Question 1
Fig. 1.1 shows the structures of seven substances, A, B, C, D, E, F and G.
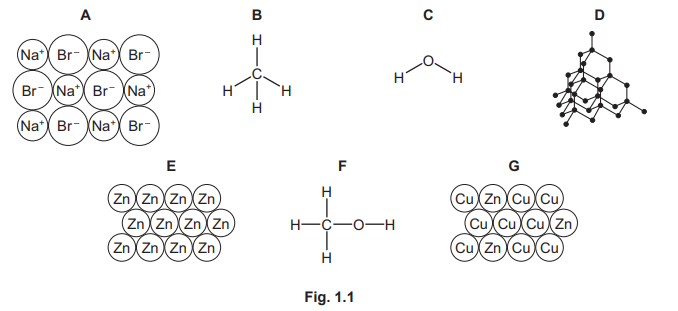
(a) Answer the following questions using only the structures in Fig. 1.1. Each structure may be used once, more than once or not at all. State which structure represents:
(i) an alloy
(ii) a substance that only conducts electricity when molten or in aqueous solution
(iii) a giant covalent structure
(iv) a compound that is a product formed in a hydrogen–oxygen fuel cell
(v) a compound with a high melting point
(vi) a gas that is responsible for increased global warming.
(b) Complete Fig. 1.2 to show the dot-and-cross diagram for structure C. Show the outer shell electrons only.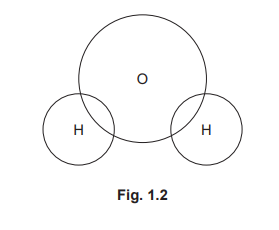
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 9.3 — Alloys and their properties (Part (a)(i))
• Topic 4.1 — Electrolysis / Topic 2.4 — Ions and ionic bonds (Part (a)(ii))
• Topic 2.6 — Giant covalent structures (Part (a)(iii))
• Topic 4.2 — Hydrogen-oxygen fuel cells (Part (a)(iv))
• Topic 2.4 — Ions and ionic bonds / Topic 2.6 — Giant covalent structures (Part (a)(v))
• Topic 10.3 — Air quality and climate (Part (a)(vi))
• Topic 2.5 — Simple molecules and covalent bonds (Part (b))
▶️ Answer/Explanation
(a)(i)
G
An alloy is a mixture of a metal with other elements. Diagram G shows a mixture of large and small atoms (representing different-sized metallic atoms like in brass or steel), which prevents layers from sliding and is characteristic of an alloy structure.
(a)(ii)
A
Structure A shows a lattice of alternating positive and negative ions (ionic compound). Ionic compounds conduct electricity only when molten or in aqueous solution because the ions are then free to move, but not when solid.
(a)(iii)
D
A giant covalent structure consists of a large network of atoms bonded by covalent bonds. Structure D represents a lattice like diamond or silicon dioxide, where each atom is bonded to many others in a repeating 3D network.
(a)(iv)
C
In a hydrogen–oxygen fuel cell, hydrogen reacts with oxygen to produce water. Structure C shows a molecule with two hydrogen atoms bonded to one oxygen atom (H₂O), which is the only chemical product of this fuel cell.
(a)(v)
A
Compounds with high melting points often have giant ionic or covalent structures. Structure A (ionic lattice) has strong electrostatic forces between oppositely charged ions, requiring large amounts of thermal energy to overcome.
(a)(vi)
B
Structure B shows a molecule of carbon dioxide (CO₂). Carbon dioxide is a greenhouse gas; it absorbs and re-emits thermal radiation, trapping heat in the atmosphere and contributing to increased global warming.
(b)
For the correct diagram:
• 1 bonding pair of electrons (2 dots/crosses) between each H and O
• 4 non-bonding electrons (2 lone pairs) on the oxygen atom
• No extra electrons on the hydrogen atoms
Oxygen has 6 outer electrons. It forms two single covalent bonds by sharing one electron with each hydrogen, completing its octet. The two remaining lone pairs are placed on the oxygen atom, while each hydrogen shares its single electron to achieve a duplet.
Question 2
(a) Table 2.1 shows the percentages by mass of the elements present in the human body.
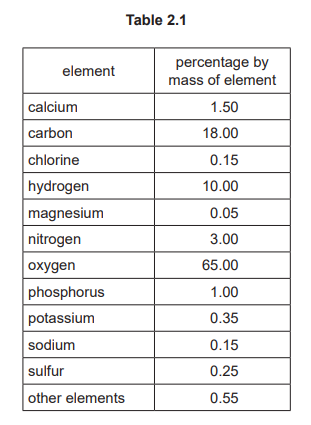
Answer these questions using information from Table 2.1.
(i) Name the non-metallic element in Table 2.1 that has the lowest percentage by mass.
(ii) Name an element in Table 2.1 that is in Period 4 of the Periodic Table.
(b) Some medicines contain a compound made of \(Mg^{2+}\) ions and \(OH^–\) ions. Name the compound made of \(Mg^{2+}\) ions and \(OH^–\) ions.
(c) Describe the observations when aqueous sodium hydroxide is added dropwise to a solution containing calcium ions until the sodium hydroxide is in excess.
observations with dropwise addition of sodium hydroxide
observations with excess sodium hydroxide
(d) Name a calcium salt that is soluble in water.
(e) Table 2.2 shows some properties of the Group I metals.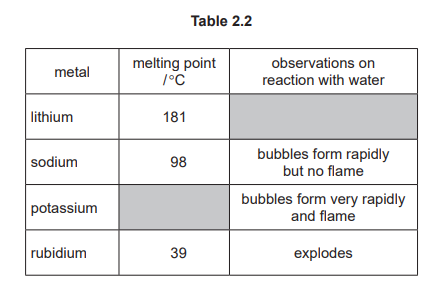
Use the information in Table 2.2 to predict:
● the melting point of potassium
● the observations when lithium reacts with water.
(f) State how the density of the Group I elements changes down the group.
(g) Sodium reacts with water to produce sodium hydroxide and a gas which pops with a lighted splint. Complete the symbol equation for this reaction.
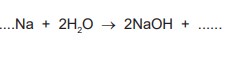
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 8.1 — Arrangement of elements (Part (a)(ii))
• Topic 8.2 — Group I properties (Parts (a)(i), (e), (f), (g))
• Topic 7.1 — Characteristic properties of acids and bases (Part (b))
• Topic 12.5 — Identification of ions and gases (Part (c))
• Topic 7.3 — Preparation of salts (Part (d))
▶️ Answer/Explanation
(a)(i)
Chlorine (Cl)
Table 2.1 shows chlorine at just 0.2% by mass, which is lower than other non-metals like carbon (18.0%), oxygen (65.0%), and hydrogen (10.0%).
(a)(ii)
Potassium (K) or Calcium (Ca)
Potassium is in Period 4, Group I, and Calcium is in Period 4, Group II. Both are listed in the table as essential elements in the human body.
(b)
Magnesium hydroxide
Ionic compounds are named by the metal cation followed by the non-metal anion. Here, \(Mg^{2+}\) is magnesium, and \(OH^–\) is hydroxide, forming the base magnesium hydroxide (\(\mathrm{Mg(OH)_2}\)).
(c)
Dropwise addition: A white precipitate forms. / Excess addition: The white precipitate remains (insoluble).
Calcium ions (\(Ca^{2+}\)) react with hydroxide ions (\(OH^–\)) to form insoluble calcium hydroxide (\(Ca(OH)_2\)). Unlike amphoteric hydroxides (e.g., Al, Zn), calcium hydroxide does not dissolve in excess sodium hydroxide.
(d)
Calcium nitrate / Calcium chloride
Using the solubility rules from the syllabus: all nitrates are soluble, and chlorides are soluble (except with lead and silver). Therefore, \(Ca(NO_3)_2\) or \(CaCl_2\) are suitable soluble calcium salts.
(e)
Melting point of potassium: 64 °C (accept any value between 41–96 °C)
Observations for lithium: Bubbles form slowly (less rapid than sodium).
Group I melting points decrease down the group; since sodium melts at 98 °C, potassium must be lower. Reactivity increases down the group, so lithium reacts less vigorously than sodium, producing hydrogen bubbles slowly.
(f)
The density increases (down the group).
Although the atoms get larger down Group I, the mass per atom increases at a faster rate than the atomic volume, resulting in a higher density for elements like potassium and cesium compared to lithium.
(g)
\(2\mathrm{Na} + 2\mathrm{H_2O} \rightarrow 2\mathrm{NaOH} + \mathrm{H_2}\)
The balanced symbol equation requires 2 sodium atoms to react with 2 water molecules, producing 2 formula units of sodium hydroxide and 1 molecule of hydrogen gas (the “pop” gas).
Question 3
Aluminium is extracted by electrolysis of its purified ore.
(a) Name the main ore of aluminium.
(b) Fig. 3.1 shows the apparatus used in the extraction of aluminium.
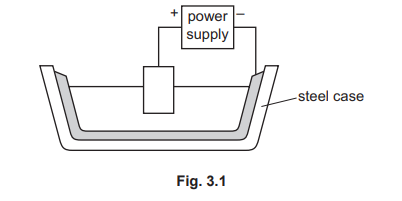
(i) Label the cathode in Fig. 3.1.
(ii) The electrolyte contains molten aluminium oxide. State the product formed at each electrode.
positive electrode: __________________
negative electrode: __________________
(c) State two physical properties that explain why aluminium is used in overhead electrical cables.
(d) Aluminium ore is purified by reacting it with sodium hydroxide. Sodium hydroxide is an alkali.
(i) State the meaning of the term alkali.
(ii) Describe how to find the pH of a dilute solution of sodium hydroxide using universal indicator paper.
(iii) A dilute solution of sodium hydroxide is added to a solution of methyl orange in acid until the sodium hydroxide is in excess.
State the colour change of the methyl orange.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 9.6 — Extraction of metals (Parts (a), (b))
• Topic 9.2 — Uses of metals (Part (c))
• Topic 7.1 — Characteristic properties of acids and bases (Part (d)(i))
• Topic 12.1 — Experimental design / Topic 7.1 — pH (Part (d)(ii))
• Topic 7.1 — Acids, bases and salts / Indicators (Part (d)(iii))
▶️ Answer/Explanation
(a)
Bauxite.
The main ore of aluminium is bauxite, which consists primarily of the minerals gibbsite (Al(OH)₃), boehmite (γ-AlO(OH)), and diaspore (α-AlO(OH)), mixed with iron oxides and clay.
(b)(i)
The cathode should be labelled on the inner carbon/graphite lining of the steel vessel (the negative terminal).
In the Hall-Héroult process, the steel container is lined with carbon which acts as the cathode (negative electrode), while large carbon blocks suspended into the electrolyte serve as the anodes (positive electrodes).
(b)(ii)
Positive electrode (anode): Oxygen
Negative electrode (cathode): Aluminium
During electrolysis, Al³⁺ ions are reduced to Al(l) at the cathode, while O²⁻ ions are oxidized at the carbon anodes to form O₂(g). The oxygen reacts with the carbon anodes, slowly burning them away.
(c)
1. Low density / lightweight.
2. Good electrical conductivity.
Overhead cables must be light to reduce sagging between pylons and minimize support structure costs, yet highly conductive to efficiently transmit electricity without excessive energy loss due to heating.
(d)(i)
An alkali is a soluble base that forms hydroxide ions (OH⁻) in aqueous solution.
While all alkalis are bases (proton acceptors), the term “alkali” specifically refers to a base that dissolves in water to release OH⁻ ions, such as sodium hydroxide (NaOH) or potassium hydroxide (KOH).
(d)(ii)
Dip a piece of universal indicator paper into the solution (or use a glass rod to transfer a drop of the solution onto the paper). Immediately compare the resulting colour to the standard pH colour chart provided with the indicator paper.
Universal indicator contains multiple dyes that produce a distinct, continuous range of colours across the pH scale, allowing a direct estimation of pH by colour matching.
(d)(iii)
From: Red / Pink to: Yellow.
Methyl orange is red in strongly acidic solutions (pH < 3.1). As the acid is neutralized by sodium hydroxide and the solution becomes alkaline (excess OH⁻), the indicator deprotonates and its structure changes, turning yellow (pH > 4.4).
Question 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 11.5 — Alkenes (Part (a))
• Topic 12.3 — Chromatography (Part (b))
• Topic 10.3 — Air quality and climate / Photosynthesis (Part (c))
▶️ Answer/Explanation
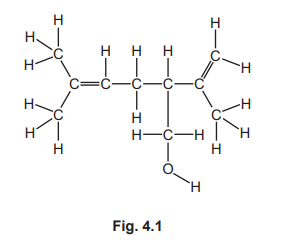
(a)
Circle around the carbon-carbon double bond (C=C) group in the displayed formula.
A carbon-carbon double bond is the functional group that defines unsaturation. Saturated compounds contain only single bonds, while unsaturated compounds contain at least one C=C double bond, which can undergo addition reactions.
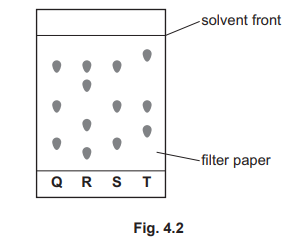
(b)(i)
R
In paper chromatography, each separated spot represents a different compound. The chromatogram for plant R shows the highest number of distinct spots (five distinct spots visible at different heights), indicating it contains the greatest number of coloured compounds.
(b)(ii)
Q and S
Both plants Q and S show identical spot patterns at exactly the same distances from the baseline. This indicates that the mixtures of coloured compounds extracted from Q and S have identical compositions and therefore contain exactly the same coloured compounds.
(b)(iii)
A solvent is a substance that dissolves a solute.
In chemistry, a solvent is the component of a solution present in the largest amount. It is responsible for dissolving the solute, forming a homogeneous mixture, and in chromatography it acts as the mobile phase to carry components along the stationary phase.
(c)(i)
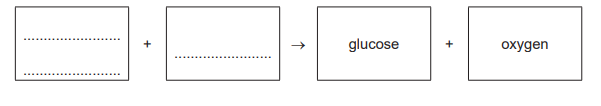
Carbon dioxide + Water → Glucose + Oxygen
During photosynthesis, plants convert carbon dioxide and water into glucose (a sugar for energy/storage) and oxygen (released as a waste product). This endothermic reaction requires light energy and takes place in the chloroplasts.
(c)(ii)
Chlorophyll (or Light/Sunlight)
Chlorophyll is the green pigment in chloroplasts essential for photosynthesis because it absorbs light energy from the sun. This energy drives the reaction that converts carbon dioxide and water into glucose and oxygen.
Question 5
(a) An atom of carbon is represented by the symbol shown.
![]()
Describe this atom of carbon in terms of:
● the position of the electrons, neutrons and protons in this atom
● the number of neutrons and number of protons
● the electronic configuration.
(b) (i) Complete the symbol equation for the incomplete combustion of carbon to produce carbon monoxide.
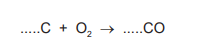
(ii) State one adverse effect of carbon monoxide.
(c) Fig. 5.1 shows the displayed formula of chromium carbonyl. 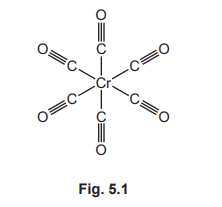
Deduce the molecular formula of chromium carbonyl.
(d) Another compound of chromium has the formula \(Na_2Cr_2C_{10}O_{10}\). Complete Table 5.1 to calculate the relative molecular mass of \(Na_2Cr_2C_{10}O_{10}\). 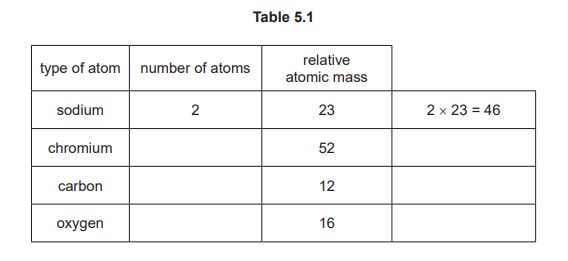
(e) Chromium can be produced by heating chromium(III) oxide, Cr2O3, with carbon.
![]()
Describe how this equation shows that chromium(III) oxide is reduced.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 2.2 — Atomic structure and the Periodic Table (Part a)
• Topic 10.3 — Air quality and climate (Part b)
• Topic 11.1 — Formulae, functional groups and terminology (Part c)
• Topic 3.2 — Relative masses of atoms and molecules (Part d)
• Topic 6.4 — Redox (Part e)
▶️ Answer/Explanation
(a)
For the correct description: protons and neutrons in nucleus; electrons outside the nucleus; 8 neutrons; 6 protons; electronic configuration 2,4.
The atom has a nucleus containing protons (positive) and neutrons (neutral), with electrons orbiting in shells. Carbon-14 has 6 protons (given by atomic number) and 8 neutrons (mass number 14 minus 6). Its electrons fill the first shell (2) and the second shell (4).
(b)(i)
For the correct equation: 2(C) + O₂ → 2(CO)
Incomplete combustion occurs when the oxygen supply is limited. For every two moles of carbon reacting with one mole of oxygen gas, two moles of carbon monoxide are produced, rather than carbon dioxide.
(b)(ii)
toxic / poisonous
Carbon monoxide binds to hemoglobin in red blood cells approximately 240 times more strongly than oxygen. This prevents oxygen from being transported around the body, leading to headaches, dizziness, unconsciousness, and eventually death.
(c)
CrC₆O₆
The displayed formula shows one central Chromium atom bonded to six Carbonyl groups (CO). Therefore, the molecular formula consists of Cr, 6 × C, and 6 × O, written as CrC₆O₆.
(d)
For the correct calculation: 430(2)
Calculate using standard relative atomic masses (Na=23, Cr=52, C=12, O=16): (2×23) + (2×52) + (10×12) + (10×16) = 46 + 104 + 120 + 160 = 430. No units are required for relative molecular mass as it is a ratio.
(e)
chromium(III) oxide loses oxygen
Reduction is defined as the loss of oxygen from a substance. In this equation, Cr₂O₃ loses oxygen atoms to become metallic Cr (the oxygen combines with Carbon to form CO), proving that the chromium(III) oxide has been reduced.
Question 6
Large pieces of solid sulfur burn in excess oxygen to produce sulfur dioxide gas.
(a) Complete the equation by adding the missing state symbol.
![]()
(b) Fig. 6.1 shows how the mass of sulfur changes as the reaction proceeds.
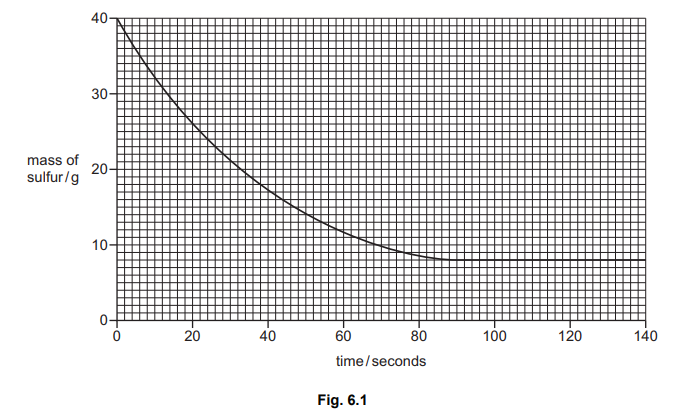
Deduce the time taken for the reaction to finish.
(c) The experiment is repeated using powdered sulfur. Describe the effect on the rate of reaction of using powdered sulfur rather than large pieces of sulfur.
(d) Sulfur dioxide reacts with oxygen in a closed container.
![]()
(i) Describe the effect, if any, each of the following has on the rate of this reaction. All other conditions stay the same.
● The temperature is decreased.
● The pressure of the gases is increased.
(ii) Changing concentration changes the rate of a reaction. Choose the correct unit of concentration from the list. Draw a circle around your chosen answer.
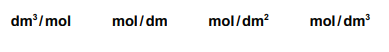
(e) Sulfur dioxide is an air pollutant.
(i) State one adverse effect of sulfur dioxide.
(ii) Emissions of sulfur dioxide can be reduced by using low-sulfur fossil fuels. State one other way of reducing sulfur dioxide emissions from fossil fuels.
(f) Aqueous sodium hydrogen sulfite releases sulfur dioxide gas at room temperature. Sulfur dioxide changes the colour of acidified potassium manganate(VII) from purple to colourless. Fig. 6.2 shows a sealed tube with a small volume of aqueous sodium hydrogen sulfite at the bottom. A piece of filter paper soaked in acidified potassium manganate(VII) is attached to the top of the tube.
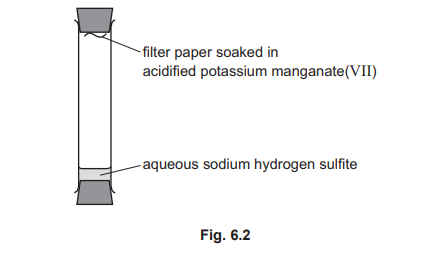
The filter paper remains purple at first. The filter paper becomes colourless after a short time. Explain these results in terms of kinetic particle theory.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 9.4 — Reactivity series (Part (a))
• Topic 6.2 — Rate of reaction (Parts (b), (c), (d)(i))
• Topic 3.2 — Relative masses of atoms and molecules (Part (d)(ii))
• Topic 10.3 — Air quality and climate (Part (e))
• Topic 1.2 — Diffusion (Part (f))
▶️ Answer/Explanation
(a)
g (for gas)
Sulfur dioxide (SO₂) is produced as a gas at room temperature and pressure, as it has a boiling point of -10°C. The complete combustion of sulfur in oxygen is S(s) + O₂(g) → SO₂(g).
(b)
90 (s)
The graph shows the mass of sulfur decreasing from 6.0 g to 0 g. The reaction is complete when the mass stops changing and reaches zero, which occurs at t = 90 seconds on the time axis.
(c)
(Rate) increases / faster reaction
Powdered sulfur has a much larger total surface area compared to a large solid piece of the same mass. This increases the frequency of collisions between reactant particles (sulfur and oxygen), leading to a higher reaction rate.
(d)(i)
● Temperature is decreased: (rate) decreases / slower reaction
● Pressure of the gases is increased: (rate) increases / faster (reaction)
Decreasing temperature reduces particle kinetic energy, lowering collision frequency and the proportion of successful collisions. Increasing pressure compresses gas particles closer together, increasing collision frequency and thus the rate.
(d)(ii)
mol / dm³
Concentration is defined as the amount of solute (in moles) per unit volume of solution (in dm³). Therefore, mol/dm³ (or mol/L) is the standard unit for measuring concentration in chemistry.
(e)(i)
Acid rain (Accept: causes breathing difficulties / respiratory problems)
Sulfur dioxide dissolves in atmospheric water vapor to form sulfurous acid (H₂SO₃) and, through oxidation, sulfuric acid (H₂SO₄), leading to acid rain which damages buildings, forests, and aquatic life.
(e)(ii)
Flue gas desulfurisation (Accept: using catalytic converters / removing sulfur before burning)
Flue gas desulfurisation typically involves spraying the waste gases with a slurry of calcium oxide (lime) or limestone, which reacts with SO₂ to form harmless calcium sulfate (gypsum), removing the pollutant before it is released.
(f)
• Diffusion / particles move / travel from the bottom to the top.
• (Movement of) particles is random / in every direction.
• Particles spread from higher concentration (at the bottom) to lower concentration (at the top).
• SO₂ particles reach / collide with the filter paper, reacting with the potassium manganate(VII) and turning it colourless.
Initially, no SO₂ has reached the top. Over time, gaseous SO₂ particles, released from the solution, move randomly and spread throughout the sealed tube via diffusion. Eventually, enough particles collide with the acidified KMnO₄ on the filter paper to reduce the purple Mn(VII) to colourless Mn(II).
Question 7
Iron is a metal. Iron has a high density, a high melting point and a high boiling point.
(a) State three other physical properties of iron.
(b) (i) State the conditions needed for iron to rust.
(ii) Rust is hydrated iron(III) oxide. State if iron(III) oxide is an acidic or basic oxide. Give a reason for your answer.
(iii) Complete this sentence about methods of preventing rusting. Rusting can be prevented by painting or………….
(c) The list shows five metals.
calcium copper iron silver sodium
Put these metals in order of their reactivity. Put the most reactive metal at the top.
(d) Complete the word equation for the reaction of calcium carbonate with nitric acid. 
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 9.1 — Properties of metals (Part a)
• Topic 9.5 — Corrosion of metals (Parts b(i), b(iii))
• Topic 7.2 — Oxides (Part b(ii))
• Topic 9.4 — Reactivity series (Part c)
• Topic 7.1 — Characteristic properties of acids and bases (Part d)
▶️ Answer/Explanation
(a)
For the correct answer (any three): lustrous/shiny, malleable, ductile, sonorous, conducts electricity/heat.
Metals like iron have a ‘sea of delocalised electrons’ which allows them to conduct electricity and heat. The metallic lattice structure also allows layers of atoms to slide, making iron malleable (can be hammered into shapes) and ductile (can be drawn into wires).
(b)(i)
Conditions: air (oxygen) and water.
For iron to rust, both oxygen from the air and water must be present simultaneously. The process is an electrochemical reaction where iron loses electrons and forms hydrated iron(III) oxide.
(b)(ii)
Iron(III) oxide is a basic oxide because iron is a metal (or it is a metal oxide).
According to syllabus section 7.2, oxides of metals (like CuO and CaO) are classified as basic oxides. They react with acids to form a salt and water.
(b)(iii)
Suitable ending: covering with plastic, greasing, or galvanising.
These methods create a barrier that prevents oxygen or water from reaching the iron’s surface. Galvanising (coating with zinc) also provides sacrificial protection if the barrier is scratched.
(c)
Order (most reactive at top): sodium, calcium, iron, copper, silver.
This order is based on the reactivity series (syllabus 9.4). Metals higher in the series lose electrons more readily to form positive ions. Sodium reacts violently with cold water, while silver does not react with water or dilute acids.
(d)
calcium nitrate + water + carbon dioxide.
This is a typical acid-carbonate neutralisation reaction (syllabus 7.1). An acid (nitric acid) reacts with a carbonate (calcium carbonate) to form a salt (calcium nitrate), water, and carbon dioxide gas.
Question 8
(a) Fig. 8.1 shows the displayed formulae of five organic compounds, V, W, X, Y and Z.
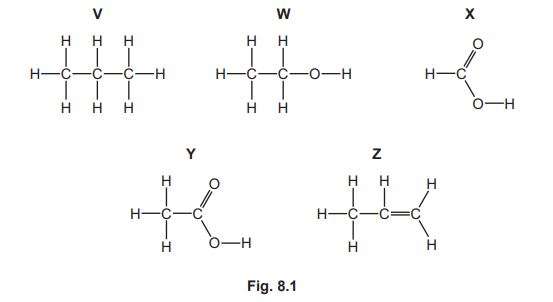
(i) State which two of the compounds, V, W, X, Y and Z, are in the same homologous series.
(ii) Explain why compound V is an alkane.
(iii) State the name of the homologous series to which compound X belongs.
(b) Ethanol can be manufactured by the catalytic addition of steam to ethene.
(i) State the temperature and pressure required for this reaction.
(ii) Name one other method of manufacturing ethanol.
(c) Describe how alkenes are manufactured from petroleum fractions.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 11.1 & 11.2 — Formulae, functional groups, and naming organic compounds (Part (a))
• Topic 11.6 — Alcohols (Part (b))
• Topic 11.5 — Alkenes & 11.3 — Fuels (Part (c))
▶️ Answer/Explanation
(a)(i)
X and Y
Both X and Y contain the carboxylic acid functional group (–COOH). X is propanoic acid (C₂H₅COOH) and Y is butanoic acid (C₃H₇COOH). They belong to the same homologous series because they share the same general formula (CnH2n+1COOH) and differ by a –CH₂– unit.
(a)(ii)
Compound V is an alkane because it is a hydrocarbon (contains only carbon and hydrogen) and it contains only single covalent bonds (it is saturated).
The displayed formula of V (ethane, C₂H₆) shows only C–C and C–H single bonds, fitting the general formula CnH2n+2, which is characteristic of alkanes.
(a)(iii)
Carboxylic acids
Compound X has the formula C₂H₅COOH. The –COOH group (carboxyl group) defines the carboxylic acid homologous series. Members of this series are weak acids that react with bases, carbonates, and alcohols (to form esters).
(b)(i)
300 (°C) and 60 (atm / 6000 kPa)
The industrial hydration of ethene (C₂H₄ + H₂O → C₂H₅OH) requires a temperature of 300 °C and a pressure of 60 atm (approximately 6000 kPa) in the presence of an acid catalyst (e.g., phosphoric acid). These conditions are chosen to optimize the rate and equilibrium yield of ethanol production.
(b)(ii)
Fermentation
An alternative method is the fermentation of glucose (C₆H₁₂O₆) using yeast enzymes at around 25–35 °C in anaerobic conditions. The process yields ethanol and carbon dioxide (C₆H₁₂O₆ → 2C₂H₅OH + 2CO₂). Fermentation uses renewable resources (sugar cane, corn) but produces a dilute, impure ethanol that requires further purification.
(c)
M1: Cracking – Large alkane molecules from petroleum fractions are heated to a high temperature (about 600–700 °C) and passed over a catalyst (e.g., zeolite, alumina, or silica).
M2: Product formation – The thermal decomposition breaks carbon–carbon bonds in the long-chain alkanes, producing smaller, more useful molecules, including alkenes (e.g., ethene, propene) and hydrogen, along with shorter alkanes.
The process is a form of thermal or catalytic cracking. The high temperature provides the activation energy for C–C bond scission. Alkenes manufactured this way are important feedstocks for making polymers (plastics) and other organic chemicals.
