Some animals live in cold places. They sometimes huddle together as shown in Fig. 1.1.

You are going to investigate the effect of huddling on heat loss from model animals. Test-tubes filled with hot water will represent the animals.
Read all the instructions but DO NOT DO THEM until you have drawn a table for your results in the space provided in 1(a)(i).
You should use the safety equipment provided while you are doing the practical work.
Raise your hand when you are ready for hot water to be poured into the beaker that is wrapped in foil.
Step 1 Half-fill one test-tube with hot water and place it in the empty beaker.
Step 2 Put the thermometer into the test-tube containing hot water.
Step 3 Measure the temperature of the hot water in the test-tube. Record this temperature in your table in 1(a)(i).
Step 4 Leave the thermometer in the test-tube and start the stop-clock.
Step 5 After one minute, measure the temperature of the hot water in the test-tube. Record the temperature in your table in 1(a)(i).
Step 6 Measure and record the temperature of the hot water in the test-tube every minute for a total of five minutes.
Step 7 Remove the thermometer from the test-tube and carefully empty the water from the test-tube into the beaker labelled waste.
Step 8 Place three test-tubes together and keep them together with the elastic band, as shown in Fig. 1.2.
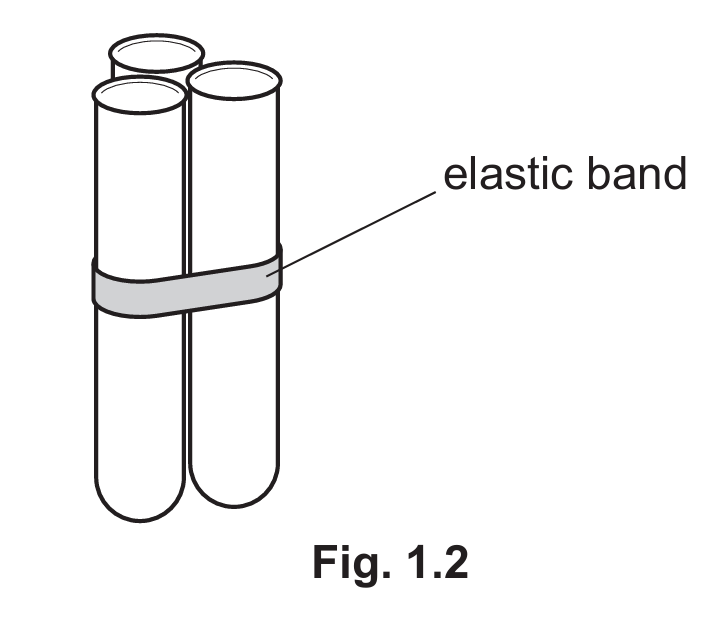
Step 9 Place the group of three test-tubes into the empty beaker. Half-fill all three test-tubes with hot water.
Step 10 Place the thermometer into any one of the three test-tubes.
Step 11 Repeat step 3 to step 7 with this group of three test-tubes.
Step 12 Remove the elastic band from the group of three test-tubes. Make a group of seven test-tubes and keep them together with the elastic band, as shown in Fig. 1.3.
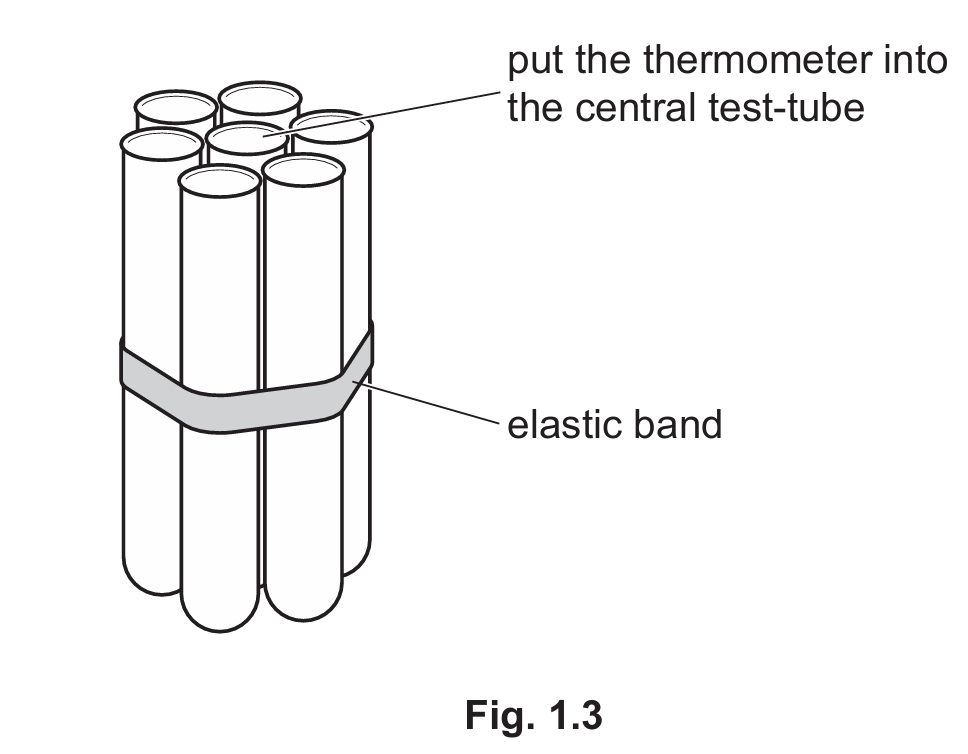
Step 13 Place the group of seven test-tubes into the empty beaker. Half-fill all seven test-tubes with hot water.
Step 14 Place the thermometer into the central test-tube.
Step 15 Repeat step 3 to step 7 with this group of seven test-tubes.
(a) (i) Prepare a table to record the results of this investigation.
(a) (ii) Plot a line graph on the grid of temperature against time. Include all three sets of data from your table in 1(a)(i) and a key.

(a) (iii) State two conclusions for this investigation.
(a) (iv) State two variables that were kept constant during this investigation.
(a) (v) Suggest two possible sources of error in this investigation.
(a) (vi) Suggest one improvement to the method that you used in this investigation.
(a) (vii) Identify one hazard for this investigation.
(b) In mammals, respiration releases heat energy to maintain an optimum temperature for enzyme activity in the body. Amylase is an enzyme that catalyses the breakdown of starch into reducing sugars. Plan an investigation to identify the temperature at which the enzyme amylase is most active.
▶️ Answer/Explanation
(a) (i)
The table should have columns for Time (minutes), Temperature for 1 test-tube (°C), Temperature for 3 test-tubes (°C), and Temperature for 7 test-tubes (°C). It should include rows for measurements at 0, 1, 2, 3, 4, and 5 minutes.
Explanation: A well-designed table is crucial for recording experimental data. It should have clear headings with units and enough rows to record all measurements. The temperature should be recorded for each configuration (1, 3, and 7 test-tubes) at each time interval.
(a) (ii)
Plot temperature on the y-axis (with units °C) and time on the x-axis (with units minutes). Use different line styles or colors for each test-tube configuration and include a key.
Explanation: The graph should clearly show how temperature changes over time for each group of test-tubes. The key helps distinguish between the different experimental conditions. The axes should be properly labeled with units.
(a) (iii)
1. Huddling reduces heat loss (more test-tubes lose heat more slowly).
2. As time increases, temperature decreases in all configurations.
Explanation: These conclusions are drawn from the expected results where grouped test-tubes would lose heat more slowly than single ones due to reduced surface area exposed to the environment.
(a) (iv)
1. Size/type of test-tubes was kept constant.
2. Total experimental time was kept constant.
Explanation: Controlling variables is essential for a fair test. Other variables that should be constant include starting temperature, volume of water, and environmental conditions.
(a) (v)
1. Volume of water might not be exactly the same in all test-tubes.
2. Starting temperature might vary between trials.
Explanation: Potential errors include measurement inaccuracies, inconsistent water volumes, or temperature fluctuations. These could affect the reliability of the results.
(a) (vi)
Repeat the investigation multiple times to calculate mean values.
Explanation: Repeating the experiment helps identify anomalies and increases the reliability of the results by reducing the impact of random errors.
(a) (vii)
Risk of burns from hot water or broken glass.
Explanation: Safety considerations are important in any experiment involving heat and glassware. Proper handling and protective equipment should be used.
(b)
Plan should include:
1. Independent variable: Different temperatures (e.g., 20°C, 30°C, 40°C, 50°C, 60°C)
2. Dependent variable: Time for starch to disappear (using iodine test) or time for reducing sugars to appear (using Benedict’s test)
3. Controlled variables: Volume/concentration of amylase and starch, pH
4. Method: Mix amylase and starch at each temperature, test samples at regular intervals
5. Safety: Use water baths for temperature control, wear goggles
Explanation: A good investigation plan identifies all key variables and describes a clear method. The temperature range should cover the expected optimum (around 37°C for human enzymes). Testing at regular intervals allows observation of the reaction rate. Safety precautions are essential when working with hot water baths.
(a) Some plants lose their leaves in the winter and grow new leaves in the spring.
A scientist measured the total leaf area of the leaves on one grapevine plant. They repeated this on 100 grapevine plants and found the mean total leaf area per plant.
This procedure was done every two months for one year. The results are shown in Fig. 2.1.
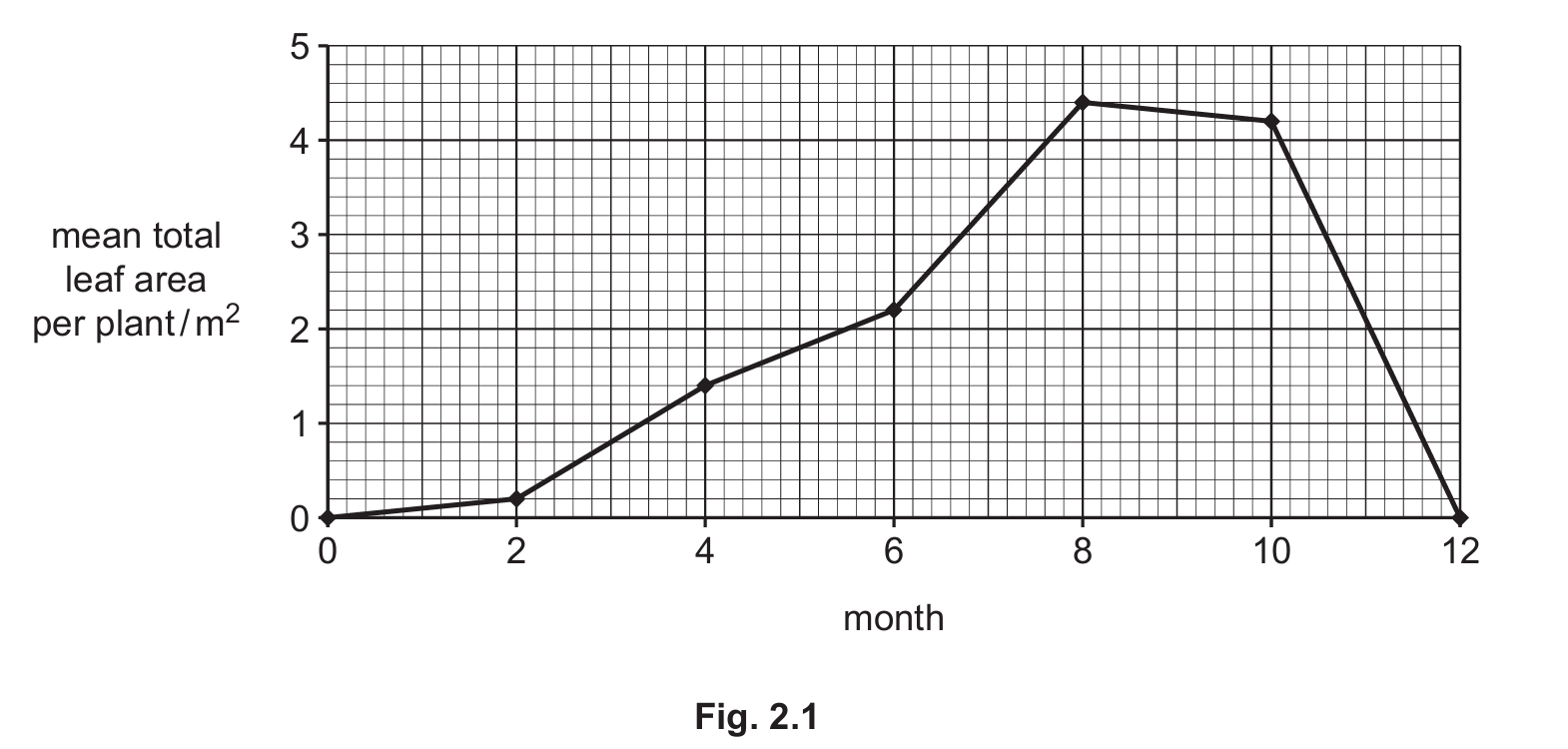
(i) Suggest why a large number of plants were sampled.
(ii) Estimate the mean total leaf area per plant for month 7. Show on Fig. 2.1 how you estimated this value.
(iii) Using the information in Fig. 2.1, calculate the percentage increase in the mean total leaf area per plant from month 4 to month 6. Give your answer to two significant figures.
(b) Fig. 2.2 shows a leaf from a grapevine plant.

Use the grid to determine the area of the grapevine leaf shown in Fig. 2.2 by counting the squares containing the leaf. Only count squares that are more than half-filled by the leaf. Include the unit.
(c) Grapevines produce fruits called grapes. A large leaf area is important when growing grapes because the leaves supply reducing sugars to the grapes.
Describe the method for testing for reducing sugars. Include the result for a positive test.
(d) Fig. 2.3 is a photograph of a leaf from a fig plant.

Make a large drawing of the leaf shown in Fig. 2.3.
(e) State one similarity and one difference between the grapevine leaf in Fig. 2.2 and the fig leaf in Fig. 2.3.
▶️ Answer/Explanation
(a)(i) To get a representative sample / to avoid bias / to identify or exclude anomalies / to increase confidence in results.
Explanation: Sampling a large number of plants (100 in this case) helps ensure the data is representative of the entire population. This reduces the impact of individual variations or outliers and increases the reliability of the results. It’s a fundamental principle in biological studies to use adequate sample sizes for meaningful conclusions.
(a)(ii) 3.3 m² (accept values between 3.2-3.4 m²).
Explanation: To estimate the value for month 7, you would locate month 7 on the x-axis, follow it up to the curve, then read across to the y-axis. The graph shows the value is approximately halfway between 3.0 and 3.5 m², hence 3.3 m² is a reasonable estimate.
(a)(iii) 57%
Explanation: From the graph, month 4 shows ~1.4 m² and month 6 shows ~2.2 m². The increase is 0.8 m². Percentage increase = (Increase/Original) × 100 = (0.8/1.4) × 100 = 57.14%, which rounds to 57% to two significant figures.
(b) 59 cm² (accept 55-66 cm²)
Explanation: To calculate the area, count all grid squares that are more than half-covered by the leaf. Each square represents 1 cm². The exact count may vary slightly depending on interpretation, but should fall within the 55-66 cm² range.
(c) Method: Add Benedict’s reagent to the sample and heat it. Positive test: Color change to green/yellow/orange/red/brown.
Explanation: Benedict’s test is used to detect reducing sugars. When heated with Benedict’s solution, reducing sugars will cause a color change from blue through green, yellow, orange to brick red, depending on the concentration of reducing sugars present. The precipitate formed is copper(I) oxide.
(d) The drawing should show:
- A clear outline of the entire leaf (single unbroken line)
- Size at least 65mm wide
- Five distinct lobes
- One main vein visible in each lobe
Explanation: Biological drawings should be large, clear and show all relevant features. The fig leaf has five distinct lobes with visible venation. The drawing should be proportional and show these key features accurately without shading or coloring.
(e) Similarity: Both leaves are lobed/have veins/have uneven edges. Difference: Grapevine leaf has serrated edges while fig leaf has smooth edges / Grapevine leaf lacks a petiole while fig leaf has one.
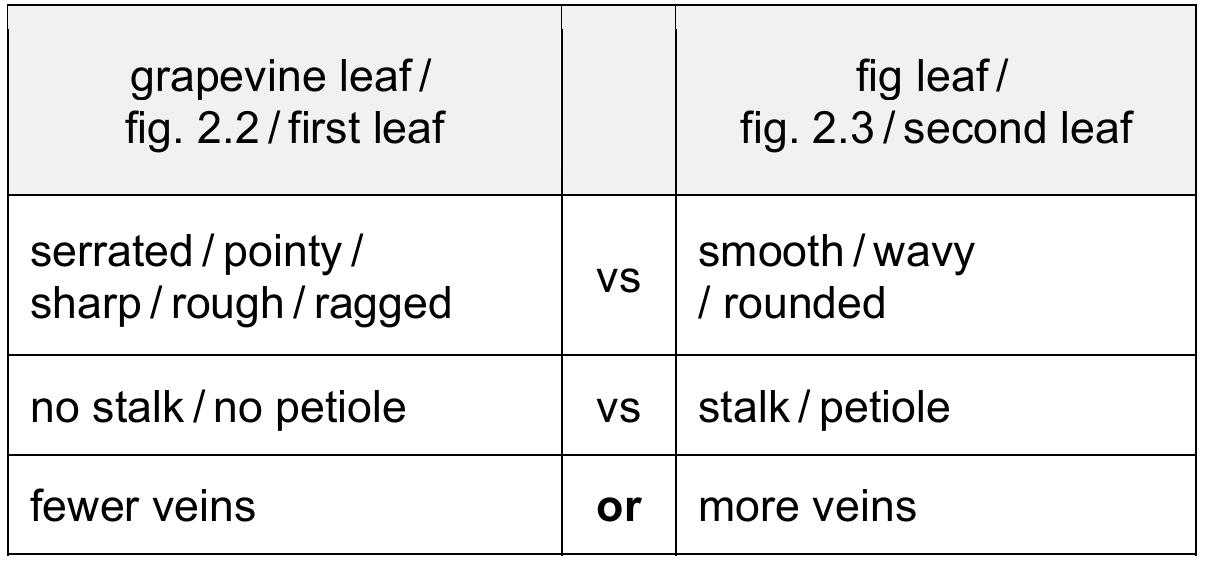
Explanation: Both leaves show the common dicotyledonous feature of being lobed with visible venation. The main differences are in the edge texture (grapevine leaves are famously serrated while fig leaves are smooth) and the presence of a petiole (leaf stalk) which is clearly visible in the fig leaf but not in the grapevine leaf shown.
