Question 1
A sealed balloon contains air. The temperature of the balloon is increased.
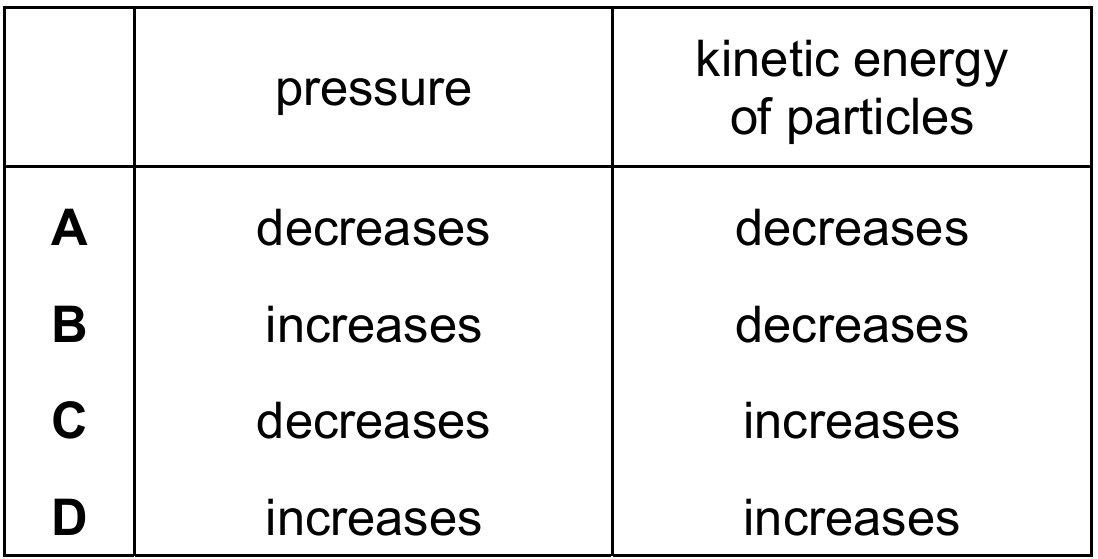
Which row describes how the pressure in the balloon and the kinetic energy of the particles in air change?
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: D
Explanation: According to the kinetic particle theory, increasing the temperature increases the average kinetic energy of the gas particles. The particles move faster and collide with the walls of the balloon more frequently and with greater force, thus increasing the pressure inside the balloon.
✅ Therefore, both pressure and kinetic energy increase.
Question 2
Ammonia gas is reacted with hydrogen chloride gas using the apparatus shown.
Solid ammonium chloride is produced.
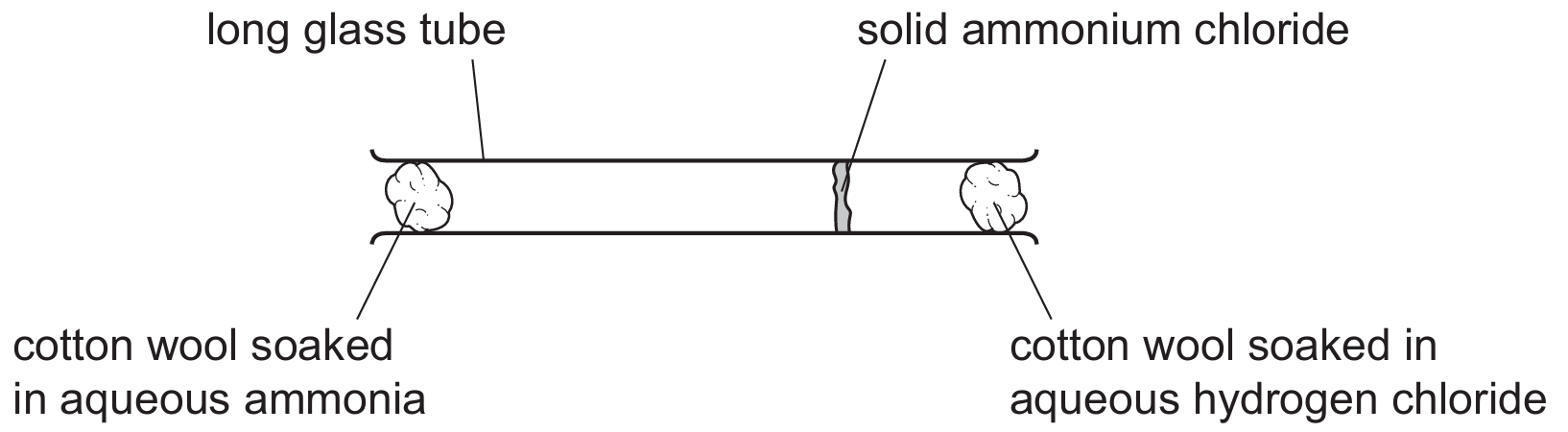
Which statement explains why the solid ammonium chloride is formed nearer to the hydrogen chloride?
A. Aqueous ammonia is a base and aqueous hydrogen chloride is an acid.
B. Ammonia molecules diffuse more slowly than hydrogen chloride molecules.
C. Hydrogen chloride has a greater molecular mass than ammonia.
D. Only ammonia molecules diffuse in all directions at the same time.
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: C
Explanation: The rate of diffusion is inversely proportional to molecular mass (Graham’s Law). \(M_r(\text{HCl}) = 1 + 35.5 = 36.5\) and \(M_r(\text{NH}_3) = 14 + 3 = 17\). Since HCl is heavier, it diffuses more slowly. The lighter NH₃ diffuses faster and travels further down the tube, so the ammonium chloride ring forms closer to the HCl source.
✅ Hydrogen chloride has a greater molecular mass than ammonia.
Question 3
The table shows information about four different particles.
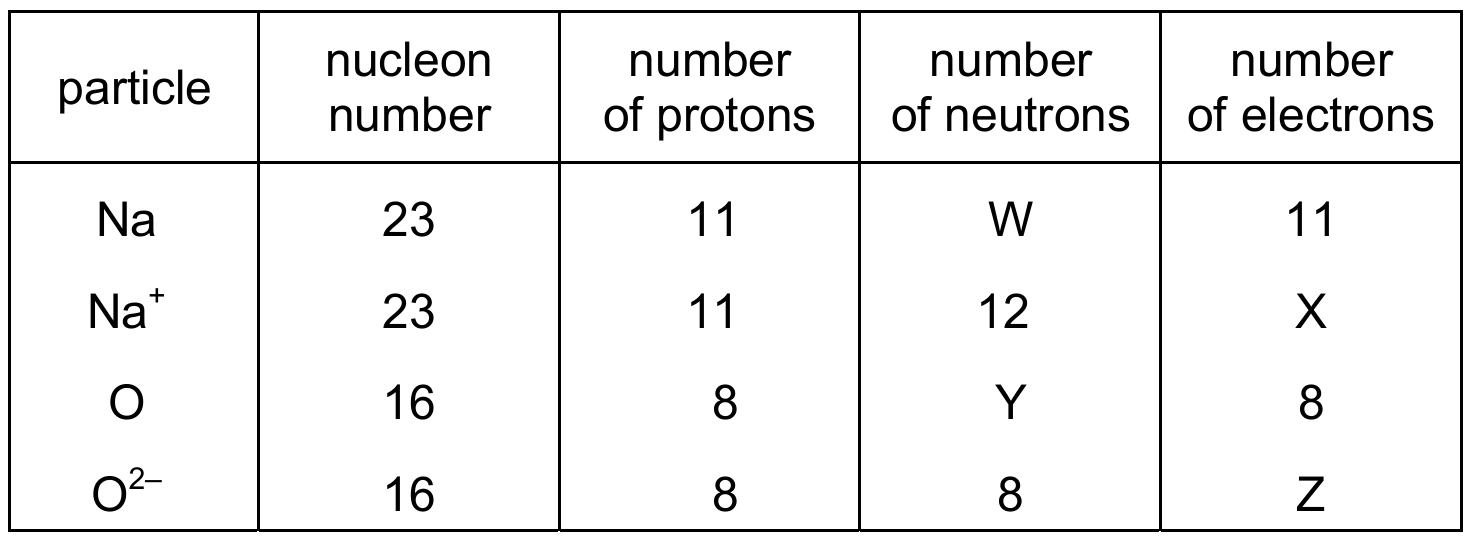
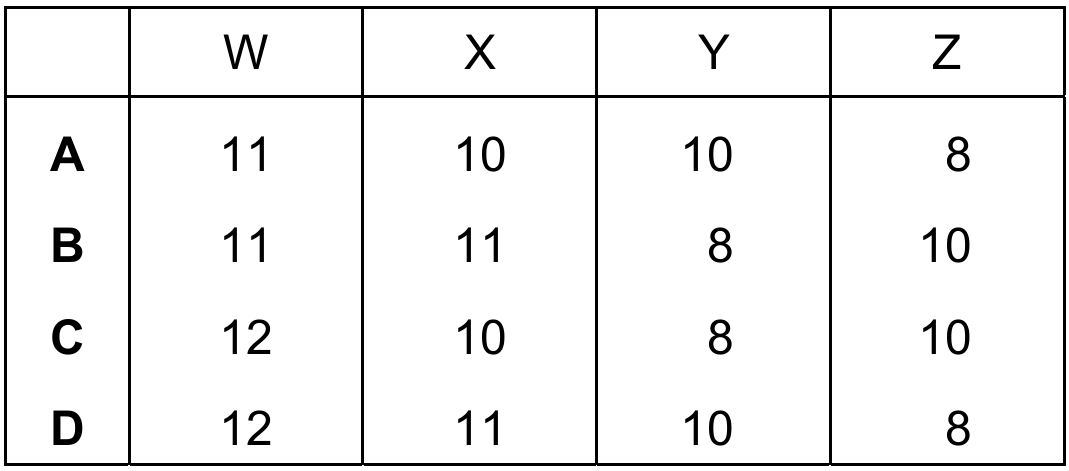
What are the values of W, X, Y and Z?
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: C (W=10, X=8, Y=8, Z=10)
Explanation:
– For \(\text{Na}^+\), electrons = number of protons – charge = \(11 – 1 = 10\) (W=10).
– For \(^{16}_{8}\text{O}\), neutrons = nucleon number – proton number = \(16 – 8 = 8\) (X=8).
– For \(\text{O}^{2-}\), neutrons = \(16 – 8 = 8\) (Y=8) and electrons = \(8 + 2 = 10\) (Z=10).
✅ Option C correctly gives W=10, X=8, Y=8, Z=10.
Question 4
Substances with giant covalent structures can be used as lubricants and as cutting tools for hard materials.
The diagrams show how the atoms are arranged in two giant covalent substances, P and Q.
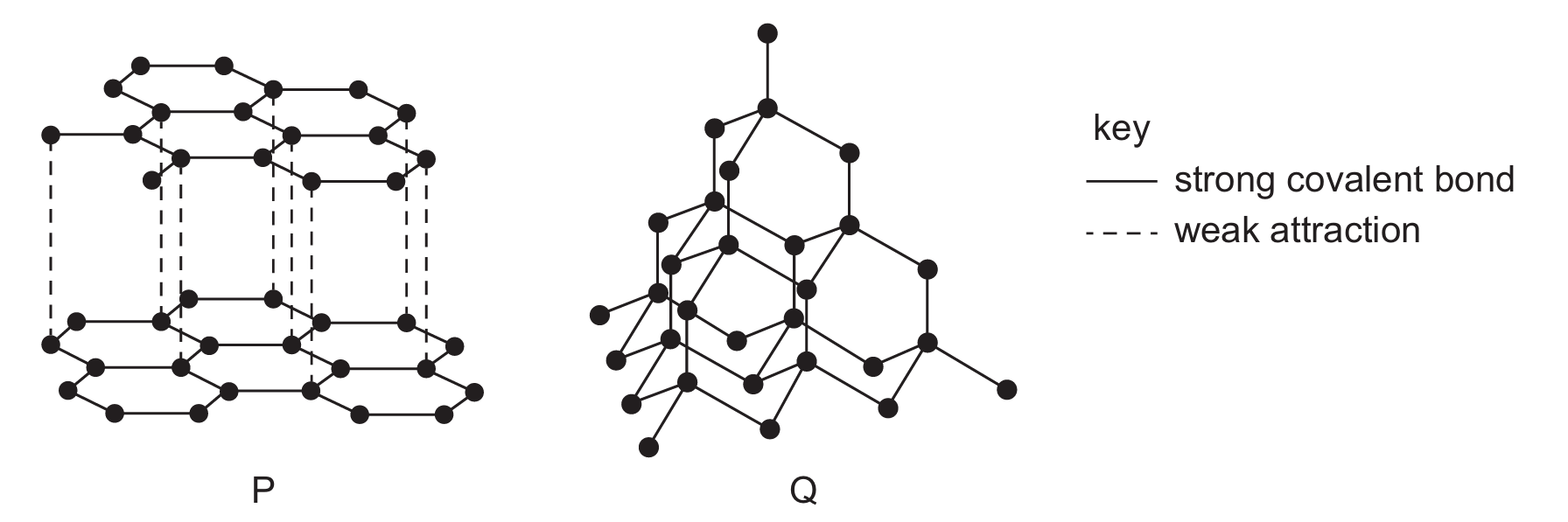
Which statement is correct?
A. Only P is used as a cutting tool and only Q is used as a lubricant.
B. Only P is used as a lubricant and only Q is used as a cutting tool.
C. P and Q are both used as cutting tools.
D. P and Q are both used as lubricants.
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: B
Explanation: Based on the syllabus (section 2.6), diamond (typically P, tetrahedral structure) is used in cutting tools due to its extreme hardness. Graphite (typically Q, layered structure) is used as a lubricant because its layers can slide over each other due to weak intermolecular forces.
✅ Therefore, only P is used as a lubricant and only Q is used as a cutting tool.
Question 5
Symbols representing four particles are shown.
\[^{40}\text{W} \quad ^{41}\text{X}^{2+} \quad ^{37}\text{Y} \quad ^{37}\text{Z}\]
The letters are not the chemical symbols.
Which particles have the same number of neutrons?
A. W and X²⁺
B. W and Z
C. X²⁺ and Y
D. Y and Z
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: D
Explanation: Number of neutrons = nucleon number – proton number. However, the proton numbers are not directly given. Since Y and Z have the same nucleon number (37) and the same symbol (Z represents the same element as Y? The question states the letters are not chemical symbols, so Y and Z are different particles with the same nucleon number but different proton numbers. For them to have the same number of neutrons, their proton numbers must also be the same, which means Y and Z are isotopes of the same element. Given the answer key, Y and Z have the same neutron number.
✅ Y and Z have the same number of neutrons.
Question 6
Which statement describes the formation of bonds in lithium bromide?
A. A lithium atom donates one electron to a bromine atom.
B. A bromine atom donates one electron to a lithium atom.
C. A lithium atom shares a pair of electrons with a bromine atom.
D. A bromine atom donates a pair of electrons to a lithium atom.
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: A
Explanation: Lithium is in Group I and has 1 electron in its outer shell. Bromine is in Group VII and has 7 electrons in its outer shell. Lithium donates its one electron to bromine, forming Li⁺ and Br⁻ ions. The electrostatic attraction between these oppositely charged ions forms the ionic bond.
✅ A lithium atom donates one electron to a bromine atom.
Question 7
Which statement explains why solid magnesium is a good conductor of electricity?
A. Magnesium has delocalised electrons that can move through the lattice.
B. Magnesium has strong attractions between ions.
C. Magnesium consists of a giant lattice of anions in a ‘sea’ of delocalised electrons.
D. Magnesium consists of metal ions that move through the lattice.
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: A
Explanation: In metallic bonding, the positive metal ions (cations) are arranged in a giant lattice surrounded by a ‘sea’ of delocalised electrons. These delocalised electrons are free to move throughout the structure, allowing them to carry an electric current.
✅ Magnesium has delocalised electrons that can move through the lattice.
Question 8
The equation for the reaction between dilute ethanoic acid and aqueous lithium hydroxide is shown.
\[\mathrm{CH_3COOH + LiOH \rightarrow CH_3COOLi + H_2O}\]
In an experiment, \(20.0\text{ cm}^3\) of \(0.050\text{ mol/dm}^3\) aqueous lithium hydroxide is exactly neutralised by \(12.5\text{ cm}^3\) of dilute ethanoic acid.
Which statement is correct?
A. The amount of lithium hydroxide is equal to \(0.050 \times \frac{1000}{20}\text{ mol}\).
B. The amount of ethanoic acid reacted is \(0.0010\text{ mol}\).
C. The concentration of the dilute ethanoic acid is \(0.16\text{ mol/dm}^3\).
D. The volume of aqueous lithium hydroxide used is \(0.20\text{ dm}^3\).
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: B
Calculation:
Moles of LiOH = concentration × volume (in dm³) = \(0.050 \times \frac{20.0}{1000} = 0.0010\text{ mol}\).
From the 1:1 reaction ratio, moles of CH₃COOH = moles of LiOH = \(0.0010\text{ mol}\).
Concentration of CH₃COOH = \(\frac{0.0010}{12.5/1000} = 0.080\text{ mol/dm}^3\).
✅ Option B is correct: the amount of ethanoic acid reacted is \(0.0010\text{ mol}\).
Question 9
The molecular formulae of four compounds are listed.
- \(\mathrm{CH_4}\)
- \(\mathrm{C_2H_4}\)
- \(\mathrm{C_2H_5OH}\)
- \(\mathrm{C_6H_{12}O_6}\)
Which molecular formulae are also the empirical formulae for the compounds?
A. 1 and 2
B. 1 and 3
C. 2 and 4
D. 3 and 4
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: B (1 and 3)
Explanation:
– \(\mathrm{CH_4}\): Ratio C:H = 1:4 (already simplest) → empirical formula is \(\mathrm{CH_4}\).
– \(\mathrm{C_2H_4}\): Ratio C:H = 2:4 simplifies to 1:2 → empirical formula is \(\mathrm{CH_2}\).
– \(\mathrm{C_2H_5OH}\): Formula is \(\mathrm{C_2H_6O}\). Ratio C:H:O = 2:6:1 (cannot simplify further) → empirical formula is \(\mathrm{C_2H_6O}\) (same as molecular).
– \(\mathrm{C_6H_{12}O_6}\): Ratio C:H:O = 6:12:6 simplifies to 1:2:1 → empirical formula is \(\mathrm{CH_2O}\).
✅ Compounds 1 and 3 have molecular formulae that are also empirical formulae.
Question 10
The equation for the reaction between copper(II) oxide, CuO, and ammonia is given.
\[3\mathrm{CuO} + 2\mathrm{NH_3} \rightarrow 3\mathrm{Cu} + 3\mathrm{H_2O} + \mathrm{N_2}\]
[M: CuO, 80; NH₃, 17; H₂O, 18; N₂, 28]
[A: Cu, 64]
Which statements about this reaction are correct?
- 80.0g of copper(II) oxide reacts exactly with 17.0g of ammonia.
- 4.0g of copper(II) oxide reacts with excess ammonia to produce 3.2g of copper.
- The mass of water produced in this reaction is greater than the mass of nitrogen.
A. 1, 2 and 3
B. 1 and 2 only
C. 1 and 3 only
D. 2 and 3 only
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: D (2 and 3 only)
Calculations:
Statement 1: 3 mol CuO (3×80=240g) reacts with 2 mol NH₃ (2×17=34g). So 80g CuO reacts with (80/240)×34 = 11.33g NH₃, not 17g. ❌
Statement 2: 3 mol CuO (240g) produces 3 mol Cu (3×64=192g). So 4g CuO produces (4/240)×192 = 3.2g Cu. ✅
Statement 3: 3 mol H₂O (3×18=54g) and 1 mol N₂ (28g). 54g > 28g, so mass of H₂O > mass of N₂. ✅
✅ Statements 2 and 3 are correct.
Question 11
How many atoms are present in 1.00 mol of argon?
A. \(2.06 \times 10^{23}\)
B. \(3.02 \times 10^{26}\)
C. \(6.02 \times 10^{23}\)
D. \(6.32 \times 10^{20}\)
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: C
Explanation: The Avogadro constant (\(N_A = 6.02 \times 10^{23}\)) defines the number of particles (atoms, molecules, or ions) in one mole of any substance. Argon is a monatomic gas, so 1.00 mol of argon contains \(6.02 \times 10^{23}\) atoms.
✅ \(6.02 \times 10^{23}\) atoms.
Question 12
Aqueous copper(II) sulfate is electrolysed using copper electrodes.
Which statement is correct?
A. Oxygen gas is produced at the positive electrode.
B. The blue colour of the solution gradually fades.
C. The concentration of copper ions in the solution stays the same.
D. The mass of the negative electrode decreases.
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: C
Explanation: With copper electrodes, the anode (positive) dissolves: \(\mathrm{Cu \rightarrow Cu^{2+} + 2e^-}\), and copper is deposited on the cathode (negative): \(\mathrm{Cu^{2+} + 2e^- \rightarrow Cu}\). The concentration of Cu²⁺ ions in solution remains constant because copper lost from the anode is replaced by deposition at the cathode. The blue colour (due to Cu²⁺) does not fade.
✅ The concentration of copper ions in the solution stays the same.
Question 13
Which row describes the reactions during the electrolysis of dilute aqueous sodium chloride?
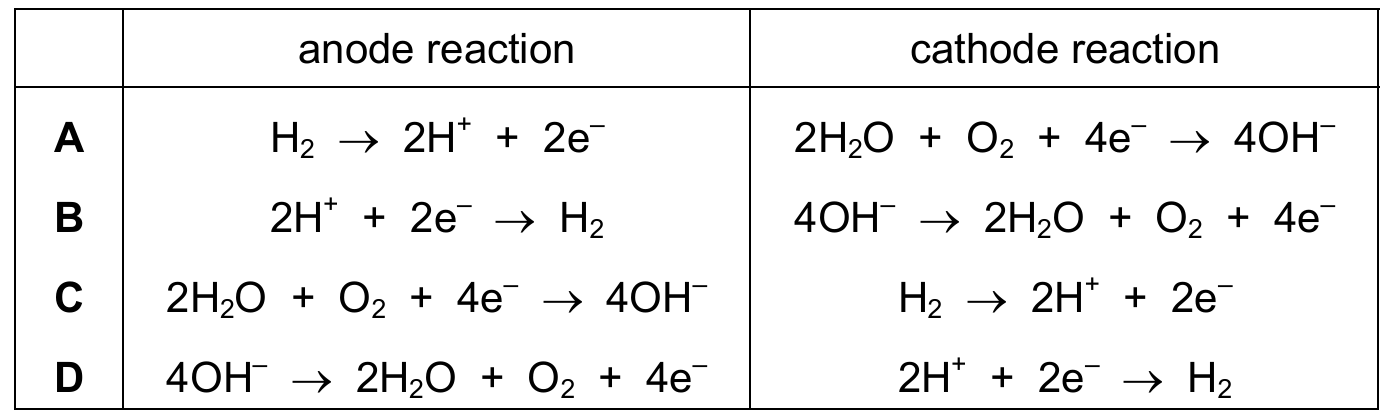
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: D
Explanation: In dilute aqueous sodium chloride, the ions present are Na⁺, Cl⁻, H⁺, and OH⁻. At the anode (positive), OH⁻ ions are preferentially oxidised: \(4\mathrm{OH^-} \rightarrow 2\mathrm{H_2O} + \mathrm{O_2} + 4e^-\). At the cathode (negative), H⁺ ions are preferentially reduced: \(2\mathrm{H^+} + 2e^- \rightarrow \mathrm{H_2}\).
✅ Row D correctly shows the anode reaction (oxidation of OH⁻) and cathode reaction (reduction of H⁺).
Question 14
Information about two reactions is given.
- The neutralisation reaction between citric acid and sodium hydrogencarbonate is endothermic.
- The displacement reaction between magnesium and carbon dioxide is exothermic.
Which statements about the two reactions are correct?
- The energy of the products formed in the neutralisation reaction is greater than the energy of the reactants.
- The energy of magnesium and carbon dioxide is greater than the energy of magnesium oxide and carbon.
- In an exothermic reaction, the energy required to break the bonds is greater than the energy released when the new bonds are formed.
A. 1, 2 and 3
B. 1 and 2 only
C. 1 and 3 only
D. 2 and 3 only
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: B (1 and 2 only)
Explanation:
Statement 1: In an endothermic reaction, energy is absorbed, so products have higher energy than reactants. ✅
Statement 2: In an exothermic reaction, energy is released, so reactants have higher energy than products. Mg + CO₂ are reactants, MgO + C are products. ✅
Statement 3: In an exothermic reaction, energy released when new bonds form is greater than energy required to break old bonds. ❌
✅ Statements 1 and 2 are correct.
Question 15
The reaction pathway diagram for a reaction is shown.
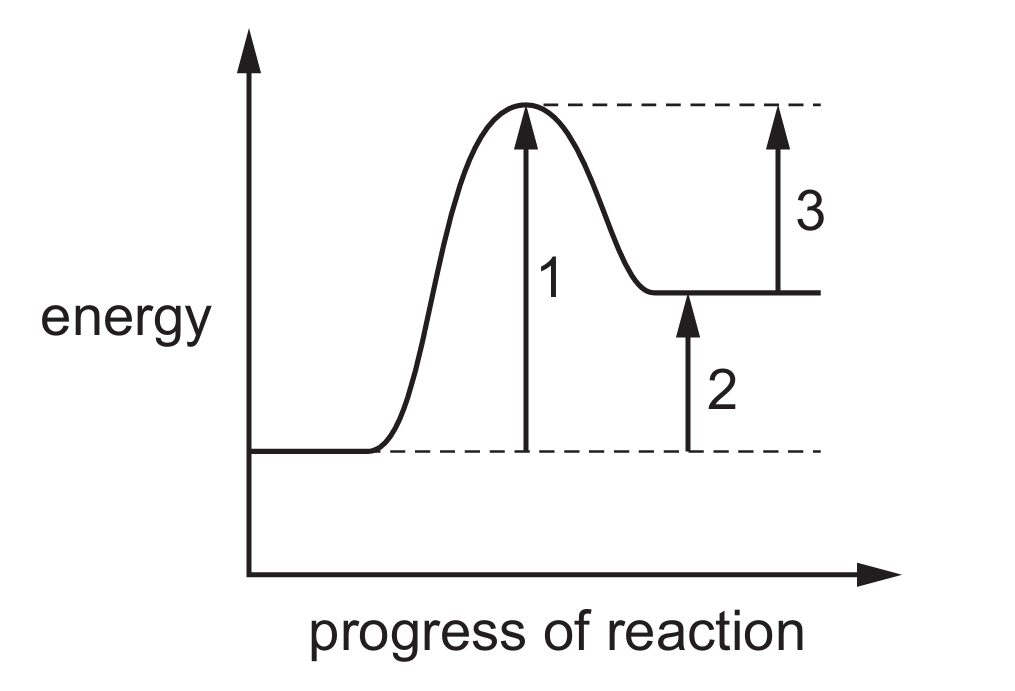
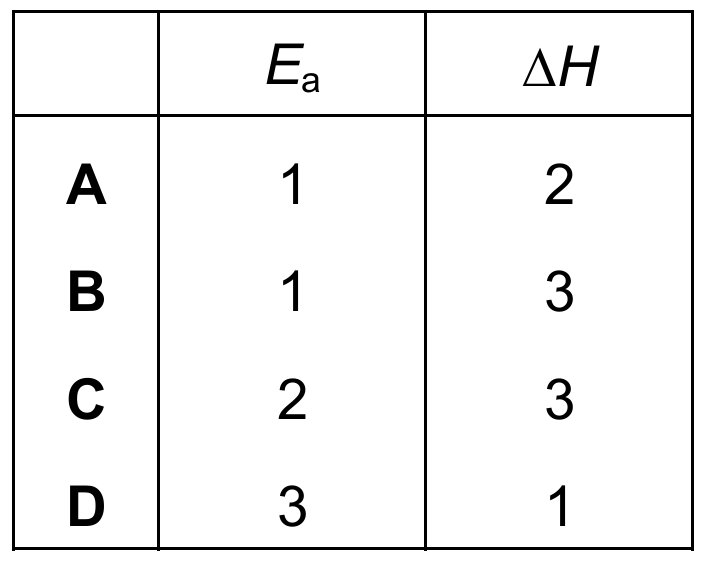
Which row identifies the activation energy, \(E_a\), and the enthalpy change, \(\Delta H\), for this reaction?
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: A
Explanation: Activation energy (\(E_a\)) is the energy difference between the reactants and the transition state (peak). In the diagram, arrow 1 shows this difference. Enthalpy change (\(\Delta H\)) is the energy difference between the reactants and products. Since the products are at a lower energy level than the reactants, the reaction is exothermic, and arrow 2 shows this difference. (Arrow 3 represents the energy difference between the transition state and products, which is not \(\Delta H\).)
✅ \(E_a = 1\), \(\Delta H = 2\).
Question 16
Hydrogen peroxide solution decomposes very slowly at room temperature to produce oxygen gas. This gas forms a rising foam when liquid detergent is added.
Five test-tubes are half-filled with hydrogen peroxide solution. A drop of liquid detergent is added to each one.
Different metal oxides are added to four of the test-tubes and the height of the foam formed after one minute is measured. The results are shown.
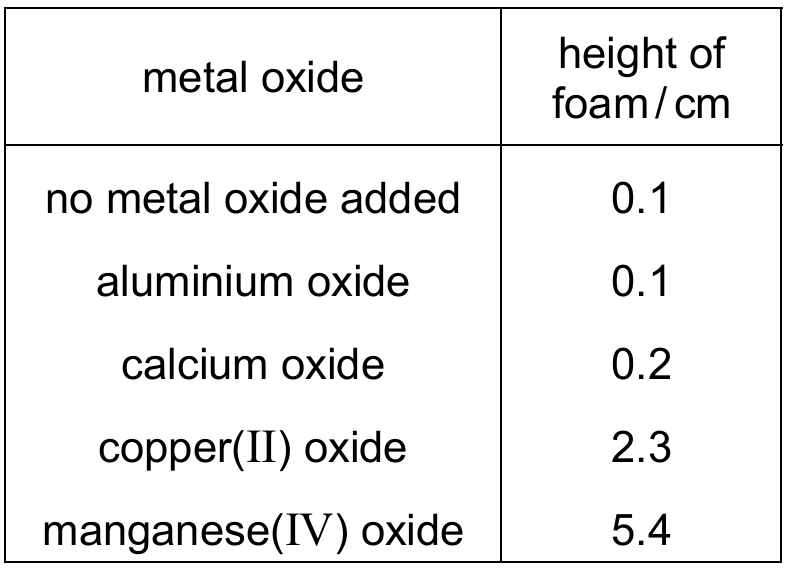
Which conclusion can be drawn from these results?
A. Metal oxides do not affect the rate of this reaction.
B. All metal oxides increase the rate of this reaction and act as catalysts.
C. Manganese(IV) oxide is the best catalyst of the four metal oxides tested.
D. Only transition element oxides increase the rate of this reaction.
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: C
Explanation: The height of foam is proportional to the rate of oxygen production. Manganese(IV) oxide produced the greatest foam height (5.4 cm), indicating it is the most effective catalyst among those tested. Aluminium oxide and calcium oxide (not transition elements) produced very little foam, similar to the control. Therefore, not all metal oxides increase the rate.
✅ Manganese(IV) oxide is the best catalyst of the four metal oxides tested.
Question 17
Which equation represents one of the reactions in the Contact process?
A. \(2\mathrm{SO_2} + \mathrm{O_2} \rightleftharpoons 2\mathrm{SO_3}\)
B. \(2\mathrm{SO_3} \rightarrow 2\mathrm{SO_2} + \mathrm{O_2}\)
C. \(\mathrm{S} + \mathrm{O_2} \rightarrow \mathrm{SO_2}\)
D. \(\mathrm{SO_2} + \mathrm{H_2O} \rightarrow \mathrm{H_2SO_3}\)
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: A
Explanation: The Contact process is used to manufacture sulfuric acid. The key reversible reaction is the oxidation of sulfur dioxide to sulfur trioxide: \(2\mathrm{SO_2(g)} + \mathrm{O_2(g)} \rightleftharpoons 2\mathrm{SO_3(g)}\). This reaction takes place at 450°C, 200 kPa, using a vanadium(V) oxide catalyst.
✅ Option A is the correct equation for one of the reactions in the Contact process.
Question 18
The ionic equation for the redox reaction between acidified manganate(VII) ions and iodide ions is shown.
\[2\mathrm{MnO_4^- (aq)} + 16\mathrm{H^+ (aq)} + 10\mathrm{I^- (aq)} \rightarrow 2\mathrm{Mn^{2+} (aq)} + 5\mathrm{I_2 (aq)} + 8\mathrm{H_2O (l)}\]
Which row shows the changes in oxidation number for manganese and for iodine during this reaction?
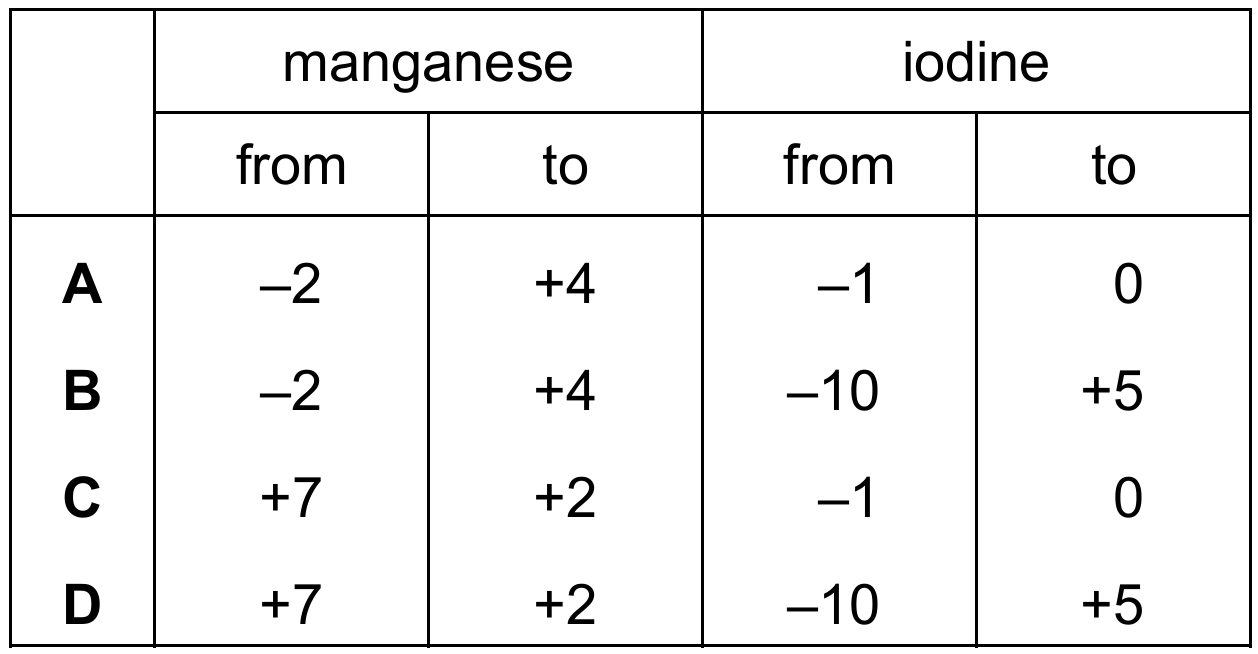
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: C
Explanation:
– In \(\mathrm{MnO_4^-}\), the oxidation number of Mn is +7 (O is -2, so Mn + (-2×4) = -1 → Mn = +7). In \(\mathrm{Mn^{2+}}\), the oxidation number is +2. So Mn goes from +7 to +2.
– In \(\mathrm{I^-}\), the oxidation number is -1. In \(\mathrm{I_2}\), the oxidation number is 0. So I goes from -1 to 0.
✅ Manganese: +7 → +2; Iodine: -1 → 0.
Question 19
In the Haber process, nitrogen and hydrogen react to form ammonia.
\[\mathrm{N_2 + 3H_2 \rightleftharpoons 2NH_3}\]
The forward reaction is exothermic. Which changes in pressure and temperature increase the yield of ammonia?
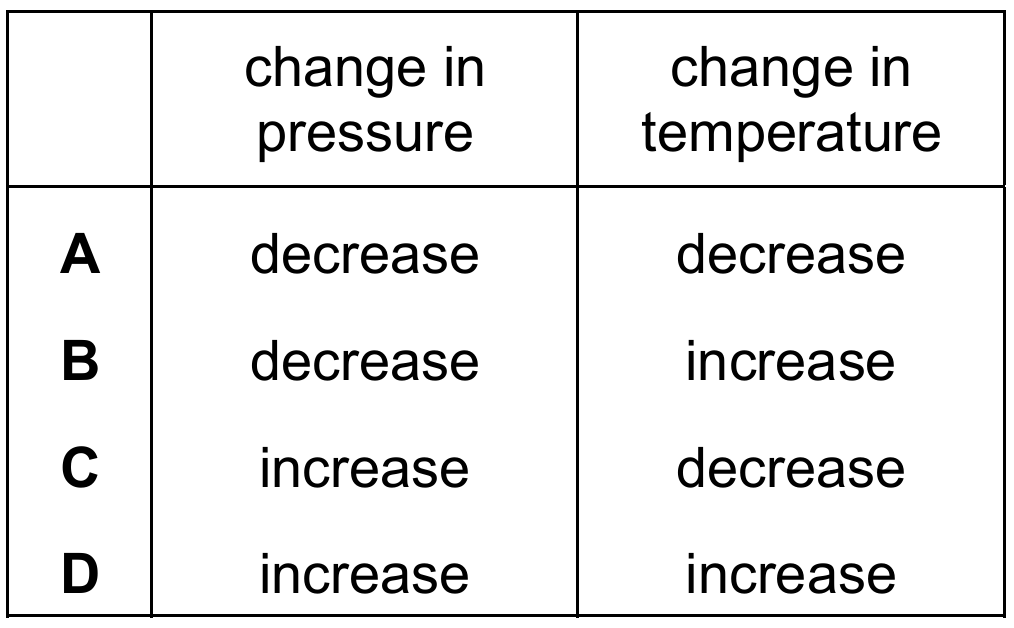
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: C
Explanation: Le Chatelier’s principle: For the exothermic forward reaction, decreasing temperature favours the forward reaction (increases yield). For the reaction with fewer moles of gas on the product side (4 moles → 2 moles), increasing pressure favours the forward reaction (increases yield).
✅ Increase pressure and decrease temperature increase the yield of ammonia.
Question 20
Which statement describes a property of ethanoic acid?
A. It does not produce carbon dioxide when added to sodium carbonate.
B. It does not produce hydrogen when added to magnesium.
C. It only donates some of its protons when added to excess aqueous sodium hydroxide.
D. It only partially dissociates when added to water.
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: D
Explanation: Ethanoic acid (CH₃COOH) is a weak acid. In aqueous solution, it only partially dissociates into H⁺ and CH₃COO⁻ ions, establishing an equilibrium: \(\mathrm{CH_3COOH(aq) \rightleftharpoons H^+(aq) + CH_3COO^-(aq)}\). Unlike strong acids, most of the ethanoic acid molecules remain undissociated.
✅ It only partially dissociates when added to water.
Question 21
A student adds different solid salts to an excess of water. The student stirs the mixtures to determine the solubilities of the salts.
The results are shown.
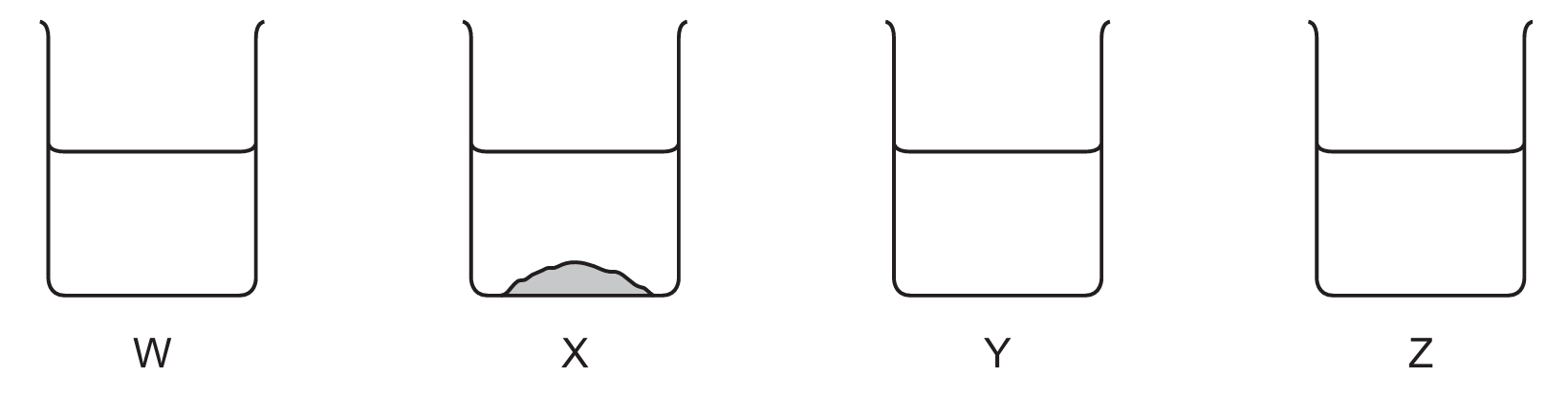
What could the four different salts be?

Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: B
Explanation: Solubility rules:
– Ammonium nitrate: soluble (all ammonium salts are soluble).
– Sodium carbonate: soluble (all sodium salts are soluble).
– Calcium sulfate: slightly soluble/insoluble (sulfates except Ba, Ca, Pb are soluble; calcium sulfate is sparingly soluble).
– Lead(II) chloride: insoluble (chlorides are soluble except lead and silver).
✅ Option B correctly matches the solubility observations from the diagram.
Question 22
Four metal oxides are listed.
- \(\mathrm{Al_2O_3}\)
- \(\mathrm{CaO}\)
- \(\mathrm{CuO}\)
- \(\mathrm{ZnO}\)
Which metal oxides are amphoteric?
A. 1 and 4
B. 1 only
C. 2, 3 and 4
D. 2 and 3 only
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: A (1 and 4)
Explanation: Amphoteric oxides react with both acids and bases to form salts and water. According to the syllabus (section 7.2), \(\mathrm{Al_2O_3}\) (aluminium oxide) and \(\mathrm{ZnO}\) (zinc oxide) are amphoteric. CaO (calcium oxide) is basic, and CuO (copper(II) oxide) is basic.
✅ \(\mathrm{Al_2O_3}\) and \(\mathrm{ZnO}\) are amphoteric.
Question 23
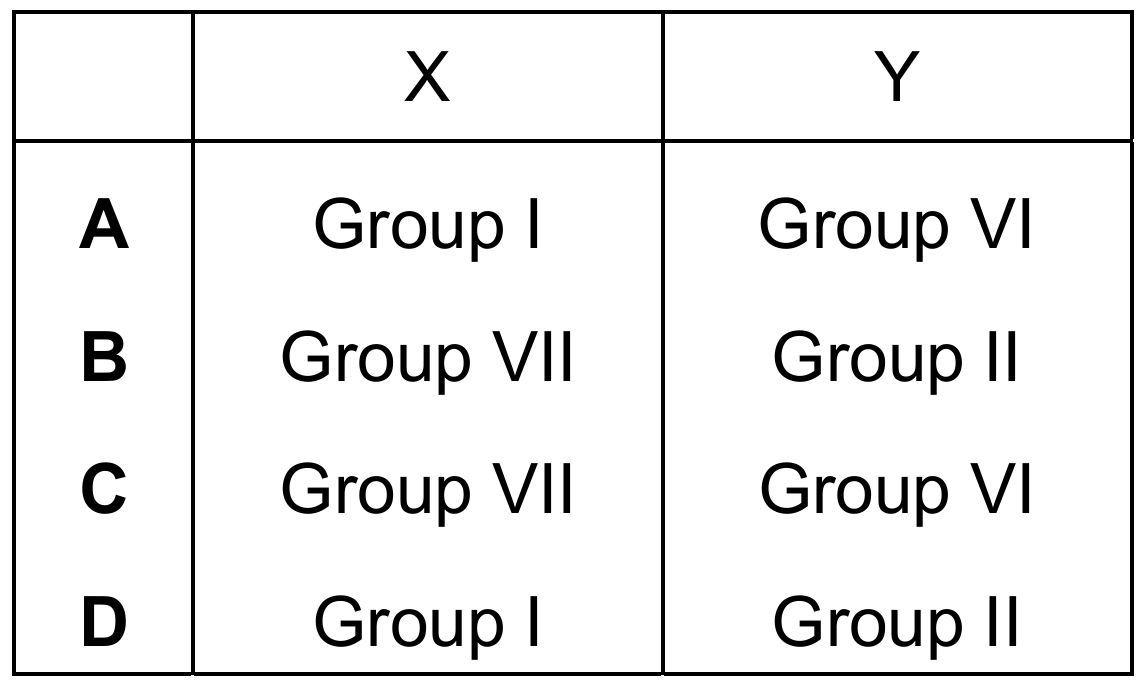
The ions formed when element X and element Y react together are shown.
- Element X forms \(X^-\) ions.
- Element Y forms \(Y^{2+}\) ions.
Which row identifies the position of X and of Y in the Periodic Table?
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: B
Explanation: Elements in Group VII (halogens) gain one electron to form \(X^-\) ions (e.g., Cl⁻). Elements in Group II (alkaline earth metals) lose two electrons to form \(Y^{2+}\) ions (e.g., Mg²⁺, Ca²⁺).
✅ X is in Group VII, Y is in Group II.
Question 24
Which properties decrease down the group of alkali metals?
- melting point
- density
- reactivity
A. 1 and 2
B. 1 only
C. 2 and 3
D. 3 only
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: B (1 only)
Explanation: Down Group I (alkali metals: Li, Na, K, Rb, Cs):
– Melting point: decreases (property 1 ✅ decreases).
– Density: generally increases (property 2 ❌ does not decrease).
– Reactivity: increases (property 3 ❌ does not decrease).
✅ Only melting point decreases down the group.
Question 25
Some halide ions are displaced from aqueous potassium halides by halogens.
In which mixtures are the halide ions displaced?
- potassium bromide and chlorine
- potassium bromide and iodine
- potassium iodide and chlorine
- potassium chloride and iodine
A. 1 and 3
B. 1 and 4
C. 2 and 3
D. 2 and 4
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: A (1 and 3)
Explanation: A more reactive halogen displaces a less reactive halide from solution. Reactivity decreases down Group VII: Cl > Br > I.
1. KBr + Cl₂: Cl displaces Br (Cl more reactive) → displacement occurs. ✅
2. KBr + I₂: I is less reactive than Br → no displacement. ❌
3. KI + Cl₂: Cl displaces I (Cl more reactive) → displacement occurs. ✅
4. KCl + I₂: I is less reactive than Cl → no displacement. ❌
✅ Displacement occurs in mixtures 1 and 3.
Question 26
What is the correct order of reactivity of the following metals, from least reactive to most reactive?
A. calcium → magnesium → zinc → copper
B. copper → zinc → magnesium → calcium
C. copper → zinc → calcium → magnesium
D. zinc → copper → magnesium → calcium
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: B
Explanation: The reactivity series (from most to least reactive): K, Na, Ca, Mg, Al, C, Zn, Fe, H, Cu, Ag, Au. Therefore, from least reactive to most reactive: Cu (least) → Zn → Mg → Ca (most).
✅ Copper → zinc → magnesium → calcium.
Question 27
The equation for the reaction between aluminium and iron(III) oxide is shown.
\[2\mathrm{Al} + \mathrm{Fe_2O_3} \rightarrow \mathrm{Al_2O_3} + 2\mathrm{Fe}\]
Which statement about this reaction is correct?
A. The aluminium acts as an oxidising agent.
B. It shows that iron is more reactive than aluminium.
C. The oxidation number of the aluminium decreases during the reaction.
D. It shows that aluminium has a greater tendency to form ions than iron.
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: D
Explanation: In this thermite reaction, aluminium displaces iron from iron(III) oxide because aluminium is more reactive than iron. Aluminium loses electrons (oxidation: Al → Al³⁺ + 3e⁻) and acts as a reducing agent. The displacement shows that aluminium has a greater tendency to form positive ions than iron.
✅ It shows that aluminium has a greater tendency to form ions than iron.
Question 28
Which row identifies methods used to prevent iron from rusting?

Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: C
Explanation: Rusting of iron requires both oxygen and water. Barrier methods prevent rusting by excluding oxygen or water. Greasing and painting are effective barrier methods. Washing with distilled water does not prevent rusting; it actually removes any protective layers and exposes the iron to water, promoting rusting.
✅ Greasing ✓, Painting ✓, Washing with distilled water ×.
Question 29
Which statement about the extraction of aluminium by electrolysis is correct?
A. Aluminium oxide is dissolved in molten cryolite to increase its melting point.
B. At the cathode, oxygen ions lose electrons to form oxygen gas.
C. The carbon in the graphite anode reacts with oxygen gas forming carbon dioxide.
D. The reaction at the anode is \(\mathrm{Al^{3+} + 3e^- \rightarrow Al}\).
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: C
Explanation: In the Hall-Héroult process, aluminium oxide (Al₂O₃) is dissolved in molten cryolite (Na₃AlF₆) to lower the melting point, not increase it. At the cathode: \(\mathrm{Al^{3+} + 3e^- \rightarrow Al}\). At the anode: oxygen ions are oxidised to oxygen gas, which reacts with the graphite (carbon) anode to form carbon dioxide: \(\mathrm{C + O_2 \rightarrow CO_2}\). This causes the anodes to be consumed and need regular replacement.
✅ The carbon in the graphite anode reacts with oxygen gas forming carbon dioxide.
Question 30
Catalytic converters are used to remove oxides of nitrogen from exhaust gases. The equation represents one of the reactions that occur in a catalytic converter.
\[2\mathrm{NO} + 2\mathrm{CO} \rightarrow \mathrm{N_2} + 2\mathrm{CO_2}\]
What is a disadvantage of using a catalytic converter to remove oxides of nitrogen?
A. CO is an essential reactant in photosynthesis.
B. \(\mathrm{CO_2}\) is a greenhouse gas.
C. \(\mathrm{N_2}\) is acidic.
D. \(\mathrm{N_2}\) is toxic.
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: B
Explanation: Catalytic converters convert toxic NO and CO into N₂ and CO₂. While this removes harmful gases, CO₂ is produced, which is a greenhouse gas contributing to global warming and climate change. N₂ is neither acidic nor toxic (it is the main component of air). CO is not essential for photosynthesis; plants require CO₂.
✅ CO₂ is a greenhouse gas, which is a disadvantage.
Question 31
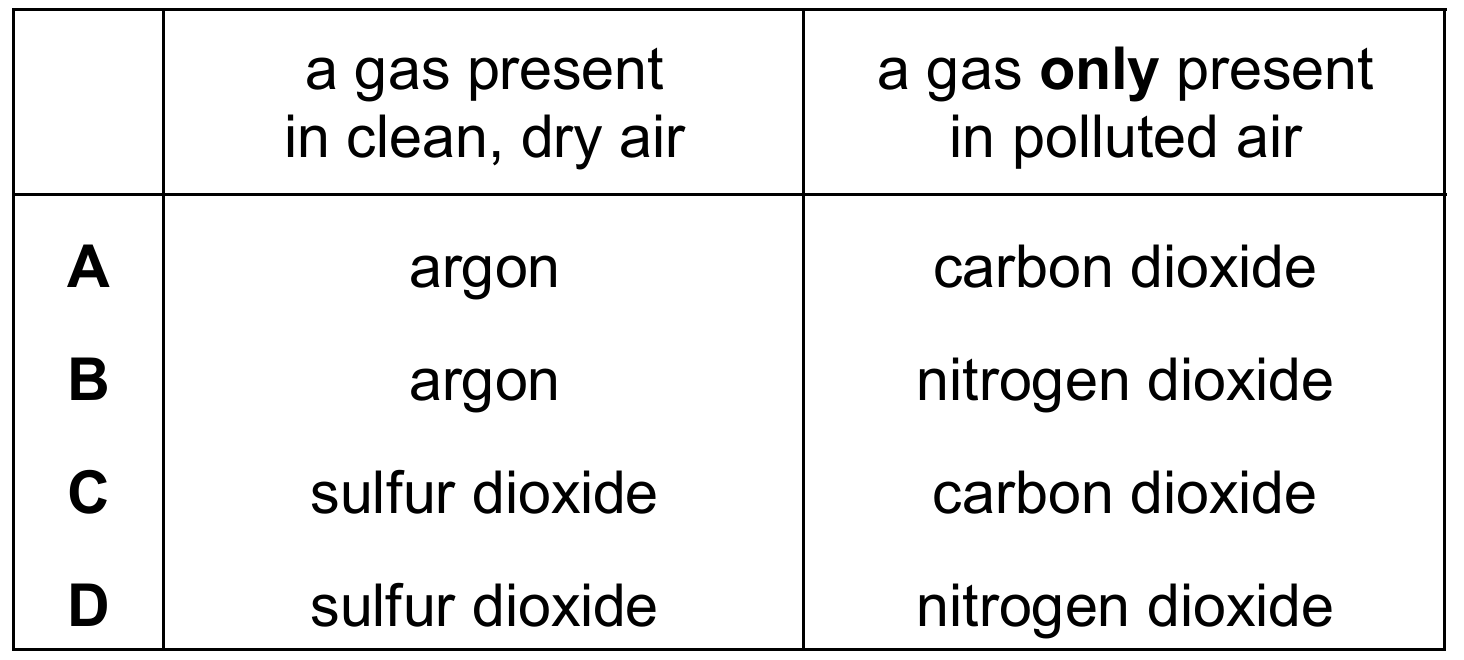
Some gases are present in clean, dry air, while other gases are only present in polluted air.
Which row is correct?
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: B
Explanation: Clean, dry air is approximately 78% nitrogen, 21% oxygen, and the remainder includes noble gases (mainly argon) and carbon dioxide. Argon (a noble gas) is present in clean air. Nitrogen dioxide (NO₂) is an air pollutant produced from car engines and is not present naturally in clean, dry air.
✅ Argon (present in clean air); Nitrogen dioxide (only in polluted air).
Question 32
Petroleum is separated by fractional distillation. Which statement about the fractions produced is correct?
A. Bottled gas for heating and cooking is obtained from the naphtha fraction.
B. Diesel oil is used as a fuel for jet aircraft.
C. Substances used to make polishes are obtained from the lubricating fraction.
D. The kerosene fraction contains many useful waxes.
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: C
Explanation: According to the syllabus (section 11.3):
– Bottled gas is from the refinery gas fraction, not naphtha. ❌
– Jet fuel is from the kerosene/paraffin fraction, not diesel oil. ❌
– The lubricating oil fraction is used for lubricants, waxes, and polishes. ✅
– Kerosene fraction is for jet fuel; waxes come from the lubricating fraction. ❌
✅ Substances used to make polishes are obtained from the lubricating fraction.
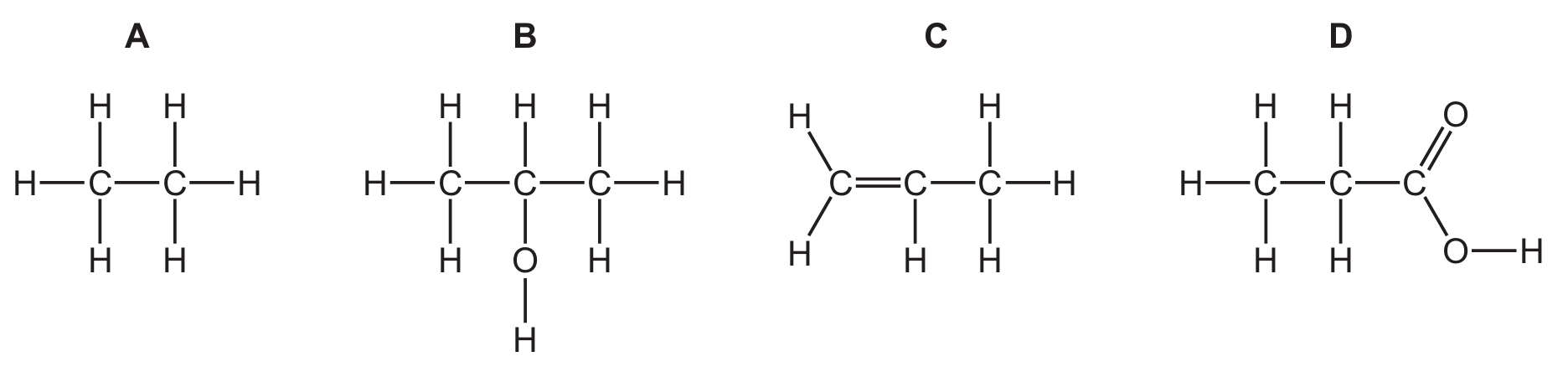
Question 33
Which structure represents a compound in the alcohol homologous series?
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: A
Explanation: Alcohols are a homologous series with the functional group -OH (hydroxyl group). The general formula for alcohols is \(\mathrm{C_nH_{2n+1}OH}\) or \(\mathrm{C_nH_{2n+2}O}\). Option A, \(\mathrm{CH_3CH_2CH_2OH}\) (propan-1-ol), contains the -OH group and is an alcohol. Option B is a carboxylic acid, option C is an ester, and option D is an alkane.
✅ \(\mathrm{CH_3CH_2CH_2OH}\) represents an alcohol.
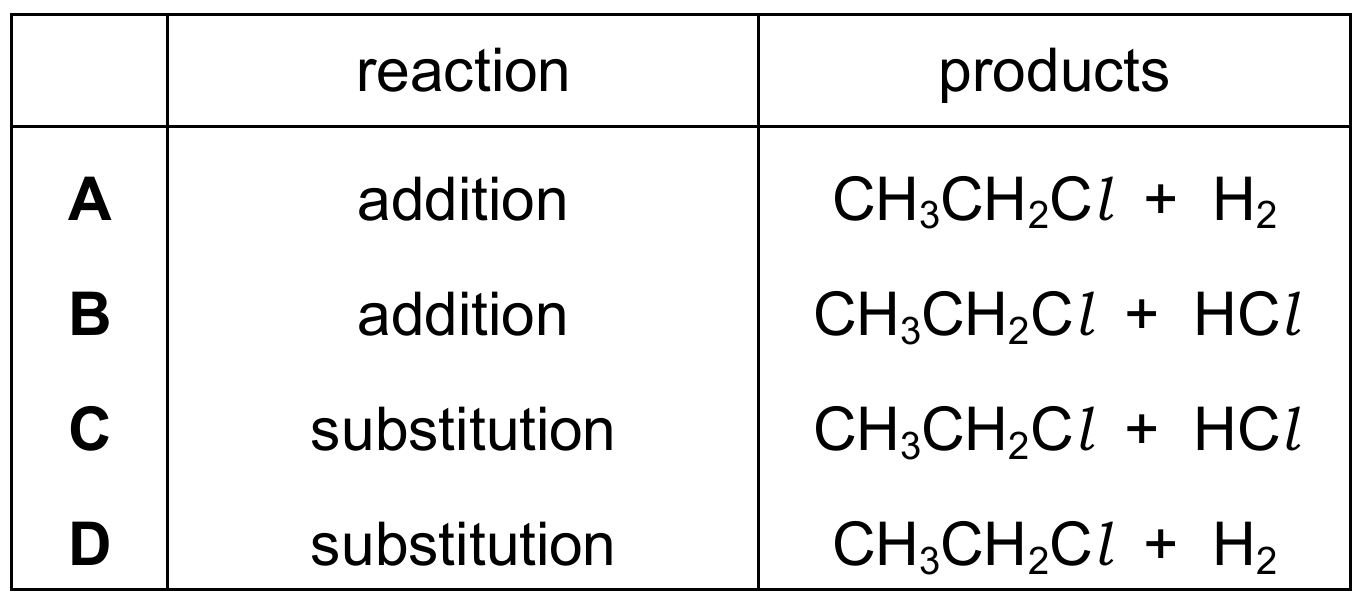
Question 34
Ethane reacts with chlorine. Which row identifies the type of reaction and the formulae of the products formed?
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: C
Explanation: Alkanes (saturated hydrocarbons) undergo substitution reactions with halogens in the presence of ultraviolet light. One hydrogen atom in ethane is replaced by a chlorine atom, forming chloroethane (CH₃CH₂Cl) and hydrogen chloride (HCl). This is a photochemical substitution reaction.
✅ Substitution reaction producing CH₃CH₂Cl + HCl.
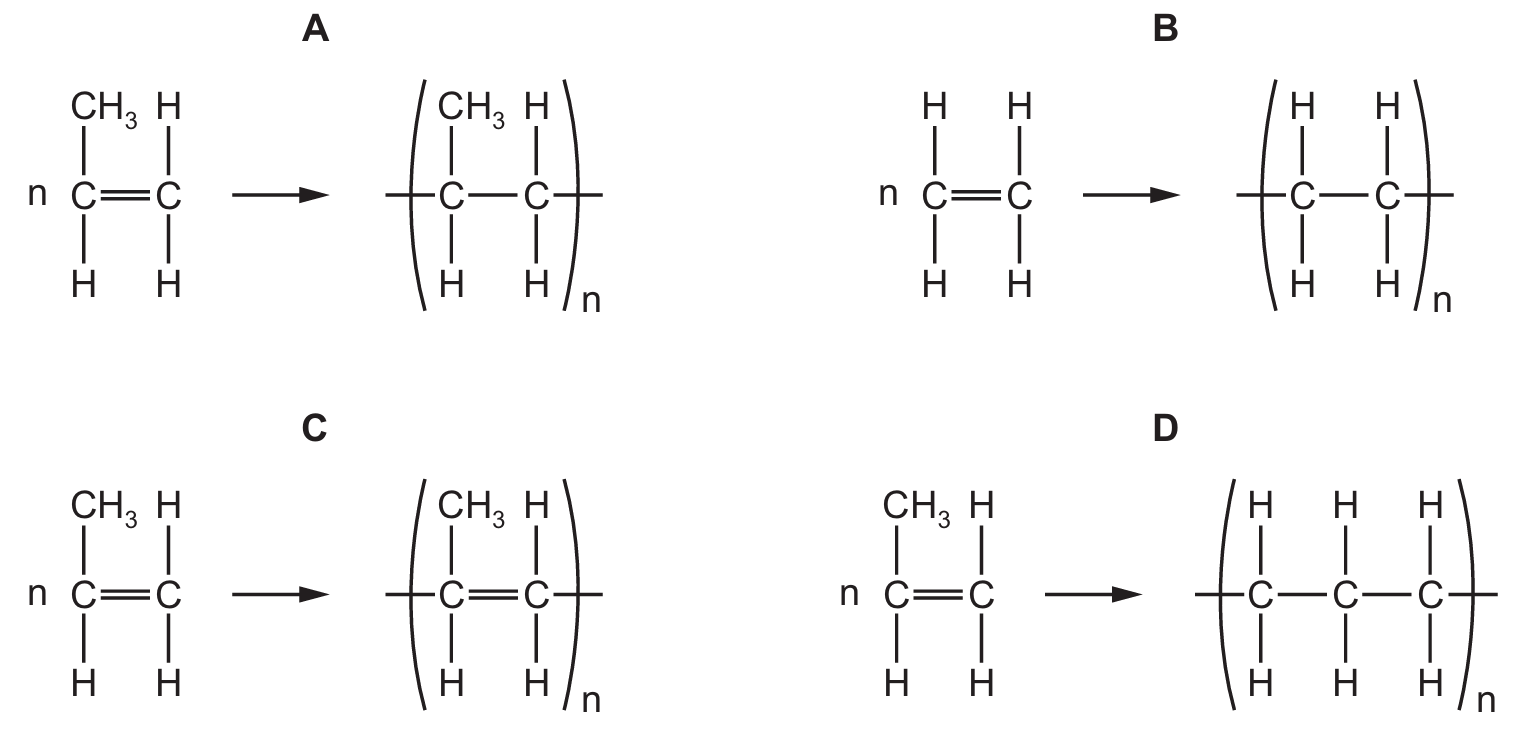
Question 35
Which equation represents the formation of poly(propene) from propene?
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: A
Explanation: Propene (CH₃CH=CH₂) is an alkene with a carbon-carbon double bond. It undergoes addition polymerisation to form poly(propene). The double bond opens up, allowing the monomer units to join together to form a long chain polymer with the repeating unit \(\mathrm{-[CH_2CH(CH_3)]-}\).
✅ \(n\mathrm{CH_3CH=CH_2} \rightarrow \mathrm{[CH_2CH(CH_3)]_n}\) is correct.
Question 36
Ethene reacts with hydrogen in an addition reaction to form ethane.
Which catalyst is used for this reaction?
A. iron
B. nickel
C. vanadium(V) oxide
D. yeast
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: B
Explanation: The addition reaction of ethene with hydrogen to form ethane (hydrogenation) requires a nickel catalyst at a temperature of about 150°C. Iron is used in the Haber process, vanadium(V) oxide in the Contact process, and yeast in fermentation.
✅ Nickel is the catalyst for this reaction.
Question 37
What is produced by the bacterial oxidation of ethanol?
A. ethene
B. sodium ethanoate
C. ethyl ethanoate
D. vinegar
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: D
Explanation: When ethanol (in alcoholic drinks or cider) is exposed to air in the presence of certain bacteria (Acetobacter), it is oxidised to ethanoic acid. The dilute solution of ethanoic acid produced is known as vinegar. This is the bacterial oxidation process: \(\mathrm{CH_3CH_2OH + O_2 \rightarrow CH_3COOH + H_2O}\).
✅ Vinegar (ethanoic acid) is produced.
Question 38
Which statement about addition polymers or condensation polymers is correct?
A. Addition polymers only contain carbon and hydrogen atoms.
B. One of the monomers used to make a polyester is a diamine.
C. Addition polymers contain carbon-carbon double bonds in the main chain.
D. Water is an additional product in the formation of a protein from amino acids.
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: D
Explanation: Proteins are natural polyamides formed by condensation polymerisation of amino acid monomers. In condensation polymerisation, a small molecule (usually water) is eliminated as the polymer forms. When two amino acids join, a water molecule is released, forming a peptide bond. Addition polymers (like poly(ethene)) contain only single C-C bonds in the main chain and can contain atoms other than C and H (e.g., PVC contains Cl). Polyesters are made from a dicarboxylic acid and a diol, not a diamine (polyamides use diamines).
✅ Water is an additional product in the formation of a protein from amino acids.
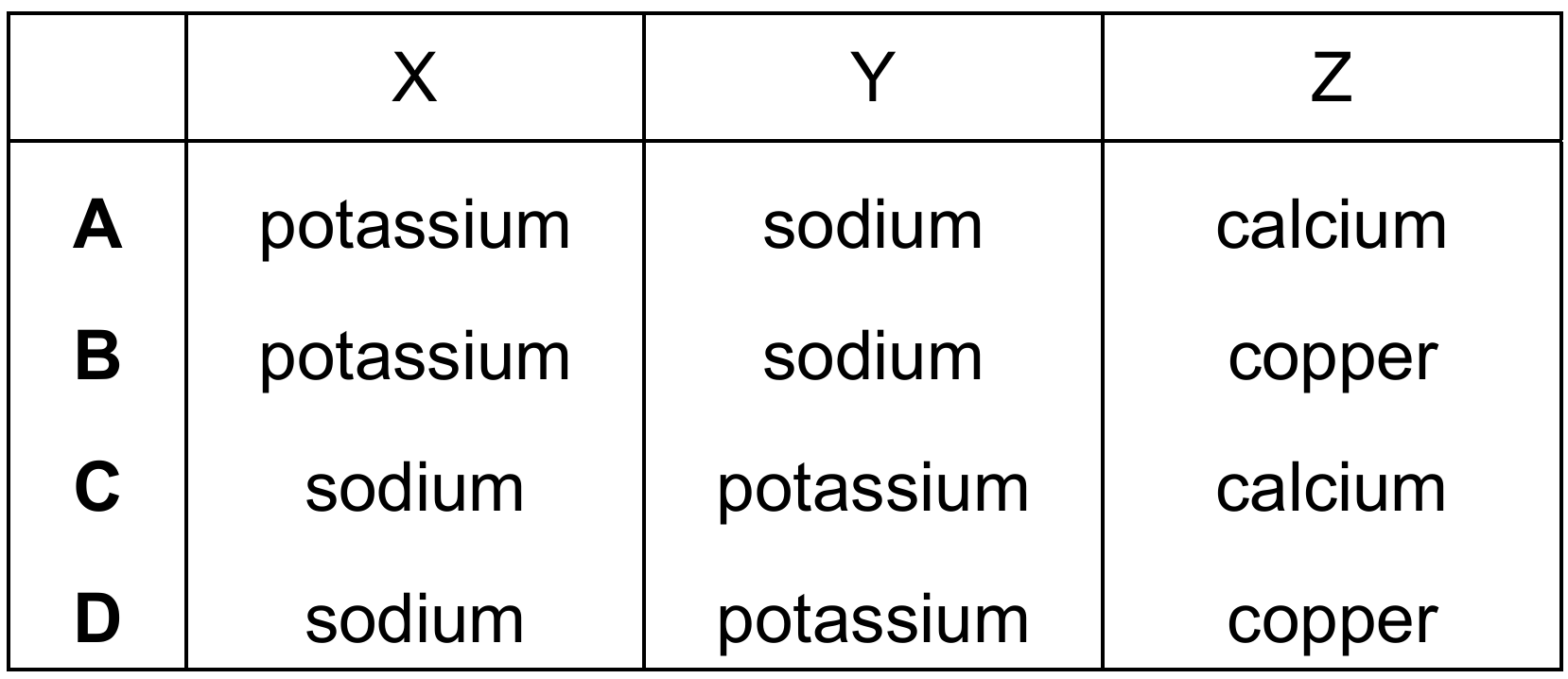
Question 39
The results of flame tests on three metal salts, X, Y and Z, are shown.
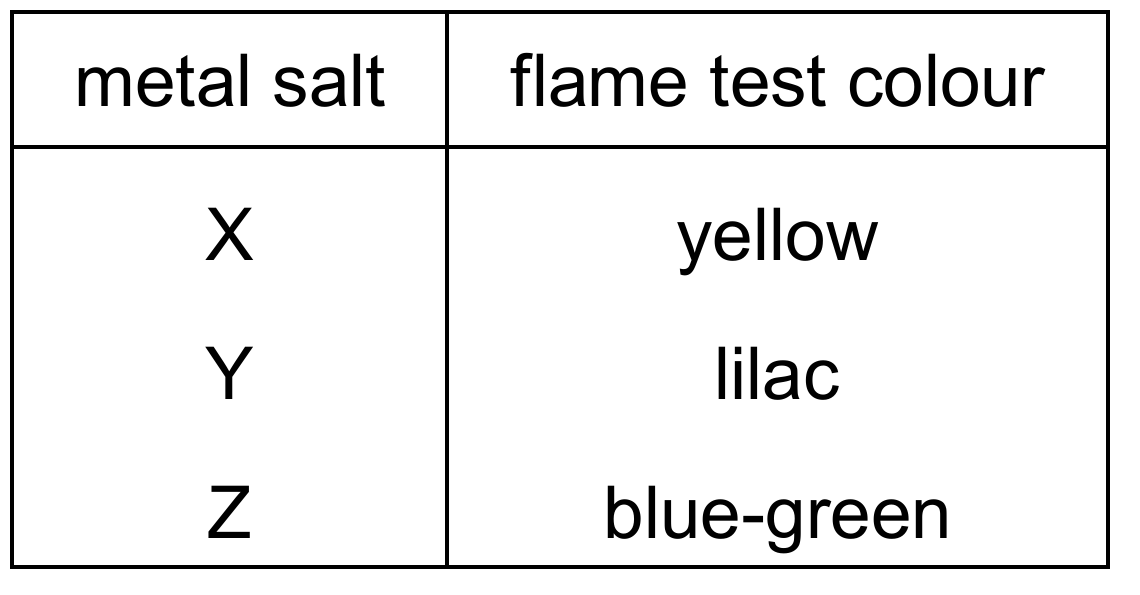
Which row identifies the cations present in X, Y and Z?
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
▶️ Answer/Explanation
Correct Answer: D
Explanation: Flame test colours for common metal ions:
– Sodium (Na⁺) gives a yellow flame.
– Potassium (K⁺) gives a lilac flame.
– Copper(II) (Cu²⁺) gives a blue-green flame.
– Calcium (Ca²⁺) gives an orange-red flame.
✅ X = sodium (yellow), Y = potassium (lilac), Z = copper(II) (blue-green).
Question 40
Ethanol can be made by fermentation of sugar, using yeast. This produces a mixture of ethanol and water. How is ethanol separated from this mixture?
A. distilling the mixture using a fractionating column
B. filtering the mixture
C. heating to evaporate most of the water, and allowing the ethanol to crystallise
D. heating to evaporate the water
Cambridge IGCSE Chemistry 0620 (2026-2028 syllabus):
• TOPIC 12.4: Separation and purification (fractional distillation to separate miscible liquids with different boiling points)
▶️ Answer/Explanation
Correct Answer: A
Explanation: The fermentation mixture contains ethanol (boiling point 78°C) and water (boiling point 100°C). Since they are miscible liquids with different boiling points, fractional distillation is used to separate them. The fractionating column allows the more volatile ethanol to vaporise first, then condense and be collected separately from water. Filtering would remove solids, not separate dissolved liquids. Ethanol does not crystallise; it is a liquid. Evaporating water would leave ethanol, but this is inefficient and less pure.
✅ Distilling the mixture using a fractionating column.
