Question 1
The boiling point of sodium is 890°C. What happens to sodium atoms as the temperature of a sample of sodium changes from 950 °C to 900°C?
A The atoms move more quickly and bonds are formed.
B The atoms move more quickly and bonds are neither broken nor formed.
C The atoms move more slowly and bonds are formed.
D The atoms move more slowly and bonds are neither broken nor formed.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (D)
Question 2
Which row shows the conditions for the particles of a gas colliding most frequently?
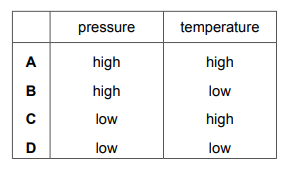
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 6.2: Rate of reaction — Describe collision theory in terms of number of particles per unit volume and frequency of collisions
▶️ Answer/Explanation
✅ Answer: B
Question 3
The diagram shows some properties of particles in an atom. To which labelled part of the diagram do electrons belong?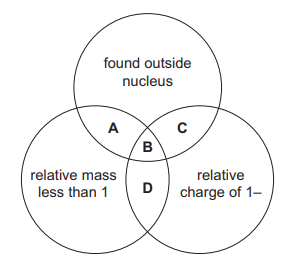
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 2.2: State the relative charges and relative masses of a proton, a neutron and an electron (Core)
▶️ Answer/Explanation
✅ Answer: B
Question 4
Some properties of substances W, X, Y and Z are shown.
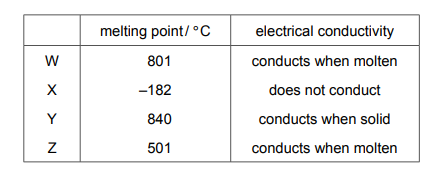
Which substances are ionic?
A. W, X and Y
B. W and Y only
C. W and Z
D. X and Z
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C) W and Z
Question 5
Atoms lose or gain electrons to become ions. Which row is correct?
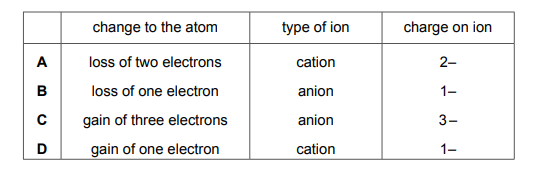
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 2.4: Ions and ionic bonds — Describe the formation of positive ions (cations) and negative ions (anions).
▶️ Answer/Explanation
✅ Answer: (C)
Question 6
A covalent molecule, M, contains four shared pairs of electrons. What is M?
A. ammonia, \(NH_3\)
B. hydrogen chloride, HCl
C. methane, \(CH_4\)
D. water, \(H_2O\)
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C)
Question 7
Which substance has a giant covalent structure?
A sodium chloride
B sodium
C ethane
D diamond
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: D
Question 8
Iron(III) oxide is reduced by carbon monoxide to produce iron and carbon dioxide. What is the balanced equation for this reaction?
A \(Fe_2O_3 + 2CO → 2Fe + 2CO_2\)
B \(Fe_2O_3 + 3CO → 2Fe + 3CO_2\)
C \(2Fe_2O_3 + 6CO → 2Fe + 6CO_2\)
D \(2Fe_2O_3 + 3CO → 4Fe + 3CO_2\)
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B)
Question 9
The equation for the reaction between magnesium and dilute hydrochloric acid is shown.
\(Mg + 2HCl \rightarrow MgCl_2 + H_2\)
Which mass of magnesium chloride is formed when 48.0 g of magnesium completely reacts with excess dilute hydrochloric acid?
A 23.8 g
B 47.5 g
C 95.0 g
D 190 g
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 3.3: The mole and the Avogadro constant — Calculate stoichiometric reacting masses (Supplement).
▶️ Answer/Explanation
✅ Answer: (D) 190 g
Question 10
Dilute sulfuric acid and lead(II) bromide are electrolysed separately. Which statements are correct?
1 Colourless gases are produced when dilute sulfuric acid is electrolysed.
2 Lead(II) bromide can be electrolysed when molten.
3 Lead is formed at the positive electrode when lead(II) bromide is electrolysed.
4 Sulfate ions are produced at the negative electrode when dilute sulfuric acid is electrolysed.
A 1 and 2
B 1 and 3
C 2 and 3
D 3 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A) 1 and 2
Question 11
Which statements about a hydrogen–oxygen fuel cell are correct?
1 The main form of energy released by the fuel cell is heat.
2 The reaction is a redox reaction.
3 An acidic gas is produced.
4 Water is the only chemical product.
A 1 and 3
B 1 and 4
C 2 and 3
D 2 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: D
Question 12
Which row describes what happens during an endothermic reaction?
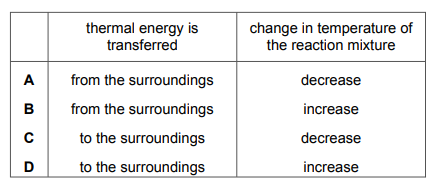
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: A
Here is the HTML code that formats your question, the syllabus topics, and a detailed solution as requested, based on the provided template and syllabus.
Question 13
Which diagram represents a chemical change?
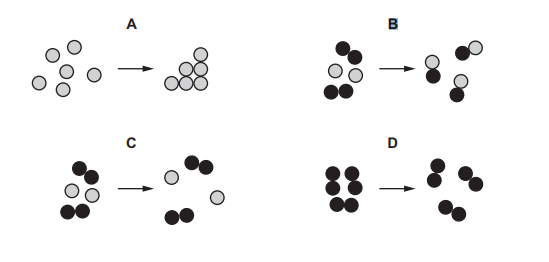
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 2.5: Simple molecules and covalent bonds — Describe the formation of covalent bonds in simple molecules (Core)
▶️ Answer/Explanation
✅ Answer: (B)
Question 14
A method used to investigate the rate of reaction of calcium carbonate with dilute hydrochloric acid under different conditions is shown. Place 50 cm³
• of dilute hydrochloric acid in a conical flask.
• Add a known volume of water to the conical flask.
• Heat the conical flask to the required temperature.
• Add 1.0 g of calcium carbonate to the conical flask.
• Measure the time taken for the reaction to finish.
Which volume of water and which temperature give the shortest time taken for the reaction to finish?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: B
Question 15
The rate of reaction between magnesium and hydrochloric acid is investigated. The total volume of hydrogen given off is measured at different times. A graph of the results is shown.
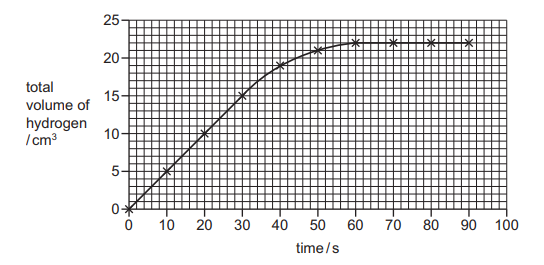
Which conclusions are correct?
1 The rate is fastest between 0 and 30 seconds.
2 The maximum volume of hydrogen given off is 22 cm³.
3 At 40 seconds, 20 cm³ of hydrogen is given off.
A. 1 and 2 only
B. 1 and 3 only
C. 2 and 3 only
D. 1, 2 and 3
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: A (1 and 2 only)
Statement 1 is correct because the slope of the graph is steepest between 0 and 30 seconds, indicating the highest rate of hydrogen production. Statement 2 is correct as the graph flattens at 22 cm³, showing the reaction is complete. Statement 3 is incorrect because at 40 seconds, the graph clearly shows a volume of approximately 21 cm³, not 20 cm³.
Question 16
Water is added to anhydrous copper(II) sulfate. Which row describes the direction of energy change and the colour change of the mixture during the reaction?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 5.1: Exothermic and endothermic reactions — State that an exothermic reaction transfers thermal energy to the surroundings leading to an increase in the temperature of the surroundings (Core)
• Topic 7.3: Preparation of salts — Define a hydrated substance as a substance that is chemically combined with water and an anhydrous substance as a substance containing no water (Core)
• Topic 10.1: Water — Describe chemical tests for the presence of water using anhydrous copper(II) sulfate (Core)
▶️ Answer/Explanation
Adding water to anhydrous copper(II) sulfate (white solid) causes it to become hydrated copper(II) sulfate (blue crystals). This is an exothermic process, meaning heat energy is released to the surroundings, making the test tube feel warm. The colour change observed is from white to blue, which is also the standard chemical test for water.
Question 17
Which equation represents an oxidation reaction?
A \(CaCO_3 \rightarrow CaO + CO_2\)
B \(4FeO + O_2 \rightarrow 2Fe_2O_3\)
C \(2NO_2 \rightarrow N_2O_4\)
D \(2P_2O_5 \rightarrow P_4O_{10}\)
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B)
Question 18
A farmer treats a field with calcium hydroxide to make it less acidic. When the farmer adds ammonium nitrate fertiliser to the field immediately after the calcium hydroxide, the two substances react. Why does this reaction make the fertiliser less effective?
A. It makes ammonia gas, so less nitrogen is absorbed by the soil.
B. It makes an acid, making the soil acidic again.
C. It makes nitrogen gas, so less nitrogen is absorbed by the soil.
D. It makes the fertiliser too strong, stopping the plants growing well.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 10.2: Fertilisers — State that ammonium salts are used as fertilisers (Core).
• Topic 6.4: Redox (implicitly, via base reaction) and Topic 7.3 Preparation of salts.
▶️ Answer/Explanation
✅ Answer: (A)
Question 19
Which statement about sodium oxide or nitrogen dioxide is correct?
A. Nitrogen dioxide is a solid at room temperature.
B. Nitrogen dioxide is acidic.
C. Sodium oxide has a lower melting point than nitrogen dioxide.
D. Sodium oxide is covalently bonded.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Nitrogen dioxide (NO₂) is a non-metal oxide which dissolves in water to form an acidic solution (nitric and nitrous acids), making it an acidic oxide. Sodium oxide (Na₂O) is an ionic metal oxide with a very high melting point due to its giant ionic lattice, while NO₂ is a simple covalent molecule that is a gas at room temperature, so NO₂ has a much lower melting point than Na₂O, not the other way around. Therefore, only statement B is correct.
Question 20
A titration method is used to prepare a pure soluble sulfate salt from dilute sulfuric acid. What is the other reagent?
A. copper(II) oxide
B. magnesium
C. sodium hydroxide
D. zinc carbonate
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C)
Question 21
Which row about elements in the Periodic Table is correct?
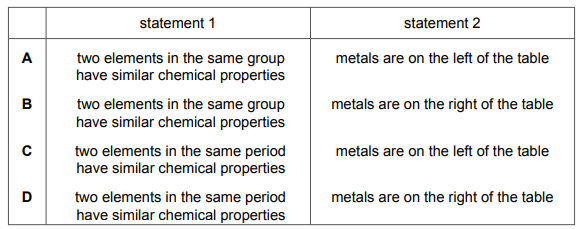
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 8.2: Group I properties — Describe alkali metals as relatively soft metals with trends down the group (Core)
• Topic 8.3: Group VII properties — Describe halogens as diatomic non-metals with trends down the group (Core)
▶️ Answer/Explanation
✅ Answer: A
Question 22
The table gives some information about three elements in Group I of the Periodic Table.

Which row identifies the melting point and the density of rubidium?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed Solution: Moving down Group I (from lithium to sodium to potassium), the melting point decreases significantly (from 180°C to 98°C to 63°C) and the density increases (from 0.53 to 0.97 to 0.86 g/cm³; note potassium is less dense than sodium, but the overall trend is increasing density with a slight anomaly). Rubidium is below potassium, so its melting point should be even lower than 63°C (option B: 39°C) and its density should be higher than 0.86 g/cm³ (option B: 1.53 g/cm³). Option A has a higher melting point, option C has a lower density, and option D has both melting point and density values inconsistent with the downward trend.
✅ Answer: (B)
Question 23
Which statement describes a transition element?
A. It is a dull grey metal that only forms white compounds.
B. It is a high-density metal with a high melting point that is used as a catalyst.
C. It is a low-density metal with a high melting point that reacts with steam to make hydrogen.
D. It is a soft, shiny silver metal that reacts vigorously with water.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B)
Question 24
The electronic configurations of helium, neon and argon are shown.
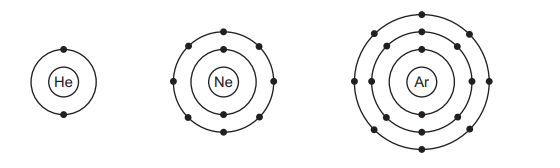
Which row describes these gases?
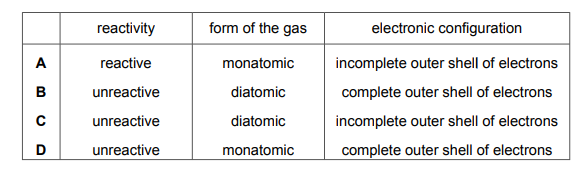
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: D
Question 25
X is a shiny silver-coloured solid at room temperature and pressure. X is a good conductor of heat and electricity when solid. Which statement about X is correct?
A. X is an ionic compound or a metallic element.
B. X is a metallic element or a non-metallic element.
C. X is an alloy or a metallic element.
D. X is an alloy or a non-metallic element.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 9.3: Alloys and their properties — Describe an alloy as a mixture of a metal with other elements, and state that alloys can be harder and stronger than the pure metals.
▶️ Answer/Explanation
✅ Answer: (C)
Question 26
Which elements can be combined to produce an alloy?
1 magnesium and aluminium
2 nitrogen and oxygen
3 iron and carbon
4 copper and zinc
A 1, 3 and 4
B 1 and 2
C 2 and 3
D 4 only
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A) 1, 3 and 4
Question 27
Three metals, L, M and N, are added separately to dilute hydrochloric acid and cold water. The results are shown.
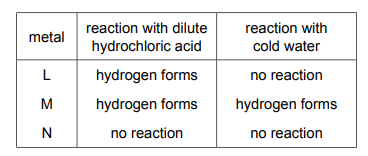
What is the order of reactivity of the metals?
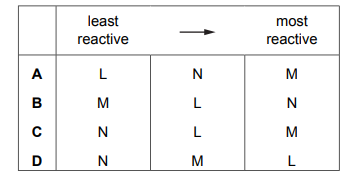
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 9.4: Supplement — Describe the relative reactivities of metals in terms of their tendency to form positive ions, by displacement reactions.
▶️ Answer/Explanation
✅ Answer: C
Question 28
Which reaction produces carbon dioxide?
A. cracking of large hydrocarbon molecules
B. photosynthesis
C. reaction of a base with a carbonate
D. thermal decomposition of calcium carbonate
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 9.6: Extraction of metals — Describe the thermal decomposition of calcium carbonate/limestone to produce calcium oxide (Core)
• Topic 11.5: Alkenes — Describe the manufacture of alkenes and hydrogen by the cracking of larger alkane molecules (Core)
• Topic 10.3: Air quality and climate — Photosynthesis word equation (Core)
▶️ Answer/Explanation
✅ Answer: (C)
Question 29
A sample of air containing four gases only is analysed. 99.0% of the sample contains the two main gases in the same percentages as in clean, dry air. The remaining 1.0% of the sample contains argon and carbon dioxide. The gas that makes up 0.1% of the sample turns limewater milky. Which row shows the percentage composition of the sample of air?
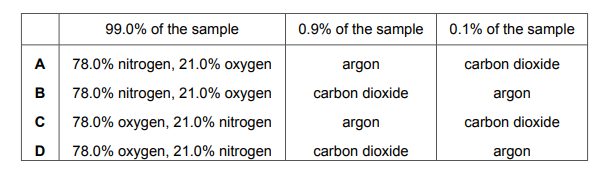
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A)
Question 30
Which substance contains two elements that are found in NPK fertilisers?
A. ammonium chloride
B. calcium hydroxide
C. potassium nitrate
D. sodium phosphate
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: C
Question 31
Which statement about sulfur is correct?
A When sulfur is burned, it produces a substance that causes acid rain.
B Sulfur is produced by the thermal decomposition of limestone.
C Compounds of sulfur make up approximately 1% of unpolluted air.
D Sulfur is a member of the family of elements called halogens.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 8.2/8.3: Group I / Group VII — sulfur is not a halogen; halogens are Group VII (Core)
• Topic 9.6: Extraction of metals — limestone decomposes to calcium oxide, not sulfur (Core)
▶️ Answer/Explanation
✅ Answer: (A)
Question 32
Questions 32
What are two adverse effects of particulates in the air?
1 acid rain
2 cancer
3 photochemical smog
4 respiratory problems
A 1 and 3
B 1 and 4
C 2 and 3
D 2 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (D)
Question 33
Which formula represents a compound that is a member of the homologous series of alkanes?
A \(C_2H_4\)
B \(C_3H_6\)
C \(C_4H_8\)
D \(C_5H_{12}\)
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 11.2: Naming organic compounds — State the type of compound present, given a molecular formula (Core)
▶️ Answer/Explanation
✅ Answer: (D) \(C_5H_{12}\)
Question 34
Which statement about ethane is correct?
A It rapidly decolourises aqueous bromine.
B It does not burn.
C It forms long-chain compounds called polymers.
D It only contains single bonds between its atoms.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (D)
Question 35
Which raw material is used to make ethanol by fermentation?
A. carbon dioxide
B. ethene
C. glucose
D. natural gas
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C)
Question 36
Which statement about ethanoic acid is correct?
A It contains a –COOH group.
B It has a pH greater than pH 7.
C It reacts with sodium carbonate to form hydrogen gas.
D It reacts with copper to form copper(II) ethanoate.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 7.1: The characteristic properties of acids and bases — Define acids as proton donors; state that aqueous solutions of acids contain H⁺ ions (Supplement).
▶️ Answer/Explanation
✅ Answer: (A)
Question 37
Which statement explains why the disposal of plastic waste leads to environmental problems?
A. Plastic waste forms toxic gases when it is burned.
B. Plastic waste contains many small molecules.
C. Plastic waste rapidly dissolves in the oceans.
D. Plastic waste reacts with both acids and bases.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: A
Question 38
Substance X and four known substances, P, Q, R and S, are analysed by chromatography. The chromatogram produced is shown.

Which statement about X is correct?
A It is a mixture of P, Q and S.
B It contains P and S only.
C It contains P, S and another unknown substance.
D It is a mixture of Q, R and S.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C)
Question 39
Copper is insoluble in water. Copper(II) oxide is a solid that is insoluble in water but reacts with dilute hydrochloric acid. Which method is used to separate copper from a mixture of copper and copper(II) oxide?
A. dissolve the mixture in water then filter
B. dissolve the mixture in water then crystallise
C. react the mixture with dilute hydrochloric acid then filter
D. react the mixture with dilute hydrochloric acid then crystallise
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 12.4: Separation and purification — Describe and explain methods of separation and purification using filtration (Core)
▶️ Answer/Explanation
✅ Answer: (C)
Question 40
A salt, S, is dissolved in water and three tests are carried out on the solution formed.

What is the identity of S?
A copper(II) chloride
B copper(II) sulfate
C iron(II) chloride
D iron(II) sulfate
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B) copper(II) sulfate
