Question 1
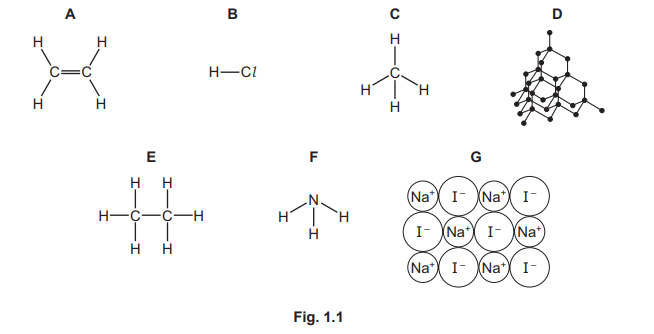
Fig. 1.1 shows the structures of seven substances, A, B, C, D, E, F and G.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 11.3 — Fuels (Part (a)(i), (a)(v))
• Topic 11.8 — Polymers (Part (a)(ii))
• Topic 2.6 — Giant covalent structures (Part (a)(iii))
• Topic 2.4/2.5 — Ionic/Simple molecular compounds (Part (a)(iv))
• Topic 7.3 — Preparation of salts / 8.4 Transition elements (Part (a)(vi))
• Topic 2.5 — Simple molecules and covalent bonds (Part (b))
▶️ Answer/Explanation
(a)(i)
C
Detailed solution: Methane (CH₄) is the main constituent of natural gas. Structure C shows a central carbon atom bonded to four hydrogen atoms (tetrahedral arrangement), which is the correct molecular structure of methane.
(a)(ii)
A
Detailed solution: The monomer for poly(ethene) (polyethylene) is ethene (C₂H₄). Structure A shows a double bond between two carbon atoms, with each carbon bonded to two hydrogen atoms, which is the correct structure of ethene (H₂C=CH₂).
(a)(iii)
D
Detailed solution: Structure D is diamond, a giant covalent lattice where each carbon atom is covalently bonded to four other carbon atoms in a three-dimensional network, making it extremely hard with a very high melting point.
(a)(iv)
G
Detailed solution: Structure G represents an ionic lattice (e.g., sodium chloride, NaCl). Ionic compounds have strong electrostatic forces between oppositely charged ions throughout the entire lattice, requiring a large amount of energy to overcome, hence they possess very high melting points.
(a)(v)
C
Detailed solution: Methane (CH₄, structure C) is produced as a waste gas from the digestion of vegetation in animals (enteric fermentation, e.g., in cows). It is a significant greenhouse gas released from agricultural processes.
(a)(vi)
G
Detailed solution: Structure G is an ionic compound (such as NaCl). It is a solid at room temperature and its ions are immobile in the lattice; however, when dissolved in water, the ions dissociate and become free to move, allowing the solution to conduct electricity.
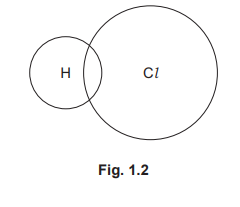
(b)
For the correct dot-and-cross diagram (2 marks):
• One bonding pair (2 electrons) shown as a cross (from H) and a dot (from Cl) between the H and Cl atoms (1)
• Six non-bonding outer electrons (shown as dots, crosses, or a mixture) placed around the chlorine atom only, and no extra electrons around the hydrogen atom (1)
Detailed solution: Structure B is hydrogen chloride (HCl). Hydrogen has 1 valence electron (cross), chlorine has 7 valence electrons (dots). They share one pair to complete H’s duplet and Cl’s octet. The remaining 6 electrons (3 lone pairs) are placed around the chlorine atom. The resulting diagram shows a single covalent bond (H:Cl) with three lone pairs on Cl.
Question 2
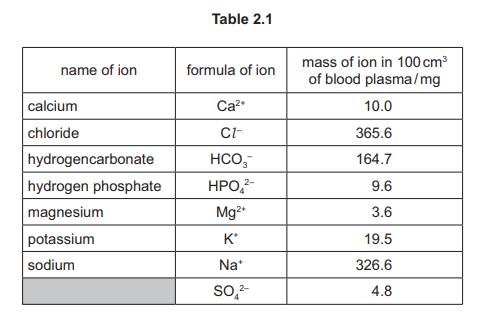
(a) Intracellular fluid is the solution between the cells in the human body. Table 2.1 shows the masses, in mg, of some ions in \(100cm^3\) of intracellular fluid.

● predict the hardness of potassium
● describe the observations when rubidium reacts with water.
2Na + …….. → …..NaH
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 8.2 — Group I properties (Part (a)(i), (e))
• Topic 8.1 — Arrangement of elements / Periodic Table (Part (a)(ii))
• Topic 12.5 — Identification of ions and gases (Part (b))
• Topic 2.1 — Elements, compounds and mixtures (Part (c))
• Topic 7.3 — Preparation of salts / solubility rules (Part (d))
• Topic 2.2 — Atomic structure and the Periodic Table / reactions (Part (f))
▶️ Answer/Explanation
(a)(i)
Calcium (Ca²⁺)
The table shows the mass of calcium ions is 0.1 mg, which is significantly lower than potassium (150 mg), sodium (10 mg), or magnesium (30 mg). A lower mass in the same volume corresponds directly to a lower concentration of that positive ion.
(a)(ii)
Hydrogen carbonate (HCO₃⁻)
Group IV of the Periodic Table contains Carbon (C). Hydrogen carbonate ions are the only ions listed in Table 2.1 that contain a Group IV element (carbon) within their molecular structure.
(b)
Add dilute hydrochloric acid (or nitric acid) followed by barium chloride solution; a white precipitate (barium sulfate) confirms sulfate ions.
Acidifying first removes carbonate or sulfite impurities that could also form precipitates with barium. The formation of an insoluble white precipitate of BaSO₄ is a positive, unambiguous test for the sulfate anion (SO₄²⁻).
(c)
NH₄Cl
Ammonium (NH₄⁺) carries a 1+ charge, and chloride (Cl⁻) carries a 1- charge. Therefore, they combine in a 1:1 ratio to form the neutral ionic compound ammonium chloride.
(d)
Lead chloride (PbCl₂)
According to general solubility rules, most chlorides are soluble except those of lead, silver, and mercury(I). Sodium chloride, ammonium chloride, and magnesium chloride are all highly soluble in water, unlike lead chloride.
(e)
Hardness: between 0.25 and 0.65 MPa (e.g., 0.4 MPa). Observations: Extremely rapid reaction producing a lilac flame, or explosion/bubbles form more rapidly than potassium.
Group I hardness decreases down the group (lithium 0.6, sodium 0.4, so potassium ~0.2-0.3). Reactivity increases down the group; rubidium reacts more violently than potassium, producing hydrogen that ignites instantly.
(f)
2Na + H₂ → 2NaH
Hydrogen gas (H₂) exists as a diatomic molecule. The equation requires 2 sodium atoms to balance the 2 sodium atoms in 2NaH, and the 2 hydrogen atoms from one H₂ molecule provide the hydrogen for the two formula units.
Question 3
(a) Molten calcium bromide is electrolysed.
(i) Define the term electrolysis.
(ii) Name an inert metal that can be used for the electrodes.
(iii) Name the product formed at each electrode.
positive electrode ………………………………………….
negative electrode …………………………………………
(b) Calcium reacts with water. An alkaline solution is produced.
(i) Name the ion which causes a solution to be alkaline.
(ii) Choose the pH value of an alkaline solution. Draw a circle around your chosen answer.
pH1 pH5 pH7 pH9
(iii) Dilute hydrochloric acid is added to a solution of litmus in alkaline solution until the acid is in excess. State the colour change of the litmus.
(c) Calcium carbonate is added to the blast furnace in the production of iron. Calcium carbonate breaks down when heated to produce calcium oxide and a gas that turns limewater milky.
(i) Name the gas that turns limewater milky.
(ii) Name the type of chemical reaction that takes place when calcium carbonate is heated.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 4.1 — Electrolysis (Part (a))
• Topic 7.1 — Characteristic properties of acids and bases / Topic 9.4 — Reactivity series (Part (b))
• Topic 9.6 — Extraction of metals (Part (c))
▶️ Answer/Explanation
(a)(i)
Electrolysis is the decomposition of an ionic compound when molten or in aqueous solution by the passage of an electric current. The process involves the movement of ions towards the oppositely charged electrodes, where they lose or gain electrons.
(a)(ii)
Platinum (carbon/graphite is also accepted). These materials are chemically inert, meaning they do not react with the electrolyte or the products of electrolysis, and they conduct electricity well due to their structure.
(a)(iii)
Positive electrode (anode): bromine. Negative electrode (cathode): calcium. In molten calcium bromide (CaBr₂), Ca²⁺ ions are reduced to Ca atoms at the cathode, while Br⁻ ions are oxidised to Br₂ molecules at the anode.
(b)(i)
Hydroxide ion (OH⁻). Alkaline solutions contain an excess of hydroxide ions compared to hydrogen ions. It is the hydroxide ion that gives the solution its characteristic properties, such as turning litmus blue and having a pH greater than 7.
(b)(ii)
pH 9. On the pH scale, values below 7 are acidic, 7 is neutral, and values above 7 are alkaline (basic). Therefore, pH 9 is clearly an alkaline value.
(b)(iii)
From blue to red (or pink). Litmus is purple in neutral solution, turns blue in alkaline conditions (due to OH⁻ ions). When excess acid is added, the H⁺ ions neutralise the OH⁻, and the excess H⁺ turns the litmus red, indicating acidic conditions.
(c)(i)
Carbon dioxide (CO₂). Carbon dioxide gas reacts with calcium hydroxide solution (limewater) to form a white precipitate of calcium carbonate. This milky appearance is the standard chemical test to confirm the presence of CO₂ gas.
(c)(ii)
Thermal decomposition. This is a chemical reaction where a single compound (calcium carbonate) breaks down into two or more simpler substances (calcium oxide and carbon dioxide) when heated. It requires an input of thermal energy (endothermic).
Question 4
(a) Chlorophyll is a coloured compound found in plants. Chlorophyll is needed for photosynthesis.
(i) Complete the word equation for photosynthesis.
(ii) State one other condition that is essential for photosynthesis.
(b) Several other coloured compounds are found in plant leaves. A student extracts a mixture of coloured compounds from a plant leaf. Fig. 4.1 shows the apparatus used to separate the coloured compounds.
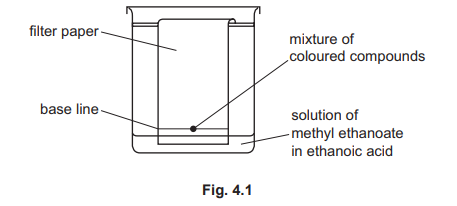
(i) Name this method of separation.
(ii) Suggest why the base line is drawn in pencil and not in ink.
(iii) The liquid used to separate the coloured compounds is a solution of methyl ethanoate in ethanoic acid. State the meaning of the term solution.
(c) Fig. 4.2 shows the displayed formula of a compound found in plant cells.
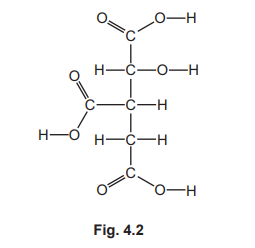
On Fig. 4.2, draw a circle around the alcohol functional group.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 10.3 — Air quality and climate / Photosynthesis (Part (a))
• Topic 12.3 — Chromatography (Part (b))
• Topic 11.1 — Formulae, functional groups and terminology (Part (c))
▶️ Answer/Explanation
(a)(i)
For the correct answer: glucose and oxygen.
Photosynthesis uses carbon dioxide and water, in the presence of chlorophyll and light energy, to produce glucose (a sugar for plant energy/storage) and oxygen (released as a by-product).
(a)(ii)
light / sunlight / energy from light
Light provides the activation energy needed to drive the endothermic reaction. Without light (even with chlorophyll), the process cannot occur.
(b)(i)
chromatography / paper chromatography
This method separates mixtures based on how far each substance travels with a solvent along a stationary phase (paper). Different coloured compounds have different solubilities and affinities for the paper.
(b)(ii)
so that it doesn’t run up the paper / ink would run up the paper / pencil is insoluble in the solvent (while ink would dissolve and move)
Pencil lead (graphite) is insoluble in the solvent and remains on the baseline. Ink would dissolve, travel up the paper with the solvent front, and obscure the separated components.
(b)(iii)
a mixture of one or more solutes dissolved in a solvent
In this context, methyl ethanoate is the solute (the substance being dissolved) and ethanoic acid is the solvent (the liquid doing the dissolving). The resulting uniform mixture is a solution.
(c)
circle around the –O–H group attached to the second carbon atom down
The alcohol functional group is a hydroxyl group (-OH) bonded to a saturated carbon atom. In the displayed formula, this appears as “O–H” connected to a carbon chain. The circled group should include the oxygen and hydrogen atoms bonded together.
Question 5
● the position of the electrons, neutrons and protons in the atom
● the number of neutrons and the number of protons
● the electronic configuration.
\(…..P + …..O_2 \rightarrow P_4O_{10}\)
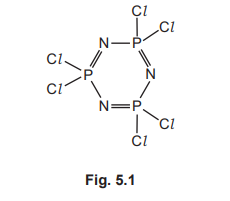 Deduce the molecular formula of this compound.
Deduce the molecular formula of this compound.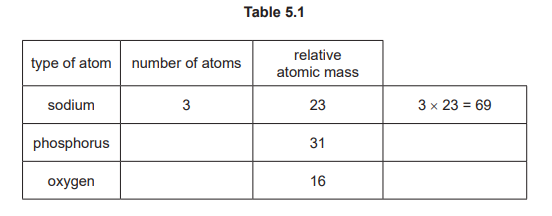
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 2.2 — Atomic structure and the Periodic Table (Part (a))
• Topic 6.4 — Redox / Topic 9.4 — Reactivity series (Part (b))
• Topic 11.1 — Formulae, functional groups and terminology (Part (c))
• Topic 3.2 — Relative masses of atoms and molecules (Part (d))
• Topic 10.1 — Water / Topic 10.2 — Fertilisers (Part (e))
▶️ Answer/Explanation
(a)
• Protons and neutrons are located in the central nucleus, while electrons orbit the nucleus in shells (energy levels).
• The atom has 15 protons (equal to the atomic number 15) and 16 neutrons (mass number 31 minus 15 protons).
• Its electronic configuration is 2,8,5, filling the first three shells with 2, 8, and 5 electrons respectively.
(b)
4P + 5O₂ → P₄O₁₀
Four phosphorus atoms are needed on the left to match the P₄ in the product. To balance the ten oxygen atoms in P₄O₁₀, five O₂ molecules (each containing two oxygen atoms) are required.
(c)
P₃N₃Cl₆
The displayed formula shows a ring of three P atoms alternating with three N atoms. Each P atom is bonded to two Cl atoms. Counting the atoms gives three Phosphorus, three Nitrogen, and six Chlorine atoms.
(d)
164
Calculation: Na₃ = 3 × 23 = 69; P = 1 × 31 = 31; O₄ = 4 × 16 = 64. Total = 69 + 31 + 64 = 164. One mark is awarded for the correct total, or for correctly calculating the oxygen contribution (64).
(e)
Fertilisers (or detergents / sewage / animal waste).
Phosphates enter waterways mainly from agricultural runoff containing phosphate-based fertilisers, or from domestic wastewater containing phosphate detergents. These excess nutrients cause eutrophication, leading to algal blooms and subsequent deoxygenation of the water.
Question 6
Dilute hydrochloric acid reacts with small pieces of calcium carbonate.
\(2HCl(aq) + CaCO_3(s) → CaCl_2(aq) + H_2O(l) + CO_2(g)\)
(a) State the meaning of the state symbol (aq).
(b) Fig. 6.1 shows how the mass of small pieces of calcium carbonate changes as the reaction proceeds. The calcium carbonate is in excess.
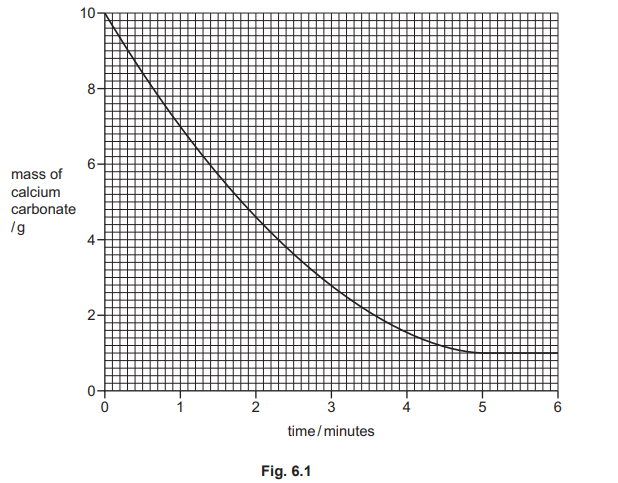
(i) Deduce the mass of calcium carbonate two minutes from the start of the reaction.
(ii) Explain how the graph shows that the calcium carbonate is in excess.
(iii) The experiment is repeated at a higher temperature. All other conditions stay the same. Draw a line on Fig. 6.1 to show how the mass of calcium carbonate changes at a higher temperature as the time increases.
(c) (i) Describe the effect, if any, on the rate of reaction when large pieces of calcium carbonate are used instead of small pieces of calcium carbonate. All other conditions stay the same.
(ii) Increasing the concentration of dilute hydrochloric acid increases the rate of reaction of dilute hydrochloric acid with calcium carbonate. Choose the correct unit of concentration from the list. Draw a circle around your chosen answer.
(d) Concentrated hydrochloric acid gives off hydrogen chloride gas. Hydrogen chloride is an acidic gas that turns damp universal indicator paper red. A long glass tube is set up as shown in Fig. 6.2.
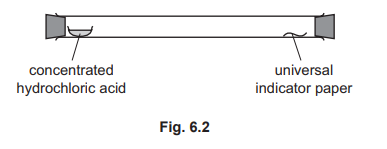
At first, the universal indicator paper does not change colour. The universal indicator paper turns red after a short time. Explain these results in terms of kinetic particle theory.
(e) Hydrogen chloride breaks down to form hydrogen and chlorine at 1500°C. The reaction is endothermic. State the meaning of the term endothermic.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 7.1 — Characteristic properties of acids and bases (Parts a, e)
• Topic 6.2 — Rate of reaction (Parts b, c)
• Topic 1.1 & 1.2 — States of matter and diffusion (Part d)
▶️ Answer/Explanation
(a)
For the correct answer:
aqueous / dissolved in water
The symbol (aq) stands for “aqueous”, which means the substance is dissolved in water to form a solution. In this reaction, hydrochloric acid (HCl) and calcium chloride (CaCl₂) are both in the aqueous state.
(b)(i)
\(4.6 \, (\mathrm{g})\)
Reading from the graph at exactly 2 minutes on the x-axis, the corresponding mass on the y-axis is approximately 4.6 g. This shows that 0.4 g of calcium carbonate has reacted by this time (starting from 5.0 g).
(b)(ii)
line levels off above 0 g / line levels off at 1 g
The mass of calcium carbonate does not drop to zero; it levels off at about 1 g. This residual mass confirms that some solid carbonate remains unreacted because all the acid has been used up, proving the solid was provided in excess.
(b)(iii)
For the correct line (2 marks):
• curve steeper and starting at 10 g (1)
• line levels off at 1 g and before 4.8 min (1)
At a higher temperature, particles have more kinetic energy, increasing the frequency and energy of collisions. The initial rate is faster (steeper slope), so the reaction finishes sooner (levels off earlier). The final mass remains the same (1 g) because the same amounts of reactants produce the same products.
(c)(i)
(rate) decreases / rate slows / slower (reaction)
Using large pieces reduces the total surface area of the solid calcium carbonate exposed to the acid. Fewer collisions occur between acid particles and the solid per second, which decreases the reaction rate according to collision theory.
(c)(ii)
\(mol / dm^3\) (or mol dm⁻³)
Concentration in chemistry is typically measured in moles per cubic decimetre (\( \mathrm{mol/dm^3} \)), which represents the amount of solute (in moles) dissolved in one cubic decimetre of solution. This unit is standard for expressing concentration in rate studies.
(d)
For the correct answer (3 marks for any 3 of the following):
• diffusion
• particles move / collide / travel
• (movement of) particles is random / in every direction
• particles spread out / particles mix
• particles hit filter paper
• (particles spread) from higher concentration to lower concentration
Initially, the hydrogen chloride gas particles are concentrated at the open end. They move randomly and diffuse down the concentration gradient along the tube. It takes time for them to travel the distance and reach the indicator paper, which then turns red upon contact with the acidic gas.
(e)
(reaction which) takes in thermal energy (from the surroundings)
An endothermic reaction absorbs heat from its surroundings. This results in a decrease in the temperature of the surroundings because the system requires energy to break chemical bonds in the reactants (e.g., breaking HCl bonds to form H₂ and Cl₂).
Question 7
Chromium and iron are transition elements. They are ductile and have high melting and boiling points.
(i) State the chemical name of rust.
(ii) An iron object is coated with plastic. Explain how this prevents the iron from rusting.
aluminium calcium copper iron zinc
Put these metals in order of their reactivity. Put the most reactive metal at the top
ZnO + C → Zn + CO
Describe how this equation shows that zinc oxide is reduced.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• Topic 8.4 — Transition elements (Part a)
• Topic 9.5 — Corrosion of metals (Part b)
• Topic 9.1 — Properties of metals / 7.1 — Acids and bases (Part c)
• Topic 9.4 — Reactivity series (Part d)
• Topic 6.4 — Redox (Part e)
▶️ Answer/Explanation
(a)
Any three from: lustrous/shiny, malleable, sonorous, conducts electricity, conducts heat, high density (any three).
Transition metals like chromium are typically hard, shiny, good conductors of heat/electricity, malleable, and ductile, with high density.
(b)(i)
Hydrated iron(III) oxide.
Rust is not pure iron(III) oxide; it contains variable amounts of water molecules (water of crystallisation) within its crystal lattice, hence “hydrated”.
(b)(ii)
The plastic coating acts as a barrier, preventing oxygen and/or water from reaching the iron surface.
Rusting requires both oxygen and water. By excluding either one, the plastic barrier stops the electrochemical process of oxidation at the iron surface.
(c)
chromium sulfate + hydrogen
Metals react with dilute sulfuric acid to form a salt (metal sulfate) and hydrogen gas. The salt is named after the metal (chromium) and the acid (sulfuric acid → sulfate).
(d)
Most reactive: calcium → aluminium → zinc → iron → copper (least reactive).
This order is determined by the reactivity series from the syllabus (Topic 9.4). Calcium reacts vigorously with cold water; aluminium, zinc, and iron react with acids to varying degrees, while copper is below hydrogen and does not displace it.
(e)
Reduction is the loss of oxygen. Zinc oxide (ZnO) loses an oxygen atom to become zinc (Zn).
In this reaction, the oxygen atom from ZnO combines with carbon (C) to form carbon monoxide (CO). Since ZnO loses oxygen, it undergoes reduction (and simultaneously, carbon is oxidised).
Question 8
This question is about hydrocarbons.
(a) Table 8.1 shows the names, formulae and boiling points of methane, ethane, propane and butane.
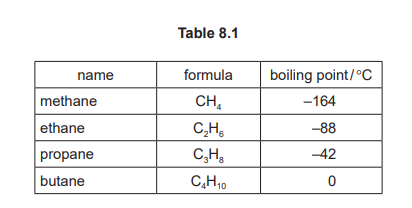
Use the information in Table 8.1 to answer these questions.
(i) Name the homologous series that includes methane, ethane, propane and butane.
(ii) State the trend in the boiling point of this homologous series as the number of carbon atoms increases.
(iii) Deduce the general formula of this homologous series.
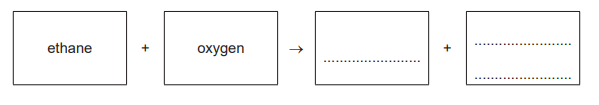
(b) Complete the word equation for the complete combustion of ethane.
(c) Long-chain hydrocarbons can be cracked to produce alkenes and hydrogen.
(i) State two conditions for cracking.
(ii) The diesel oil fraction from the fractional distillation of petroleum can be used for cracking. Give one other use of the diesel oil fraction.
(d) Alkene molecules can react together to produce polymers. Define the term polymer.
▶️ Answer/Explanation
(a)(i)
Alkanes.
Methane, ethane, propane, and butane are the first four members of the alkane homologous series, characterized by single carbon-carbon bonds (saturated hydrocarbons).
(a)(ii)
The boiling point increases (as the number of carbon atoms increases).
As the chain length increases, the molecules have larger surface areas, leading to stronger intermolecular forces (London forces) that require more energy to overcome.
(a)(iii)
\( \mathrm{C_nH_{2n+2}} \).
For alkanes, where ‘n’ is the number of carbon atoms, the number of hydrogen atoms follows the formula \(2n+2\) to satisfy the tetravalency of carbon, as seen in methane (CH₄, n=1) and butane (C₄H₁₀, n=4).
(b)
ethane + oxygen → carbon dioxide + water.
Complete combustion of a hydrocarbon (containing only C and H) in excess oxygen always produces carbon dioxide and water, releasing a large amount of energy.
(c)(i)
1. High temperature.
2. Catalyst (e.g., zeolite or aluminium oxide).
Cracking is a thermal decomposition process that uses high temperatures and a catalyst to break down large alkane molecules into smaller, more useful alkenes and alkanes.
(c)(ii)
Fuel for diesel engines / diesel engine fuel.
The diesel oil/gas oil fraction is primarily used as a fuel in compression-ignition (diesel) engines for buses, trucks, trains, and in some cars.
(d)
A polymer is a large molecule built up from many smaller, repeating units called monomers.
During addition polymerisation, unsaturated alkene monomers open their double bonds and join together to form very long, saturated chains without producing any other products.
