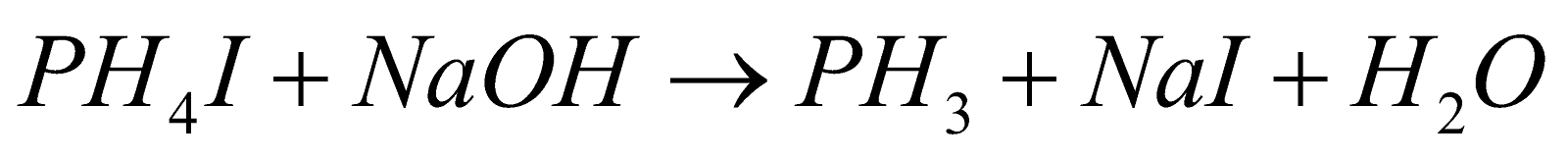
PROPERTIES
- Physical Properties : Colourless, highly poisonous, with rotten fish odour gas, slightly soluble in water
- Basic nature :
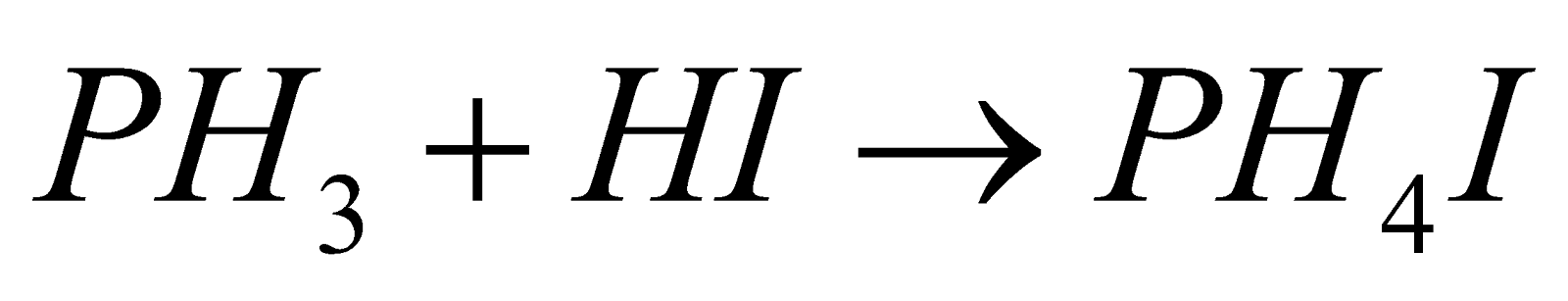
- Decomposition :
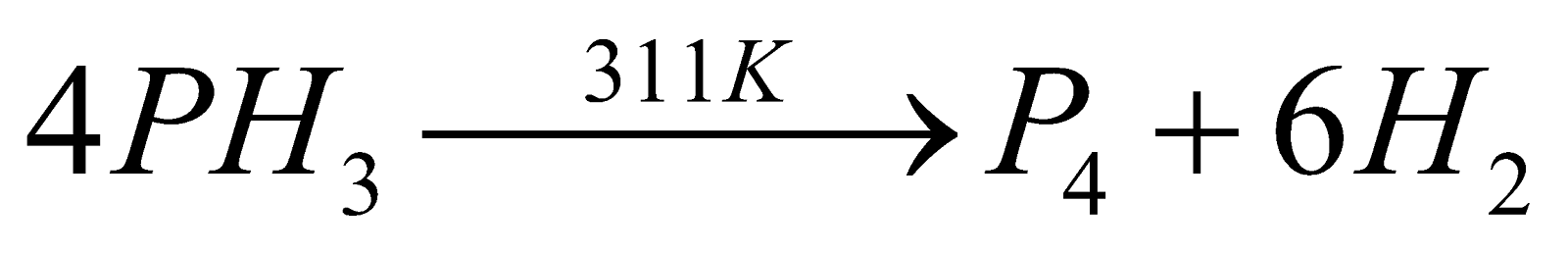
- Combustibility :
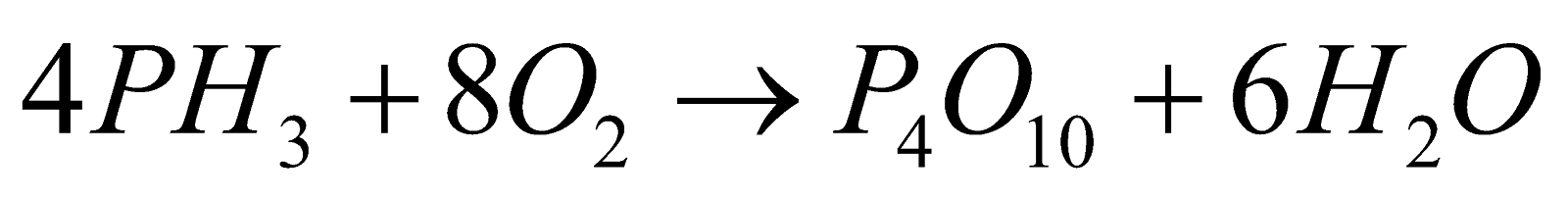
- With metallic salts :
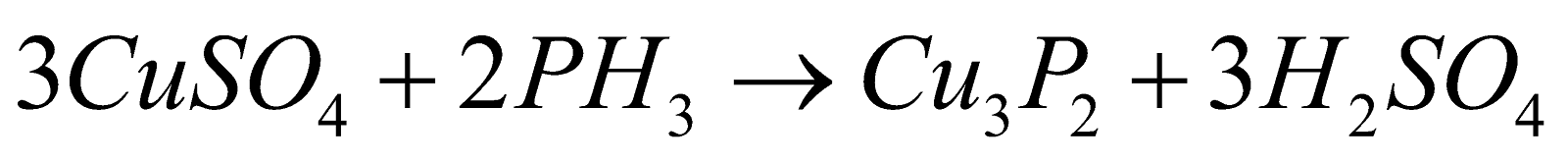
- With chlorine :
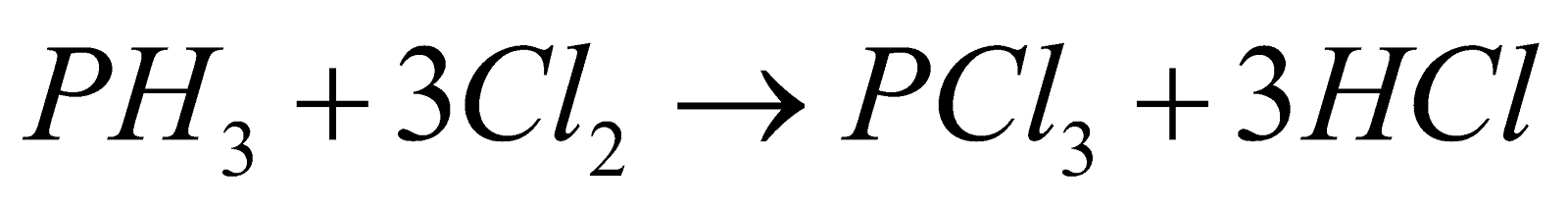
USES
- Holme’s signals : A mixture of CaC2 and Ca3P2 when treated with water, phosphine is liberated which catches fire and lights up acetylene. Burning gases serve the purpose of a signal. They are used in ships.
- Smoke screen : Ca3P2 is used smoke screen. PH3 obtained from it catches fire to give the needed smoke.
- Celphos : It is trade name of AlP, aluminium phosphide and used as fumigant. In presence of moisture it gives PH3 which kills insects and pests
- Rat poison : Zinc phosphide Zn3P2 is a rat poison, which gives PH3
ORTHOPHOSPHORIC ACID (H3PO4)
PREPARATION
PROPERTIES
- Physical Properties – Colourless syrupy liquid
- Action of heat
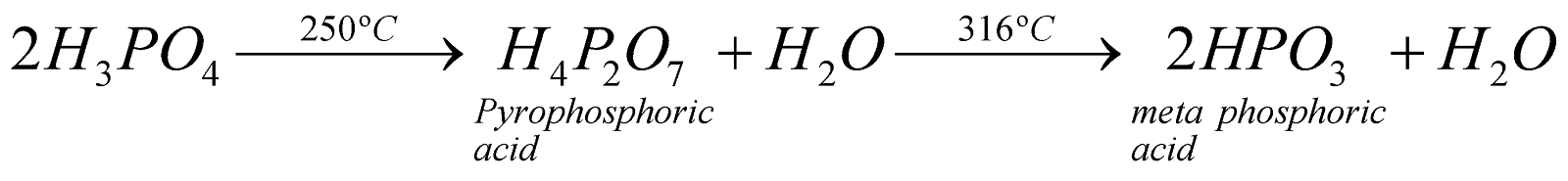
- It is tribasic and ionises in three steps
USES
- For preparation of HBr, HI in laboratory
- For preparing metaphosphoric acid
- Stabiliser for H2O2
ORTHOPHOSPHORUS ACID (H3PO3)
PREPARATION
PROPERTIES
- Physical Properties – It crystallises as deliquescent white solid
- Acidic nature :
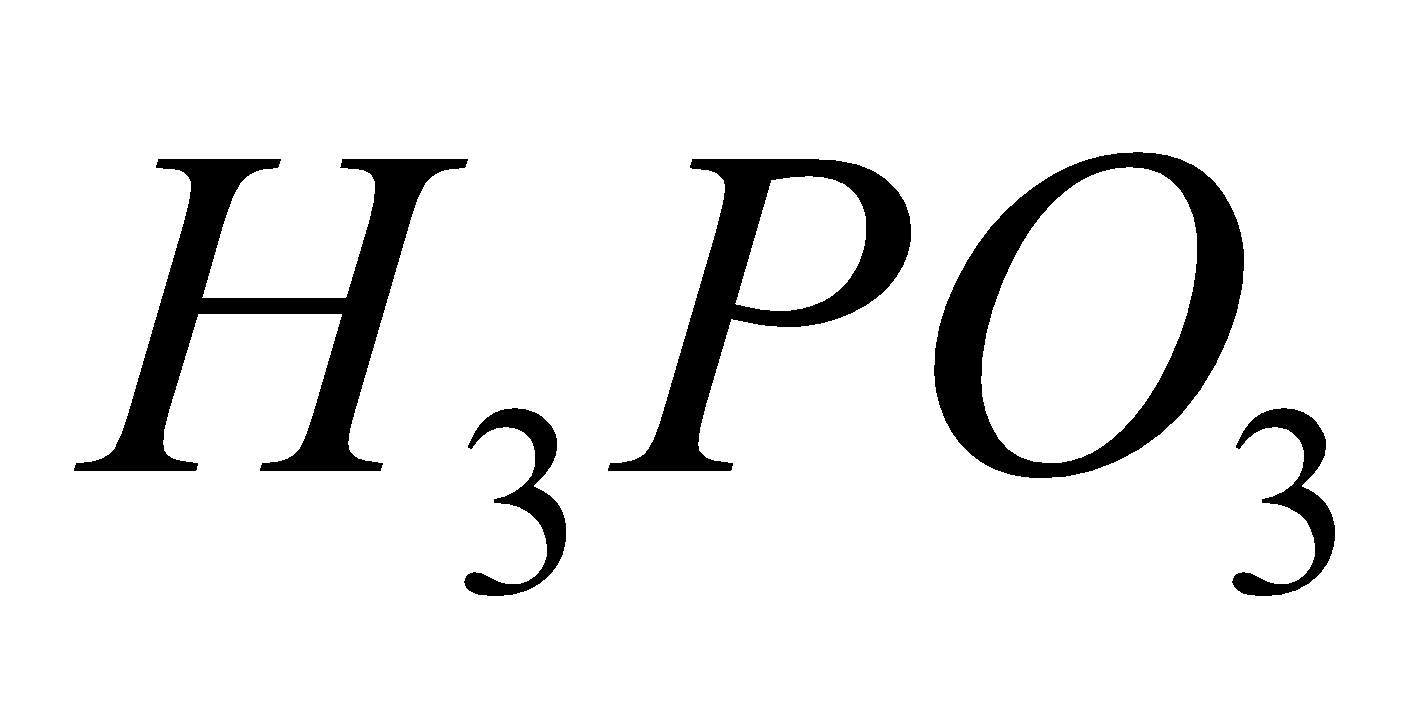 ⇌
⇌ 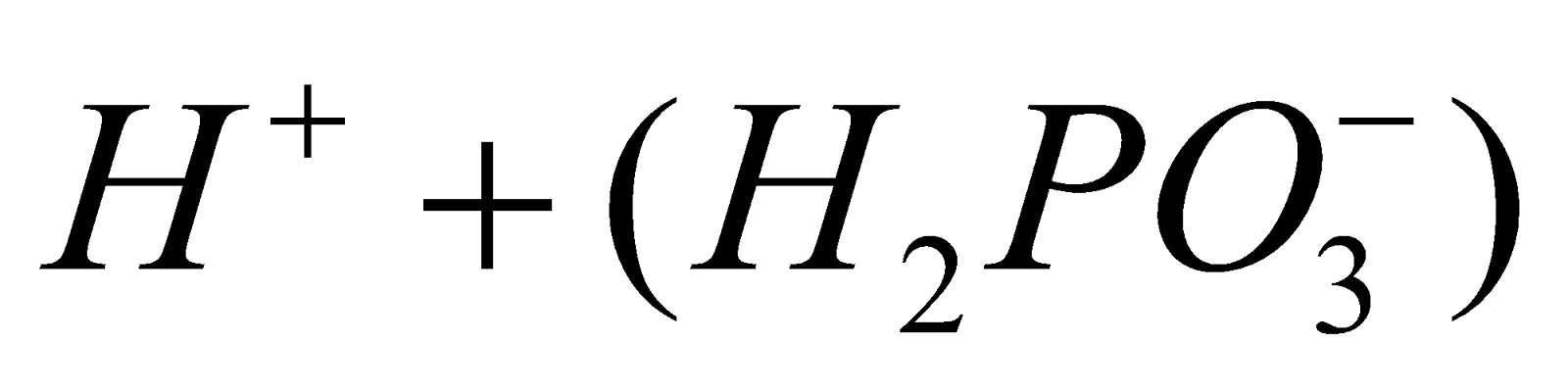 ⇌
⇌ 
- Decomposition :
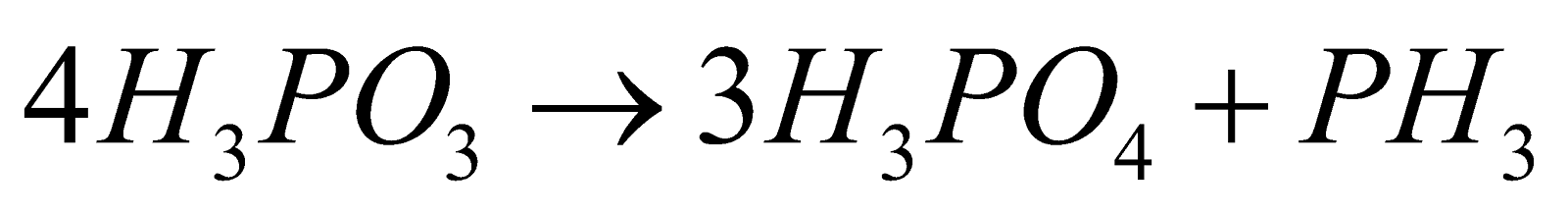 . This reaction is disproportionation
. This reaction is disproportionation - Reducing nature :
USES
FERTILIZERS
- Primary nutrients : which are consumed in large quantities eg. Nitrogen, Phosphorus and Potassium
- Secondary nutrients : Calcium and Magnesium
- Micro nutrients : which are required in minute quantities eg. Copper, Zinc, Manganese, Boron, Molybdenum, chlorine and Iron
TYPES OF FERTILIZERS
- Urea
- Ammonium sulphate (Sindri fertilizer)
- Basic Calcium nitrate
(nitrate of lime or Norwegian saltpetre)
- Calcium cyanamide
- Super phosphate of lime
- Phosphatic slag or Thomas slag
- Triple super phosphate
- Nitrophos
POTASH FERTILIZERS
- Ammoniated superphosphate
It is prepared by spraying ammoniation solution (NH4NO3 = 65.0%, NH3 = 21.7% and H2O = 13.27%) on superphosphate
