Question 1
Sodium chloride is a liquid at 900°C. Which row describes the arrangement and the motion of the particles in sodium chloride at 900°C?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: D
Question 2
Hydrogen chloride gas, HCl, reacts with ammonia gas, NH₃, to form solid ammonium chloride. The apparatus is set up as shown. After a few minutes, a white cloud of solid ammonium chloride forms where the two gases meet.

The experiment is repeated using hydrogen bromide gas, HBr, in place of hydrogen chloride. How far along the tube does the white cloud of solid ammonium bromide form?
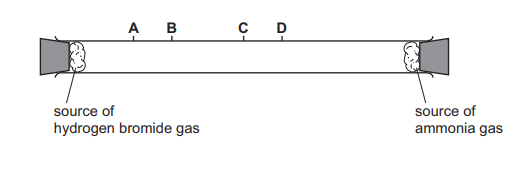
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A) Position A
Detailed solution: The rate of diffusion of a gas is inversely proportional to the square root of its relative molecular mass (Graham’s law). HBr (Mr ≈ 81) is heavier than HCl (Mr ≈ 36.5), so it diffuses more slowly. The ammonia (NH₃, Mr ≈ 17) diffuses fastest. The reaction front forms where the two gases meet. Since HBr is slower, NH₃ travels further down the tube before meeting HBr, meaning the cloud forms closer to the HBr end — position A.
Question 3
Substances P and Q both conduct electricity. P is a mixture of two different types of atom. Q is made of only one type of atom. Which row describes P and Q?
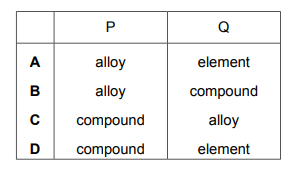
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 2.7: Metallic bonding — Describe metallic bonding as the electrostatic attraction between positive ions and delocalised electrons (Supplement); Explain good electrical conductivity (Supplement)
• Topic 9.1: Properties of metals — Compare the general physical properties of metals and non‑metals, including electrical conductivity (Core)
▶️ Answer/Explanation
✅ Answer: (A)
Question 4
An atom of element R contains 15 protons, 16 neutrons and 15 electrons. What is R?
A gallium
B phosphorus
C sulfur
D zinc
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: B (phosphorus)
Question 5
Which molecule contains a double covalent bond between two atoms of the same element?
A. carbon dioxide
B. ethanol
C. ethene
D. nitrogen
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 11.5: Alkenes — State that the bonding in alkenes includes a double carbon-carbon covalent bond (unsaturated hydrocarbons).
▶️ Answer/Explanation
Ethene (C₂H₄) contains a carbon-carbon double covalent bond (C=C), where the bond is formed between two atoms of the same element (carbon).
• Carbon dioxide (A) has double bonds, but between carbon and oxygen (different elements).
• Ethanol (B) contains only single covalent bonds (C–C, C–H, C–O, O–H).
• Nitrogen (D) contains a triple covalent bond (N≡N), not a double bond.
Question 6
Silicon(IV) oxide is a covalently bonded compound. Which statements are correct?
1 Silicon atoms form four single bonds in silicon(IV) oxide.
2 Oxygen atoms form two double bonds in silicon(IV) oxide.
3 Silicon(IV) oxide has a high melting point.
4 Silicon(IV) oxide contains one silicon atom and four oxygen atoms.
A 1 and 2
B 1 and 3
C 2 and 3
D 3 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 2.6: Giant covalent structures — Describe the similarity in properties between diamond and silicon(IV) oxide, related to their structures (Supplement)
▶️ Answer/Explanation
✅ Answer: (B) 1 and 3
Question 7
The equation for the reaction of iron(III) oxide with carbon monoxide is shown.
\(Fe_2O_3 + 3CO → 2Fe + 3CO_2\)
What is the percentage yield of iron when 16.8 g of carbon monoxide reacts completely with iron(III) oxide to form 8.96g of iron?
A 26.7%
B 40.0%
C 53.3%
D 80.0%
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 3.3: Calculate percentage yield, given appropriate data (Supplement)
▶️ Answer/Explanation
Step 2: From the equation, 3 mol CO → 2 mol Fe, so moles of Fe (theoretical) = 0.600 × (2/3) = 0.400 mol.
Step 3: Mass of Fe (theoretical) = 0.400 × 56 = 22.4 g. Percentage yield = (8.96 / 22.4) × 100% = 40.0%.
✅ Answer: (B) 40.0%
Question 8
What is the volume of 14.5g of gaseous butane, C₄H₁₀, at room temperature and pressure?
A. 96.0 cm³
B. 6.0 cm³
C. 96.0 dm³
D. 6.0 dm³
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: D
Question 9
Aqueous copper(II) sulfate is electrolysed using copper electrodes.
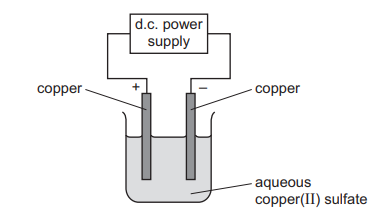
Which statement describes what happens during the electrolysis?
A Copper atoms gain electrons at the cathode and copper(II) ions lose electrons at the anode.
B Electrons move in the external circuit from the positive electrode to the negative electrode.
C Copper(II) ions move through the electrolyte from the cathode to the anode.
D Copper is formed at the cathode and oxygen is formed at the anode.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 4.1: Electrolysis — Construct ionic half-equations for reactions at the anode (to show oxidation) and at the cathode (to show reduction) (Supplement)
▶️ Answer/Explanation
With copper electrodes, copper atoms at the anode lose electrons (oxidize) to form Cu²⁺ ions, which go into solution; at the cathode, Cu²⁺ ions gain electrons (reduce) to deposit copper metal. Electrons always flow in the external circuit from the negative electrode (anode) to the positive electrode (cathode), making option B correct. Option A has incorrect charges (copper atoms lose electrons at anode). Option C is wrong because Cu²⁺ ions move from anode to cathode, not cathode to anode. Option D is incomplete as no oxygen forms; the anode dissolves, producing copper ions.
Question 10
Electrolysis is carried out on concentrated aqueous potassium bromide using inert electrodes. Which products are formed at the anode and the cathode?
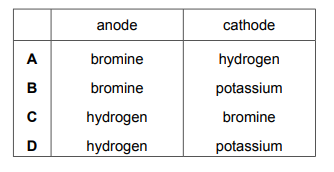
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A)
“`
Question 11
Hydrogen–oxygen fuel cells and gasoline are each used to power cars. Which statement describes an advantage of using hydrogen–oxygen fuel cells in cars in comparison with gasoline engines?
A. Hydrogen is a non-renewable resource.
B. Hydrogen is produced during the fractional distillation of petroleum.
C. Hydrogen–oxygen fuel cells do not release carbon dioxide.
D. Hydrogen–oxygen fuel cells need electricity to work.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C)
Question 12
Plant cells use energy from sunlight for photosynthesis. Which row describes and explains the energy change that occurs?
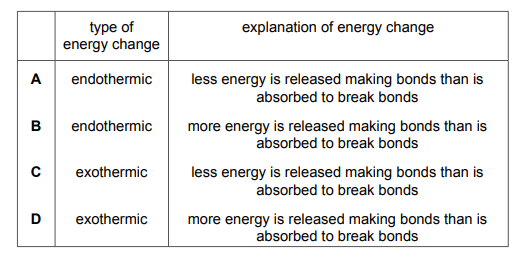
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A)
Question 13
Two different experiments are done to find the enthalpy change, ΔH, of each reaction. The temperature of each reaction mixture is measured at the beginning of the reaction and after 3 minutes.
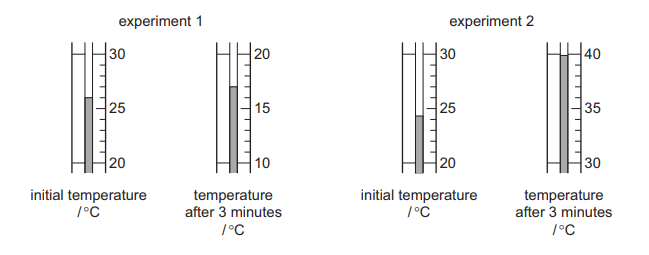
Which row gives the correct sign for the value of ΔH for each experiment and identifies if the reaction is endothermic or exothermic? 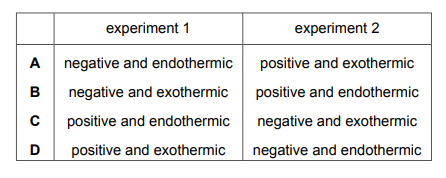
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C)
Question 14
The reaction pathway diagram for an endothermic reaction is shown. u, v and w are known energy values
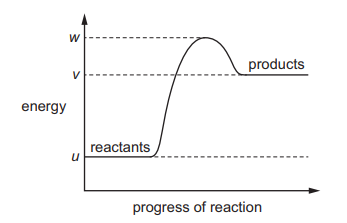
Which diagram shows the reaction pathway diagram when a catalyst is used in the reaction? 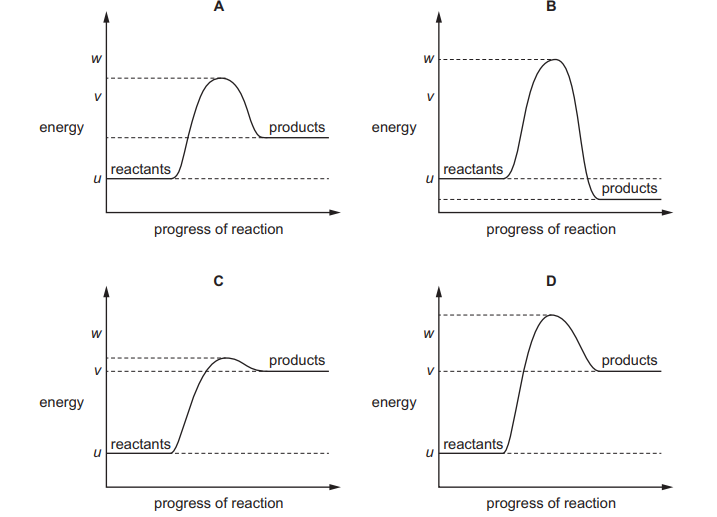
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C)
Question 15
Which process is a physical change?
A burning a piece of magnesium
B reacting calcium carbonate with hydrochloric acid
C melting an ice cube
D the rusting of an iron nail
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C)
Question 16
The Contact process is used to convert sulfur dioxide to sulfur trioxide. Vanadium(V) oxide is the catalyst in this process.
![]()
The forward reaction in this equilibrium is exothermic. Which statements about this process are correct?
1 The catalyst increases the rate of both the forward and backward reactions.
2 A low pressure increases the yield of sulfur trioxide.
3 A low pressure is used to keep the costs low.
4 A high temperature increases the yield of sulfur trioxide.
A 1 and 2
B 1 and 3
C 2 and 4
D 3 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 6.2: Rate of reaction — State that a catalyst increases rate of reaction and is unchanged; describe effect of pressure (Core and Supplement)
▶️ Answer/Explanation
✅ Answer: (B) 1 and 3
Question 17
The equation for the decomposition of hydrogen peroxide, \(H_2O_2\), is shown.
![]()
In an experiment, the total volume of oxygen produced is \(100cm^3\). The experiment is repeated using 1.00g of a solid catalyst. All other conditions remain the same. Which row describes the total volume of oxygen and the mass of the catalyst at the end of the second experiment?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 6.2 (Supplement): State that a catalyst decreases the activation energy, \(E_{\mathrm{a}}\) of a reaction.
▶️ Answer/Explanation
✅ Answer: (B) — 100 cm³ of oxygen and 1.00 g of catalyst.
“`
Question 18
Nitrogen reacts with hydrogen to form ammonia in the presence of an iron catalyst. The reaction is reversible.
![]()
Which statement about this reaction is correct?
A. The iron catalyst decreases the activation energy of only the forward reaction.
B. When equilibrium is reached, the forward reaction has stopped.
C. When the pressure changes, the concentration of ammonia at equilibrium remains constant.
D. Nitrogen and hydrogen never completely convert to ammonia.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: D
Question 19
The equation for a redox reaction is shown.
![]()
Potassium iodide is …… 1 …… agent in this reaction because iodide ions …… 2 …… electrons.
Which words complete gaps 1 and 2?
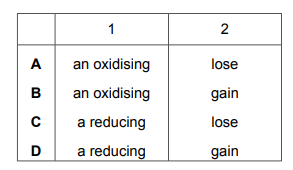
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 6.4 (Supplement): Define oxidation in terms of loss of electrons and reduction in terms of gain of electrons.
▶️ Answer/Explanation
✅ Answer: (C) reducing / lose
Question 20
Element E is in Group II of the Periodic Table. Which row describes element E and its oxide?
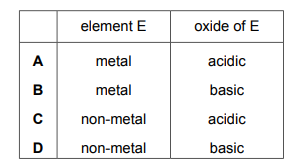
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 8.2: Group I properties — Predict the properties of other elements in a group (applied to Group II by extension).
• Topic 7.2: Oxides — Classify oxides as acidic or basic related to metallic and non-metallic character.
▶️ Answer/Explanation
✅ Answer: (B)
Question 21
Lead(II) iodide is formed as a precipitate in the reaction shown.
![]()
Which method is used to separate the lead(II) iodide from the mixture?
A crystallisation
B distillation
C evaporation
D filtration
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 7.3: Preparation of salts — Describe the preparation of insoluble salts by precipitation (Supplement).
▶️ Answer/Explanation
✅ Answer: D (filtration)
“`
Question 22
Which statement explains why sulfur, S, has similar chemical properties to selenium, Se?
A. They both have the same number of electrons in their outer electron shell.
B. They are both solids at room temperature and pressure.
C. They are both non-metals.
D. They both form negative ions.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A)
Question 23
Atoms of sodium, rubidium and element Q each have one outer shell electron. Some properties of these elements are shown.
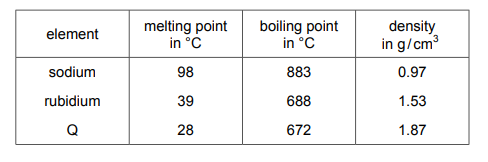
What is Q?
A hydrogen
B lithium
C potassium
D caesium
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (D) caesium
Question 24
Which statement about elements in Group VIII of the Periodic Table is correct?
A They all have a full outer shell of electrons.
B They all react with Group I elements to form ionic compounds.
C They are all diatomic molecules.
D They are all liquids at room temperature and pressure.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A)
Question 25
The electrical conductivity of magnesium is tested. Magnesium is then added to dilute sulfuric acid. A gas, W, is produced. Which row describes the electrical conductivity of magnesium and identifies W?
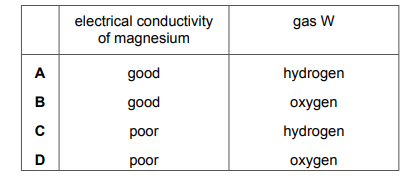
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 7.1: The characteristic properties of acids and bases — Describe the characteristic properties of acids in terms of their reactions with metals (Core).
• Topic 6.4: Redox — Identify redox reactions as reactions involving gain and loss of oxygen (Core).
▶️ Answer/Explanation
Magnesium is a metal, and metals have delocalized electrons that can move freely, making them good conductors of electricity in both solid and molten states. When magnesium reacts with dilute sulfuric acid (a typical metal-acid reaction), it displaces hydrogen, producing hydrogen gas (W) and magnesium sulfate. Therefore, the correct row shows “good” conductivity and the gas as “hydrogen”.
Question 26
Aluminium metal is extracted from its purified ore by electrolysis. Which statement about the electrolyte in this process is correct?
A. The electrolyte is purified molten bauxite only.
B. The electrolyte is purified bauxite dissolved in molten cryolite.
C. The electrolyte is purified molten cryolite only.
D. The electrolyte is purified cryolite dissolved in molten bauxite.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Purified aluminium oxide (from bauxite) has a very high melting point (over 2000°C), making electrolysis of the molten oxide alone impractical and expensive. It is therefore dissolved in molten cryolite (sodium aluminium fluoride), which lowers the operating temperature to about 950°C while maintaining electrical conductivity. Option B correctly describes this mixture as the electrolyte; the other options are incorrect because pure bauxite or pure cryolite are not used alone as the electrolyte in the Hall-Héroult process.
✅ Answer: (B)
Question 27
Part of the reactivity series is shown.

Which metals are represented by X and Y?
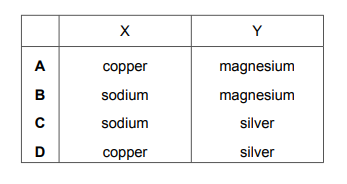
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✔️ Correct Option: B (X = Zinc, Y = Iron)
The reactivity series provided shows Magnesium (most reactive) at the top and Copper/Silver (least reactive) at the bottom. Comparing the positions: Zinc is placed between Magnesium and Iron in the standard reactivity series (Mg > Zn > Fe > Cu). Iron is less reactive than Zinc but more reactive than Copper. Therefore, the only pair that fits the sequence Mg → X → Y → Cu → Ag is X = Zinc and Y = Iron. Option A has Calcium which is more reactive than Magnesium; Option C has Aluminium which is between Mg and Zn but then has Lead incorrectly ordered; Option D has Tin and Lead, which are both less reactive than Iron but would be out of order.
✅ Answer: (B)
Question 28
Some substances found in water extracted from a river are listed.
1 plastics
2 nitrates
3 oxygen
Which substances are harmful to aquatic life?
A. 1 and 2
B. 1 and 3
C. 2 only
D. 3 only
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A) 1 and 2
Question 29
Car engines which use gasoline as a fuel produce oxides of nitrogen. Oxides of nitrogen are removed from the exhaust gases. Which statements about the formation or removal of oxides of nitrogen are correct?
1 Gasoline reacts with nitrogen in the air to produce oxides of nitrogen.
2 Gasoline contains nitrogen.
3 Nitrogen and oxygen react at high temperatures to produce oxides of nitrogen.
4 Nitrogen monoxide, NO, reacts with carbon monoxide, CO, in a catalytic converter.
A 1 and 2
B 1 and 3
C 2 and 4
D 3 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Statements 1 and 2 are incorrect because gasoline is a hydrocarbon (contains carbon and hydrogen, not nitrogen). The intense heat inside a car engine causes nitrogen and oxygen from the air to react directly, forming oxides of nitrogen (Statement 3). Once formed, these pollutants are removed in a catalytic converter where nitrogen monoxide (NO) reacts with carbon monoxide (CO) to form harmless nitrogen and carbon dioxide (Statement 4).
Question 30
Which structures represent a pair of structural isomers?
1 \(CH_3CH_2CH_2CH_2CH_2CH_3\)
2 \(CH_3CH_2CH_2CH_2CH(CH_3)_2\)
3 \(CH_3CH(CH_3)CH_2CH_2CH_3\)
4 \(CH_3CH(CH_3)CH_2CH_2CH_2CH_3\)
A 1 and 2
B 1 and 3
C 2 and 4
D 3 and 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: B (1 and 3)
Question 31
Alkanes are a homologous series of hydrocarbons. The table shows the names and boiling points of the first four members of this series.
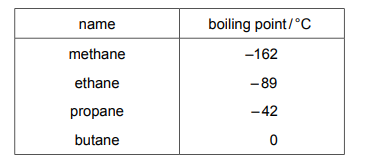
Pentane is the next member of the series. Which row gives the molecular formula and the boiling point of pentane? 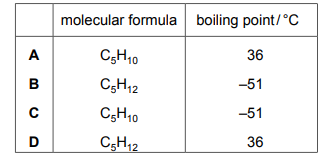
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 11.4: Alkanes — State that alkanes are saturated hydrocarbons with single covalent bonds (Core)
• Topic 11.1: Formulae, functional groups and terminology — General formula of alkanes CnH2n+2 (Core)
▶️ Answer/Explanation
✅ Answer: D
Question 32
Which displayed formula represents the ester formed by the reaction of propan-1-ol with ethanoic acid?
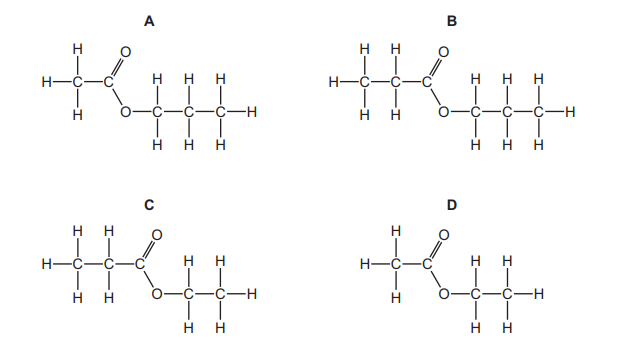
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 11.2: Naming organic compounds — Name and draw displayed formulae of unbranched esters made from unbranched alcohols and carboxylic acids, each containing up to four carbon atoms (Supplement)
▶️ Answer/Explanation
✅ Answer: A
Question 33
Ethanol can be manufactured by fermentation and by the catalytic addition of steam to ethene. Which statement identifies an advantage of using one of these methods?
A. Catalytic addition requires a high temperature and pressure.
B. Ethanol produced by fermentation is extracted by distillation.
C. Fermentation is a batch process.
D. The raw material in fermentation is a renewable resource.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (D)
Question 34
Which statement about carboxylic acids is correct?
A They react with alkalis to form a salt and water.
B They react with metals to form a salt and water.
C They react with metal carbonates to form a salt, water and hydrogen.
D The general formula for carboxylic acids is \(C_nH_{2n+1O}H\).
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 7.1: The characteristic properties of acids and bases — Describe neutralisation (Core); Define acids as proton donors (Supplement)
▶️ Answer/Explanation
✅ Answer: A
Question 35
The structure of a polymer is shown.
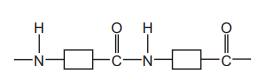
Which statement about this polymer is correct?
A. Alkenes are polymerised to make the polymer.
B. It is a polyester.
C. It is an addition polymer.
D. Water is produced when the polymer is made.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (D) Water is produced when the polymer is made.
Question 36
Methane undergoes substitution reactions with chlorine and complete combustion with excess oxygen. Which row about the two reactions is correct?

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 6.2: Rate of reaction — Describe the effect on the rate of reaction of changing temperature (Core)
▶️ Answer/Explanation
The substitution reaction of methane with chlorine requires ultraviolet light for initiation, making it photochemical. Complete combustion of methane with excess oxygen is highly exothermic, and a catalyst is not required (the flame itself provides the activation energy). Row D correctly states the reaction type (substitution) and that combustion is not catalytic. Rows A and B are incorrect because combustion is addition (not substitution) but still not catalytic. Row C incorrectly claims substitution is not photochemical.
Question 37
What is used to identify the end-point of an acid–base titration?
A. balance
B. measuring cylinder
C. indicator
D. volumetric pipette
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (C)
Question 38
Four pure substances, P, Q, R and S, are tested using chromatography. The same solvent is used each time. The table shows the distance moved by each substance and by the solvent from the baseline.
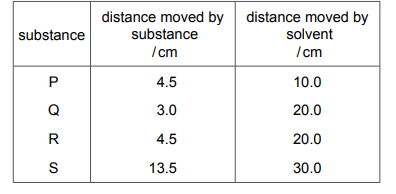
Which two substances are identical?
A P and R
B P and S
C Q and R
D Q and S
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (B) P and S
Question 39
A substance is tested with three different reagents. Which row shows the results obtained with aqueous iron(II) nitrate?
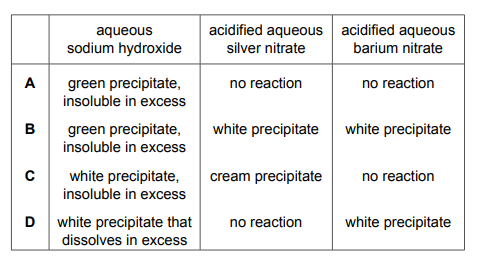
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A)
Question 40
A student carries out a flame test on a sample. The flame colour observed is light green. Which ion is present in the sample?
A. \(Ba^{2+}\)
B. \(Ca^{2+}\)
C. \(Li^+\)
D. \(K^+\)
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
✅ Answer: (A)
