Question 1
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
When water vapour changes to rain, it is changing from a gas to a liquid. This process is called condensing. The water vapour molecules lose energy and come closer together to form liquid water droplets. Boiling and evaporating are liquid to gas changes, while freezing is liquid to solid. So condensing is the correct answer.
Question 2

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
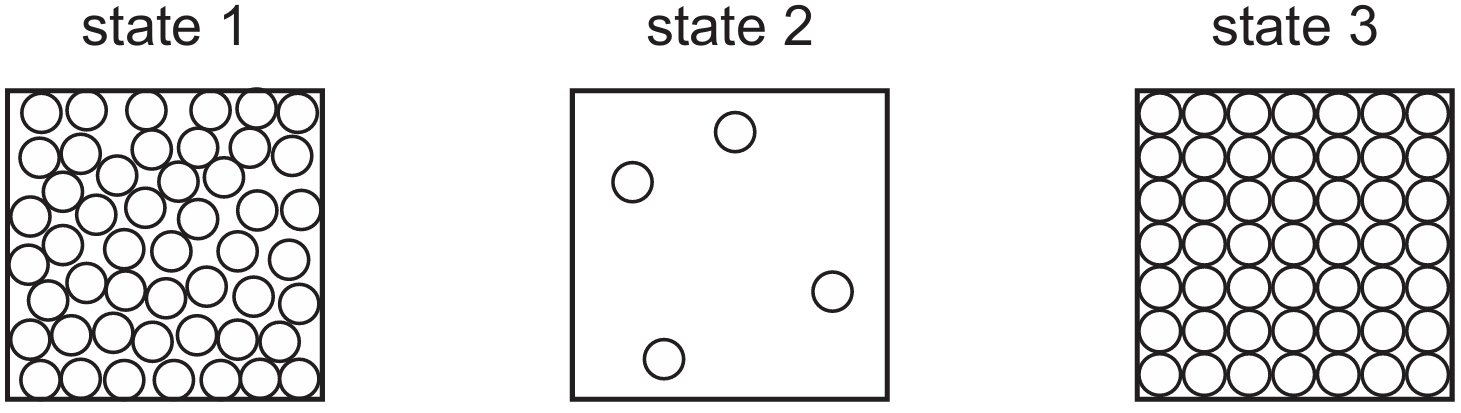
The diagrams show three states: 1 = gas (particles far apart, random motion), 2 = liquid (particles close, random arrangement), 3 = solid (particles close, regular arrangement). When a solid (3) changes to a gas (1), this is sublimation. During this change, the particles gain energy and their motion increases significantly. Therefore, for change 3→1, energy increases and particle motion increases, which matches row C.
Question 3
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
The number of protons determines the identity of an element, and this number is called the atomic number. Since the atom has 19 protons, its atomic number is 19. Looking at the Periodic Table, the element with atomic number 19 is potassium (K). Calcium has 20 protons, strontium has 38 protons, and yttrium has 39 protons. So Q is definitely potassium.
Question 4
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
A neutron is a subatomic particle located in the nucleus of an atom. It has a relative mass of approximately 1 (the same as a proton), but it carries no electrical charge, meaning its relative charge is 0. An electron has a relative mass of about 1/1840 and a charge of -1. A proton has a relative mass of 1 and a relative charge of +1. The nucleus contains both protons and neutrons, so it has a positive charge and a mass greater than 1. Only the neutron fits both criteria.
Question 5
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
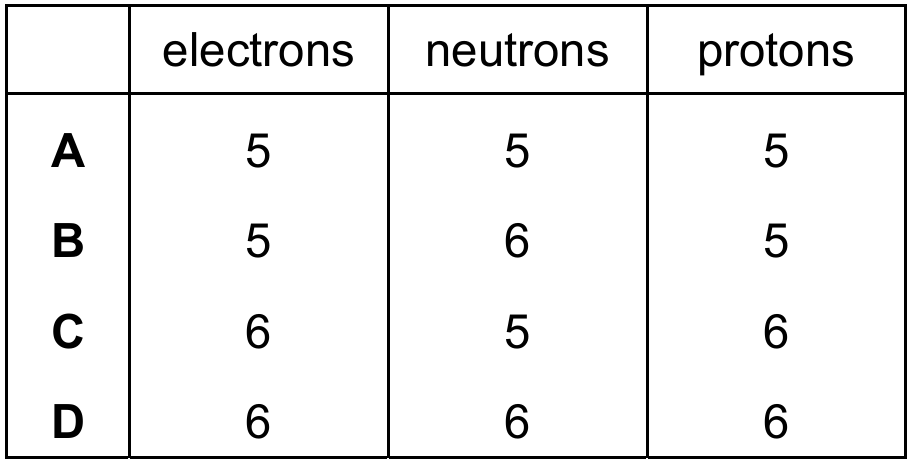
$^{11}_{5}\mathrm{B}$ has atomic number 5, so any isotope of boron must have 5 protons. In a neutral atom, electrons equal protons, so 5 electrons. $^{11}_{5}\mathrm{B}$ itself has 11 – 5 = 6 neutrons. The question asks for a particle which is an isotope of $^{11}_{5}\mathrm{B}$. An isotope has the same number of protons (5) but a different number of neutrons. Option A (5 electrons, 5 neutrons, 5 protons) represents boron-10 ($^{10}_{5}\mathrm{B}$), which is indeed a different isotope of boron. Option B represents $^{11}_{5}\mathrm{B}$ itself, while options C and D have the wrong number of protons.
Question 6
- It is formed from potassium anions and iodide cations.
- It is a good electrical conductor when molten or in aqueous solution.
- Potassium atoms share electrons with iodine atoms.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Potassium iodide (KI) is an ionic compound. Statement 1 is incorrect: potassium forms positive ions (cations, K⁺), while iodide forms negative ions (anions, I⁻). Statement 2 is correct: like other ionic compounds, potassium iodide conducts electricity when molten or dissolved in water because the ions become free to move. Statement 3 is incorrect: potassium and iodine form an ionic bond by transferring electrons, not sharing them. Therefore, only statement 2 is correct.
Question 7
- argon
- methane
- iron(III) oxide
- chlorine
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Shared pairs of electrons are found in covalent bonds. Argon is a noble gas that exists as single atoms with no chemical bonds, so it has no shared pairs. Iron(III) oxide is an ionic compound where electrons are transferred from iron to oxygen. Methane (CH₄) has four C–H covalent bonds, each containing a shared pair of electrons. Chlorine exists as Cl₂ molecules with one shared pair of electrons between the two chlorine atoms. So only methane and chlorine contain shared pairs of electrons.
Question 8
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Diamond is an allotrope of carbon where each carbon atom is covalently bonded to four other carbon atoms, forming a continuous three-dimensional giant lattice structure. This makes it extremely hard with a very high melting point. Ammonia (NH₃), carbon dioxide (CO₂), and water (H₂O) are all simple molecular compounds made of small molecules held together by weak intermolecular forces, not giant covalent structures.
Question 9

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Sodium carbonate has the correct formula Na₂CO₃ because sodium ions are Na⁺ and carbonate ions are CO₃²⁻, so two sodium ions are needed to balance the charge. Option A is wrong: cobalt(II) chloride should be CoCl₂, not Cu₂Cl (copper is a different element altogether). Xenon is a monatomic noble gas, so its formula is simply Xe, not Xe₂. Ammonium sulfate contains NH₄⁺ and SO₄²⁻ ions, so the correct formula is (NH₄)₂SO₄, not NH₄SO₄.
Question 10
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
The relative formula mass of MgCO₃ is given as 84. The relative formula mass of MgO is 24 (Mg) + 16 (O) = 40. According to the equation, 84 g of MgCO₃ produces 40 g of MgO. To find the mass produced from 21.0 g, we use the ratio: (40 ÷ 84) × 21.0 = (40 × 21.0) ÷ 84 = 840 ÷ 84 = 10.0 g. So 10.0 g of magnesium oxide is formed.
Question 11
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
When concentrated aqueous sodium chloride (brine) is electrolysed, the ions present are Na⁺, Cl⁻, H⁺, and OH⁻. At the positive electrode (anode), oxidation occurs and negative ions are attracted. In a concentrated solution, chloride ions (Cl⁻) are discharged in preference to hydroxide ions (OH⁻). So chlorine gas (Cl₂) is produced as the main product at the anode. Hydrogen forms at the cathode, and sodium is not produced in aqueous solution.
Question 12
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Air contains about 21% oxygen by volume. This means 21 cm³ of oxygen is present in every 100 cm³ of air. To get 630 dm³ of oxygen, we set up the proportion: 21% of total air = 630 dm³. So total air volume = (630 ÷ 21) × 100 = 30 × 100 = 3000 dm³. Therefore, a minimum of 3000 dm³ of clean, dry air is needed to supply 630 dm³ of oxygen.
Question 13
- In an endothermic reaction, the temperature of the surroundings increases.
- In an exothermic reaction, thermal energy is taken in from the surroundings.
- In the reaction pathway diagram for an exothermic reaction, the energy level of the products is lower than the energy level of the reactants.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Statement 1 is wrong because endothermic reactions take in energy from the surroundings, making the surroundings cooler. Statement 2 is wrong because exothermic reactions transfer energy out to the surroundings; it is endothermic reactions that take in energy. Statement 3 is correct: in an exothermic reaction, the products end up with less energy than the reactants started with, so on the energy level diagram, the products are drawn lower down than the reactants. Only statement 3 is correct.
Question 14
- The metal melts.
- Hydrogen is produced.
- Steam is produced.
- The pH of the solution increases.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
A physical change is one where no new substance is formed. The metal melting (statement 1) is simply a change of state from solid to liquid caused by the heat of the reaction – the metal itself does not change into a new substance. Steam being produced (statement 3) is water evaporating due to the heat released, again just a change of state. Hydrogen being produced (statement 2) and the pH increasing due to alkali formation (statement 4) both involve the creation of new chemical substances, so these are chemical changes.
Question 15
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
A catalyst works by providing an alternative reaction pathway with a lower activation energy, which speeds up the rate of reaction. Crucially, the catalyst itself is not consumed during the reaction – it remains chemically unchanged. Therefore, its mass is exactly the same before and after the reaction. A catalyst does not change the overall enthalpy change (so it does not make the reaction more exothermic), does not affect the final yield of products, and is definitely not used up.
Question 16
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
In this reaction, copper(II) oxide (CuO) loses its oxygen to become copper (Cu). Loss of oxygen is the definition of reduction, so CuO is reduced. Carbon (C) gains oxygen to become carbon dioxide (CO₂) – gain of oxygen is oxidation, so carbon is oxidised. CO₂ is a product and is not being oxidised further. Copper is a product of reduction, not being oxidised. So the only correct statement is that CuO is reduced.
Question 17

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Thymolphthalein is an indicator that is colourless in acids and turns blue in alkaline solutions. Methyl orange turns red in acidic solutions and yellow in alkaline solutions. Aqueous sodium hydroxide (NaOH) is an alkali. Therefore, in NaOH, thymolphthalein will appear blue and methyl orange will appear yellow. This perfectly matches row D.
Question 18

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
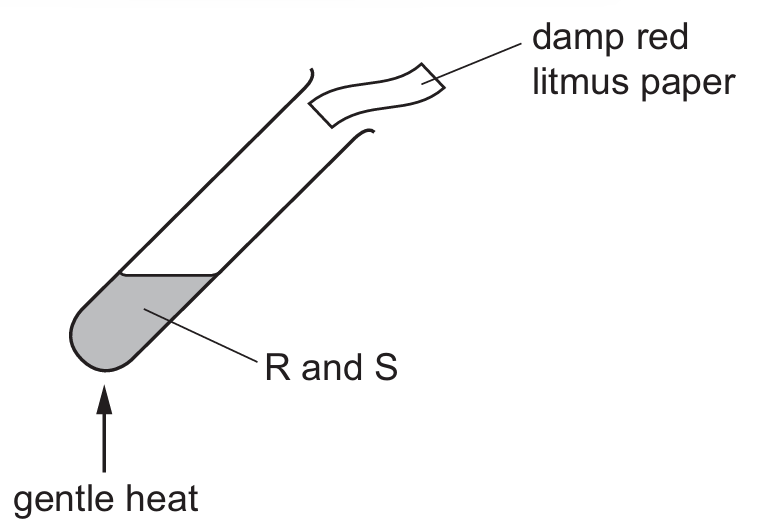
Damp red litmus paper turning blue indicates the presence of an alkaline gas, which is ammonia (NH₃). Ammonia is produced when a base reacts with an ammonium salt upon heating. Therefore, one substance must be a base (a basic oxide) and the other must be an ammonium salt (ammonium chloride, NH₄Cl). The basic oxide reacts with ammonium chloride to release ammonia gas, which then turns the red litmus paper blue.
Question 19
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Copper(II) sulfate is a soluble salt that can be prepared by reacting dilute sulfuric acid with an excess of copper(II) oxide (a base) or copper(II) carbonate (a metal carbonate). Copper metal is not reactive enough to directly displace hydrogen from dilute acids. Copper(II) hydroxide (alkali) could be used but is less common. The reaction between an acid and a metal carbonate is a standard method that produces the salt, water, and carbon dioxide gas.
Question 20
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Elements in Group II have two electrons in their outermost shell. They lose these two electrons to achieve a stable noble gas configuration, forming ions with a 2+ charge (e.g., Mg²⁺, Ca²⁺). Option A is wrong because elements are arranged by increasing atomic number, not atomic mass. Option B is wrong because in Group VII, reactivity decreases down the group. Option C is wrong because elements in the same group (vertical column), not period (horizontal row), have similar chemical properties.
Question 21
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
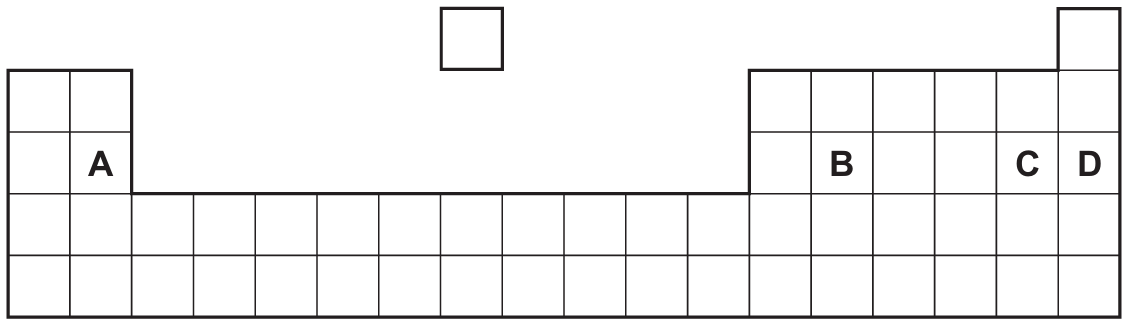
Metallic character decreases as you move from left to right across a period and increases as you move down a group. In the section of the Periodic Table shown, the element located furthest to the left and lowest down will have the most pronounced metallic properties. Based on the diagram, sodium (Na) in Group I, Period 3 is the element that best fits this description among the highlighted elements.
Question 22
- It has a higher melting point than caesium.
- It has a lower density than sodium.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
In Group I, as you go down the group, melting points decrease and densities generally increase. Caesium is near the bottom, so it has a relatively low melting point. For E to have a higher melting point than caesium, it must be above it in the group. Sodium has a certain density; for E to have a lower density than sodium, it must be above sodium. The only Group I element above both sodium and caesium is lithium (Li).
Question 23
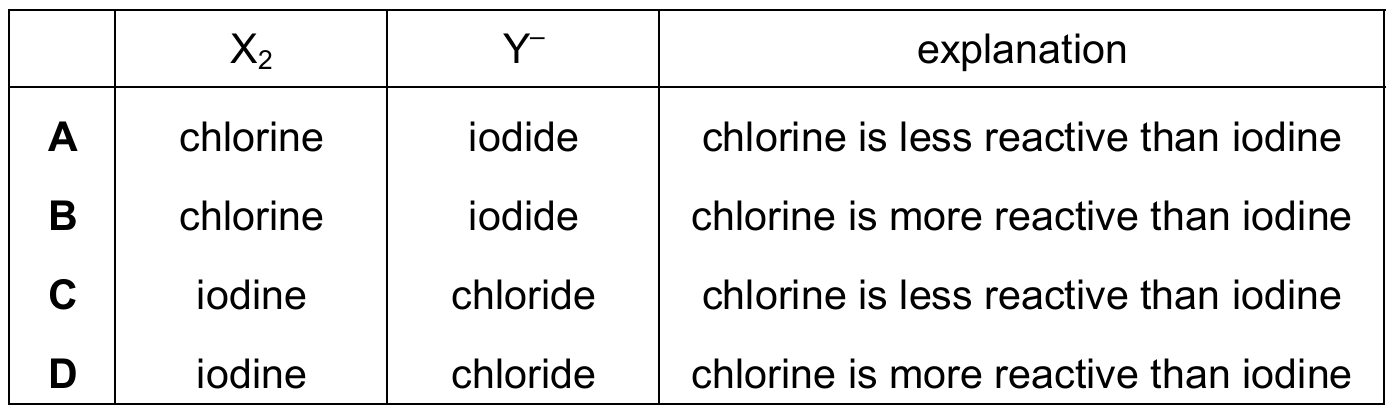
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
In Group VII, a more reactive halogen can displace a less reactive halogen from its halide compound. Chlorine is higher up the group and therefore more reactive than iodine. So chlorine (X₂) will displace iodide ions (Y⁻) from solution, forming chloride ions and iodine. The reaction takes place precisely because chlorine is more reactive than iodine, making row B the correct choice.
Question 24

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
The properties listed—a very high melting point (1555°C), a very high boiling point, formation of a coloured oxide (brown), and use as a catalyst—are all characteristic features of transition elements. Group I metals have low melting points and are not used as catalysts in their elemental form. Group VII elements are non-metals. Group VIII elements are noble gases, which are unreactive and do not form oxides. The description fits perfectly with transition metals like iron.
Question 25

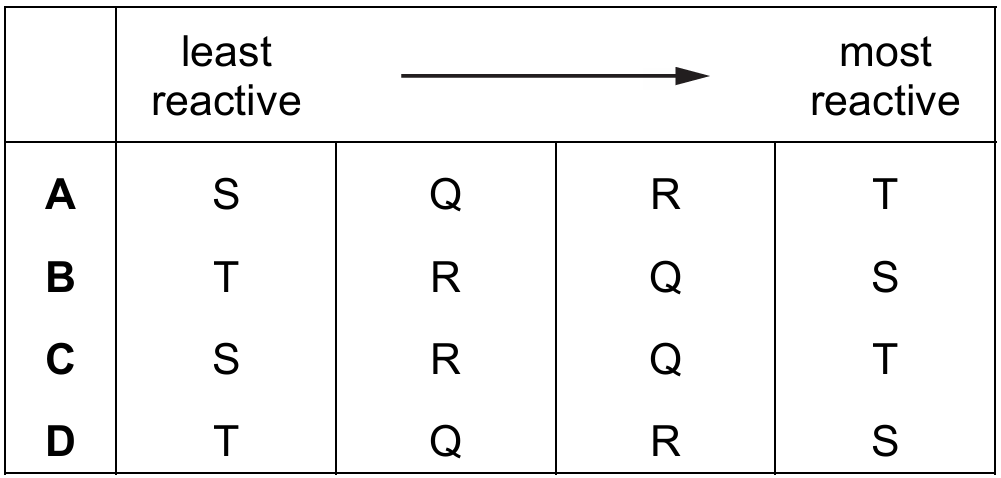
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Metal S shows no reaction with either cold water or acid, so it is the least reactive. Metal T reacts vigorously with both, so it is the most reactive. Metal Q reacts slowly with water and acid, while metal R shows no reaction with water but does fizz with acid. A metal that can react with water is more reactive than one that cannot, so Q is more reactive than R. The correct order from least to most reactive is therefore S, R, Q, T.
Question 26
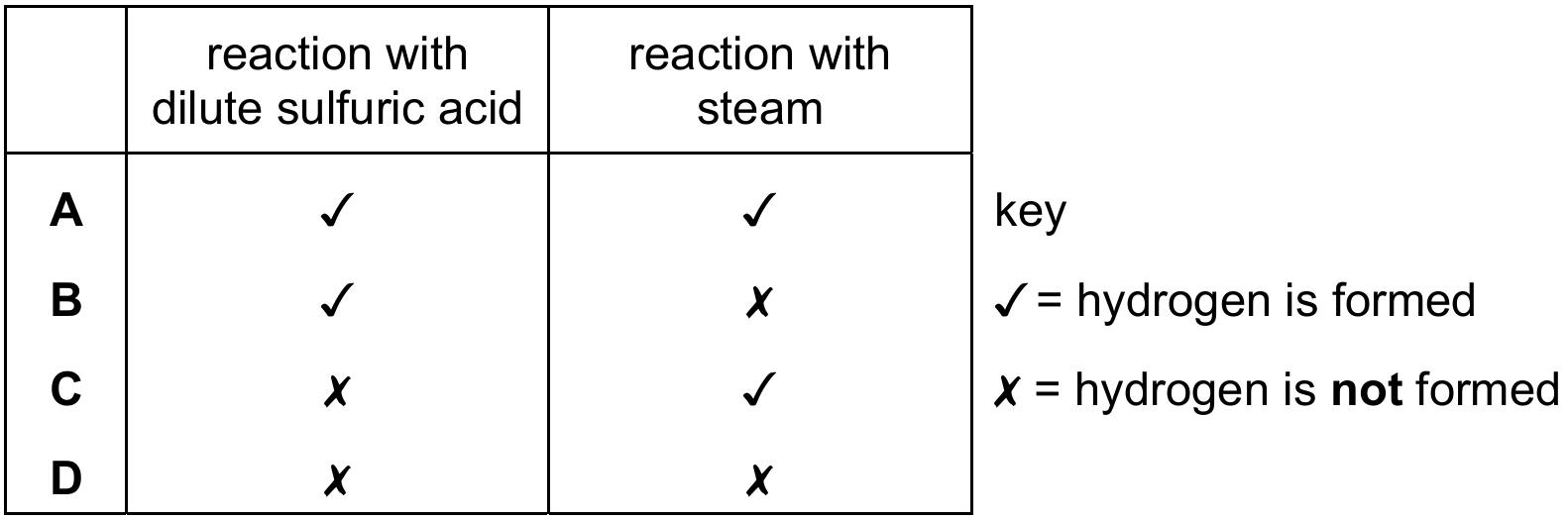
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Magnesium is a reactive metal. With dilute sulfuric acid, it reacts to form magnesium sulfate and hydrogen gas. The equation is: Mg + H₂SO₄ → MgSO₄ + H₂. When magnesium reacts with steam, it forms magnesium oxide and also releases hydrogen gas: Mg + H₂O → MgO + H₂. So hydrogen is formed as a product in both reactions.
Question 27
- It is more reactive than calcium.
- The main ore of aluminium is bauxite.
- It can be extracted from its oxide using carbon.
- Brass is an alloy of aluminium and copper.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Statement 1 is false: in the reactivity series, calcium is above aluminium, meaning calcium is more reactive. Statement 2 is true: bauxite (Al₂O₃) is indeed the main ore from which aluminium is extracted. Statement 3 is false: aluminium is too reactive to be extracted by reduction with carbon; it must be extracted by electrolysis. Statement 4 is false: brass is an alloy of copper and zinc, not aluminium. Therefore, only statement 2 is correct.
Question 28
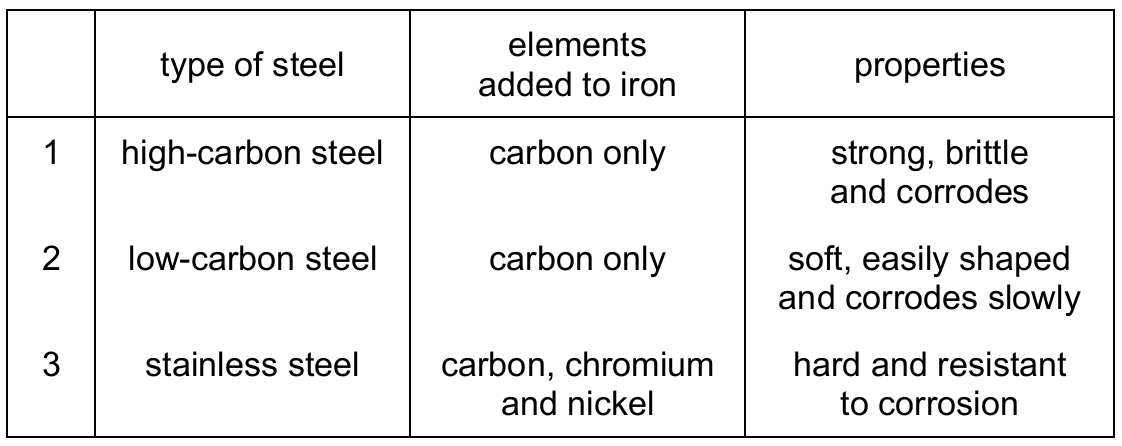
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Cutlery needs to be hard, strong, and resistant to corrosion (rusting) since it comes into contact with water and food. Stainless steel (type 3) is an alloy containing chromium and nickel, which makes it hard and highly resistant to rust. High-carbon steel (type 1) is brittle and corrodes, while low-carbon steel (type 2) is too soft and still corrodes. Therefore, only stainless steel is suitable for cutlery.
Question 29
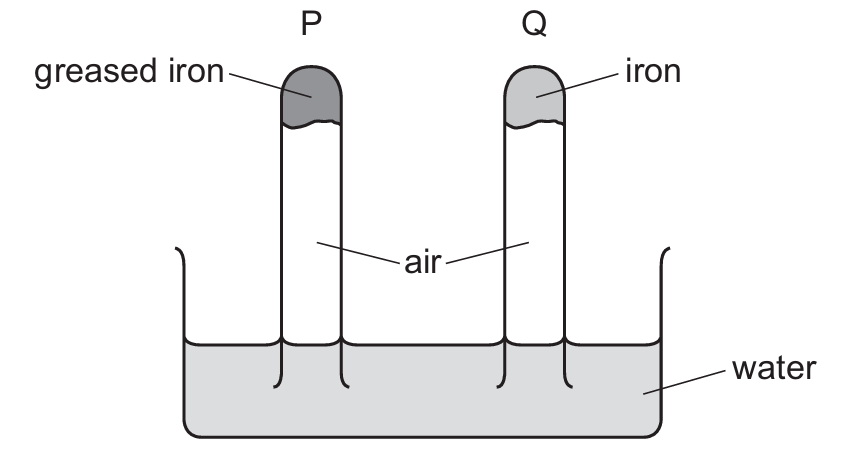

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Rusting of iron requires both water and oxygen. In tube P, a drying agent (anhydrous calcium chloride) absorbs all moisture, so there is no water present. Without water, rusting cannot occur, and no oxygen is consumed, so the water level remains unchanged. In tube Q, water is present along with air, so rusting takes place. Rusting consumes oxygen from the air, reducing the pressure inside the tube, and as a result, water from the beaker rises up the tube.
Question 30
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Plants need three primary elements: nitrogen (N), phosphorus (P), and potassium (K). We need two compounds that between them contain all three. Potassium nitrate (KNO₃) provides nitrogen and potassium. Calcium phosphate (Ca₃(PO₄)₂) provides phosphorus. Together, they supply all three essential elements: N, P, and K. The other combinations either miss one element or do not provide all three between the two compounds.
Question 31
- anhydrous cobalt(II) chloride
- anhydrous copper(II) sulfate
- litmus
- methyl orange
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Anhydrous cobalt(II) chloride is blue and turns pink when it comes into contact with water. Anhydrous copper(II) sulfate is white and turns blue when hydrated by water. Both are classic chemical tests used to detect the presence of water. Litmus and methyl orange are acid-base indicators; they change colour based on pH, not specifically due to water. So substances 1 and 2 are correct.
Question 32
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
When methane (CH₄) burns in a plentiful supply of oxygen, it undergoes complete combustion to produce carbon dioxide and water. However, when the oxygen supply is limited, incomplete combustion occurs, producing carbon monoxide (CO) and/or carbon (soot). Carbon monoxide is a toxic, colourless, odorless gas. Hydrogen is not produced, and sulfur dioxide would require sulfur in the fuel, which methane does not contain.
Question 33
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
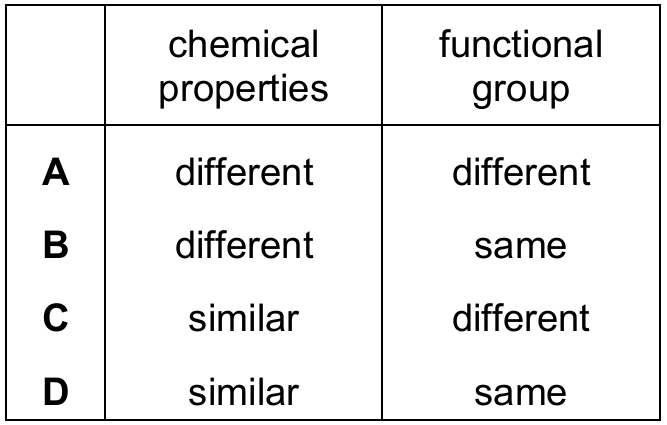
A homologous series is a group of organic compounds that share the same functional group and, because of this, exhibit very similar chemical properties. For example, all alkanes are unreactive except in combustion and substitution, all alkenes undergo addition reactions, etc. Therefore, compounds belonging to the same homologous series will always have similar chemical properties and the same functional group.
Question 34

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Compound Z has the molecular formula C₄H₁₀, which fits the general formula for alkanes (CₙH₂ₙ₊₂). This means Z is butane, an alkane. Alkanes are saturated hydrocarbons that generally only undergo combustion and substitution reactions. They do not react with steam. With chlorine, in the presence of ultraviolet (UV) light, alkanes undergo a substitution reaction where a hydrogen atom is replaced by a chlorine atom.
Question 35
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
In the fractionating column, the smallest molecules with the lowest boiling points are collected at the top. The order of fractions from lowest boiling point to highest is: refinery gas (lowest), then gasoline, naphtha, kerosene, diesel oil, fuel oil, lubricating oil, and bitumen (highest). Option C correctly lists gasoline → naphtha → kerosene → diesel oil, which is in order of increasing boiling point.
Question 36
- Cracking produces petrol for cars.
- Cracking only produces short-chain alkenes.
- Cracking produces alkenes used to make polymers.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Cracking is the process of breaking down large alkane molecules into smaller, more useful hydrocarbons. Statement 1 is correct because cracking produces shorter-chain alkanes that are used in petrol. Statement 2 is incorrect because cracking produces both short-chain alkanes and alkenes, not just alkenes. Statement 3 is correct because the alkenes produced (like ethene and propene) are used as monomers to manufacture polymers such as poly(ethene).
Question 37
- hydrocarbon
- monomer
- saturated
- unreactive
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Ethene (C₂H₄) is a hydrocarbon because it is composed only of carbon and hydrogen atoms. It is a monomer because it can be polymerised to form poly(ethene). However, ethene contains a carbon-carbon double bond (C=C), which makes it an unsaturated compound, not saturated. The double bond also makes ethene fairly reactive, as it readily undergoes addition reactions. So ethene is described as a hydrocarbon and a monomer.
Question 38
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
To measure a volume as precise as $25.00\,\mathrm{cm}^3$ (i.e., to two decimal places), a high-precision apparatus is needed. A burette is graduated in $0.1\,\mathrm{cm}^3$ intervals, allowing the user to read volumes to the nearest $0.05\,\mathrm{cm}^3$, making it suitable for such accurate measurements. A beaker gives only approximate volumes, a measuring cylinder is less precise (typically to the nearest $0.5\,\mathrm{cm}^3$), and a dropping pipette is not calibrated for specific volumes.
Question 39
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
When excess solid magnesium oxide is added to dilute nitric acid, the acid reacts to form soluble magnesium nitrate and water, but the unreacted magnesium oxide remains as an insoluble solid. To remove this excess solid from the liquid, we use filtration. The mixture is poured through filter paper in a funnel; the solid remains on the filter paper (as residue) and the clear solution (filtrate) passes through.
Question 40
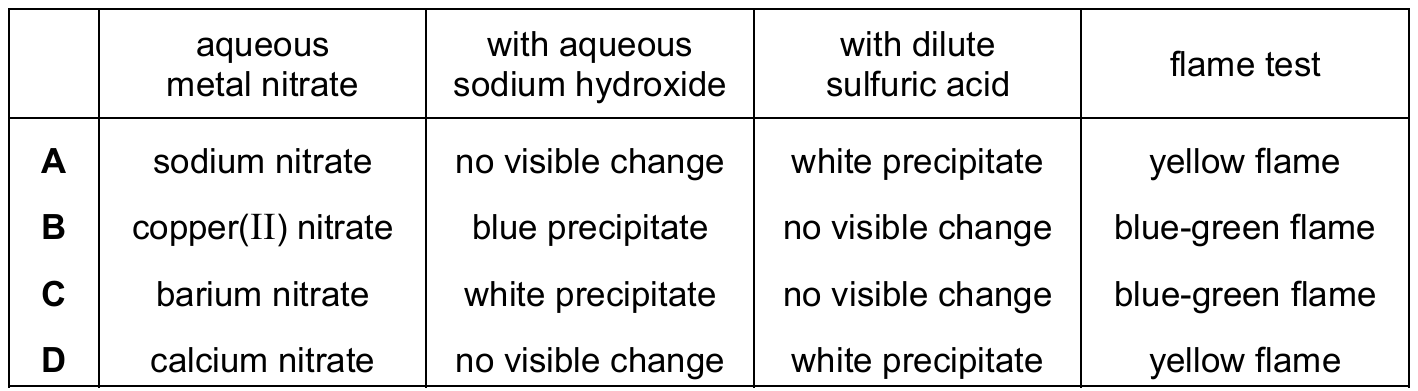
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Copper(II) ions (Cu²⁺) react with aqueous sodium hydroxide to form a light blue precipitate of copper(II) hydroxide, which is insoluble in excess. Copper(II) sulfate is soluble, so with dilute sulfuric acid there is no visible change (no precipitate). In a flame test, copper(II) ions produce a characteristic blue-green flame. Sodium nitrate would not form a precipitate with sulfuric acid; barium nitrate gives a white precipitate with sulfuric acid and a light green flame; calcium nitrate gives a white precipitate with sodium hydroxide and an orange-red flame.
