Question 1
Which two processes are required to change ice into steam?
A boiling and melting
B boiling and freezing
C condensing and melting
D condensing and freezing
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A)
Question 2
Which row describes how the volume of a gas changes when the temperature increases, or when the pressure increases?
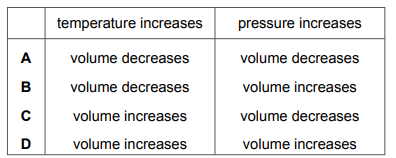
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: C
Question 3
Four substances in a mixture are listed.
• calcium nitrate
• iron(II) sulfate
• oxygen
• water
Which statement describes the mixture?
A It contains 6 elements.
B It contains 3 compounds and 1 element.
C It contains 2 compounds and 2 elements.
D It contains 4 compounds.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B)
“`
Question 4
An ion is represented by the symbol \( _{8}^{18}\textrm{O}^{2-}\). Which statements about this ion are correct?
1 The ion contains 8 electrons.
2 The ion contains 10 neutrons.
3 The ion contains 8 protons.
A 1 and 2
B 1 and 3
C 2 only
D 2 and 3
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 2.3: Isotopes — Interpret and use symbols for atoms and ions.
▶️ Answer/Explanation
✅ Answer: D
Question 5
What is the meaning of the term nucleon number?
A the number of neutrons in the nucleus of an atom
B the number of protons in the nucleus of an atom
C the total number of protons and electrons in the nucleus of an atom
D the total number of protons and neutrons in the nucleus of an atom
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (D) the total number of protons and neutrons in the nucleus of an atom
Question 6
Which row describes the electrical conductivity of covalent and ionic compounds when solid and when molten?
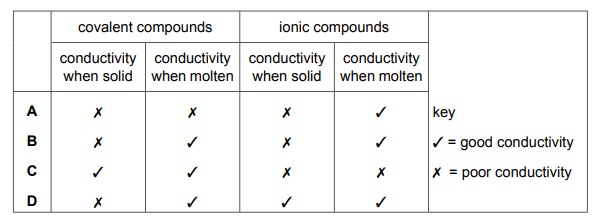
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 2.4: Ions and ionic bonds — Describe the properties of ionic compounds: high melting points and boiling points, good electrical conductivity when aqueous or molten and poor when solid (Core)
▶️ Answer/Explanation
✅ Answer: (A)
Question 7
Which statement about the bonding in sodium chloride is correct?
A The sodium and chlorine atoms share pairs of electrons.
B The chlorine atoms give electrons to the sodium atoms to form positive and negative ions.
C There is covalent bonding between sodium and chlorine.
D The positive and negative ions have noble gas electronic configurations.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: D
Question 8
The diagram shows the arrangement of carbon atoms in a giant covalent structure.

Which row identifies the substance and describes a use of this substance?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B)
“`
Question 9
Which statement is the correct definition for molecular formula?
A an atom or group of atoms that determine the chemical properties of a compound
B a structure which shows all the atoms and all the bonds in a compound
C the number and arrangement of different atoms in one gram of a compound
D the number and type of different atoms in one molecule of a compound
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (D)
Question 10
A compound, T, has the formula \(CH_3Cl\). Three statements about this compound are listed.
1 A molecule of the compound contains five atoms.
2 A molecule of the compound contains five different elements.
3 The relative molecular mass of the compound is 50.5.
Which statements are correct?
A 1, 2 and 3
B 1 and 2 only
C 1 and 3 only
D 2 and 3 only
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 3.2: Relative masses of atoms and molecules — Define relative molecular mass, \(M_{r}\) as the sum of the relative atomic masses (Core).
• Topic 2.2: Atomic structure and the Periodic Table — Interpret and use symbols for atoms and ions (Core).
▶️ Answer/Explanation
✅ Answer: (C) 1 and 3 only
“`
Question 11
Iron water taps are often electroplated with a layer of chromium. Which statements explain why iron water taps are electroplated?
1 It improves the appearance of the taps.
2 It increases the strength of the taps.
3 It prevents the corrosion of the taps.
4 It improves the electrical conductivity of the taps.
A 1 and 3
B 1 and 4
C 2 and 3
D 2 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A) 1 and 3
Question 12
Molten caesium bromide is electrolysed using inert electrodes. Which row identifies the product at each electrode?
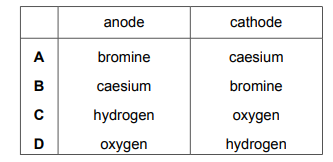
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: A
Question 13
Which equation represents the overall reaction in a hydrogen–oxygen fuel cell?
A \(4H + O_2 → 2H_2O\)
B \(2H_2 + O_2 → 2H_2O\)
C \(H_2 + O_2 → H_2O\)
D \(2H + O → H_2O\)
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B)
Question 14
Which statements about endothermic reactions are correct?
1 The energy of the products is greater than the energy of the reactants.
2 The energy of the reactants is greater than the energy of the products.
3 The temperature of the surroundings increases during the reaction.
4 The temperature of the surroundings decreases during the reaction.
A 1 and 3
B 1 and 4
C 2 and 3
D 2 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B)
Question 15
Which process is a physical change?
A firework exploding
B burning wood
C chocolate melting
D iron rusting
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C) chocolate melting
Question 16
Magnesium reacts with dilute hydrochloric acid, HCl, to produce hydrogen gas. Which row identifies the reaction conditions that give the fastest rate of reaction?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B)
Question 17
Which reaction produces a white-coloured substance?
A. adding water to anhydrous cobalt(II) chloride
B. adding water to anhydrous copper(II) sulfate
C. heating hydrated cobalt(II) chloride
D. heating hydrated copper(II) sulfate
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 10.1: Water — Describe chemical tests for the presence of water using anhydrous cobalt(II) chloride and anhydrous copper(II) sulfate (Core).
▶️ Answer/Explanation
✅ Answer: (D)
Question 18
In a blast furnace, iron is extracted when iron(III) oxide reacts with carbon monoxide. The equation is shown.
\(Fe_2O_3 + 3CO \rightarrow 2Fe + 3CO_2\)
Which substance is oxidised and which is reduced in this reaction?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
In the reaction \(Fe_2O_3 + 3CO \rightarrow 2Fe + 3CO_2\), oxidation is the gain of oxygen and reduction is the loss of oxygen. Carbon monoxide (CO) gains an oxygen atom to become carbon dioxide (CO₂), so it is oxidised. Iron(III) oxide (Fe₂O₃) loses its oxygen to become elemental iron (Fe), so it is reduced.
✅ Answer: A (Carbon monoxide is oxidised, Iron(III) oxide is reduced)
Question 19
Which row shows the colours of litmus and methyl orange with solutions of acids or bases?
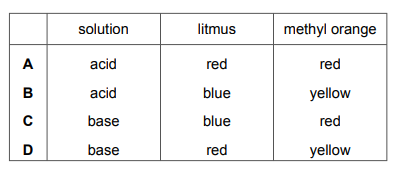
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A)
Question 20
Aqueous sodium hydroxide is reacted with excess dilute hydrochloric acid. Which ion causes the resulting mixture to be acidic?
A. \(Na^+\)
B. \(H^+\)
C. \(OH^-\)
D. \(Cl^-\)
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B)
Question 21
Universal indicator is added to an aqueous solution of oxide X. The indicator changes colour from green to red. What is X?
A. MgO
B. CaO
C. K2O
D. NO2
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 7.2: Oxides — Classify oxides as acidic (including NO2) or basic (including MgO, CaO, K2O).
▶️ Answer/Explanation
Question 22
Which rows identify two aqueous salts which react together to produce a precipitate?

A 1 and 4
B 1 and 3
C 2 and 3
D 2 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 12.5: Identification of ions and gases — Tests for aqueous cations and anions using precipitation reactions
▶️ Answer/Explanation
✅ Answer: (B) 1 and 3
Question 23
Which statements about elements in the Periodic Table are correct?
1. Elements in the same group have the same number of electrons in their outer shell.
2. Elements in the same period have the same number of occupied electron shells.
3. The elements are arranged in order of their atomic mass.
4. Every period contains eight elements.
A. 1 and 2
B. 1 and 3
C. 2 and 4
D. 3 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 8.1: Explain similarities in the chemical properties of elements in the same group in terms of their electronic configuration (Core)
▶️ Answer/Explanation
✅ Answer: (A)
Question 24
Four mixtures each contain a halogen and an aqueous sodium halide. Which row describes what happens in the mixtures shown?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 6.4: Redox — Identify redox reactions as involving gain and loss of electrons (Supplement)
• Topic 2.5: Simple molecules and covalent bonds — Describe the formation of covalent bonds in halogen molecules (Core)
▶️ Answer/Explanation
✅ Answer: C
Question 25
Which row describes a transition element?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: C
Question 26
Which statement about the use of metals is correct?
A. Aluminium has a high strength and high density so is used to make aircraft.
B. Copper has a low melting point so is used in electrical wiring.
C. Aluminium is resistant to corrosion so is used in food containers.
D. Zinc is used to make the alloy stainless steel which is used in cutlery.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed Explanation: Aluminium forms a thin, protective oxide layer that prevents further reaction with air and moisture, making it highly resistant to corrosion. This property is ideal for food containers, ensuring that the metal does not react with or contaminate the food. Option A is false because aluminium is used in aircraft due to its low density, not high density. Option B is false because copper is used for wiring due to its excellent electrical conductivity and ductility, not a low melting point. Option D is false because stainless steel is an alloy of iron with chromium and nickel; zinc is used for galvanising iron to prevent rusting (sacrificial protection).
Question 27
Which statements explain why stainless steel is used in cutlery?
1 It is resistant to rusting.
2 It is a hard material.
3 It is a pure metal.
A 1, 2 and 3
B 1 and 2 only
C 1 and 3 only
D 2 and 3 only
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: B (1 and 2 only)
Question 28
Four different metals are separately mixed with an equal volume of dilute hydrochloric acid. The table shows the rate of effervescence for each metal.

What is the order of reactivity of the four metals starting with the most reactive?
A iron → magnesium → calcium → copper
B magnesium → calcium → copper → iron
C copper → iron → magnesium → calcium
D calcium → magnesium → iron → copper
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
The rate of effervescence (bubbling) indicates how quickly hydrogen gas is produced, which corresponds to the metal’s reactivity. A very fast rate indicates a more reactive metal, while no reaction indicates a metal that is less reactive than hydrogen. Calcium shows the fastest rate, followed by magnesium, then iron (slowest of the three that react), and copper produces no bubbles. Therefore, the correct order starting with the most reactive is calcium → magnesium → iron → copper.
✅ Answer: D
Question 29
Which statement about the rusting of iron is correct?
A The rusting of iron forms hydrated iron(II) oxide.
B Barrier methods prevent rusting by excluding nitrogen and water.
C A piece of iron submerged in water will not rust.
D Coating with plastic is a barrier method that prevents iron rusting.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: D
Question 30
Water is extracted from a river for use in a domestic water supply. Some treatments for domestic water are listed.
• chlorination
• sedimentation and filtration
• treatment with carbon
Which statement about these treatments is correct?
A. Filtration is used to remove soluble substances.
B. Treatment with carbon is used to remove unpleasant odours.
C. Chlorination is used to remove unpleasant tastes.
D. Sedimentation is used to kill microbes.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B)
Question 31
A farmer knows his soil needs phosphorus and potassium. He has a choice of four fertilisers.
1 \(NH_4NO_3\)
2 \((NH_4)_3PO_4\)
3 \(KNO_3\)
4 \((NH_2)_2CO\)
Which fertilisers should he use?
A 1 and 2
B 1 and 4
C 2 and 3
D 3 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C) 2 and 3
Question 32
Which strategies are useful in reducing the production of acid rain?
1 planting trees
2 using catalytic converters in motor vehicles
3 reducing livestock farming
4 using low-sulfur fuels
A 1 and 3
B 1 and 4
C 2 and 3
D 2 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (D)
Question 33
The structures of four organic compounds, W, X, Y and Z, are shown.
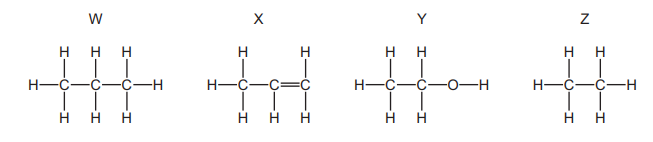
Which compounds are members of the same homologous series?
A W and X
B W and Z
C X and Y
D Y and Z
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 11.2: Naming organic compounds — State the type of compound present, given a chemical name ending in -ane, -ene, -ol, or -oic acid or from a molecular formula or displayed formula.
▶️ Answer/Explanation
✅ Answer: (B) W and Z
Question 34
Which molecular formula represents an alkene?
A. \(C_2H_6O\)
B. \(C_2H_6\)
C. \(CH_4\)
D. \(C_3H_6\)
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (D)
Question 35
Which row identifies the petroleum fractions used to reduce the friction between metal parts in engines and as a fuel in cars?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: D
Question 36
Four fuels are listed.
1 ethanol
2 coal
3 hydrogen
4 natural gas
Which fuels are fossil fuels?
A 1, 2 and 3
B 1 and 3 only
C 2 and 4
D 4 only
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 10.3: Air quality and climate — Source of carbon dioxide from complete combustion of carbon-containing fuels (Core)
▶️ Answer/Explanation
✅ Answer: (C)
Question 37
Aqueous bromine is added to two test-tubes. Excess hexene is added to one test-tube. Excess hexane is added to the second test-tube. Which row describes the observation in each test-tube?
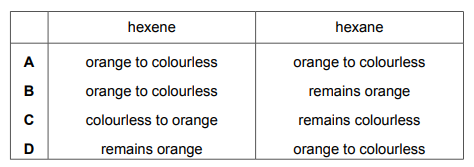
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 11.4: Alkanes — Describe the properties of alkanes as being generally unreactive, except in terms of combustion and substitution by chlorine (Core)
▶️ Answer/Explanation
✅ Correct Answer: B (Hexene: decolourises; Hexane: no change)
Question 38
The structure of a compound, G, is shown. G is in the same homologous series as ethanoic acid.

Which row describes an aqueous solution of G? 
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 11.7: Carboxylic acids — Describe the reaction of ethanoic acid with metals, bases, carbonates (Core).
• Topic 7.1 (Supplement): Define a weak acid as an acid that is partially dissociated in aqueous solution.
▶️ Answer/Explanation
✅ Answer: (C) — The row that correctly describes the solution as a weak acid with a pH in the range 4–6.
Question 39
When zinc reacts with dilute sulfuric acid, hydrogen gas is produced. Which apparatus is needed to investigate the effect of temperature on the rate of this reaction?
1 thermometer
2 stop-watch
3 volumetric pipette
4 gas syringe
A 1 and 2
B 1 and 4
C 2 and 3
D 3 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B) 1 and 4
Question 40
Two tests are carried out on substance Z.
test 1 A flame test produces a red flame.
test 2 Z is dissolved in water and dilute nitric acid is added, followed by aqueous silver nitrate. A yellow precipitate is produced.
What is substance Z?
A lithium bromide
B lithium iodide
C sodium bromide
D sodium iodide
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: B (lithium iodide)
