Question 1
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
In solids, particles are packed very closely together in a regular arrangement, with hardly any space between them. In liquids, particles are also close together but can move around each other, so the distance between them is similar to that in solids. In gases, however, the particles are spread far apart with large distances between them. Therefore, the states where particles are a similar distance apart are solid and liquid only, making option B the correct choice.
Question 2
- The volume of gas decreases.
- The gas molecules move faster.
- The pressure inside the cylinder increases.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
When a sealed container of fixed shape (constant volume) is heated, the gas molecules gain kinetic energy and therefore move faster, so statement 2 is correct. Since the volume cannot change due to the fixed shape of the container, the faster-moving particles collide with the walls more frequently and with greater force, causing the pressure inside to increase, so statement 3 is also correct. Statement 1 is incorrect because the volume cannot decrease in a container with a fixed shape. Thus, statements 2 and 3 are the changes that take place, making option C the right answer.
Question 3
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
When a sodium chloride crystal is dropped into water, the ionic bonds in the crystal lattice are broken as the water molecules surround the $\text{Na}^+$ and $\text{Cl}^-$ ions, so dissolving occurs. Once dissolved, the sodium and chloride ions spread out from an area of high concentration to areas of lower concentration until they are evenly distributed throughout the water, which is the process of diffusion. So both dissolving and diffusion take place, making option A the correct answer.
Question 4
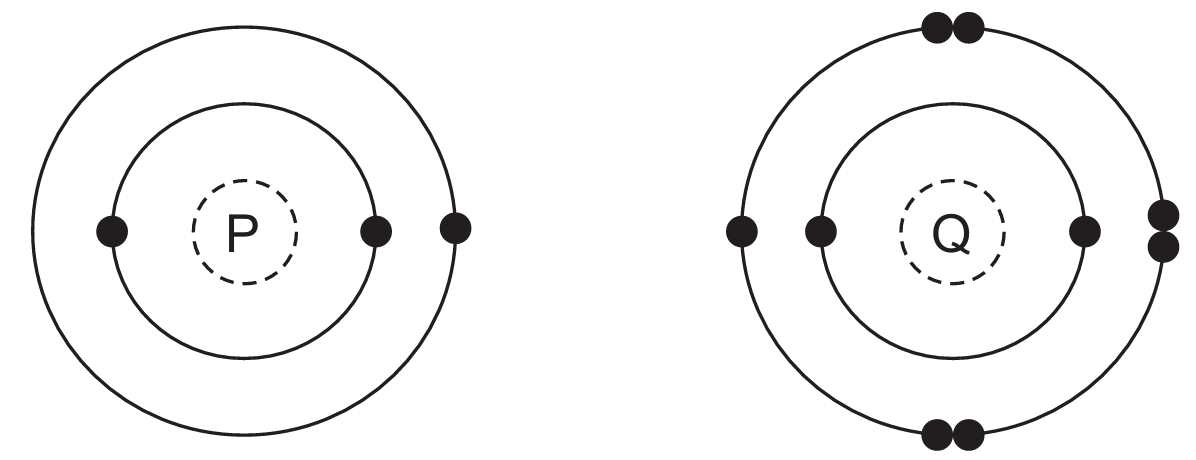
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• TOPIC 2.5: Simple molecules and covalent bonds (State that a covalent bond is formed when a pair of electrons is shared between two atoms leading to noble gas electronic configurations)
▶️ Answer/Explanation
Detailed solution:
Looking at the electronic structures, atom P has 6 electrons in its outer shell and needs to share 2 more electrons to achieve a full outer shell, while atom Q has 7 electrons in its outer shell and needs to share 1 more electron. Since both atoms need to gain electrons to achieve noble gas configurations, they will share electrons, forming covalent bonds. To satisfy the electron requirements, one atom of P will share electrons with two atoms of Q, resulting in the formula $\text{PQ}_2$. Therefore, the compound has covalent bonding with formula $\text{PQ}_2$, making option A correct.
Question 5
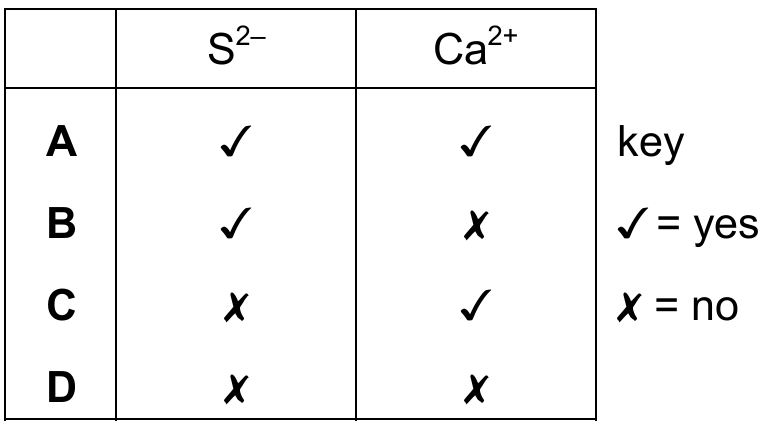
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
A sulfur atom ($\text{S}$) has proton number 16, with electronic configuration 2,8,6. When it gains 2 electrons to form the $\text{S}^{2-}$ ion, the configuration becomes 2,8,8, so $\text{S}^{2-}$ matches. A calcium atom ($\text{Ca}$) has proton number 20, with electronic configuration 2,8,8,2. When it loses 2 electrons to form the $\text{Ca}^{2+}$ ion, the configuration also becomes 2,8,8. Since both ions can have the 2,8,8 configuration, the correct choice is option A where both are marked yes.
Question 6
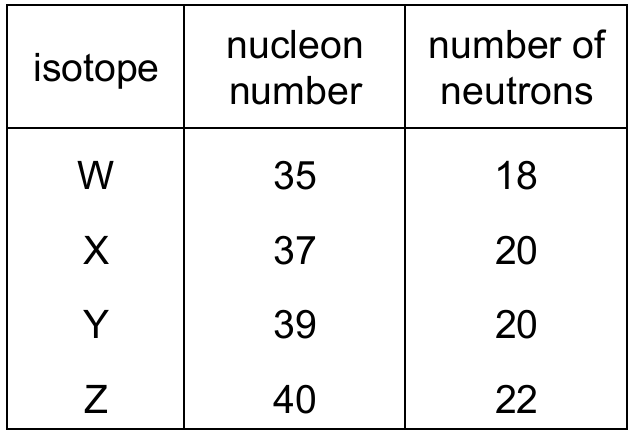
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
To find the proton number of each isotope, subtract the number of neutrons from the nucleon number. For W: $35 – 18 = 17$ protons; X: $37 – 20 = 17$ protons; Y: $39 – 20 = 19$ protons; Z: $40 – 22 = 18$ protons. Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons. Since W and X both have 17 protons (but different nucleon numbers), they are isotopes of the same element, making statement A correct. The other statements are incorrect: X and Y have different proton numbers so they are different elements, Y and Z have different proton numbers, and Z has 18 protons while Y has 19, so Z does not have a higher proton number than Y.
Question 7
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
An ionic bond is the electrostatic force of attraction that holds together positively charged ions (cations) and negatively charged ions (anions) in an ionic compound. This attraction is very strong, which explains why ionic compounds have high melting and boiling points. Option A correctly describes it as a strong electrostatic attraction between cations and anions. Option B is wrong because ionic bonds are between ions, not between protons and electrons, and options C and D are incorrect because ionic bonds are strong, not weak.
Question 8
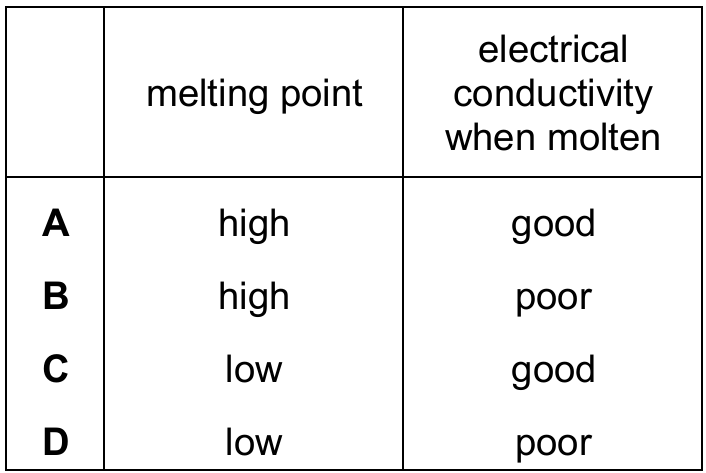
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Simple molecular compounds consist of small molecules held together by weak intermolecular forces, which means only a small amount of energy is needed to overcome these forces, resulting in low melting points. Additionally, simple molecular compounds do not contain free ions or delocalised electrons to carry charge, so they are poor conductors of electricity even when molten. Looking at the table, only row D shows both a low melting point and poor electrical conductivity, making it the correct description of simple molecular compounds.
Question 9
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Diamond has a giant covalent structure where each carbon atom is strongly bonded to four other carbon atoms in a tetrahedral arrangement, making it the hardest natural substance known. Since boron nitride has a similar structure to diamond, it will share similar properties. Diamond is very hard, does not conduct electricity, is insoluble in water, and is used in cutting tools—not as a lubricant (that’s graphite). Therefore, the property boron nitride is most likely to have is that it is very hard, making option D correct.
Question 10
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
The molecular formula tells us exactly which elements are present in a molecule and how many atoms of each element there are. For example, the molecular formula of water, $\text{H}_2\text{O}$, shows that one molecule contains two hydrogen atoms and one oxygen atom—giving both the type and number of atoms. Option A is close but incomplete because it only mentions the total number of atoms without specifying the type, while options B and C describe other concepts. Therefore, option D is the most accurate definition of molecular formula.
Question 11
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
To calculate the relative formula mass of $\mathrm{Ca(OH)_2}$, we add up the relative atomic masses of all the atoms present. Calcium (Ca) has an atomic mass of 40, oxygen (O) is 16, and hydrogen (H) is 1. The formula contains one Ca, two O atoms, and two H atoms: $M_r = 40 + 2(16) + 2(1) = 40 + 32 + 2 = 74$. So the relative formula mass of calcium hydroxide is 74, giving option C as the correct answer.
Question 12
- Electroplating improves the appearance of the object.
- Electroplating insulates the metal object.
- Electroplating makes the metal object more resistant to corrosion.
- Electroplating makes the metal object softer.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Electroplating involves coating a metal object with a thin layer of another metal, such as silver or chromium, using electrolysis. This process improves the appearance of the object by giving it a shiny, attractive surface, so statement 1 is correct. It also makes the object more resistant to corrosion by protecting the underlying metal from air and moisture, so statement 3 is correct. Electroplating does not insulate the object—metals conduct electricity—and it doesn’t make the object softer; in fact, it often makes the surface harder. Therefore, statements 1 and 3 are the correct reasons, making option B the right choice.
Question 13
- The overall equation for the reaction is $2H_{2} + O_{2} \rightarrow 2H_{2}O$.
- The hydrogen required comes from the air.
- The waste gas causes respiratory problems.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Statement 1 is correct: the overall reaction in a hydrogen-oxygen fuel cell is indeed $2\text{H}_2 + \text{O}_2 \rightarrow 2\text{H}_2\text{O}$, producing water as the only chemical product. Statement 2 is incorrect because hydrogen does not come from the air—air contains very little hydrogen; the hydrogen must be manufactured or supplied separately. Statement 3 is also incorrect because the only waste product from a hydrogen-oxygen fuel cell is water, which does not cause respiratory problems. Therefore, only statement 1 is correct, making option B the right answer.
Question 14
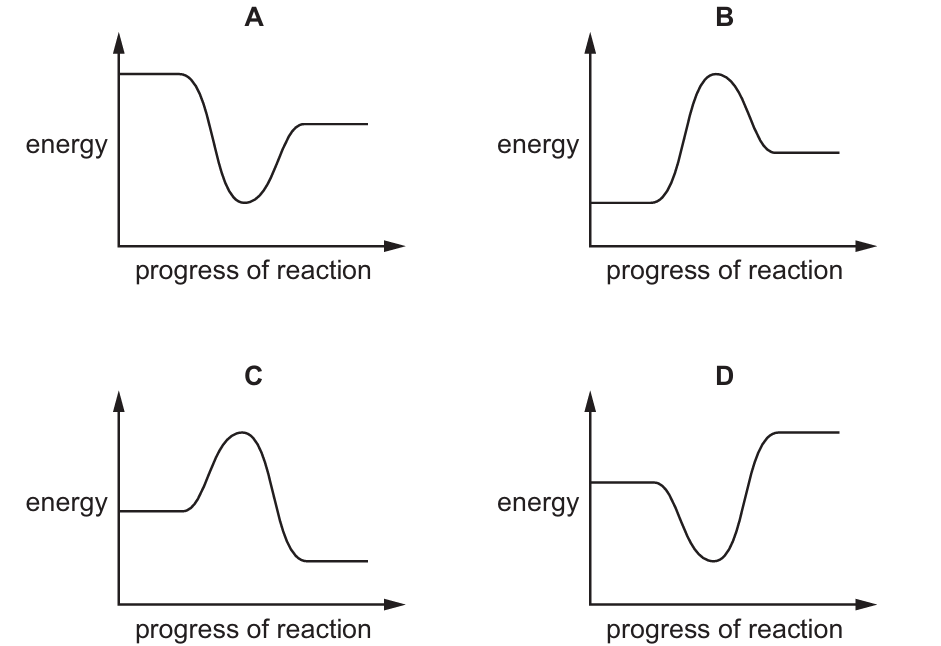
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
In a reaction pathway diagram, an endothermic reaction is one where the products are at a higher energy level than the reactants, meaning energy has been absorbed from the surroundings. Looking at the four diagrams, option B shows the products at a higher energy level than the reactants, with the arrow pointing upward from reactants to products, indicating that energy is taken in. This is the defining feature of an endothermic reaction pathway, so option B is correct.
Question 15
- rusting of iron
- condensing of steam
- dissolving of sugar in water
- cracking of alkanes
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
A physical change is one where no new substance is formed—the change is easily reversible and involves changes in state or dissolving. Rusting of iron (1) is a chemical change because a new substance, hydrated iron(III) oxide, is formed. Cracking of alkanes (4) is also a chemical change because large hydrocarbon molecules are broken down into smaller, different molecules. However, condensing of steam (2) only involves a change of state from gas to liquid, and dissolving sugar in water (3) does not produce a new substance—both are physical changes. So changes 2 and 3 are physical, making option C correct.
Question 16
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Blue copper(II) sulfate crystals are hydrated, containing water of crystallisation with the formula $\text{CuSO}_4\cdot5\text{H}_2\text{O}$. Heating drives off this water, leaving white anhydrous copper(II) sulfate. This is a reversible reaction: $\text{CuSO}_4\cdot5\text{H}_2\text{O} \rightleftharpoons \text{CuSO}_4 + 5\text{H}_2\text{O}$. When the white powder is left in air, it absorbs moisture (water vapour) from the air, which reverses the reaction and reforms the blue hydrated crystals. Option D correctly explains that water is absorbed from the air and causes the reaction to reverse.
Question 17
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
In this reaction, carbon reacts with silver oxide: $\text{C} + 2\text{Ag}_2\text{O} \rightarrow \text{CO}_2 + 4\text{Ag}$. Reduction is defined as the loss of oxygen. Silver oxide ($\text{Ag}_2\text{O}$) loses its oxygen to carbon, forming silver metal. Since silver oxide loses oxygen during the reaction, it is the substance that is reduced. Carbon gains oxygen (forming carbon dioxide) so carbon is oxidised. Therefore, option D is correct.
Question 18

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Base Q dissolves in water to form an alkaline solution. Alkalis contain $\text{OH}^-$ (hydroxide) ions, which are responsible for their basic properties. Thymolphthalein is an indicator that turns blue in alkaline conditions (pH above about 9.3) and is colourless in acidic conditions. Since aqueous Q is alkaline, the thymolphthalein will turn blue, and this colour change is due to the presence of $\text{OH}^-$ ions. Therefore, option C correctly shows the colour as blue and the ion as $\text{OH}^-$.
Question 19
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
When a base such as calcium hydroxide reacts with an ammonium salt such as ammonium chloride, ammonia gas is produced. The reaction is: $\text{Ca(OH)}_2 + 2\text{NH}_4\text{Cl} \rightarrow \text{CaCl}_2 + 2\text{H}_2\text{O} + 2\text{NH}_3$. Warming the mixture speeds up the reaction and helps drive off the ammonia gas, which can be recognised by its pungent smell and its ability to turn damp red litmus paper blue. This is a standard test for ammonium salts—heating them with a base produces ammonia gas, making option D the correct answer.
Question 20
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Elements in the same group of the Periodic Table have the same number of electrons in their outermost shell. It is these outer shell electrons that determine how an element reacts chemically, because chemical reactions involve the transfer or sharing of outer electrons. For example, all Group I elements have one electron in their outer shell, and all Group VII elements have seven electrons in their outer shell. Options A and C are incorrect because electrons are not in the nucleus and neutrons do not determine chemical properties. Option D is wrong because neutrons are not found in shells at all. Therefore, option B is the correct explanation.
Question 21
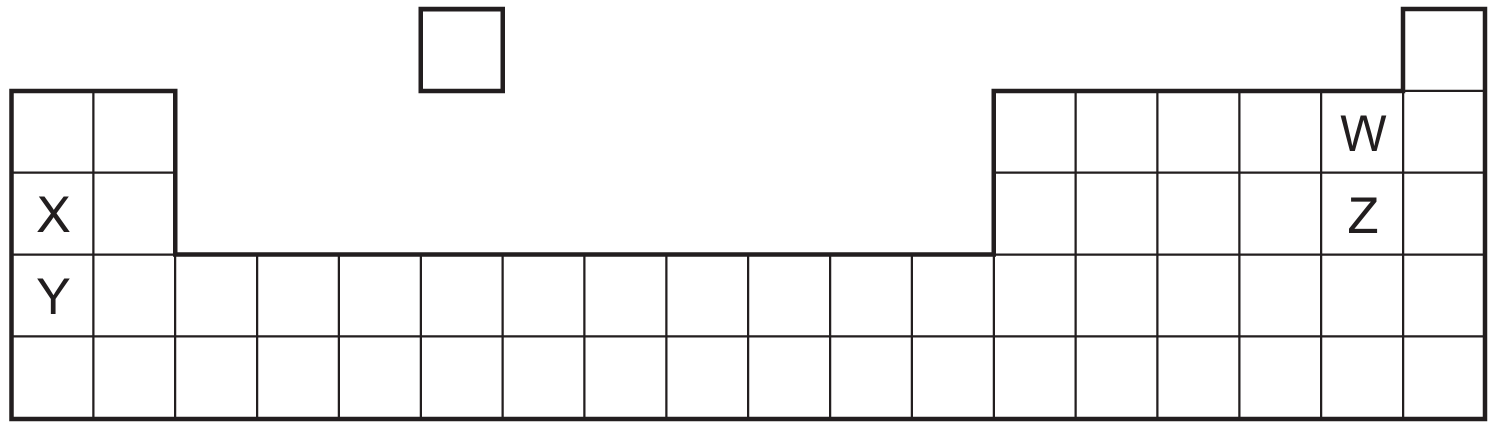
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
• TOPIC 8.2: Group I properties (Describe the Group I alkali metals… general trends down the group, limited to: increasing reactivity)
• TOPIC 8.3: Group VII properties (Describe the Group VII halogens… general trends down the group, limited to: decreasing reactivity)
▶️ Answer/Explanation
Detailed solution:
Looking at the positions in the Periodic Table, W and X are Group I alkali metals, while Y and Z are Group VII halogens. For Group I metals, reactivity increases down the group, so Y is more reactive than W (Y is below W). Looking at X and Z: X is a Group I metal and Z is a Group VII halogen in the same period. Across a period, metals are on the left and are more reactive than non-metals on the right in terms of losing electrons. However, the diagram shows relative positions, and Y (being lower down Group I) is more reactive than X (higher up), so Y is more reactive than X. For non-metals, reactivity decreases down Group VII, so W (above Z) would be a different element. Based on the diagram arrangement, Y is more reactive than X, and W is more reactive than Z, making option C the correct statement.
Question 22
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Let’s check each option using solubility rules. In option A: ammonium carbonate is soluble (ammonium salts are soluble), silver chloride is insoluble (only silver and lead chlorides are insoluble), and sodium sulfate is soluble (sodium salts are soluble)—so A is incorrect. In option B: barium sulfate is insoluble (barium sulfate is an exception), but calcium chloride is soluble (most chlorides are soluble)—so B is incorrect. In option C: calcium nitrate is soluble (all nitrates are soluble)—so C is incorrect. In option D: calcium sulfate is insoluble (calcium sulfate is an exception among sulfates), magnesium hydroxide is insoluble (most hydroxides are insoluble except Group I and ammonium), and silver chloride is insoluble (silver chloride is an exception among chlorides). All three in option D are indeed insoluble, so D is the correct answer.
Question 23
- It is a mixture.
- It is a compound.
- It is an alloy.
- It conducts electricity.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
18 carat gold is made up of 75% gold and 25% copper mixed together physically, not chemically combined, so it is a mixture (statement 1 correct) and not a compound (statement 2 incorrect). Because it is a mixture of metals, it is specifically called an alloy (statement 3 correct). Like pure gold and copper, 18 carat gold contains delocalised electrons that can move freely through the structure, so it conducts electricity (statement 4 correct). Therefore, statements 1, 3, and 4 are all correct, making option B the right choice.
Question 24
F does not react with water or dilute acid, but the oxide of F is reduced by carbon.
The oxide of G is not reduced by carbon, but G reacts vigorously with cold water.
H does not react with water or steam, but reacts with dilute acid.
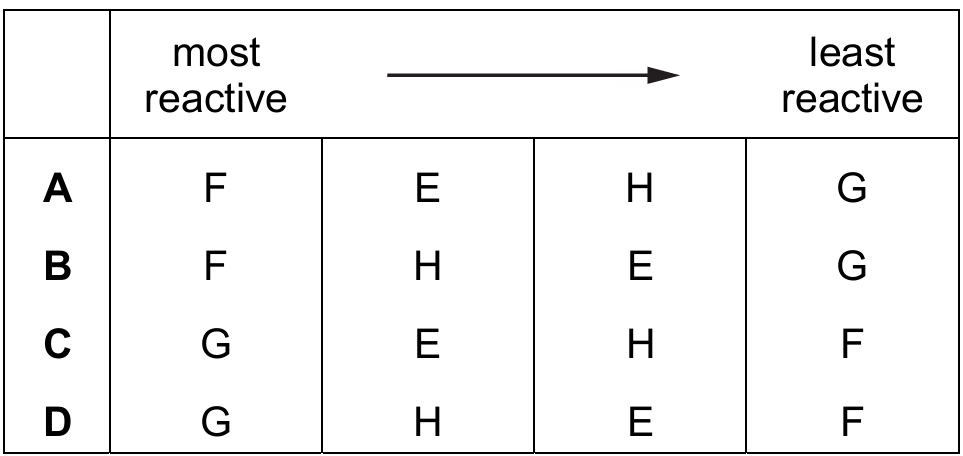
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Let’s rank the metals based on the given information. G reacts vigorously with cold water—this is characteristic of the most reactive metals like potassium or sodium, so G is the most reactive. E reacts with steam but not cold water—this places it below G but still quite reactive, like magnesium. H reacts with dilute acid but not with water or steam—this puts it lower in the reactivity series, like zinc or iron. F does not react with water or dilute acid, and its oxide can be reduced by carbon—this means F is the least reactive, below carbon in the reactivity series, like copper. So the order from most to least reactive is: G, E, H, F, which matches option C.
Question 25
- some metal compounds
- plastics
- dissolved oxygen
- nitrates and phosphates from fertilisers and detergents
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Some metal compounds (1) found in natural water, such as those containing calcium and magnesium, provide essential minerals that are needed by aquatic organisms for growth and development, so they can be beneficial. Dissolved oxygen (3) is absolutely essential for aquatic life—fish and other organisms use it for respiration. Plastics (2) are harmful to aquatic life as they can choke animals or release toxic substances, and nitrates and phosphates (4) can lead to eutrophication which depletes oxygen and damages aquatic ecosystems. Therefore, only substances 1 and 3 are beneficial, making option A correct.
Question 26
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
In the treatment of domestic water supplies, activated carbon (also called activated charcoal) is used to remove tastes and odours from the water. Carbon works through a process called adsorption, where impurities stick to the large surface area of the carbon particles. Chlorine is used for disinfection (killing microbes), not for removing tastes and odours. Calcium and copper are not used for this purpose in water treatment. Therefore, the element used to remove tastes and odours is carbon, making option B correct.
Question 27
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
NPK fertilisers contain the elements nitrogen (N), phosphorus (P), and potassium (K) to promote healthy plant growth. Looking at the options, sodium nitrate ($\text{NaNO}_3$) contains nitrogen, which is one of the three essential elements in NPK fertilisers. Nitrates and ammonium salts are commonly used as sources of nitrogen in fertilisers. Aluminium sulfate, magnesium chloride, and calcium ethanoate do not contain nitrogen, phosphorus, or potassium in forms suitable for fertilisers. Therefore, sodium nitrate is the only salt that could be used as a component of an NPK fertiliser, making option A correct.
Question 28
- carbon dioxide
- oxygen
- light
- water
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Photosynthesis is the process by which plants make glucose. The word equation is: carbon dioxide + water → glucose + oxygen, and this reaction requires light energy captured by chlorophyll. So carbon dioxide (1), light (3), and water (4) are all essential for photosynthesis to occur. Oxygen (2) is not essential for photosynthesis—it is actually a product of the reaction, not a requirement. Therefore, substances 1, 3, and 4 are essential, making option B the correct choice.
Question 29

Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Looking at the reaction sequence: $\text{CaCO}_3 \xrightarrow{\text{M}} \text{CO}_2 \xrightarrow{\text{N}} \text{CH}_4$. Reaction M involves calcium carbonate producing carbon dioxide. This occurs when calcium carbonate is heated strongly, breaking it down into calcium oxide and carbon dioxide: $\text{CaCO}_3 \rightarrow \text{CaO} + \text{CO}_2$. This is thermal decomposition (breaking down a compound using heat). For reaction N, carbon dioxide is converted to methane ($\text{CH}_4$). Methane burns in oxygen to produce carbon dioxide (combustion), but going from $\text{CO}_2$ to $\text{CH}_4$ would require a different process. However, in the context of this question, reaction N involves methane and relates to its combustion reaction. Therefore, M is thermal decomposition and N is combustion, making option B correct.
Question 30
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Alkanes are saturated hydrocarbons with the general formula $\text{C}_n\text{H}_{2n+2}$. Let’s check each compound: $\text{C}_2\text{H}_2$: for $n=2$, the alkane formula would be $\text{C}_2\text{H}_6$, so this is not an alkane (it’s an alkyne). $\text{C}_3\text{H}_8$: for $n=3$, $\text{C}_3\text{H}_{2(3)+2} = \text{C}_3\text{H}_8$, so this is an alkane. $\text{C}_4\text{H}_8$: for $n=4$, the alkane formula would be $\text{C}_4\text{H}_{10}$, so this is not an alkane (it’s an alkene). $\text{C}_5\text{H}_{10}$: for $n=5$, the alkane formula would be $\text{C}_5\text{H}_{12}$, so this is not an alkane (it’s an alkene or cycloalkane). Only one compound, $\text{C}_3\text{H}_8$, fits the alkane general formula, so option A is correct.
Question 31
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Pentane ($\text{C}_5\text{H}_{12}$) is an alkane—a saturated hydrocarbon with only single covalent bonds. Alkanes are generally unreactive but do undergo substitution reactions with halogens like chlorine. In this reaction, a chlorine atom replaces (substitutes) one hydrogen atom in the pentane molecule: $\text{C}_5\text{H}_{12} + \text{Cl}_2 \rightarrow \text{C}_5\text{H}_{11}\text{Cl} + \text{HCl}$. This is a photochemical reaction requiring ultraviolet light for activation. Alkanes cannot undergo addition reactions because they have no double bonds, and polymerisation and cracking involve different processes entirely. So the correct answer is substitution, option C.
Question 32
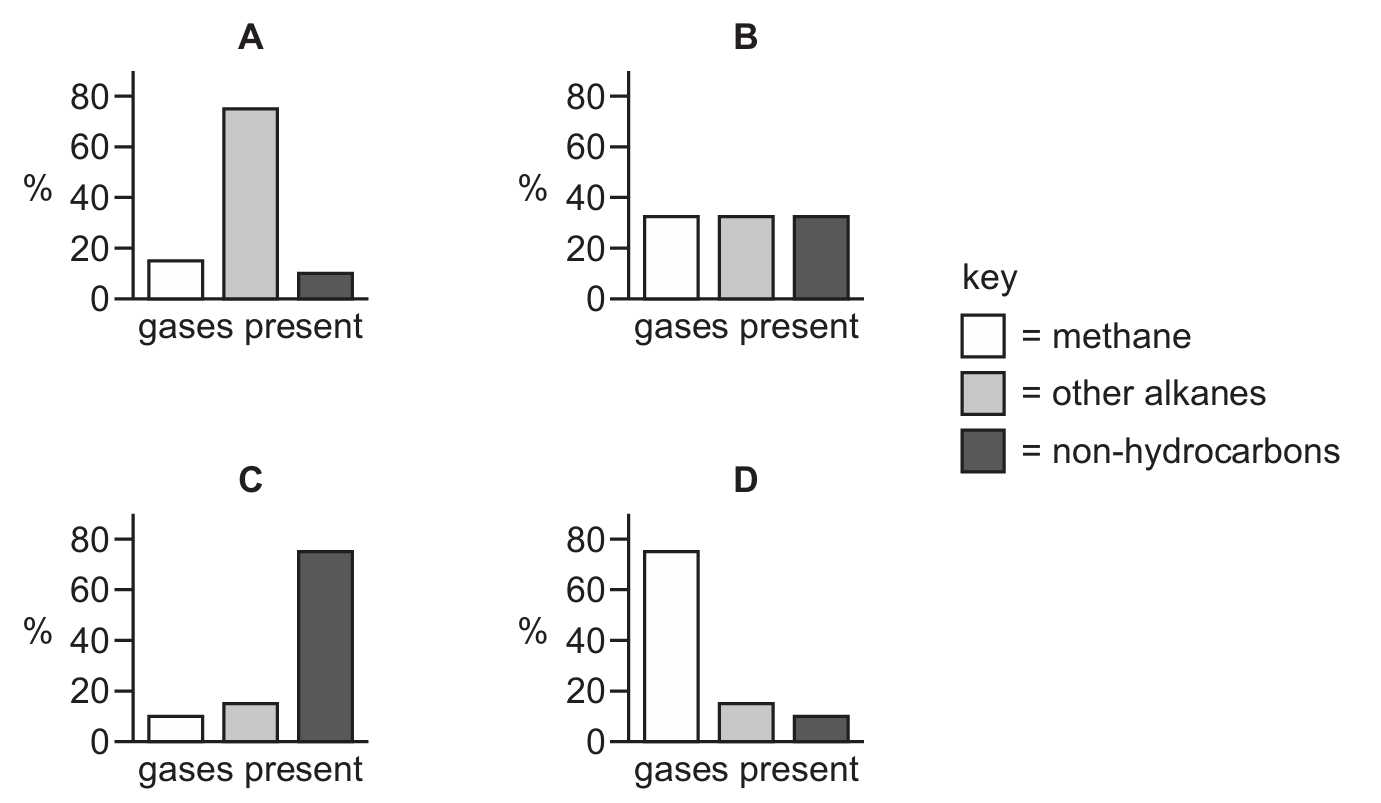
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Natural gas is primarily composed of methane ($\text{CH}_4$), typically making up around 80-95% of its composition. The remaining percentage consists of smaller amounts of other hydrocarbons such as ethane, propane, and butane. Looking at the bar charts, option D shows methane as the largest component by far, taking up the majority of the bar, with only a small portion dedicated to other hydrocarbons. This accurately represents the composition of natural gas, where methane is the main constituent. The other options show different distributions that do not match the true composition of natural gas.
Question 33
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Let’s check each statement. A is wrong: alcohols have the functional group $-\text{OH}$ (hydroxyl), while carboxylic acids have $-\text{COOH}$ (carboxyl). B is incorrect: fermentation occurs at 25–35°C, not 300°C (that’s the temperature for the catalytic addition of steam to ethene). C is incorrect: ethanol is produced by the addition of steam (water) to ethene, not hydrogen—adding hydrogen to ethene would produce ethane. D is correct: when alcohols combust, they react with oxygen to produce carbon dioxide and water: $\text{C}_2\text{H}_5\text{OH} + 3\text{O}_2 \rightarrow 2\text{CO}_2 + 3\text{H}_2\text{O}$. So water is indeed produced, making option D the right choice.
Question 34
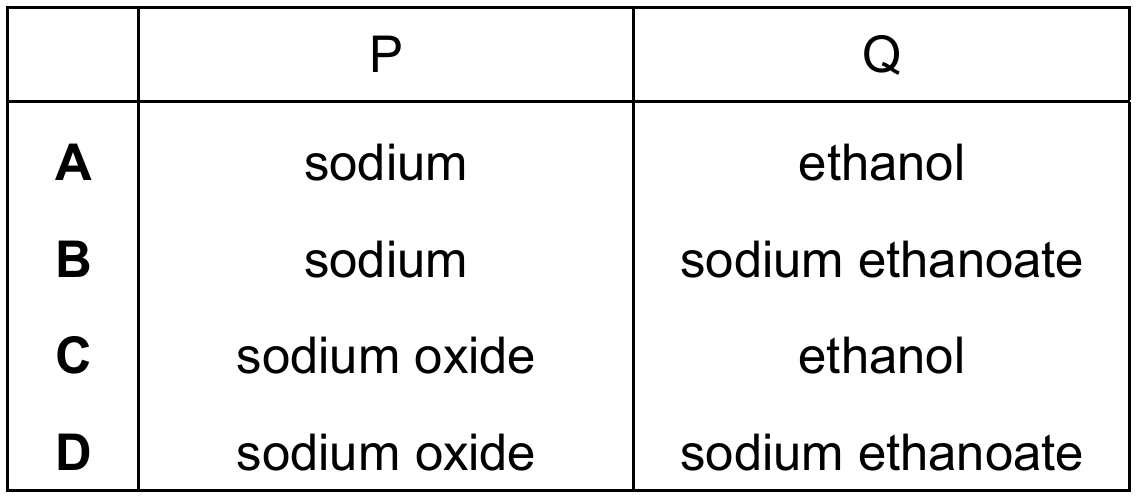
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Ethanoic acid reacts with reactive metals like sodium to produce a salt and hydrogen gas: $2\text{CH}_3\text{COOH} + 2\text{Na} \rightarrow 2\text{CH}_3\text{COONa} + \text{H}_2$. The salt formed is sodium ethanoate (Q). If P were sodium oxide (a base), the reaction would be: $2\text{CH}_3\text{COOH} + \text{Na}_2\text{O} \rightarrow 2\text{CH}_3\text{COONa} + \text{H}_2\text{O}$, producing water, not hydrogen. Since hydrogen is produced as shown in the equation, P must be a reactive metal, sodium, and Q is the salt sodium ethanoate. So option B correctly identifies P as sodium and Q as sodium ethanoate.
Question 35
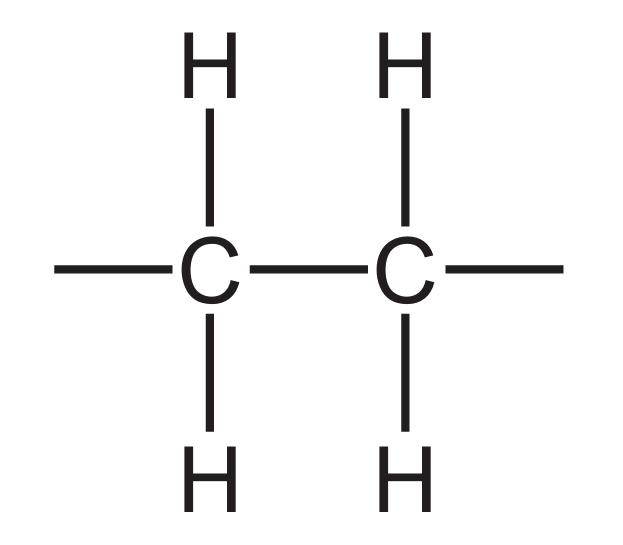
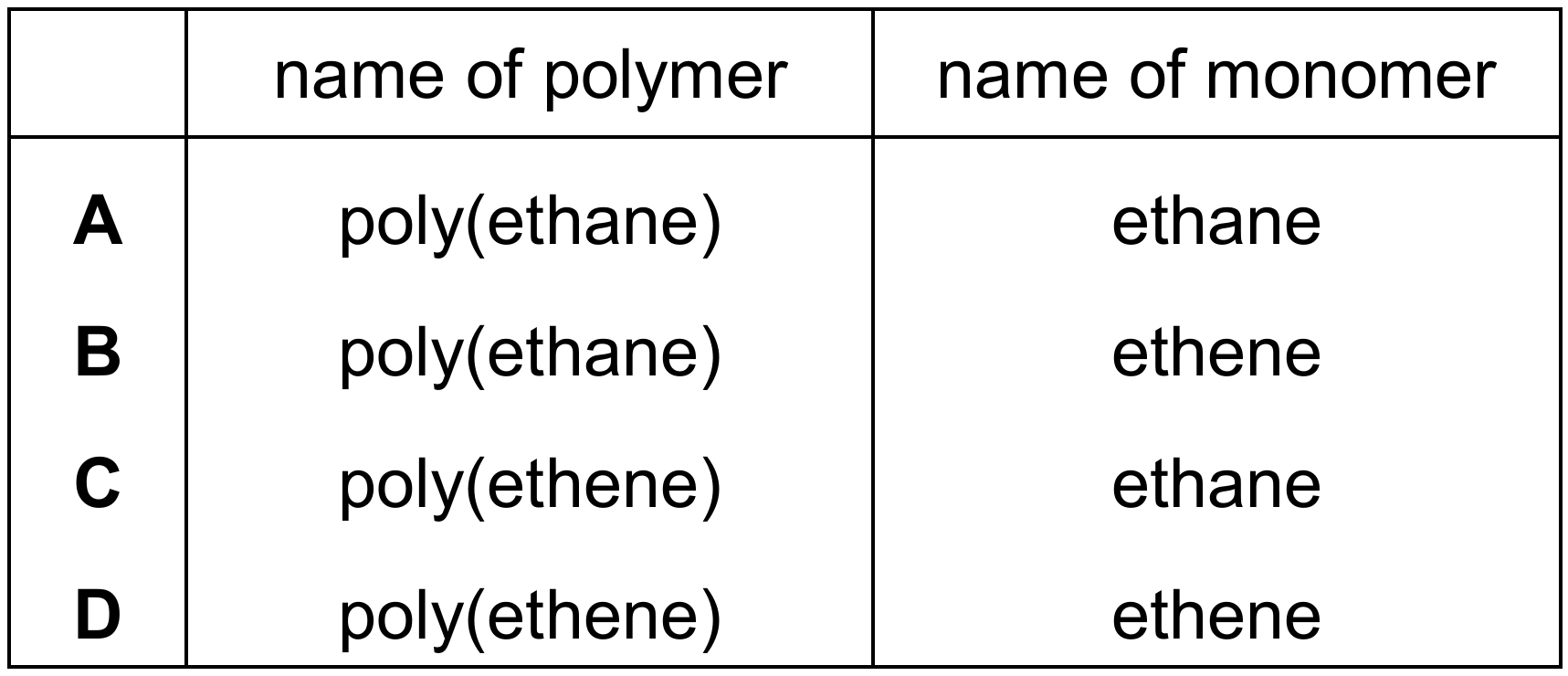
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
The repeat unit shown in the diagram is $-\text{CH}_2-\text{CH}_2-$, which is the repeat unit of poly(ethene), commonly known as polythene. The monomer used to make this polymer is ethene ($\text{CH}_2=\text{CH}_2$), which undergoes addition polymerisation where the carbon-carbon double bond breaks open and the molecules link together. Ethane ($\text{C}_2\text{H}_6$) is an alkane with only single bonds and cannot undergo addition polymerisation. Therefore, the polymer is poly(ethene) and the monomer is ethene, making option D the correct pairing.
Question 36
- It is a poor conductor of electricity.
- It does not react with water.
- It is resistant to oxidation.
- It has low density.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Plastics persist in the environment because they are not easily broken down. Property 2 (does not react with water) means that plastics won’t dissolve or be washed away by rain, rivers, or oceans—they simply persist. Property 3 (resistant to oxidation) means that plastics don’t degrade or break down when exposed to oxygen in the air, unlike many natural materials that rot or rust. These two properties directly explain why plastics accumulate and are difficult to remove from the environment. Being a poor conductor of electricity (1) and having low density (4) are properties of plastics but don’t directly explain why they are environmentally persistent. Therefore, properties 2 and 3 are the key reasons, making option C correct.
Question 37
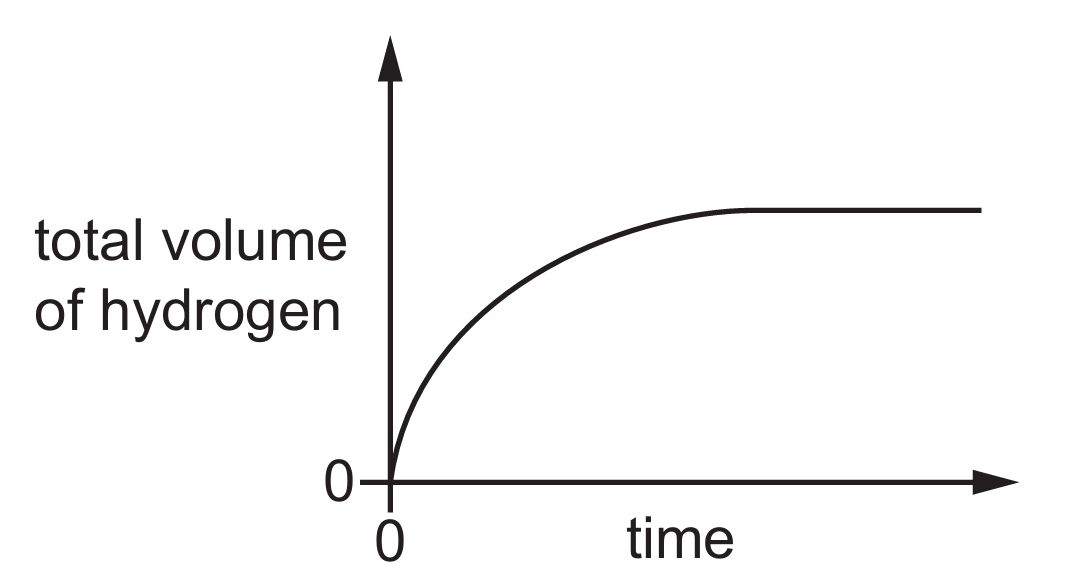
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
The graph shows the volume of hydrogen gas produced over time during a chemical reaction. To measure the volume of gas produced, a gas syringe is the most appropriate apparatus—it allows accurate measurement of gas volume as the reaction proceeds. The stop-watch is used to measure time intervals. A beaker cannot accurately measure gas volume, a pipette is used for measuring and transferring specific volumes of liquids, and a test-tube is used for holding small amounts of substances but cannot measure gas volume. Therefore, the other item of apparatus needed alongside the stop-watch is a gas syringe, making option B correct.
Question 38

- S contains only two dyes.
- X is insoluble in ethanol.
- S contains Y and Z.
- S contains W.
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
Looking at the chromatogram, dye X has not moved from the baseline (origin), meaning it is insoluble in the solvent (ethanol)—it did not dissolve and therefore could not travel up the paper. So statement 2 is correct. The sweet S contains two spots: one that aligns with W and one that appears near the bottom. The spot aligning with W indicates S contains dye W, so statement 4 is correct. However, S does not contain dyes Y or Z, as there are no spots aligning with those positions—so statement 3 is incorrect. Statement 1 says S contains only two dyes, but the chromatogram may show additional components. Based on what we can see, statements 2 and 4 are correct, making option C the right choice.
Question 39
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
When sodium hydroxide is added to different cations, characteristic precipitates form. Both aluminium and zinc ions form white precipitates with sodium hydroxide, but these precipitates dissolve in excess sodium hydroxide (forming aluminate and zincate ions respectively). Calcium ions also form a white precipitate of calcium hydroxide, but crucially this precipitate is insoluble in excess sodium hydroxide. Chromium(III) ions form a green precipitate, not white. Since the question states the precipitate is white and insoluble in excess sodium hydroxide, the cation must be calcium ($\text{Ca}^{2+}$), making option B correct.
Question 40
Most-appropriate topic codes (Cambridge IGCSE Chemistry 0620):
▶️ Answer/Explanation
Detailed solution:
The key observation is that damp red litmus paper turns white. Ammonia turns damp red litmus paper blue, not white. Carbon dioxide has no effect on litmus paper (though it turns limewater milky). Oxygen has no effect on litmus paper. Chlorine gas, however, is a powerful bleaching agent—it first turns damp litmus paper red (because chlorine dissolves in water to form an acidic solution), and then bleaches it, turning it white. The bleaching action is due to the oxidising property of chlorine. Therefore, the gas that turns damp red litmus paper white is chlorine, making option C correct.
