The three states of matter are solid, liquid and gas.
(a) Substances can change from one state to another.
The box gives some words about changes of state.

Complete the table by giving the correct word from the box for each change of state.
You may use each word once, more than once or not at all.
| Change of state | Name of change |
|---|---|
| from liquid to solid | |
| from gas to liquid | |
| from solid to gas |
(b) The diagram shows two states of matter for a substance.
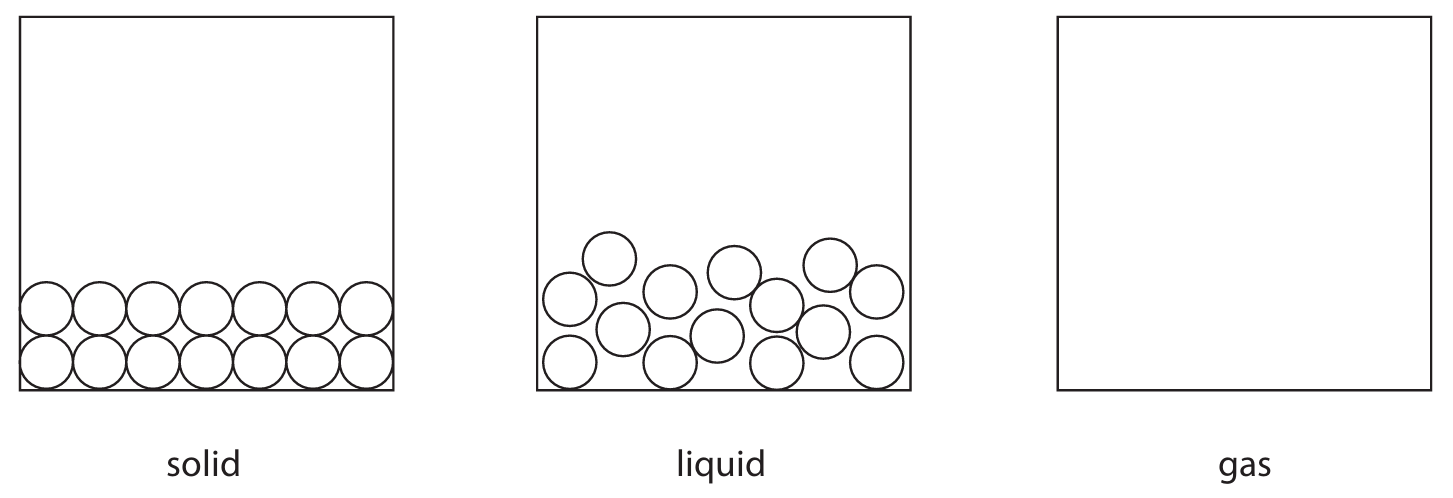
Each circle represents an atom of the substance.
(i) Complete the diagram by drawing six circles to represent atoms in the gas state.
(ii) Which statement is correct about this substance?
A) the atoms move randomly in the gas state
B) the atoms move randomly in the solid state
C) the atoms are in fixed positions in the gas state
D) the atoms are in fixed positions in the liquid state
(c) Complete the equation, by adding the state symbols, for the conversion of water into ice.
\[ \text{H}_2\text{O}( \text{……} ) \rightarrow \text{H}_2\text{O}( \text{……} ) \]
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1.1–1.7C: Particle theory, interconversions, diffusion, solubility — conceptual basis for all parts
▶️ Answer/Explanation
(a)
| Change of state | Name of change |
|---|---|
| from liquid to solid | freezing |
| from gas to liquid | condensing |
| from solid to gas | sublimation |
Explanation: Freezing is the change from liquid to solid. Condensing (or condensation) is the change from gas to liquid. Sublimation is the direct change from solid to gas without passing through the liquid state.
(b)(i)
The diagram should show six circles of similar size, randomly arranged and not touching, with at least one circle in the top and bottom of the box to represent gas particles in random motion.
(b)(ii) A (the atoms move randomly in the gas state)
Explanation:
• B is not correct because atoms in a solid vibrate but do not move randomly.
• C is not correct because atoms in a gas are not in fixed positions.
• D is not correct because atoms in a liquid can move past each other and are not in fixed positions.
(c) \[ \text{H}_2\text{O}(l) \rightarrow \text{H}_2\text{O}(s) \]
Explanation: Water in the liquid state (l) freezes to become ice in the solid state (s).
The table gives some information about five gases.
| Name | Formula | Boiling point in °C |
|---|---|---|
| chlorine | \( \text{Cl}_2 \) | -35 |
| hydrogen chloride | \( \text{HCl} \) | -114 |
| oxygen | \( \text{O}_2 \) | -183 |
| nitrogen | \( \text{N}_2 \) | -196 |
| hydrogen | \( \text{H}_2 \) | -253 |
(a) Use gases from the table to answer these questions. You may use each gas once, more than once or not at all.
(i) Name the gas needed for combustion.
(ii) Name the gas that is most abundant in air.
(iii) Name the gas that can bleach damp litmus paper.
(b) Explain why hydrogen chloride is classified as a compound.
(c) Why does chlorine have the highest boiling point of the gases in the table?
A) chlorine has the strongest covalent bonds between its atoms
B) chlorine has the strongest covalent bonds between its molecules
C) chlorine has the strongest ionic bonds between its atoms
D) chlorine has the strongest forces of attraction between its molecules
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(b): Group 7 (halogens) — part (a)(iii)
• 1(b): Elements, compounds and mixtures — part (b)
• 1(g): Covalent bonding — part (c)
• 1(a): States of matter — part (c)
▶️ Answer/Explanation
(a)(i) oxygen (ALLOW \( \text{O}_2 \)) (1)
(a)(ii) nitrogen (ALLOW \( \text{N}_2 \)) (1)
(a)(iii) chlorine (ALLOW \( \text{Cl}_2 \)) (1)
(b) (2 marks)
• M1: (Hydrogen chloride) has (atoms of) two / different elements (ALLOW two different atoms) (1)
• M2: (chemically) bonded/joined / (chemically) combined together (1)
(c) D (chlorine has the strongest forces of attraction between its molecules) (1)
• A is not the correct answer because covalent bonds are not broken when chlorine boils.
• B is not the correct answer because covalent bonds do not occur between molecules.
• C is not the correct answer because chlorine does not have ionic bonds.
Total = 6 marks
This question is about the rusting of iron.
(a) (i) Name the two substances needed for iron to rust.
(ii) Give the chemical name for rust.
(b) The diagram shows two methods used to prevent iron from rusting.

Method A will work only if the paint coating is not scratched.
Method B will work even if the zinc coating is scratched.
(i) Explain how method A prevents iron from rusting.
(ii) Give the name of method B.
(iii) Explain how method B prevents iron from rusting even when the zinc coating is scratched.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(d): Reactivity series (Redox) — Spec. point 2.20: understand the terms oxidation, reduction, redox, oxidising agent, reducing agent in terms of gain or loss of oxygen and loss or gain of electrons.
▶️ Answer/Explanation
(a)(i)
1. oxygen (or air, O2)
2. water (or moisture, H2O)
(a)(ii)
(Hydrated) iron(III) oxide (or ferric oxide, Fe2O3·xH2O).
(b)(i)
The paint acts as a barrier / protective layer. This prevents water / oxygen / air from reaching the iron surface and reacting with it.
(b)(ii)
Galvanising (or sacrificial protection).
(b)(iii)
Zinc is more reactive than iron / has a greater tendency to lose electrons. When scratched, the zinc oxidises / corrodes instead of the iron, acting as a sacrificial anode. This means the zinc loses electrons (is oxidised) in preference to the iron, protecting the iron from rusting.
A student does an experiment to identify the dyes in inks A, B, C, D and E.
The student uses this apparatus:
- a piece of chromatography paper
- a beaker
- a solvent
- inks A, B, C, D and E
(a) Describe how the student should set up the apparatus and do the experiment. You may use a diagram to help your answer.
(b) The chromatogram shows the results for inks A, B, C, D and E.
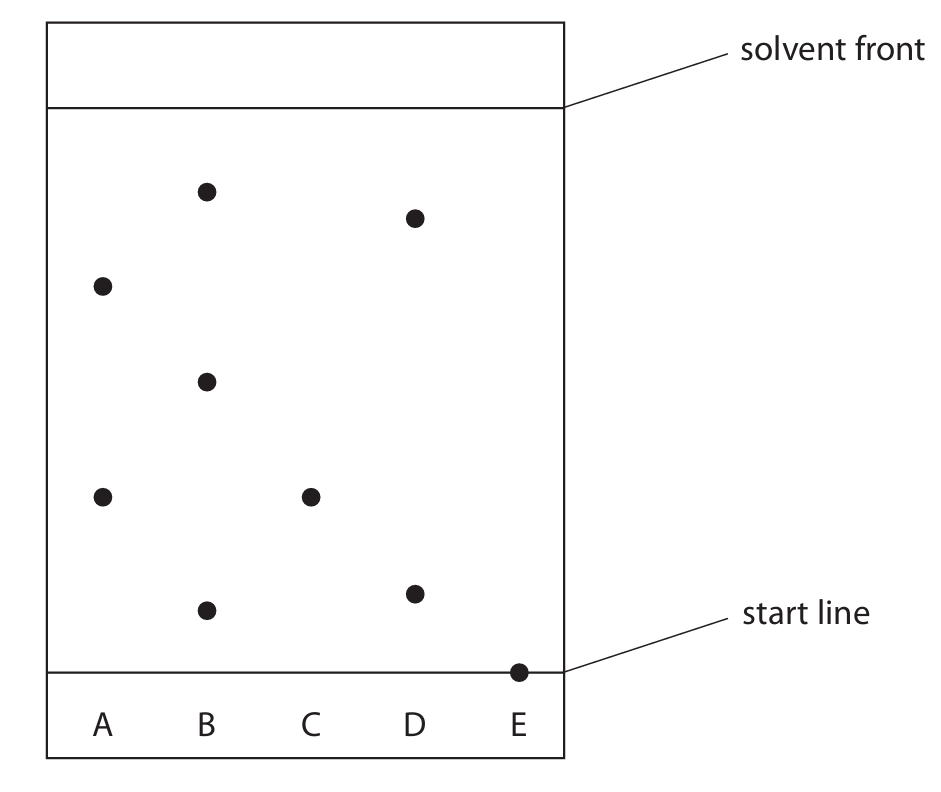
(i) Explain which ink contains a dye that is insoluble in the solvent.
(ii) Explain which inks contain the same dye.
(iii) Describe how the student can use the chromatogram to determine the \( R_f \) value for the dye in ink C. You do not need to calculate the \( R_f \) value.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• Specification points 1.10, 1.11, 1.12, 1.13: Separation techniques and chromatography.
▶️ Answer/Explanation
(a) (5 marks)
M1: Draw a line in pencil (just above the bottom of the paper) – the start line.
M2: Put a small spot of each ink on the line (ensuring they do not touch and are above the solvent level initially).
M3: Pour a small amount of the chosen solvent (e.g., water, ethanol) into the beaker.
M4: Place the paper in the beaker so the start line/spots are above the solvent level.
M5: Leave the setup until the solvent has risen up the paper (nearly to the top) – the solvent front.
Note: All marks can be awarded from a clearly labelled diagram showing these steps. The solvent must not touch the spots initially.
(b)(i) (2 marks)
M1: Ink E.
M2: Because its spot remained on the start line / did not travel up the paper / has an \( R_f \) value of 0.
Explanation: A dye that is insoluble in the solvent will not dissolve and cannot be carried up the chromatography paper by the moving solvent.
(b)(ii) (2 marks)
M1: Inks A and C.
M2: Because they both have a spot at the same height / travelled the same distance from the start line / have the same \( R_f \) value.
Explanation: In chromatography, substances that are the same chemical will travel the same distance under identical conditions.
(b)(iii) (2 marks)
M1: Measure the distance from the start line to the centre of the spot for ink C.
M2: Measure the distance from the start line to the solvent front.
M3: The \( R_f \) value is calculated using the formula:
\( R_f = \frac{\text{distance moved by substance (spot C)}}{\text{distance moved by solvent (solvent front)}} \)
Note: The question asks for a description of the method, not a calculation. The student would then substitute their measurements into this formula.
Hydrogen peroxide decomposes to form water and oxygen.
This decomposition is shown in the equation.
\[ 2H_2O_2 \rightarrow 2H_2O + O_2 \]
(a) Give the test for oxygen gas.
(b) The rate of reaction is increased by adding a suitable catalyst.
Describe how a catalyst increases the rate of a reaction.
(c) A student uses this apparatus to investigate the rate of decomposition of hydrogen peroxide solution.
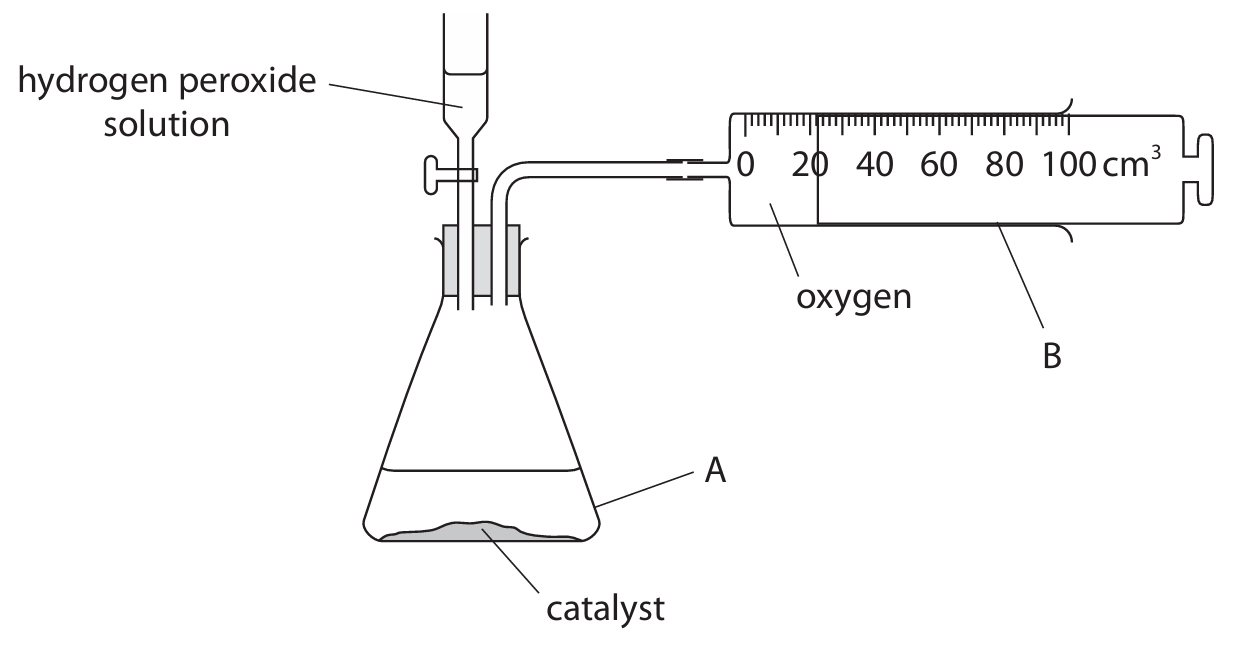
The student adds 100 cm\(^3\) of hydrogen peroxide solution at 20°C to the apparatus labelled A.
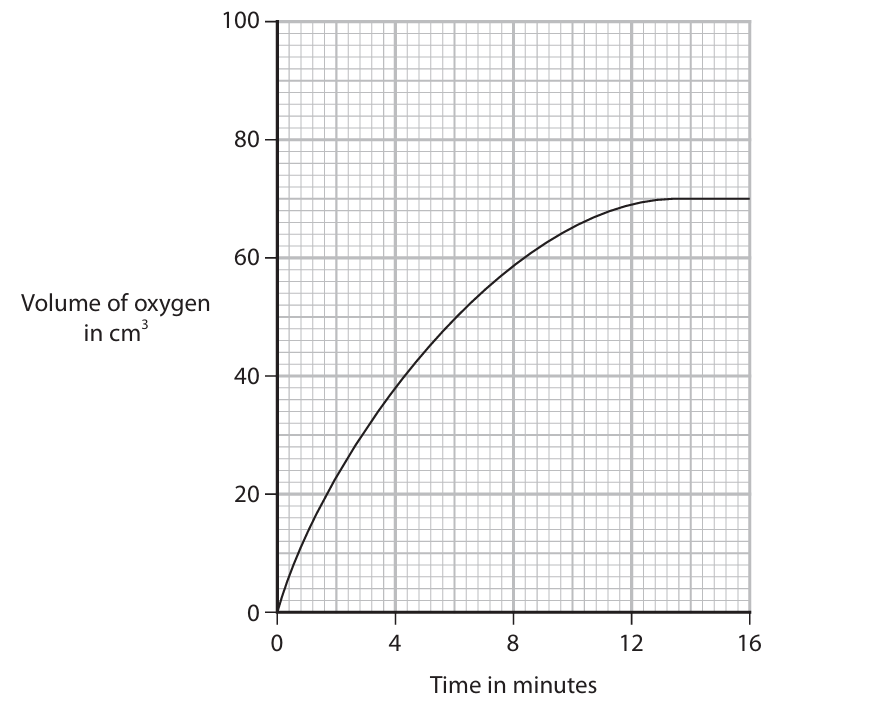
The student records the volume of oxygen gas collected every minute for 16 minutes.
The graph shows the student’s results.
(i) Name the pieces of apparatus labelled A and B.
(ii) Determine the volume of oxygen gas produced in the first 4 minutes.
Show on the graph how you obtain your answer.
(iii) Determine the rate of reaction at 8 minutes by drawing a suitable tangent to the curve.
Include the units for the rate of reaction.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 3(b): Rates of reaction — parts (b), (c)(ii), (c)(iii)
• 3(b): Rates of reaction – practical investigation — part (c)(i)
• Appendix 4: Mathematical skills (Graphs) — part (c)(iii)
▶️ Answer/Explanation
(a) Oxygen relights a glowing splint.
Explanation: This is the standard chemical test for oxygen gas.
(b)
- A catalyst provides an alternative pathway/route for the reaction. (M1)
- This alternative pathway has a lower activation energy. (M2)
Explanation: By lowering the activation energy, more reactant particles possess sufficient energy to react upon collision, thus increasing the rate of reaction.
(c)(i)
- A: (Conical) flask
- B: (Gas) syringe
(c)(ii)
- Draw a vertical line from 4 minutes on the time axis up to the curve. (M1)
- From that point, draw a horizontal line to the volume axis.
- Read the volume: 38 cm\(^3\) (allow 37–39 cm\(^3\)). (M2)
Explanation: The volume of gas produced at a specific time is read directly from the curve on the graph.
(c)(iii)
- Draw a tangent to the curve at the point where time = 8 minutes. The tangent should only touch the curve at that one point. (M1)
- Choose two points far apart on the tangent line and record their coordinates, e.g., (x1, y1) and (x2, y2). (M2)
- Calculate the rate using the formula: \(\text{rate} = \frac{\Delta y}{\Delta x} = \frac{y_2 – y_1}{x_2 – x_1}\).
Example using points (4 min, 20 cm³) and (12 min, 55 cm³) on the tangent:
\(\text{rate} = \frac{55 – 20}{12 – 4} = \frac{35}{8} = 4.375\) (M3) - Rate = 4.4 cm\(^3\)/min (to 2 significant figures). Units: \(\textbf{cm}^3\textbf{/min}\) (or cm³/min, cm³ min⁻¹, cm³/s, cm³ s⁻¹). (M4)
Explanation: The rate of reaction at a specific point is given by the gradient of the tangent to the curve at that point. The gradient represents the change in volume per unit time.
This question is about ionic and covalent compounds.
(a) The table gives the formulae of some positive ions, some negative ions and some compounds containing these ions.
| NH\(_4^+\) | Zn\(^{2+}\) | Al\(^{3+}\) | |
|---|---|---|---|
| Cl\(^-\) | NH\(_4\)Cl | ZnCl\(_2\) | |
| SO\(_4^{2-}\) | (NH\(_4\))\(_2\)SO\(_4\) | Al\(_2\)(SO\(_4\))\(_3\) | |
| N\(^{3-}\) | Zn\(_3\)N\(_2\) | AlN |
(i) Complete the table by giving the three missing formulae.
(ii) Give the name of the compound with the formula Al\(_2\)(SO\(_4\))\(_3\).
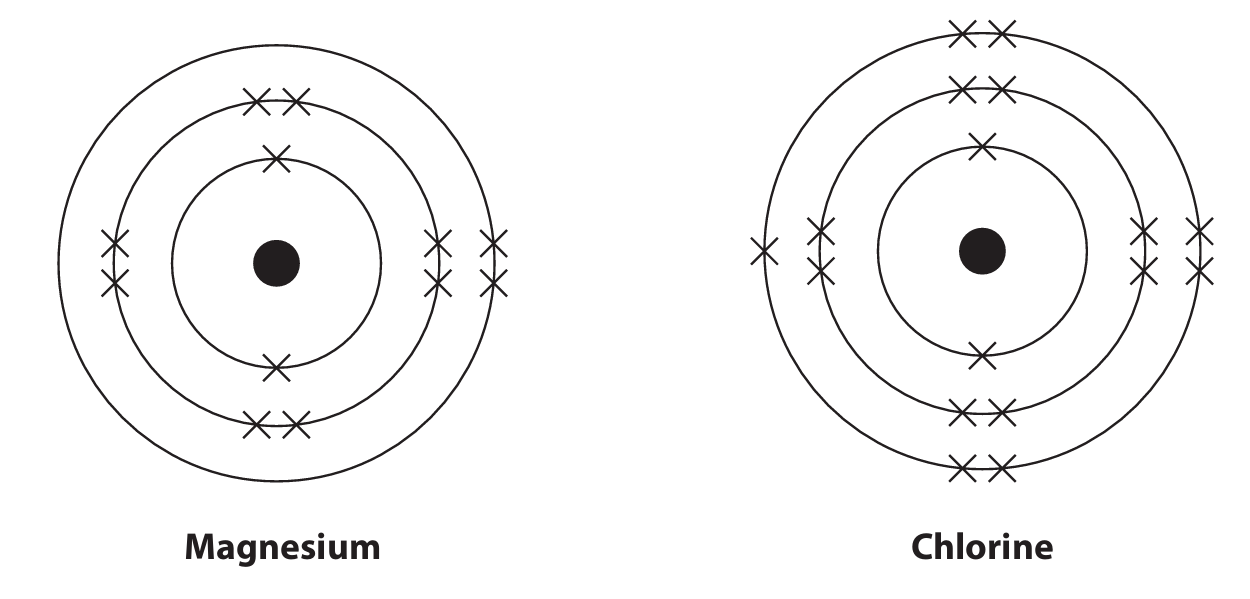
(b) Describe, in terms of electrons, what happens when magnesium atoms and chlorine atoms form magnesium chloride.
(c) (i) Complete the dot-and-cross diagram to show the outer shell electrons in a molecule of ammonia.

(ii) Describe the forces of attraction in a covalent bond.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(g): Covalent bonding — parts (c)(i), (c)(ii)
• 1(e): Chemical formulae, equations and calculations — part (a)(i)
▶️ Answer/Explanation
(a)(i)
• AlCl\(_3\)
• ZnSO\(_4\)
• (NH\(_4\))\(_3\)N
Explanation: The formula is determined by balancing the charges of the ions. For example, Al\(^{3+}\) and Cl\(^-\) combine in a 1:3 ratio to give AlCl\(_3\).
(a)(ii)
• Aluminium sulfate
Explanation: The compound consists of aluminium ions (Al\(^{3+}\)) and sulfate ions (SO\(_4^{2-}\)).
(b)
• Magnesium loses electrons.
• Chlorine gains electrons.
• Specifically, each magnesium atom loses two electrons, and two chlorine atoms each gain one electron.
• This results in the formation of Mg\(^{2+}\) and Cl\(^-\) ions, which attract each other to form magnesium chloride (MgCl\(_2\)).
Explanation: Magnesium, a Group 2 metal, readily loses its two outer shell electrons to achieve a stable full outer shell (like a noble gas). Chlorine, a Group 7 non-metal, readily gains one electron to achieve a stable full outer shell. The transfer of electrons creates oppositely charged ions that are held together by strong electrostatic forces (ionic bonding).
(c)(i)
• The dot-and-cross diagram for ammonia (NH\(_3\)) should show:
– A nitrogen atom with five outer shell electrons.
– Three hydrogen atoms, each with one electron.
– Three covalent bonds, each formed by a shared pair of electrons (one from N, one from H) between the nitrogen and each hydrogen.
– A lone pair (two non-bonding electrons) on the nitrogen atom.
Explanation: Nitrogen needs three more electrons to complete its outer shell, which it achieves by sharing electrons with three hydrogen atoms. The lone pair on nitrogen is responsible for ammonia’s basic properties.
(c)(ii)
• The force of attraction in a covalent bond is an electrostatic attraction.
• It acts between the shared pair(s) of electrons and the nuclei of the bonded atoms.
Explanation: The shared electrons are attracted to the positively charged nuclei of both atoms involved in the bond. This mutual attraction holds the atoms together. It is important to distinguish this intramolecular force from intermolecular forces, which are between separate molecules.
Magnesium sulfate crystals have the formula \(\text{MgSO}_4 \cdot x\text{H}_2\text{O}\). A student uses this apparatus to heat tube A and collect water of crystallisation in tube B.
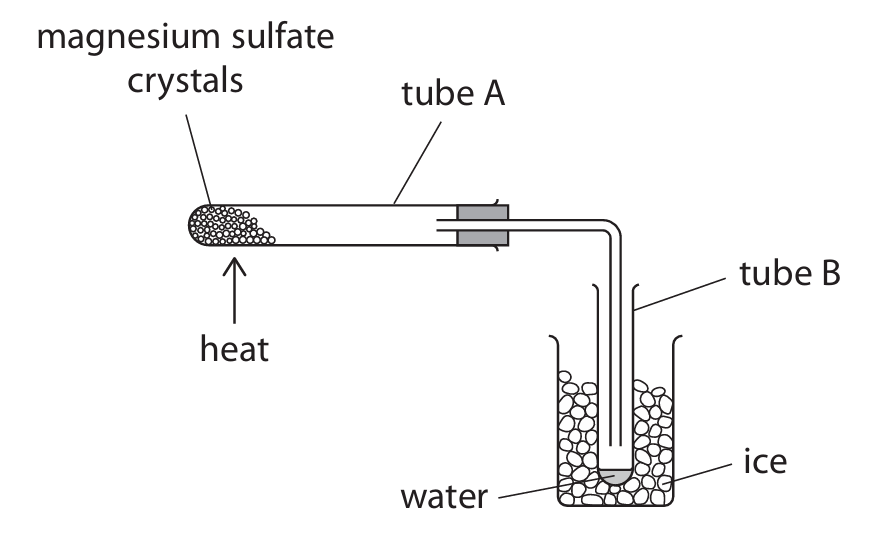
The student records the mass of tube B at the start and every minute for 6 minutes. The table gives the student’s results.
| Time in minutes | Mass of tube B in g |
|---|---|
| 0 | 15.8 |
| 1 | 16.5 |
| 2 | 17.6 |
| 3 | 19.2 |
| 4 | 20.4 |
| 5 | 22.1 |
| 6 | 22.1 |
(a) (i) Give a reason why the student surrounds tube B with ice. (1 mark)
(ii) State how the student knows when all the water of crystallisation has been given off. (1 mark)
(b) Describe a chemical test the student can use to show that the liquid in tube B contains water. (2 marks)
(c) The decomposition of hydrated magnesium sulfate is shown in the equation.
\[ \text{MgSO}_4 \cdot x\text{H}_2\text{O} \rightarrow \text{MgSO}_4 + x\text{H}_2\text{O} \]
At the end of the reaction, the mass of \(\text{MgSO}_4\) remaining in the tube is 6.0 g.
Use this mass and the results in the table to calculate \(x\) in \(\text{MgSO}_4 \cdot x\text{H}_2\text{O}\).
[For \(\text{MgSO}_4\), \(M_r = 120\)]
[For \(\text{H}_2\text{O}\), \(M_r = 18\)] (4 marks)
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(a): States of matter — part (a)(i) condensation, part (a)(ii) practical observation of mass change
• 2(h): Chemical tests — part (b) test for water
• Appendix 6: Suggested practical investigations — determine the formula of a hydrated salt by heating (related to spec point 1.36)
▶️ Answer/Explanation
(a)(i) • To condense the water vapour / steam / gas (into liquid).
Explanation: The ice cools tube B, causing the gaseous water released from the crystals to condense back into liquid so it can be collected and measured.
(a)(ii) • When the mass doesn’t change / is constant / stops increasing / (the last) two readings are the same.
Explanation: The mass of tube B increases as water condenses into it. When no more water is being produced from the crystals, the mass stabilizes.
(b) • Add anhydrous / white copper(II) sulfate. (1)
• It turns from white to blue. (1)
Alternative: Add anhydrous cobalt chloride. It turns from blue to pink.
Explanation: These substances are chemical tests for the presence of water. The colour change is a positive result.
(c) Calculation:
- Mass of water collected = Final mass of tube B – Initial mass of tube B = \(22.1 \, \text{g} – 15.8 \, \text{g} = 6.3 \, \text{g}\).
- Moles of \(\text{MgSO}_4\) = \(\frac{\text{mass}}{M_r} = \frac{6.0}{120} = 0.05 \, \text{mol}\).
- Moles of \(\text{H}_2\text{O}\) = \(\frac{6.3}{18} = 0.35 \, \text{mol}\).
- Ratio \(x = \frac{\text{moles of } \text{H}_2\text{O}}{\text{moles of } \text{MgSO}_4} = \frac{0.35}{0.05} = 7\).
Therefore, \(x = 7\).
Explanation: The formula is \(\text{MgSO}_4 \cdot 7\text{H}_2\text{O}\). The calculation uses the mole concept and the given relative formula masses to find the ratio of water to magnesium sulfate.
Mark Scheme Summary (from provided image):
M1: mass of water = 6.3 g (1)
M2: moles of \(\text{MgSO}_4\) = 0.05 (1)
M3: moles of \(\text{H}_2\text{O}\) = 0.35 (1)
M4: \(x = 7\) (1)
Award full marks for answer 7 alone.
A student uses this apparatus to investigate the rate of reaction between marble chips and dilute hydrochloric acid.
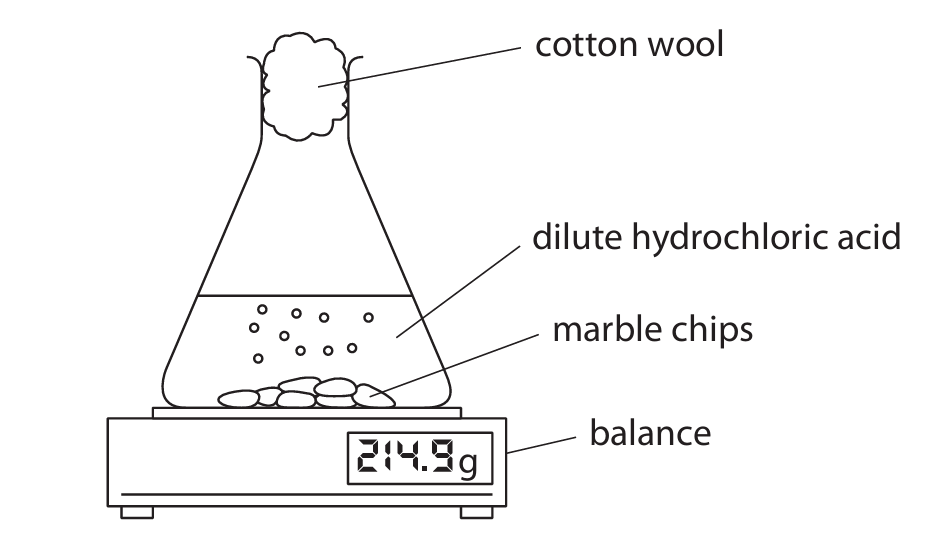
This is the equation for the reaction.
\[ \text{CaCO}_3(\text{s}) + 2\text{HCl}(\text{aq}) \rightarrow \text{CaCl}_2(\text{aq}) + \text{H}_2\text{O(l)} + \text{CO}_2(\text{g}) \]
(a) Give a reason why the reading on the balance decreases during the reaction.
(b) Explain why the rate of reaction is greatest at the start.
(c) After 10 minutes, the reaction stops even though there are marble chips remaining. Give a reason why the reaction stops.
(d) The student repeats the experiment with hydrochloric acid at a higher temperature to investigate the effect on the rate of reaction.
(i) Give two variables that should be controlled to make sure the results are valid.
(ii) Explain the effect of increasing the temperature of hydrochloric acid on the rate of reaction.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(e): Chemical formulae, equations and calculations — context from the reaction equation
• 3.9–3.16: Practical investigation of rates of reaction — experimental context
▶️ Answer/Explanation
(a) Carbon dioxide (a gas) escapes/is lost/is released through the cotton wool. (1 mark)
Explanation: The product \(\text{CO}_2\) is a gas. As it leaves the flask, the total mass of the flask and its contents decreases, so the balance reading drops.
(b)
M1: The concentration (of hydrochloric acid) is highest at the start. (1 mark)
M2: So there are more collisions per unit time / more frequent collisions. (1 mark)
Explanation: According to collision theory, the rate depends on the frequency of successful collisions. A higher acid concentration means more \(\text{H}^+\) ions are available to collide with the marble chips (\(\text{CaCO}_3\)) surface.
(c) The hydrochloric acid has been used up. (1 mark)
Explanation: Marble chips are in excess. The acid is the limiting reactant. Once all the \(\text{H}^+\) ions have reacted, the reaction cannot continue, even with solid \(\text{CaCO}_3\) remaining.
(d)(i) Any two from: (1 mark each)
1. (Same) mass of marble chips.
2. (Same) surface area/size of marble chips.
3. (Same) concentration of hydrochloric acid.
4. (Same) volume of hydrochloric acid.
Explanation: To fairly investigate the effect of temperature alone, all other factors that affect the rate (concentration, surface area, mass/volume of reactants) must be kept constant.
(d)(ii)
M1: The rate of reaction increases. (1 mark)
M2: Particles have more (kinetic) energy / more particles have energy greater than or equal to the activation energy. (1 mark)
M3: So there are more successful collisions per unit time / more frequent successful collisions. (1 mark)
Explanation: Increasing temperature increases the average kinetic energy of the reactant particles (\(\text{H}^+\) ions and \(\text{CaCO}_3\) surface). A greater proportion of collisions then possess the required activation energy, and the collisions are more energetic, leading to a higher rate.
Total marks: 9
A student uses this apparatus to investigate the heat energy released when a liquid fuel burns.
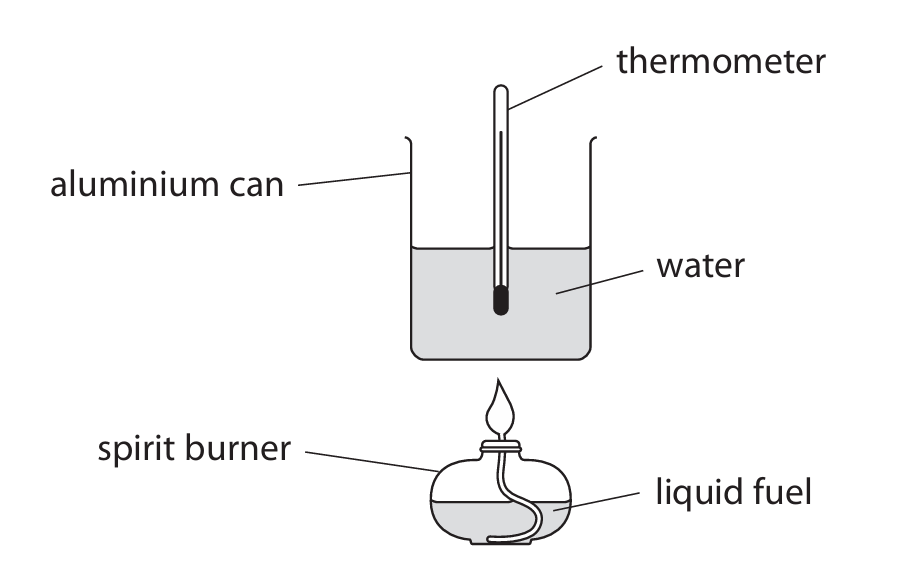
This is the student’s method.
- measure the mass of the spirit burner and fuel
- add 100 g of water to the aluminium can
- measure the temperature of the water
- use the spirit burner to heat the water until the temperature rises by 50°C
- measure the new mass of the spirit burner and fuel
(a) Give a reason why the student uses an aluminium can rather than a glass beaker.
(b) When the fuel burns, a black solid forms on the aluminium can.
(i) Identify the black solid.
(ii) Give a reason why the black solid forms.
(c) (i) Show that the heat energy needed to raise the temperature of 100 g of water by 50 °C is approximately 20 000 J.
[for water, \( c = 4.2 \, \text{J/g/°C} \)]
(ii) The student burns 1.84 g of ethanol.
Calculate the molar enthalpy change, \(\Delta H\), in kJ/mol, for the combustion of ethanol.
Include a sign in your answer.
[for ethanol, \( M_r = 46 \)]
(d) In a different experiment, the student burns 3.7 g of butanol.
This is an equation for the incomplete combustion of butanol.
\[C_4H_9OH(l) + \text{……}O_2(g) \rightarrow 2CO_2(g) + 2CO(g) + 5H_2O(g)\]
(i) Complete the equation for the incomplete combustion of butanol.
(ii) Calculate the total amount, in moles, of gas produced by the incomplete combustion of 3.7 g of butanol.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 4(b): Crude Oil / Combustion — parts (b)(i), (b)(ii)
• 1(e): Chemical formulae, equations and calculations — parts (c)(i), (c)(ii), (d)(ii)
• 4(e): Alcohols — parts (c)(ii), (d)
• 1(e): Chemical equations — part (d)(i)
▶️ Answer/Explanation
(a) aluminium is a better conductor (of heat) than glass / aluminium has a higher thermal conductivity than glass.
Note: A comparison is needed. Reject answers about insulation.
(b)(i) carbon / soot / C
(b)(ii) incomplete combustion occurs OR the supply of oxygen/air is limited.
(c)(i)
\( Q = mc\Delta T \)
\( Q = 100 \times 4.2 \times 50 \)
\( Q = 21000 \, \text{J} \) (≈ 20000 J)
Note: Full marks are awarded for showing the correct calculation (2 marks). The value 21000 J is accepted as approximately 20000 J.
(c)(ii)
M1: Heat energy \( Q = 21000 \, \text{J} = 21 \, \text{kJ} \)
M2: Moles of ethanol burned: \( \frac{1.84}{46} = 0.04 \, \text{mol} \)
M3: Enthalpy change per mole: \( \frac{21}{0.04} = 525 \, \text{kJ/mol} \)
M4: Since combustion releases heat, \( \Delta H = -525 \, \text{kJ/mol} \)
Note: Using 20 kJ gives \( \Delta H = -500 \, \text{kJ/mol} \), which is also accepted.
(d)(i)
Balanced equation: \( C_4H_9OH(l) + 5O_2(g) \rightarrow 2CO_2(g) + 2CO(g) + 5H_2O(g) \)
Note: Allow multiples if all coefficients are scaled consistently.
(d)(ii)
M1: \( M_r \) of butanol \( (C_4H_9OH) = (4 \times 12) + (9 \times 1) + 16 + 1 = 74 \)
M2: Moles of butanol: \( \frac{3.7}{74} = 0.05 \, \text{mol} \)
M3: From the equation, 1 mole of butanol produces \( 2 + 2 + 5 = 9 \) moles of gas.
Moles of gas from 0.05 mol butanol: \( 0.05 \times 9 = 0.45 \, \text{mol} \)
Answer: amount of gas = \( 0.45 \, \text{mol} \)
This question is about alkanes and alkenes.
(a) Ethane and ethene each react with bromine, but under different conditions.
(i) Give a reason why ethane does not undergo an addition reaction with bromine.
(ii) Complete the equation for the substitution reaction between ethane and bromine, including an essential condition for the reaction.
\( \text{C}_2\text{H}_6 + \text{Br}_2 \rightarrow \text{……} + \text{……} \)
(iii) Give the colour change that occurs when ethene reacts with bromine water. (1)
(b) The diagram shows the displayed formulae of two compounds in the homologous series of alkenes.
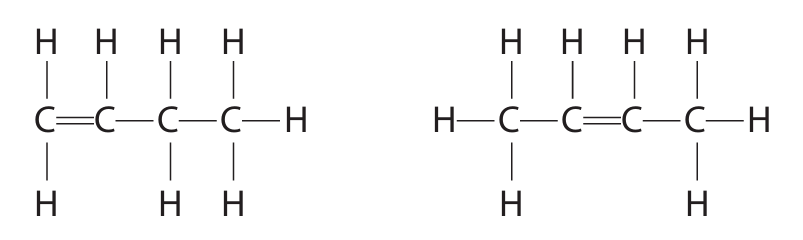
(i) Compounds in the same homologous series have the same general formula. Give two other characteristics of compounds in the same homologous series.
(ii) Explain why the alkenes in the diagram are isomers.
(iii) Draw the displayed formula of another alkene that is an isomer of these two alkenes.
(c) The reaction used to make poly(ethene) can be represented by this equation.
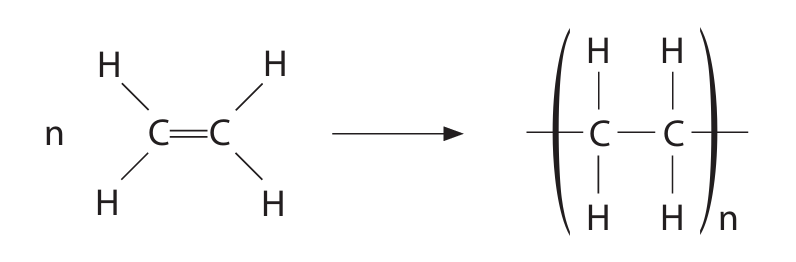
Describe the differences between the reactant and the product in this reaction. Refer to carbon chain length, type of bond and state of matter in your answer.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 4(d): Alkenes — parts (a)(iii), (b)(i), (b)(ii), (b)(iii)
• 4(a): Introduction to Organic Chemistry — parts (b)(i) [homologous series], (b)(ii) [isomerism]
• 4(h): Synthetic Polymers — part (c) [poly(ethene) formation]
▶️ Answer/Explanation
(a)(i) Any one from:
- Ethane is saturated.
- Ethane has no double bonds / only single bonds.
- It cannot add bromine across a double bond.
Explanation: Alkanes like ethane are saturated hydrocarbons, meaning they contain only single C–C bonds. Addition reactions require a double bond (unsaturated) to break and add atoms across it.
(a)(ii) \[ \text{C}_2\text{H}_6 + \text{Br}_2 \rightarrow \text{C}_2\text{H}_5\text{Br} + \text{HBr} \]
Condition: ultraviolet (UV) light / ultraviolet radiation.
Explanation: Alkanes undergo free-radical substitution with halogens in the presence of UV light, which provides the energy to break the Br–Br bond and initiate the chain reaction.
(a)(iii) Orange/yellow/brown to colourless / decolourises.
Explanation: Ethene (an alkene) reacts rapidly with bromine water in an addition reaction, forming colourless 1,2-dibromoethane. This is a standard test for unsaturation.
(b)(i) Any two from:
- They have the same functional group.
- They show a gradation in physical properties (e.g., boiling point increases with chain length).
- They have similar chemical properties.
- Each successive member differs by \( \text{CH}_2 \).
(b)(ii)
• They have the same molecular formula (\( \text{C}_4\text{H}_8 \)).
• They have different structural / displayed formulae (different arrangement of atoms).
Explanation: Both are alkenes with four carbons and eight hydrogens, but the position of the double bond differs (but-1-ene vs but-2-ene), making them structural isomers.
(b)(iii) Any other alkene with formula \( \text{C}_4\text{H}_8 \), e.g., methylpropene (2-methylprop-1-ene):
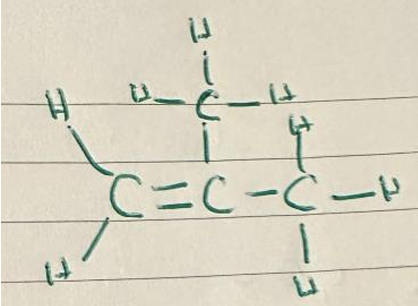
Allow E/trans isomer:
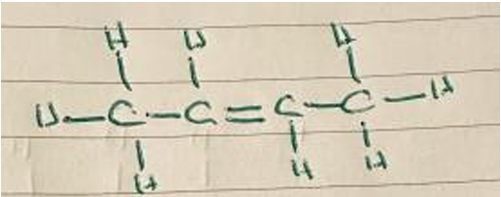
(c) Description covering three points:
- Carbon chain length: The chain length is longer in poly(ethene) / poly(ethene) has a very long chain / many repeating units / polymer chain, whereas ethene is a short-chain monomer.
- Type of bond: The reactant (ethene) contains a C=C double bond / is unsaturated, while the product (poly(ethene)) contains only single (covalent) bonds / is saturated / has no double bond.
- State of matter: Ethene is a gas (at room temperature) and poly(ethene) is a solid.
Explanation: This is an addition polymerisation reaction. Many ethene monomers (small, gaseous, unsaturated molecules with C=C bonds) join together under suitable conditions to form a long-chain saturated solid polymer with only C–C single bonds.
This question is about the reactions of some compounds of lead.
(a) Lead can be extracted from lead(II) sulfide, PbS, in two stages.
Stage 1: lead(II) sulfide is heated in air and reacts with oxygen to produce lead(II) oxide, PbO, and sulfur dioxide.
Stage 2: lead(II) oxide is heated with carbon in a furnace.
(i) Write a chemical equation for the reaction in stage 1.
(ii) Give a reason why sulfur dioxide should not be released into the atmosphere.
(iii) This is the equation for stage 2:
\( 2\text{PbO} + \text{C} \rightarrow 2\text{Pb} + \text{CO}_2 \)
A mass of 892 tonnes of lead(II) oxide is heated in a furnace with an excess of carbon.
Calculate the maximum mass, in tonnes, of carbon dioxide that could be released into the atmosphere.
\( [1 \text{ tonne} = 1 \times 10^6 \text{ g}] \)
\( [\text{For PbO, } M_r = 223; \text{ for CO}_2, M_r = 44] \)
(iv) The diagram shows the structures of lead(II) sulfide and sulfur dioxide.
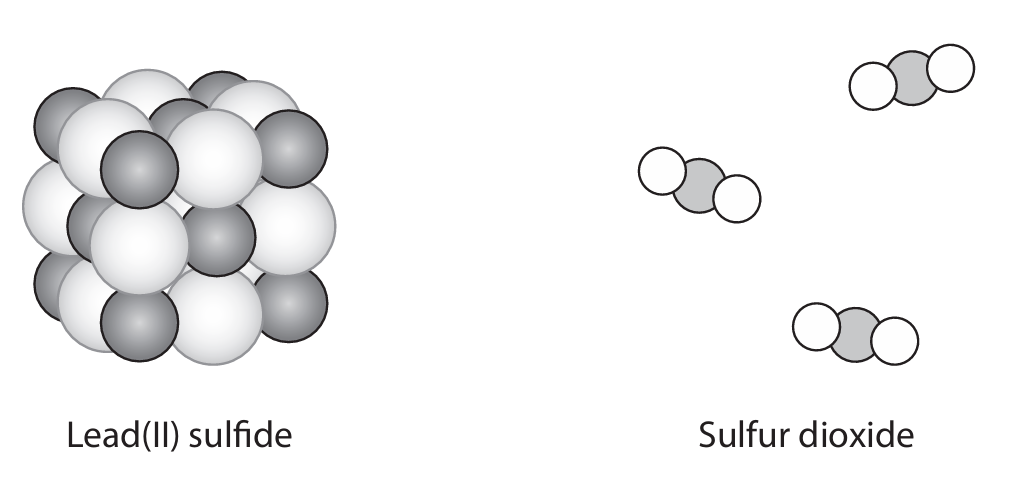
Explain, in terms of bonding and structure, why lead(II) sulfide is a solid with a very high melting point at room temperature and why sulfur dioxide is a gas at room temperature.
(b) A different oxide of lead contains 90.7% by mass of lead and 9.3% by mass of oxygen.
Determine the empirical formula of this oxide of lead.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(e): Extraction and uses of metals — context of parts (a)(i), (a)(iii)
• 4(c)/4(d): Crude oil / Alkenes — part (a)(ii) effect of sulfur dioxide (acid rain)
• 1(f): Ionic bonding — part (a)(iv) lead(II) sulfide structure
• 1(g): Covalent bonding — part (a)(iv) sulfur dioxide structure
• 1.33 / 1(e): Empirical and molecular formulae — part (b)
▶️ Answer/Explanation
(a) (i) \( 2\text{PbS} + 3\text{O}_2 \rightarrow 2\text{PbO} + 2\text{SO}_2 \)
Explanation: The correct formulae for oxygen (\(\text{O}_2\)) and sulfur dioxide (\(\text{SO}_2\)) are needed (M1). The equation must then be balanced correctly (M2).
(a) (ii) (Sulfur dioxide causes) acid rain / breathing problems.
Explanation: Accept named breathing problems (e.g., asthma) or other effects of acid rain such as killing fish, damaging stonework, or killing plants.
(a) (iii) 88 tonnes
Working:
M1: Moles of lead(II) oxide = \( \frac{892\,000\,000}{223} = 4\,000\,000 \text{ mol} \)
M2: From the equation \(2\text{PbO} \rightarrow \text{CO}_2\), moles of \(\text{CO}_2\) = \( \frac{4\,000\,000}{2} = 2\,000\,000 \text{ mol} \)
M3: Mass of \(\text{CO}_2\) = \( 2\,000\,000 \times 44 = 88\,000\,000 \text{ g} = 88 \text{ tonnes} \)
Explanation: All steps are dependent. Calculations can be done in megamoles. 88 tonnes scores full marks.
(a) (iv)
Lead(II) sulfide:
• Giant ionic structure/lattice (M1).
• Strong (ionic) bonds / strong electrostatic forces between oppositely charged ions (M2).
• These bonds/forces take a lot of energy to break/overcome (M3). Do not mention molecules, covalent bonds, or intermolecular forces.
Sulfur dioxide:
• Simple molecular / covalent structure (M4).
• Weak intermolecular forces / weak forces between molecules (M5).
• These forces take little energy to overcome (M6). Do not mention ions or ionic bonds.
Explanation: Award marks for clear comparisons of structure and bonding. M3 depends on M2; M6 depends on M5.
(b) Empirical formula = \(\text{Pb}_3\text{O}_4\)
Working:
M1: Moles of Pb = \( \frac{90.7}{207} \approx 0.438 \)
Moles of O = \( \frac{9.30}{16} \approx 0.581 \)
M2: Ratio of moles \( \text{Pb} : \text{O} = 0.438 : 0.581 \)
M3: Simplify ratio: divide by smallest (\(0.438\)) → \(1 : 1.33\)
M4: Multiply by 3 to get whole numbers → \(3 : 4\) → Empirical formula is \(\text{Pb}_3\text{O}_4\).
Explanation: The answer must be to at least 2 significant figures. Allow ecf (error carried forward) from a consistent ratio to produce a plausible formula.
