Questions 1
Which statement about a solid, a liquid or a gas is correct?
A A solid has a fixed shape and can be compressed.
B A liquid takes the shape of the container it is in and can be compressed.
C A solid has no fixed shape and cannot be compressed.
D A gas takes the shape of the container it is in and can be compressed.
▶️Answer/Explanation
Ans D
Questions 2
Which diagram represents a mixture of compounds?
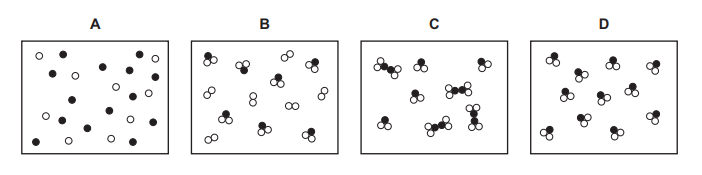
▶️Answer/Explanation
Ans C
Questions 3
Four ions are listed.
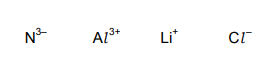
Which pair of ions have the same electronic configuration?
A \(N^{3–}\) and \(Li^+\)
B \(Al^{3+}\) and \(N^{3–}\)
C \(Cl^–\) and \(Al^{3+}\)
D \(Li^+\) and \(Cl^–\)
▶️Answer/Explanation
Ans B
Questions 4
Which statement about isotopes is correct?
A Atoms with different numbers of electrons are isotopes of each other.
B Atoms with the same mass numbers are isotopes of each other.
C Isotopes of the same element have different numbers of neutrons.
D Isotopes of the same element have different numbers of protons.
▶️Answer/Explanation
Ans C
Questions 5
Which statement about the ions formed by the elements in Group VII of the Periodic Table is correct?
A All the ions have the same charge of 1–.
B All the ions have the same number of electron shells.
C Each ion is formed by losing one electron.
D Each ion has seven electrons in its outer electron shell.
▶️Answer/Explanation
Ans A
Questions 6
Which row describes the properties of potassium bromide?
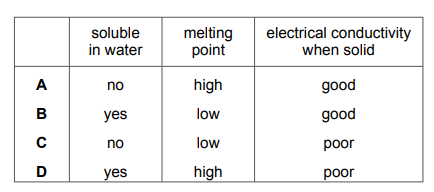
▶️Answer/Explanation
Ans D
Questions 7
Which statement explains why graphite is used as a lubricant?
A Each carbon atom in graphite forms three bonds.
B The bonding in graphite is covalent.
C The carbon atoms are arranged in hexagons.
D There are weak forces of attraction between the layers of carbon atoms
▶️Answer/Explanation
Ans D
Questions 8
What is the balanced equation for the reaction between magnesium and dilute sulfuric acid?
A Mg + \(H_2SO_4\) → \(MgSO_4\) +\( H_2\)
B Mg + \(2H_2SO_4\) → \(Mg(SO_4)_2\) + \(2H_2\)
C 2Mg + \(H_2SO_4\) → \(Mg_2SO_4\) + \(H_2\)
D 2Mg + \(3H_2SO_4\) → \(Mg_2(SO_4)_3\) + \(3H_2\)
▶️Answer/Explanation
Ans A
Questions 9
The relative atomic mass, Ar, of an element is the average mass of the isotopes of that element compared to another particle. Which particle is used for this comparison?
A a proton
B an atom of 12 C
C an atom of 40 Ca
D an atom of 1 H
▶️Answer/Explanation
Ans B
Questions 10
The equations for two reactions are shown.
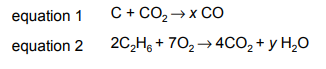
Which row shows the value of x, the value of y and the equations that are for redox reactions?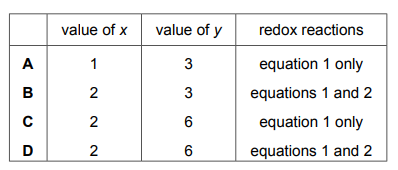
▶️Answer/Explanation
Ans D
Questions 11
Concentrated aqueous sodium chloride is electrolysed using graphite electrodes. What is the product formed at the cathode?
A chlorine
B hydrogen
C oxygen
D sodium
▶️Answer/Explanation
Ans B
Questions 12
Which row describes the changes that occur when metals burn in oxygen?
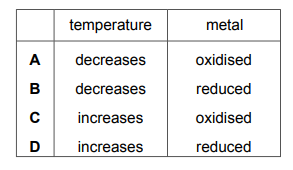
▶️Answer/Explanation
Ans C
Questions 13
When calcium carbonate is heated strongly, carbon dioxide gas is produced. Which words describe the type of change that occurs?
A endothermic and chemical
B endothermic and physical
C exothermic and chemical
D exothermic and physical
▶️Answer/Explanation
Ans A
Questions 14
Which row about a hydrogen–oxygen fuel cell is correct?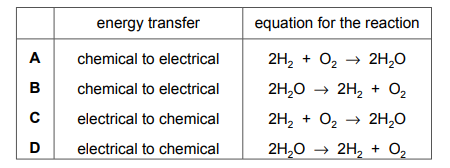
▶️Answer/Explanation
Ans A
Questions 15
Which list contains only chemical changes?
A melting, evaporating, dissolving
B rusting, freezing, cracking
C neutralisation, polymerisation, combustion
D boiling, condensing, distillation
▶️Answer/Explanation
Ans C
Questions 16
Excess calcium carbonate is added to \(50 cm^3\) of dilute hydrochloric acid of different concentrations in two separate experiments. The volume of gas produced in experiment 1 and in experiment 2 is measured every 30 seconds. The results are shown.

Which diagram represents the data collected? 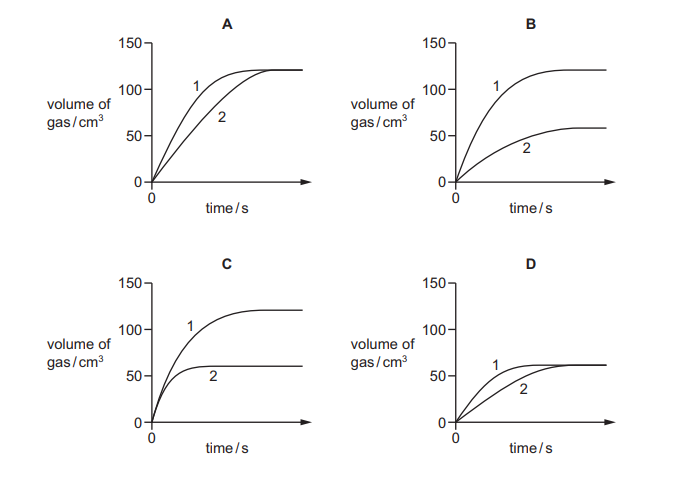
▶️Answer/Explanation
Ans B
Questions 17
An experiment is set up as shown. The mass of the conical flask and its contents is measured at 30-second intervals.
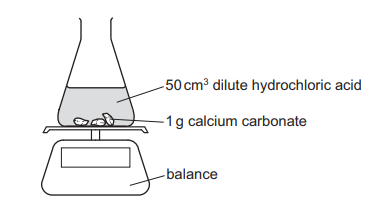
Which statement about the reaction and changes to the reaction conditions is correct?
A Adding \(10 cm^3\) of water to the \(50 cm^3\) of acid increases the rate of the reaction.
B Increasing the size of the pieces of calcium carbonate increases the rate of the reaction.
C Increasing the temperature increases the rate of the reaction.
D The mass of the conical flask and its contents increases as carbon dioxide is formed.
▶️Answer/Explanation
Ans C
Questions 18
Which reaction is reversible?
A reaction of aqueous sodium hydroxide with dilute hydrochloric acid
B formation of anhydrous copper(II) sulfate from hydrated copper(II) sulfate
C oxidation of methane to form carbon dioxide and water
D combustion of sulfur to form sulfur dioxide
▶️Answer/Explanation
Ans B
Questions 19
Silver oxide reacts with magnesium to make silver and magnesium oxide.
![]()
Which substance is oxidised in this reaction?
A magnesium
B magnesium oxide
C silver
D silver oxide
▶️Answer/Explanation
Ans A
Questions 20
Compound X dissolves in water to form an aqueous solution. Methyl orange is added to aqueous compound X. The methyl orange turns red. What is compound X?
A sodium carbonate
B copper(II) oxide
C potassium oxide
D sulfur dioxide
▶️Answer/Explanation
Ans D
Questions 21
Dilute hydrochloric acid reacts with aqueous sodium hydroxide in a neutralisation reaction. Which two ions are involved in this neutralisation reaction?
![]()
▶️Answer/Explanation
Ans B
Questions 22
The table shows some properties of some of the elements in Group I of the Periodic Table.
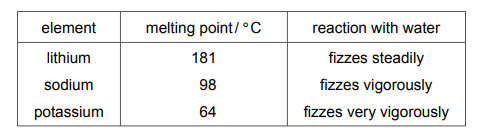
Rubidium is also an element in Group I of the Periodic Table. Which row describes the properties of rubidium? 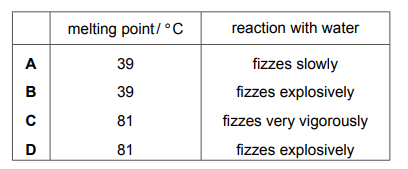
▶️Answer/Explanation
Ans B
Questions 23
Copper(II) sulfate crystals are blue. They are made by adding an excess of copper(II) oxide to sulfuric acid. The mixture is heated and stirred. The mixture is then filtered and the filtrate is allowed to evaporate, leaving blue crystals. Why is filtration necessary?
A to remove soluble impurities
B to remove sulfuric acid
C to remove the blue crystals
D to remove unreacted copper(II) oxide
▶️Answer/Explanation
Ans D
Questions 24
Which barium salts are soluble in water?
1 barium carbonate
2 barium chloride
3 barium nitrate
4 barium sulfate
A 1 and 2
B 1 and 4
C 2 and 3
D 3 and 4
▶️Answer/Explanation
Ans C
Questions 25
Which statement about the properties of elements in Group I or in Group VII is correct?
A Bromine displaces iodine from an aqueous solution of potassium iodide.
B Chlorine, bromine and iodine are diatomic gases at room temperature.
C Lithium, sodium and potassium are soft non-metals.
D Lithium, sodium and potassium have an increasing number of electrons in their outer
shells.
▶️Answer/Explanation
Ans A
Questions 26
Rubidium and strontium are both in Period 5 of the Periodic Table. Rubidium is in Group I. Strontium is in Group II. Which statement about these elements is correct?
A Each element has five electrons in its outer electron shell.
B The atomic number of rubidium is greater than the atomic number of strontium.
C Rubidium forms the \(Rb^+\) ion; strontium forms the \(Sr^{2+}\) ion.
D Electrolysis of molten rubidium chloride and of molten strontium chloride produces hydrogen.
▶️Answer/Explanation
Ans C
Questions 27
A company needs a metal with a low density which resists corrosion and is a good electrical conductor. Which metal should it use?
A aluminium
B iron
C magnesium
D sodium
▶️Answer/Explanation
Ans A
Questions 28
Which statement about stainless steel is correct?
A All atoms in stainless steel are the same size.
B Stainless steel is a mixture of copper and zinc.
C Stainless steel is an iron compound.
D Stainless steel is stronger than pure iron.
▶️Answer/Explanation
Ans D
Questions 29
Which statements explain why a water tap made of steel is electroplated with copper?
1 It improves the conductivity of the tap.
2 It improves the tap’s resistance to corrosion.
3 It increases the density of the tap.
A 1 and 2
B 1 and 3
C 2 only
D 3 only
▶️Answer/Explanation
Ans C
Questions 30
The table shows the results of separately heating four metals with oxides of different metals. The four metals are iron, copper, magnesium and Y. The results are shown.
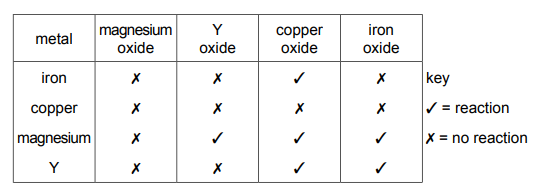
What is the order of reactivity of the metals, least reactive first? 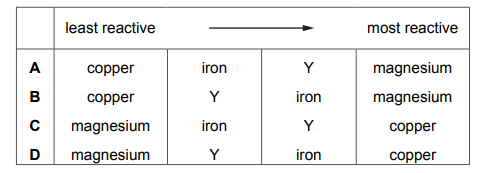
▶️Answer/Explanation
Ans A
Questions 31
Which metal is most easily obtained from its ore?
A aluminium
B copper
C calcium
D iron
▶️Answer/Explanation
Ans B
Questions 32
Why is distilled water used in chemical experiments rather than tap water?
A Distilled water contains fewer chemical impurities.
B Distilled water has a better colour.
C Distilled water has a higher boiling point.
D Distilled water is a better solvent.
▶️Answer/Explanation
Ans A
Questions 33
Which type of compound is also the name of a homologous series?
A carbonate
B carboxylic acid
C halide
D hydroxide
▶️Answer/Explanation
Ans B
Questions 34
The fractional distillation of petroleum is shown.
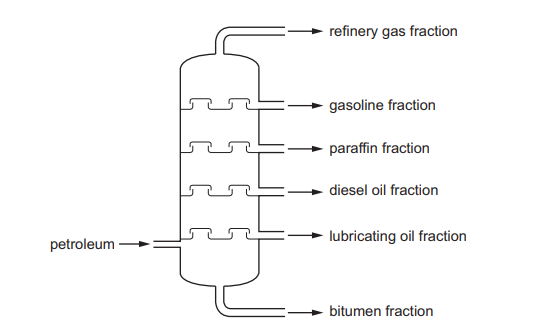
Which fraction is the least volatile?
A bitumen
B diesel oil
C gasoline
D refinery gas
▶️Answer/Explanation
Ans A
Questions 35
Which formula represents an unsaturated hydrocarbon?
A \(C_3H_6\)
B \(C_3H_8\)
C \(C_4H_{10}\)
D \(C_5H_{12}\)
▶️Answer/Explanation
Ans A
Questions 36
The structure of an organic compound is shown.
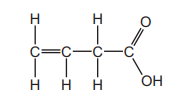
The compound is tested separately with thymolphthalein and with aqueous bromine. Which row describes the final colour observed for each test?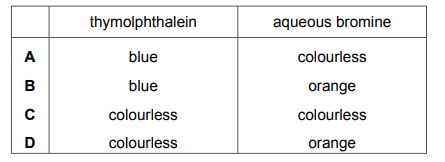
▶️Answer/Explanation
Ans C
Questions 37
Which statement describes methane?
A It is an alcohol.
B It is an unsaturated molecule.
C It contains carbon, hydrogen and oxygen atoms only.
D Its molecules contain four single covalent bonds.
▶️Answer/Explanation
Ans D
Questions 38
Which row explains why plastics such as poly(ethene) cause pollution?
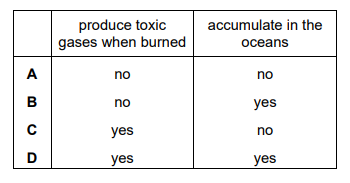
▶️Answer/Explanation
Ans D
Questions 39
Two experiments are described.
experiment 1 A large mass of copper(II) sulfate is stirred into a beaker of water. After a few minutes, undissolved crystals are visible on the bottom of the
beaker.
experiment 2 Sea water is distilled. Distilled water and solid impure salt are separated into two containers.
Which statement is correct?
A In experiment 1, the undissolved crystals are the filtrate.
B In experiment 1, the water is the solute.
C In experiment 2, sea water boils at 100 °C.
D In experiment 2, the impure salt is a residue
▶️Answer/Explanation
Ans D
Questions 40
An aqueous sample of X is heated with aqueous sodium hydroxide and small pieces of aluminium. A gas is produced which turns damp red litmus paper blue. Aqueous sodium hydroxide is added to a second aqueous sample of X. A pale green precipitate is observed. What is X?
A ammonium nitrate
B chromium(II) chloride
C iron(II) nitrate
D iron(II) sulfate
▶️Answer/Explanation
Ans C
