Question 1
Which statement about gases is correct?
A Gases are difficult to compress when pressure is applied.
B The particles in gases are close together.
C The particles in gases have a random arrangement.
D The particles in gases move slowly past each other.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C)
Question 2
A sample of argon gas is heated in a closed container. Which row describes what happens to the pressure and the size of the argon atoms?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 2.2: Atomic structure and the Periodic Table — Describe the structure of the atom as a central nucleus containing neutrons and protons surrounded by electrons in shells (Core)
▶️ Answer/Explanation
According to the kinetic particle theory, heating a gas in a closed container increases the average kinetic energy of its particles. This leads to more frequent and forceful collisions with the container walls, causing the pressure to increase (eliminating rows B and C). However, the size of individual argon atoms is a fixed property of the element and does not change with temperature; only the space between atoms or their speed changes. Therefore, both increased pressure and unchanged atomic size are correctly described in row D.
Question 3
Which statement is correct?
A. Air is a mixture of gaseous elements only.
B. Alloys are formed when a metal is ionically bonded to other elements.
C. Carbon dioxide is a mixture of carbon and oxygen.
D. Potassium bromide is an ionic compound.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 2.4: Ions and ionic bonds — Describe the formation of ionic bonds between elements from Group I and Group VII (Core).
▶️ Answer/Explanation
✅ Answer: D
Question 4
Which row identifies methods of testing the purity of a compound?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 12.3: Chromatography — Interpret simple chromatograms to identify pure and impure substances (Core)
▶️ Answer/Explanation
✅ Answer: D
Question 5
Which row shows the number of protons, neutrons and electrons in the ion \( _{8}^{18}\textrm{O}^{2-}\)?
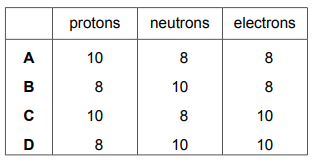
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 2.4: Ions and ionic bonds — Describe the formation of positive ions, known as cations, and negative ions, known as anions (Core).
▶️ Answer/Explanation
✅ Answer: D
Question 6
Isotopes of the same element have some features that are the same and some that are different. Which row shows the features that are the same and those that are different?
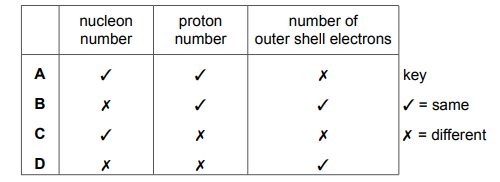
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Isotopes have the same number of protons (and hence electrons) but different numbers of neutrons. The number of electrons determines chemical properties, so chemical properties are the same. The different mass number affects physical properties like density and mass. Row B correctly identifies the same chemical properties and different physical properties.
Question 7
The electronic configurations of four atoms, W, X, Y and Z, are shown.
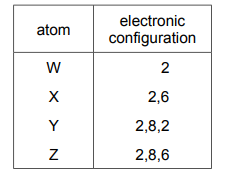
Which atoms form an ion with a charge of 2– when they react?
A W and Y
B W only
C X and Z
D Y only
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 2.4: Ions and ionic bonds — Describe the formation of negative ions, known as anions (Core).
▶️ Answer/Explanation
✅ Answer: (C)
Question 8
Which statement about bonding is correct?
A All the atoms in \(CH_4\), \(NH_3\) and \(H_2O\) molecules have noble gas electronic configurations.
B Calcium chloride is a covalent molecule.
C Group I metals gain electrons when they bond with Group VII elements.
D Oxide ions in calcium oxide are positively charged.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: A
Question 9
Which statement about diamond is correct?
A. It is a giant covalent structure consisting of carbon atoms and each atom is bonded to four other atoms.
B. It is a giant covalent structure consisting of flat sheets of carbon atoms.
C. It is a structure held together by ionic bonds and each ion is bonded to four other ions.
D. It is a structure held together by ionic bonds and each ion is bonded to three other ions.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A)
Question 10
Which row shows the correct formulae of lead(II) bromide and butane?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 11.2: Naming organic compounds — Name and draw the displayed formulae of methane and ethane (Core); butane is an alkane with formula C₄H₁₀ (Extension of understanding from alkanes CₙH₂ₙ₊₂, Topic 11.1).
▶️ Answer/Explanation
Lead(II) bromide is an ionic compound. Lead (Pb) in the +2 oxidation state forms the ion Pb²⁺, and bromine forms the ion Br⁻. To balance the charges, one Pb²⁺ requires two Br⁻ ions, giving the formula PbBr₂. Butane is a saturated hydrocarbon (alkane) with four carbon atoms. The general formula for alkanes is CₙH₂ₙ₊₂, so for n=4, the formula is C₄H₁₀. Only row B correctly pairs PbBr₂ and C₄H₁₀.
Question 11
Calcium phosphate forms when calcium chloride and sodium phosphate solutions react together.
\(x CaCl_2 + y Na_3PO_4 → 2Ca_3(PO_4)_2 + 12NaCl\)
Which values of x and y balance the equation?
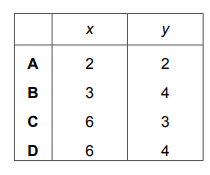
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 3.3: The mole and the Avogadro constant — Calculate stoichiometric reacting masses (Supplement).
▶️ Answer/Explanation
✅ Answer: D
Question 12
What is the definition of relative molecular mass, \(M_r\)?
A It is the average mass of the isotopes in a compound.
B It is the sum of the atomic numbers in a compound.
C It is the sum of the relative atomic masses in a compound.
D It is the total number of atoms in a compound.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C)
Question 13
In an experiment, a molten compound is broken down using electricity. Which row identifies the negative electrode and the general name for the molten compound being broken down?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: C
Question 14
Hydrogen–oxygen fuel cells can be used to power vehicles. What is produced by the fuel cells?
1 carbon dioxide
2 electricity
3 water
A 1, 2 and 3
B 1 and 2 only
C 1 and 3 only
D 2 and 3 only
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: D (2 and 3 only)
Question 15
Three reactions are described.
1 An acid is added to substance H. Rapid fizzing happens and the temperature decreases.
2 When substance J is ignited, it produces large quantities of heat.
3 Substance K reacts slowly with air and becomes warmer.
Which reactions are endothermic?
A 1, 2 and 3
B 1 and 2 only
C 1 only
D 2 and 3 only
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C) 1 only
Question 16
Which of the processes produces a physical change?
A thermal decomposition of calcium carbonate
B addition of sodium chloride to water
C addition of magnesium to hydrochloric acid
D combustion of sodium
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B)
Question 17
A known mass of gaseous ammonia and excess gaseous hydrogen chloride react together to make solid ammonium chloride. Line X shows the total mass of ammonium chloride produced over time. The reaction is repeated at a higher pressure. All other conditions are kept the same. Line Y shows the total mass of ammonium chloride produced over time at the higher pressure. Which diagram is correct?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 6.2 (Supplement): Describe and explain the effect on the rate of reaction of changing the pressure of gases using collision theory
• Topic 6.3: Reversible reactions and equilibrium — Predict and explain how changing pressure affects the position of equilibrium (Supplement)
▶️ Answer/Explanation
✅ Answer: (D)
Question 18
The equation for the hydration of anhydrous copper(II) sulfate is shown.
\(CuSO_4 + 5H_2O \rightarrow CuSO_4 \cdot 5H_2O\)
Which colour change is observed in this reaction?
A. blue to white
B. white to blue
C. pink to blue
D. blue to pink
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 7.3: Preparation of salts — Define a hydrated substance as a substance that is chemically combined with water and an anhydrous substance as a substance containing no water (Core)
• Topic 10.1: Water — Describe chemical tests for the presence of water using anhydrous copper(II) sulfate (Core)
▶️ Answer/Explanation
✅ Answer: (B)
Question 19
Which of the reactions show the underlined substance being reduced?
1 \(\underline{CuO} + H_2 → Cu + H_2O\)
2 \(2Mg + \underline{O_2} → 2MgO\)
3 \(\underline{MgO} + 2HCl → MgCl_2 + H_2O\)
A 1 and 2
B 1 only
C 2 and 3
D 3 only
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A)
Question 20
Four different solutions are separately tested with blue litmus and with methyl orange. Each solution is known to be either acidic or alkaline. The results are shown.
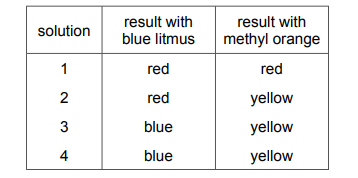
Which statement is correct?
A Solutions 1 and 4 are acidic.
B Solutions 1 and 2 are alkaline.
C Solutions 3 and 4 are acidic.
D Solutions 3 and 4 are alkaline.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (D)
Question 21
Which statement about sulfur dioxide or calcium oxide is correct?
A Calcium oxide is an acid.
B Calcium oxide turns thymolphthalein yellow.
C Sulfur dioxide is a base.
D Sulfur dioxide turns thymolphthalein colourless.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 7.2: Oxides — Classify oxides as acidic (including SO₂) or basic (including CaO) related to metallic and non-metallic character (Core)
▶️ Answer/Explanation
✅ Answer: (D)
Question 22
Which substances can be used to make pure crystals of sodium sulfate?
A. potassium sulfate and sodium hydroxide
B. sodium carbonate and sulfuric acid
C. sodium nitrate and magnesium sulfate
D. sulfuric acid and sodium chloride
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B)
Question 23
Which statements describe changes that occur from left to right across a period of the Periodic Table?
1 The atomic number of the elements increases.
2 The metallic character of the elements decreases.
3 The physical state of the elements changes from gas to solid.
A 2 only
B 1 and 2
C 1 and 3
D 2 and 3
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B) 1 and 2
Question 24
Part of the Periodic Table is shown. Which element is the most reactive non-metal?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: D
Question 25
An alloy contains aluminium, copper, magnesium, manganese, silver and zirconium. Which row identifies the number of transition elements in the alloy and the relative density of the transition elements compared to sodium?
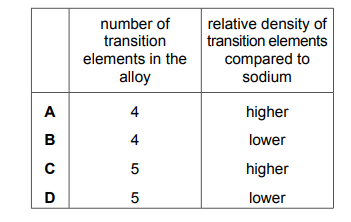
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 9.1 (Properties of Metals): Compare the general physical properties of metals and non-metals, including density.
▶️ Answer/Explanation
✅ Answer: (A)
Question 26
Which statement about the halogens and their compounds is correct?
A The colour of the element gets lighter going down Group VII.
B The elements get less dense going down Group VII.
C When chlorine is added to sodium iodide solution, iodine is formed.
D When iodine is added to sodium bromide solution, bromine is formed.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed Solution: In Group VII (halogens), reactivity decreases down the group. A more reactive halogen (chlorine) will displace a less reactive halogen (iodine) from its salt (sodium iodide), producing iodine. Thus, when chlorine is added to sodium iodide solution, iodine is formed (solution turns brown). Option A is incorrect because colours get darker (paler yellow-green to dark grey-black). Option B is incorrect because density increases down the group. Option D is incorrect because iodine is less reactive than bromine and cannot displace it.
Question 27
Which row describes an element in Group VIII of the Periodic Table?
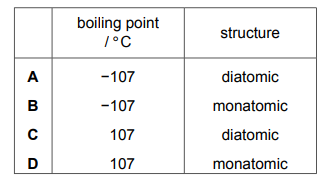
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: B
Question 28
Magnesium reacts with dilute hydrochloric acid. Which gas is given off in this reaction?
A carbon dioxide
B chlorine
C hydrogen
D oxygen
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 7.1: Characteristic properties of acids — Reaction of acids with metals.
▶️ Answer/Explanation
✅ Answer: C (hydrogen)
Question 29
Which statement about aluminium is correct?
A Aluminium is easy to extract from its ore because it is near the bottom of the reactivity series.
B Aluminium is formed when aluminium oxide is heated with carbon.
C Bauxite is an important ore of aluminium.
D Hematite is an important ore of aluminium.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C)
Question 30
Which statement shows that a liquid is pure water?
A It boils at 100°C.
B It has a pH value of 7.
C It turns blue cobalt(II) chloride pink.
D It turns white copper(II) sulfate blue
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 10.1: Water — Describe chemical tests for the presence of water using anhydrous cobalt(II) chloride and anhydrous copper(II) sulfate; Explain how to test for the purity of water using melting point and boiling point.
▶️ Answer/Explanation
✅ Answer: (A)
Question 31
Which compound can be added to ammonium sulfate to make an NPK fertiliser?
A. \((NH_4)_3PO_4\)
B. \(KNO_3\)
C. \(K_3PO_4\)
D. \(CO(NH_2)_2\)
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C)
Question 32
Four gases found in air are listed.
1 carbon dioxide
2 carbon monoxide
3 methane
4 sulfur dioxide
Which gases lead directly to global warming when their concentrations are increased?
A 1 and 3
B 1 and 4
C 2 and 3
D 2 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: A
Question 33
The structures of four organic compounds are shown.
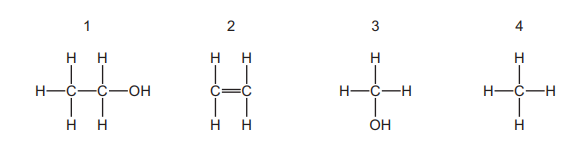
Which compounds are members of the same homologous series?
A 1, 2 and 3
B 1 and 2 only
C 1 and 3 only
D 3 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 11.2: Naming organic compounds — State the type of compound present given a displayed formula.
▶️ Answer/Explanation
✅ Answer: (C)
Question 34
Which of the statements about propene are correct?
1 Propene contains only single bonds.
2 Propene decolourises aqueous bromine.
3 Propene is obtained by cracking large alkanes.
4 Propene is a hydrocarbon.
A 1, 3 and 4
B 1 only
C 2, 3 and 4
D 2 and 4 only
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C) 2, 3 and 4
Question 35
Petroleum is a mixture. Which method is used to separate petroleum into its components?
A chromatography
B cracking
C filtration
D fractional distillation
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: D (fractional distillation)
Question 36
The equation for a reaction that produces ethanol is shown.
ethene + steam → ethanol
Which type of reaction does the equation represent?
A. addition
B. combustion
C. fermentation
D. polymerisation
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 11.6: Alcohols — Describe the manufacture of ethanol by catalytic addition of steam to ethene at 300°C and 6000 kPa in the presence of an acid catalyst (Core)
▶️ Answer/Explanation
✅ Answer: (A)
Question 37
Four statements about ethene or poly(ethene) are listed.
1 Poly(ethene) is produced by an addition reaction.
2 Ethene is a monomer.
3 Poly(ethene) is a monomer.
4 Poly(ethene) decolourises aqueous bromine.
Which statements are correct?
A 1 and 2
B 1 and 3
C 2 and 4
D 3 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A) 1 and 2
Question 38
Sea water contains dissolved sodium chloride. Which method is used to obtain pure water from sea water?
A. chromatography
B. distillation
C. evaporation
D. filtration
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B)
Question 39
Which piece of apparatus is used to measure exactly 21.4 cm³ of water?
A. a 25 cm³ beaker
B. a 25 cm³ pipette
C. a 50 cm³ burette
D. a 50 cm³ measuring cylinder
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 4: Details of the assessment — Candidates should be able to take readings from apparatus with appropriate precision and select the most appropriate apparatus or method for the task.
▶️ Answer/Explanation
✅ Answer: (C)
Question 40
An aqueous solution of compound M is tested. The results are shown.
• a lilac colour in a flame test
• a white precipitate when tested with acidified barium nitrate
What is compound M?
A copper(II) chloride
B copper(II) sulfate
C potassium carbonate
D potassium sulfate
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: D (potassium sulfate)
