Students investigated the effect of temperature on the activity of amylase.
Amylase is an enzyme that catalyses the breakdown of starch to form reducing sugars.
The students used this method:
- Use a pen to label a spotting tile as shown in Fig. 1.1.
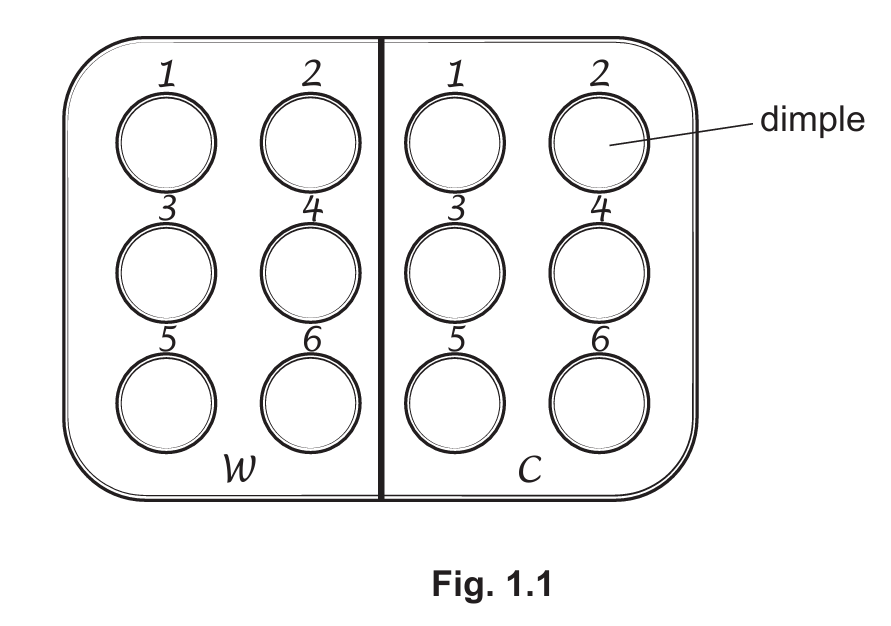
- Put one drop of iodine solution into each of the dimples on the spotting tile.
- Put 2 cm³ of 2% amylase solution into two test-tubes.
- Label a third test-tube W and a fourth test-tube C.
- Put 2 cm³ of starch suspension into test-tube W and into test-tube C.
- Label a beaker W and put approximately 200 cm³ of warm water into beaker W.
- Put test-tube W and one of the test-tubes containing 2 cm³ of 2% amylase solution into beaker W. Leave both test-tubes in beaker W for three minutes.
- After three minutes, pour the 2% amylase solution into test-tube W and start the stop-clock.
- Wait for 30 seconds and then use a pipette to remove a sample of the liquid from test-tube W. Put two drops of this sample into the dimple labelled W1 on the spotting tile. Record the colour of the liquid in dimple W1.
- Repeat step 9 using dimple W2. Continue taking and testing samples of liquid from test-tube W at 30-second intervals, using the remaining dimples W3, W4, W5 and W6. Record the colour of the liquid in each dimple. Stop the stop-clock and reset it to zero.
- Label a beaker C and put approximately 200 cm³ of cold water into beaker C.
- Put test-tube C and the other test-tube containing 2 cm³ of 2% amylase solution into beaker C. Leave both test-tubes in beaker C for three minutes.
- After three minutes, pour the 2% amylase solution into test-tube C and start the stop-clock.
- Repeat step 9 and step 10 with test-tube C and the dimples labelled C1, C2, C3, C4, C5 and C6.
The results of the investigation are shown in Fig. 1.2.
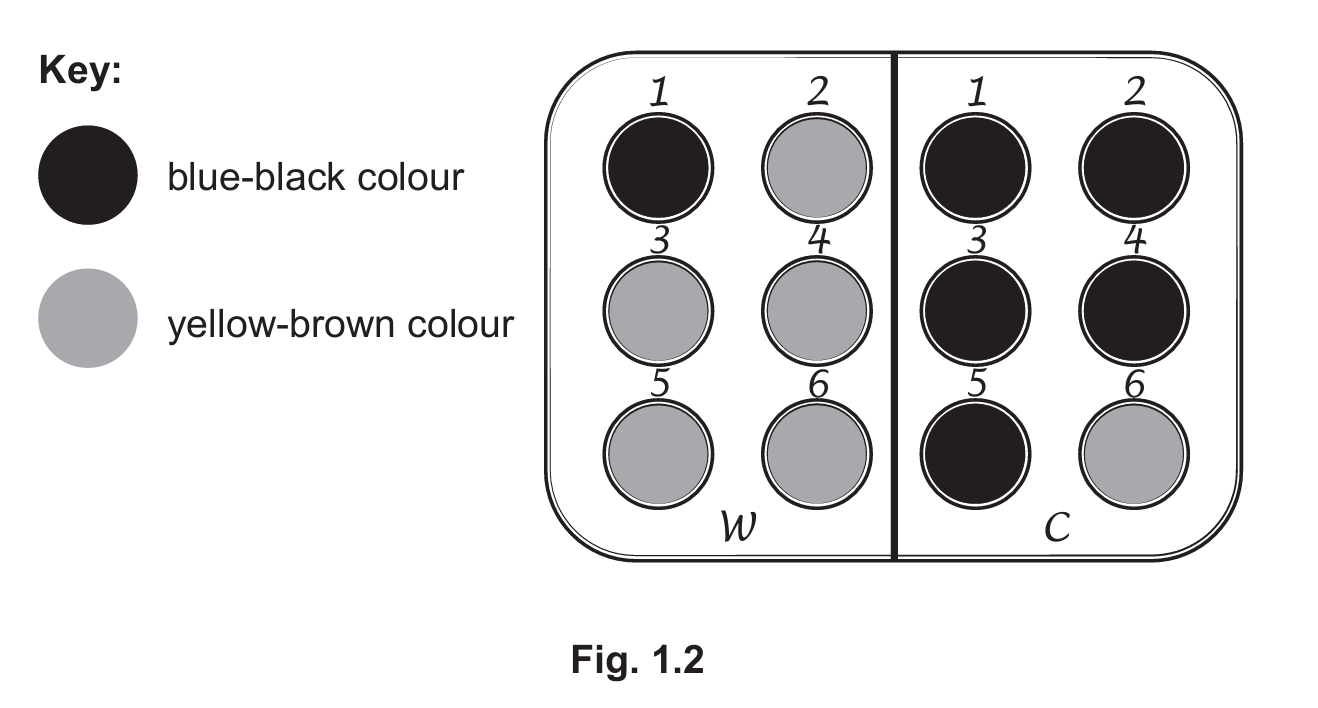
(a) (i) Prepare a table and record the results shown in Fig. 1.2.
(ii) State a conclusion for the results shown in Fig. 1.2.
(iii) State the independent variable in this investigation.
(iv) State three variables that were kept constant in this investigation.
(b) (i) Explain why the method used in this investigation does not allow the students to obtain an accurate time for the breakdown of starch.
(ii) The temperature of the water in the beakers during the investigation was a source of error. Describe how you could improve the method to reduce this error.
(iii) The test-tubes were left in the beakers of warm or cold water for three minutes in step 7 and step 12 before the amylase solution was poured into the starch suspension. Explain why the test-tubes were left in the beakers for three minutes.
▶️ Answer/Explanation
(a)(i) Table of results:
| Dimple | Warm water (W) | Cold water (C) |
|---|---|---|
| 1 | blue-black | blue-black |
| 2 | blue-black | blue-black |
| 3 | yellow-brown | blue-black |
| 4 | yellow-brown | blue-black |
| 5 | yellow-brown | blue-black |
| 6 | yellow-brown | blue-black |
Explanation: The table should have clear headings for each column (Dimple, Warm water, Cold water) and accurately record the color changes observed in each dimple. The warm water samples show a quicker change from blue-black to yellow-brown, indicating faster starch breakdown at higher temperatures.
(a)(ii) Conclusion: The higher temperature (warm water) resulted in faster breakdown of starch by amylase compared to the lower temperature (cold water).
Explanation: The color change from blue-black to yellow-brown occurs sooner in the warm water samples, showing that enzyme activity is temperature-dependent and works faster at higher temperatures within its optimal range.
(a)(iii) Independent variable: Temperature (of the water in the beakers).
Explanation: This is the variable that was deliberately changed (warm vs. cold water) to observe its effect on the enzyme activity.
(a)(iv) Controlled variables (any three):
- Number of drops of iodine solution used
- Volume of amylase solution (2 cm³)
- Concentration of amylase solution (2%)
- Volume of starch suspension (2 cm³)
- Time intervals between sampling (30 seconds)
- Number of drops of sample added to iodine solution
Explanation: These variables were kept constant to ensure that any changes observed in the rate of starch breakdown were solely due to the temperature difference and not other factors.
(b)(i) Limitation of method: The color change might occur between sampling times, making it difficult to pinpoint the exact time of complete starch breakdown.
Explanation: Since samples are only taken every 30 seconds, the precise moment when all starch is broken down could be missed. The method provides approximate rather than exact timing.
(b)(ii) Improving temperature control: Use a thermostatically controlled water bath to maintain a constant temperature throughout the experiment.
Explanation: The warm water in the beaker would naturally cool over time, affecting enzyme activity. A water bath with temperature control would eliminate this source of error by maintaining a stable temperature.
(b)(iii) Reason for 3-minute wait: To allow the test-tube contents to reach the same temperature as the water in the beaker (thermal equilibration).
Explanation: This waiting period ensures that both the enzyme solution and starch suspension are at the intended experimental temperature before mixing, making the results more reliable and comparable.
(a) Milk contains fats. The enzyme lipase catalyses the breakdown of fats to form fatty acids and glycerol. The fatty acids cause the pH of the milk to decrease.
Plan an investigation to determine the effect of lipase concentration on the breakdown of fats in milk.
(b) The emulsion test is used to test a sample of food for fat.
Describe the method you would use to do the emulsion test.
▶️ Answer/Explanation
(a)
Investigation Plan:
- Independent Variable: Use at least two different concentrations of lipase enzyme (e.g., 0.5%, 1%, 2% solutions).
- Dependent Variable: Measure either:
- The pH after a set time period (e.g., 10 minutes) using a pH meter or pH paper
- OR the time taken to reach a specific pH value (e.g., pH 4.5)
- Control Variables:
- Keep the volume of milk constant for all tests
- Use the same type/source of milk (same fat content)
- Maintain constant temperature (e.g., using water bath at 37°C)
- Use the same volume of lipase solution for each concentration
- Control Experiment: Include a negative control with boiled lipase (to show reaction requires active enzyme) or water instead of enzyme.
- Method Details:
- Measure initial pH of milk before adding lipase
- Add measured volume of lipase solution to milk and start timer
- Record pH at regular intervals or note time when target pH is reached
- Repeats: Perform each concentration test at least twice (three trials total) for reliability
- Safety: Wear goggles and gloves when handling enzymes
(b)
Emulsion Test Method:
- Take a small sample of the food to be tested (solid foods should be crushed first).
- Add the food sample to a test tube containing about 2 cm³ of ethanol.
- Shake the test tube vigorously for about 1 minute to dissolve any fats present.
- Allow any solid particles to settle or filter the mixture.
- Pour the ethanol solution into another test tube containing an equal volume of distilled water.
- Observe the result:
- Positive result: A cloudy white emulsion forms at the top of the liquid, indicating presence of fat
- Negative result: The solution remains clear, indicating no fat present
Explanation: The emulsion test works because fats are soluble in ethanol but not in water. When the ethanol solution is added to water, the fats come out of solution, forming tiny droplets that scatter light, creating the characteristic cloudy emulsion. The cloudiness is directly proportional to the amount of fat present in the sample.
Fig. 3.1 is a photograph of a type of seaweed called bladder wrack. The bladders help the seaweed float in water.
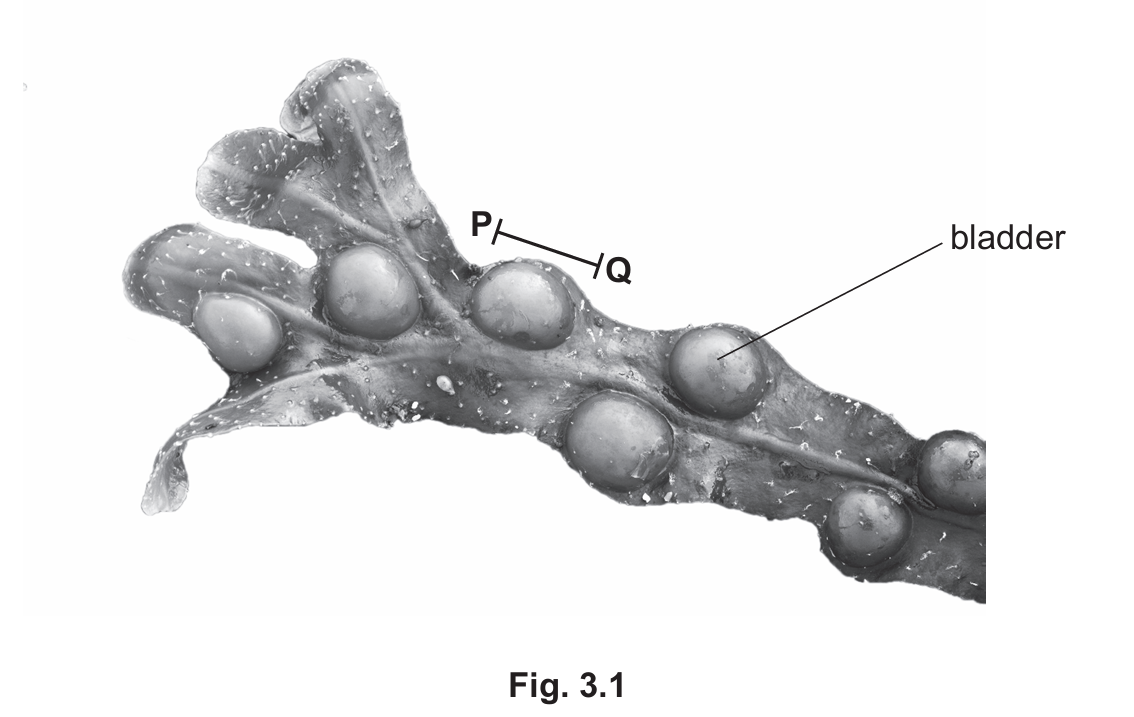
(a) (i) Draw a large diagram of the bladder wrack seaweed shown in Fig. 3.1.
(ii) Line PQ on Fig. 3.1 represents the length of one bladder on the bladder wrack seaweed. The actual length of the bladder is 19 mm.
Measure the length of line PQ on Fig. 3.1.
length of line PQ …… mm
Calculate the magnification of the photograph using the formula and your measurement.
\[ \text{magnification} = \frac{\text{length of line PQ}}{\text{actual length of the bladder}} \]
Give your answer to two decimal places.
(iii) Seaweeds are species of algae that live in the sea. Fig. 3.2 shows photographs of bladder wrack seaweed and a different species of seaweed called egg wrack. The photographs are the same magnification.
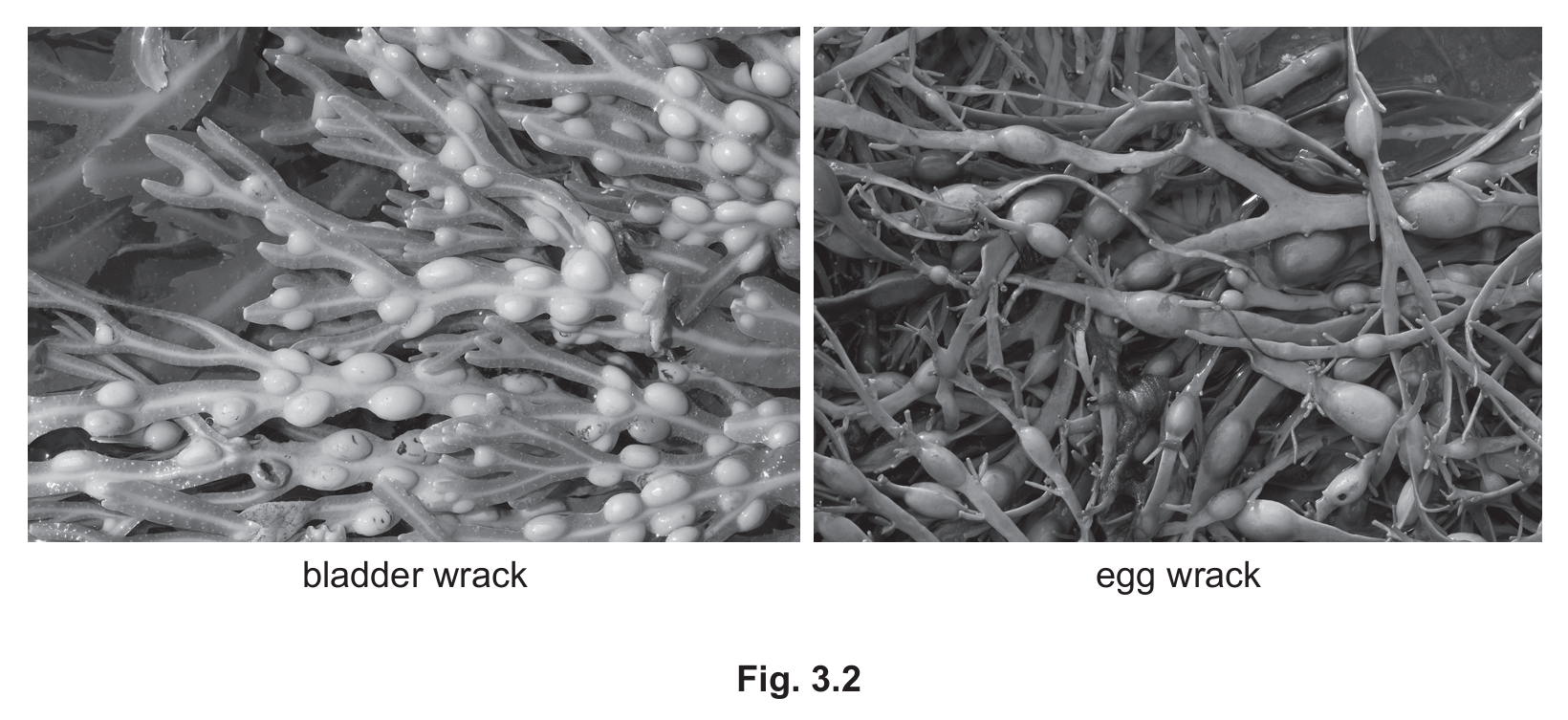
State two ways, visible in Fig. 3.2, that bladder wrack is different from egg wrack.
(b) Bladder wrack is found on the seashore and is exposed to the air when it is not covered by water at certain times of day.
Students investigated how rapidly bladder wrack lost water. They used this method:
- Three samples of bladder wrack were collected.
- The samples were blotted with tissue to remove any water on the surface of the seaweed.
- The initial mass of each sample was recorded.
- The samples were hung from a piece of string stretched between two stands.
- The mass of each sample was recorded every 30 minutes for the first two hours and then every hour for a further three hours.
(i) Suggest two variables that the students should keep constant during their investigation to ensure that the results are valid.
Table 3.1 shows the initial masses recorded by the students and the final masses recorded after five hours.
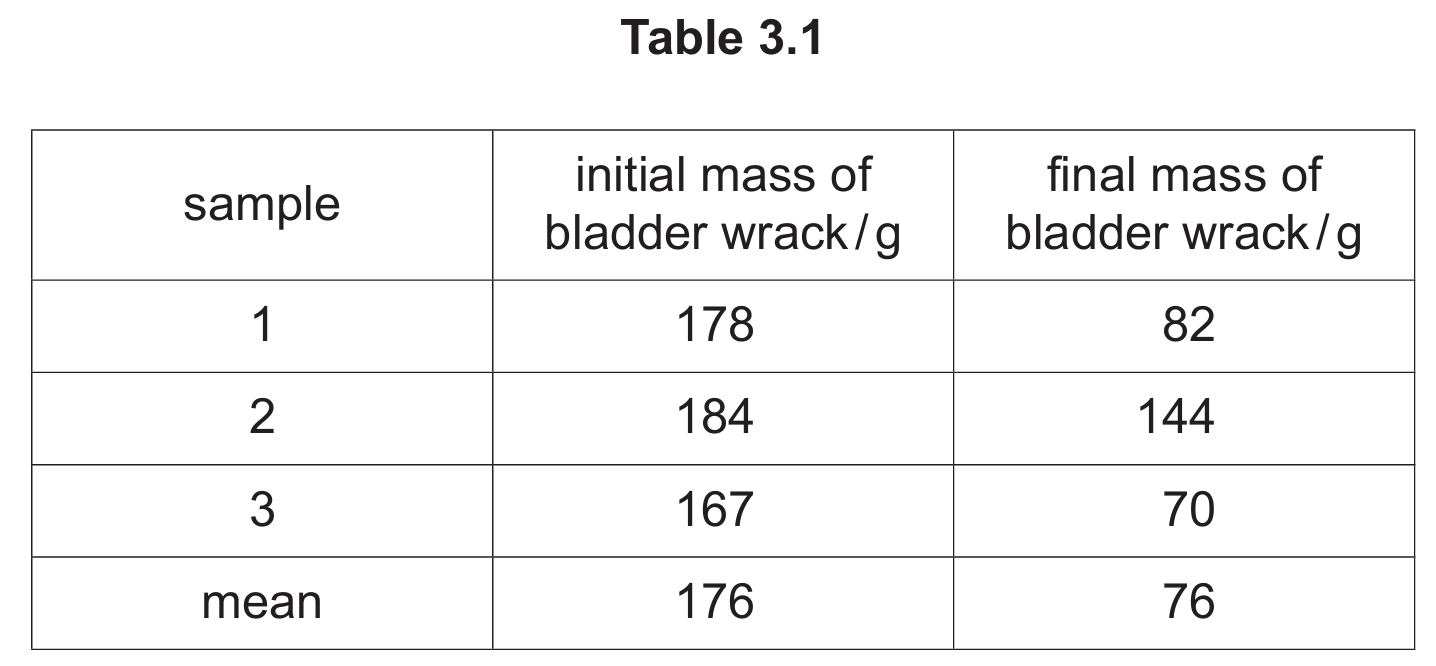
(ii) One of the final masses recorded is anomalous. State what is meant by an anomalous result.
(iii) Describe how the students calculated the mean value for the final mass of the bladder wrack.
(iv) Using the information in Table 3.1, calculate the mean percentage decrease in the mass of the bladder wrack samples after five hours. Give your answer to two significant figures.
(c) The students repeated their investigation using egg wrack seaweed. Table 3.2 shows the mean percentage decrease in the mass of the egg wrack samples throughout the investigation.
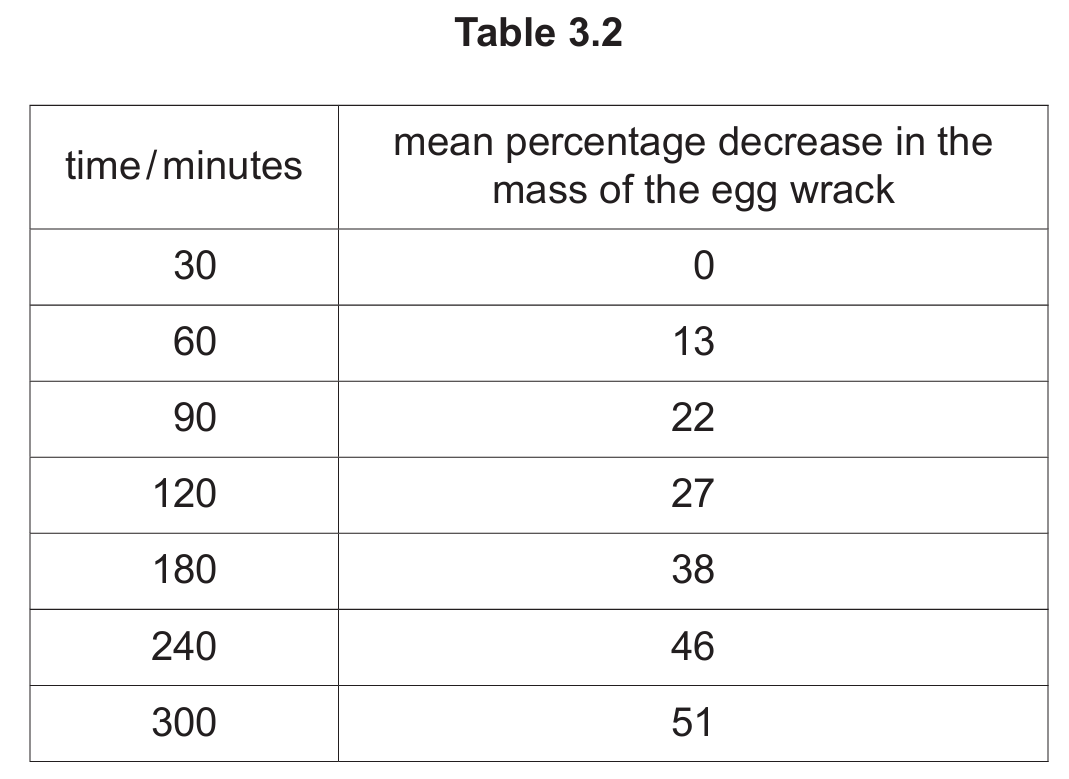
Using the data in Table 3.2, plot a line graph on the grid to show the effect of time on the mean percentage decrease in the mass of the egg wrack.

(d) Many people eat seaweed. State the names of the reagents that can be used to test seaweed for protein and vitamin C.
▶️ Answer/Explanation
(a) (i)
Answer: The diagram should have:
- A single clear outline with no shading
- Frond length at least 114 mm
- 7 bladders drawn
- Central midrib shown as a double line at least up to the point where it branches
(a) (ii)
Answer:
Measured length of line PQ: 12 mm (accept 11-13 mm)
Magnification calculation: \[ \frac{12}{19} = 0.63 \] (to two decimal places)
Explanation: First, measure line PQ carefully using a ruler. The actual length is given as 19 mm. Magnification is calculated by dividing the measured length by the actual length. Here, 12 mm ÷ 19 mm = 0.63 when rounded to two decimal places.
(a) (iii)
Answer: Two differences from:
- Bladder wrack has more bladders
- Bladders are closer together in bladder wrack
- Bladder wrack has bladders in pairs on either side of midrib (egg wrack has single bladders)
- Bladder wrack has a central midrib
- Bladder wrack divides into two at the end (more branched)
Explanation: When comparing the two seaweeds, bladder wrack shows distinct features like paired bladders along a central midrib, while egg wrack has fewer, more spaced-out bladders without the prominent midrib structure.
(b) (i)
Answer: Two variables to keep constant:
- Temperature
- Humidity
- Wind speed
- Species/type of seaweed
- Size/length/surface area of samples
Explanation: To ensure valid results, environmental conditions like temperature and humidity must be constant as they affect water loss. Also, using the same species and similar sized samples eliminates variability due to biological differences.
(b) (ii)
Answer: A result that doesn’t fit the pattern/trend.
Explanation: Sample 2’s final mass of 144g is anomalous because it’s much higher than the other two samples (82g and 70g), breaking the consistent pattern of water loss shown by the other samples.
(b) (iii)
Answer: They excluded the anomalous result (sample 2) and calculated the mean of samples 1 and 3 only.
Explanation: The mean was calculated as (82 + 70) ÷ 2 = 76g, intentionally leaving out the 144g value from sample 2 which was clearly inconsistent with the other results.
(b) (iv)
Answer: 57%
Explanation: Calculation steps:
1. Mean initial mass = 176g (given)
2. Mean final mass = 76g (excluding anomalous result)
3. Mass decrease = 176 – 76 = 100g
4. Percentage decrease = (100 ÷ 176) × 100 = 56.81%
Rounded to two significant figures: 57%
(c)
Answer: The graph should have:
- X-axis labeled “time/minutes” with scale 0-300
- Y-axis labeled “mean percentage decrease in mass” with scale 0-60%
- All seven points accurately plotted (±½ small square)
- A smooth curve connecting the points
Explanation: The graph shows a gradual increase in water loss over time, starting at 0% and reaching 51% after 300 minutes. The curve should show the rate slowing down as time progresses.
(d)
Answer:
Protein: Biuret reagent
Vitamin C: DCPIP
Explanation: Biuret reagent turns purple in the presence of proteins. DCPIP (dichlorophenolindophenol) changes from blue to colorless when reduced by vitamin C, making it a specific test for this vitamin.
