This question is about oxygen.
(a) Oxygen can be stored in tanks as a liquid or as a gas.
(i) Complete the diagram to show the arrangement of six more particles in a gas.

(ii) Give a reason why a tank can store much more oxygen as a liquid.
(iii) Identify a hazard when storing oxygen as a gas.
(b) Sulfur burns in oxygen to form sulfur dioxide.
(i) Give one observation that can be made when sulfur burns in oxygen.
(ii) Some universal indicator is added to sulfur dioxide. Explain the final colour of the universal indicator.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(c): Gases in the atmosphere / 2.11: Combustion of elements in oxygen — parts (b)(i), (b)(ii)
▶️ Answer/Explanation
(a)(i) Six particles should be arranged randomly. (Maximum of two particles should touch.)
Mark scheme note: Allow between 5 and 10 particles.
(a)(ii) Particles are (much) closer together.
Mark scheme note: Allow “the density is (much) greater / less space between particles / packed more tightly”.
(a)(iii) Any one from:
• High pressure
• Risk of explosion
• Oxidising agent
• Risk in contact with combustible materials
Mark scheme note: Allow “risk of fire”. Ignore “flammable”.
(b)(i) Blue flame.
Mark scheme note: Allow “choking / pungent gas produced / sulfur darkens”. Ignore any original colour.
(b)(ii)
M1: (Final colour) red/orange/yellow.
M2: An acidic solution / an acid / sulfurous acid is formed.
Mark scheme note: Allow “sulfuric acid” or “sulfur dioxide is acidic”. Accept “the solution formed contains hydrogen ions”.
This question is about mixtures and compounds.
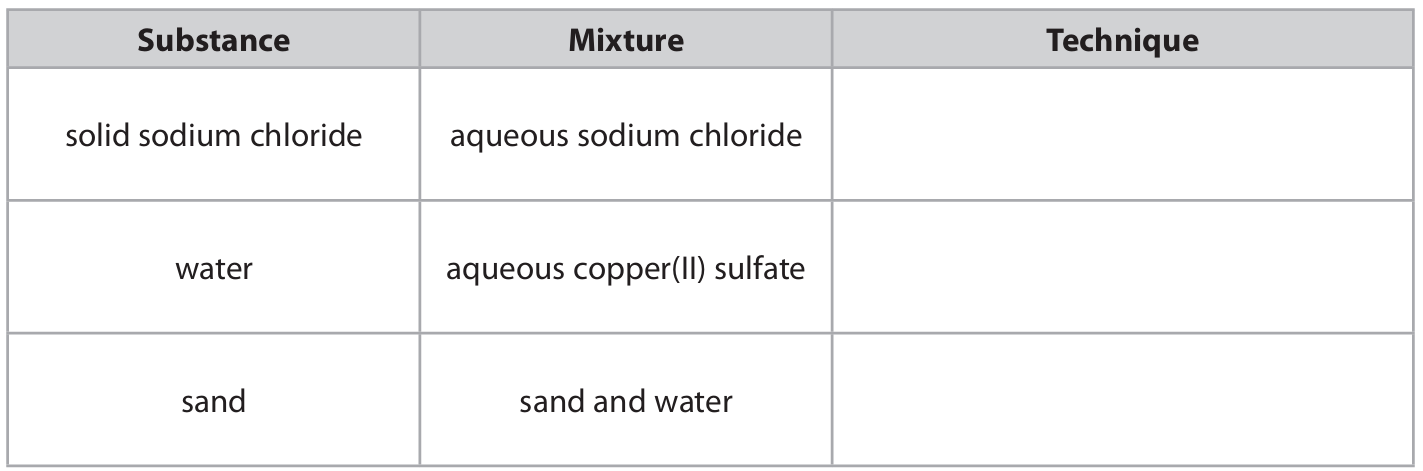
(a) The box gives some techniques used to separate mixtures.

The table lists some substances and mixtures. Complete the table using words from the box to show the best technique to obtain the named substance from each mixture. Each technique may be used once, more than once or not at all.
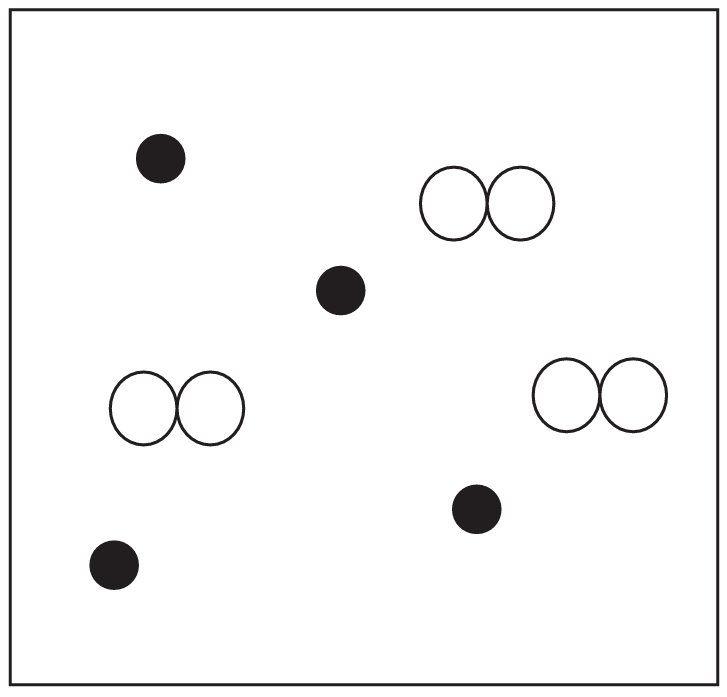
(b) The diagram below represents a mixture. State how the box represents a mixture.
(c) Food colourings are mixtures of food dyes.
A student uses paper chromatography to separate the food dyes contained in food colouring D.
The student places spots of three food dyes A, B and C and food colouring D on chromatography paper.
The diagram shows the appearance of the paper after the experiment.

Describe the composition of food colouring D.
(d) A compound has the formula \(\mathrm{Ca(HCO_3)_2}\).
(i) Determine the number of different elements in \(\mathrm{Ca(HCO_3)_2}\).
(ii) Determine the number of atoms in the formula of \(\mathrm{Ca(HCO_3)_2}\).
Most-appropriate topic codes (Edexcel IGCSE Chemistry – 4CH1):
• 1(b) Elements, compounds and mixtures – parts (a), (b), definitions and identification
• 1(b) Separation techniques – part (a), chromatography in (c)
• 1(e) Chemical formulae, equations and calculations – part (d), formula analysis
▶️ Answer/Explanation
(a) Table completion:
- Solid sodium chloride from aqueous sodium chloride → crystallisation
- Water from aqueous copper(II) sulfate → simple distillation
- Sand from sand and water → filtration
Award 1 mark per correct technique. “Distillation” without “simple” is not accepted.
(b) The box contains two different types of particles / two different substances / more than one type of particle.
Do not accept “compounds”.
(c) Any two of the following:
- Food colouring D contains three food dyes.
- Food colouring D contains dyes A and C.
- Food colouring D does not contain dye B.
- Food colouring D contains another dye which is not A, B or C.
(d)(i) 4 different elements: Ca, H, C, O.
(d)(ii) Total number of atoms = \(1\ \text{Ca} + 2 \times (1\ \text{H} + 1\ \text{C} + 3\ \text{O}) = 1 + 2 \times 5 = 11\) atoms.
(a) The diagram represents an atom of an element.
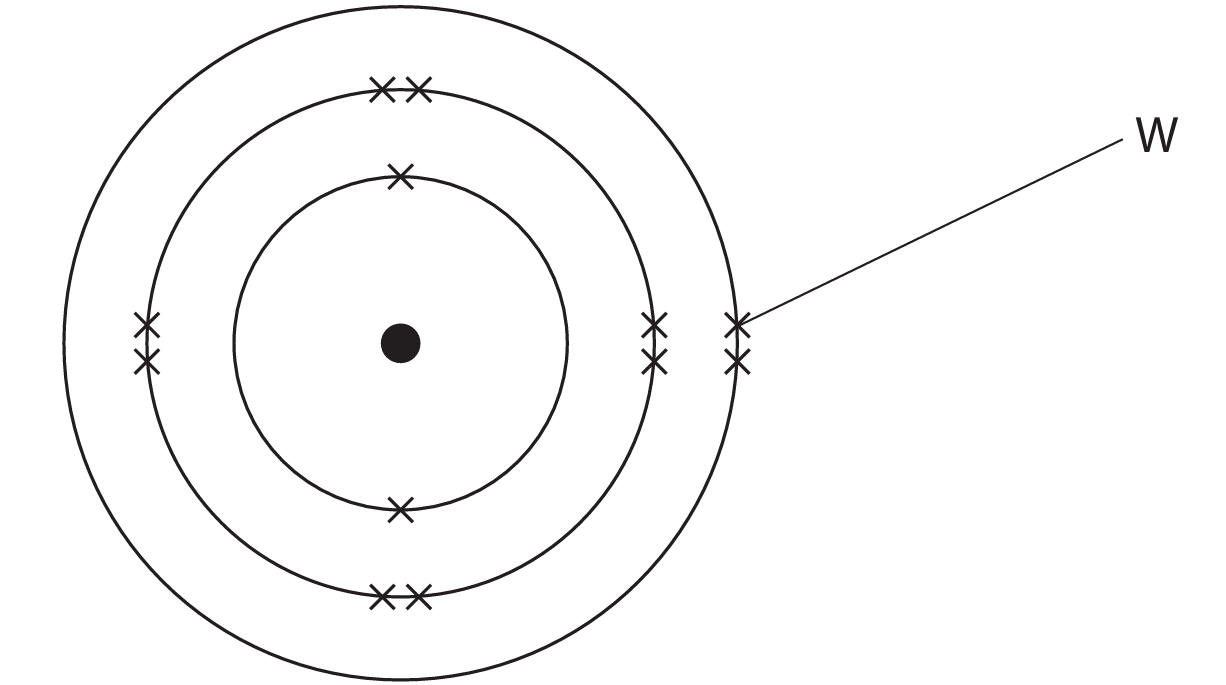
(i) What is the particle labelled W?
(ii) An atom of this element contains 13 neutrons. What is the mass number of this element?
(iii) State why atoms have no overall charge.
(iv) What is the charge on the ion usually formed from this element?
(b) There are two isotopes of the element lithium. \({}^6_3\text{Li} \quad {}^7_3\text{Li}\)
(i) State why both isotopes react in the same way.
(ii) A sample of lithium contains 7.60% \( {}^6_3\text{Li} \) and 92.4% \( {}^7_3\text{Li} \).
Calculate the relative atomic mass, \( A_r \), of this sample of lithium.
Give your answer to two decimal places.
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry):
• 1(f): Ionic bonding — part (a)(iv)
• 1(c): Isotopes — part (b)(i)
• 1(c): Relative atomic mass — part (b)(ii)
▶️ Answer/Explanation
(a)(i) A (electron)
B is not the correct answer because neutrons exist in the nucleus.
C is not the correct answer because particle W is not a nucleus.
D is not the correct answer because protons exist in the nucleus.
(a)(ii) C (25)
A is not the correct answer as the proton number is 12.
B is not the correct answer as the number of neutrons is 13.
D is not the correct answer as the number of protons, neutrons and electrons is not 49.
(a)(iii) (atoms have the) same number of protons and electrons
(a)(iv) B (2+)
A is not the correct answer as this atom will not form an ion with a + charge.
C is not the correct answer as this atom will not form an ion with a – charge.
D is not the correct answer as this atom will not form an ion with a 2- charge.
(b)(i) (both isotopes have) the same number of electrons / the same electron configuration.
IGNORE same number of protons / same number of electrons in the outer shell.
(b)(ii)
M1: \( (7.60 \times 6) + (92.4 \times 7) \)
M2: \( 692.4 \div 100 = 6.924 \)
M3: \( 6.92 \)
Answer of 6.92 scores 3 marks.
ACCEPT \( (6 \times 0.076) + (7 \times 0.924) \).
ALLOW ecf if use of 6 and 7 in calculation.
This question is about gases.
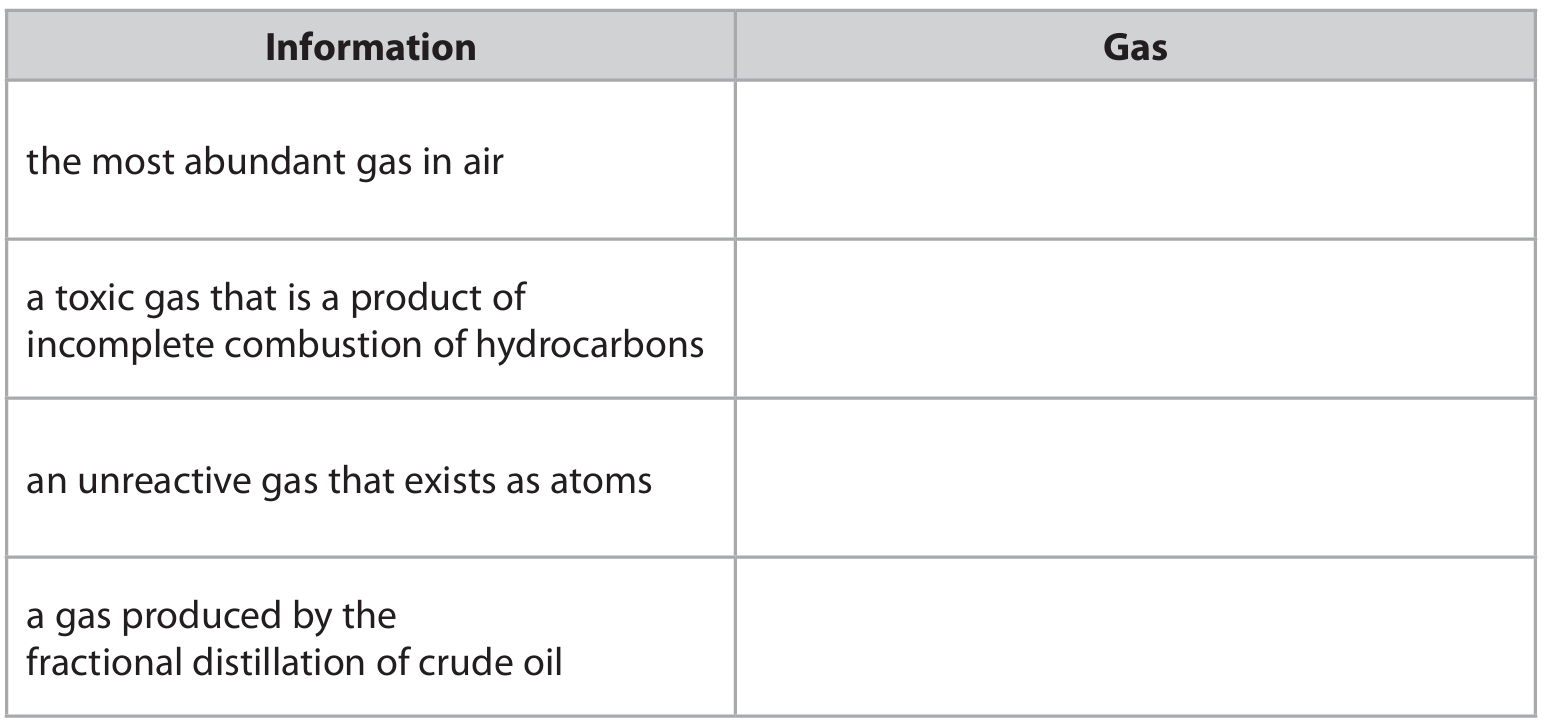
(a) The table gives information about some gases.
Complete the table by choosing a gas from the box that matches the information.

You may use each gas once, more than once, or not at all.
(b) A teacher uses this apparatus to determine the percentage of oxygen in a sample of air.
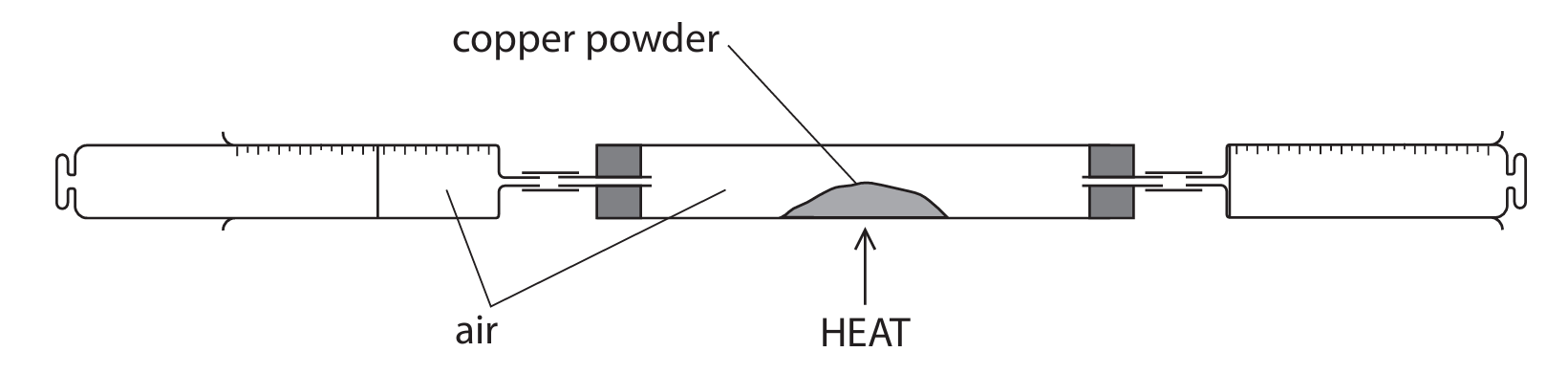
This is the teacher’s method.
- record the total volume of air in the apparatus
- heat the copper powder
- use the syringes to pass air over the heated copper powder several times
- allow the remaining gas to cool and record its volume
The copper powder turns black.
(i) State why the copper powder turns black.
(ii) Give a reason why the remaining gas is allowed to cool before its volume is recorded.
(iii) At the start of the experiment, the total volume of air in the apparatus is \(138 \, \text{cm}^3\).
At the end of the experiment, the volume of gas remaining is \(108 \, \text{cm}^3\).
Calculate the percentage of oxygen in the sample of air.
Assume that all the oxygen has reacted.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 4(b): Crude oil — part (a) last row
• 4(c)/(d): Alkanes / Alkenes — part (a) second row (combustion)
• 2(d): Reactivity series — part (b) reaction of copper with oxygen
• 1(e): Chemical formulae, equations and calculations — part (b)(iii) percentage calculation
▶️ Answer/Explanation
(a)

(b)(i) copper(II) oxide / CuO / copper oxide forms
(b)(ii) the volume of a gas changes with temperature / gas expands when hot / gas shrinks when cools (OWTTE)
(b)(iii)
M1: \(138 – 108 = 30 \, \text{cm}^3\) (volume of oxygen reacted)
M2: \(\frac{30}{138} \times 100 = 21.7\%\)
Answer: \(21.7\%\) (accept \(21.74\%\), \(22\%\))
This question is about iron.
(a) One problem with iron is that it rusts.
(i) Name the two substances that iron reacts with when it rusts.
(ii) State what type of reaction occurs when iron rusts.
(b) Iron can be prevented from rusting by painting or by coating with zinc.
(i) Explain how painting prevents iron from rusting.
(ii) Name the process used to coat iron with zinc.
(iii) Explain why a layer of zinc protects iron from rusting, even if the layer of zinc is scratched.
(c) Iron is formed when aluminium reacts with iron(III) oxide. This is the equation for the reaction.
\[2Al + Fe_2O_3 \rightarrow Al_2O_3 + 2Fe\]
(i) Explain what this reaction shows about the relative reactivities of aluminium and iron.
(ii) Explain which substance acts as an oxidising agent in this reaction.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 2(d): Oxidation and reduction — parts (a)(ii), (c)(ii)
• 2(d): Rusting and its prevention — parts (a)(i), (b)(i), (b)(ii), (b)(iii)
▶️ Answer/Explanation
(a)(i)
M1: water
M2: oxygen
Answers can be in either order. ALLOW moisture / H₂O; air / O₂.
(a)(ii)
oxidation
ALLOW oxidisation / oxidising / redox.
(b)(i)
M1: Paint acts as a barrier / layer.
M2: It prevents air / oxygen / water from getting to / reacting with the iron.
(b)(ii)
galvanising
ALLOW galvanisation. IGNORE sacrificial protection.
(b)(iii)
M1: Zinc is more reactive than iron.
M2: Therefore, zinc is oxidised / reacts with oxygen / loses electrons more readily / in preference to / instead of iron.
ALLOW corrodes instead of iron. REJECT zinc rusts.
(c)(i)
M1: Aluminium is more reactive than iron.
M2: Because aluminium displaces iron from iron(III) oxide.
(c)(ii)
M1: Iron(III) oxide (is the oxidising agent).
M2: Iron(III) oxide donates oxygen to aluminium / takes electrons from aluminium / causes aluminium to be oxidised.
ALLOW iron oxide / iron ions / Fe³⁺ ions.
A student uses this apparatus to investigate the rate of reaction when hydrogen peroxide solution decomposes.
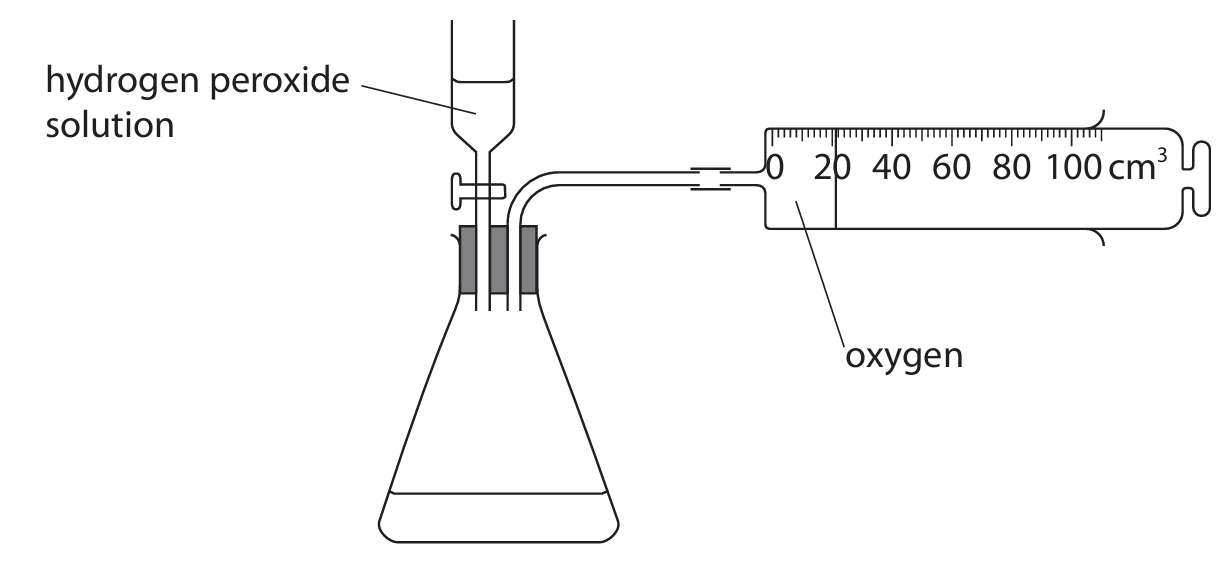
This is the equation for the reaction.
\[ 2H_2O_2 \rightarrow 2H_2O + O_2 \]
(a) Give a reason why the gas that collects in the gas syringe is not pure oxygen.
(b) The rate of reaction can be increased by adding a catalyst to the hydrogen peroxide solution.
Describe how a catalyst increases the rate of a reaction.
(c) These solids catalyse the decomposition of hydrogen peroxide solution.
- lead(IV) oxide
- manganese(IV) oxide
Describe a method that the student could use to find out which solid is the more effective catalyst.
(d) A student investigates the decomposition of a solution of hydrogen peroxide at different temperatures. The graph shows how the total volume of oxygen collected in the syringe changes with time when the solution is at a temperature of 20°C.
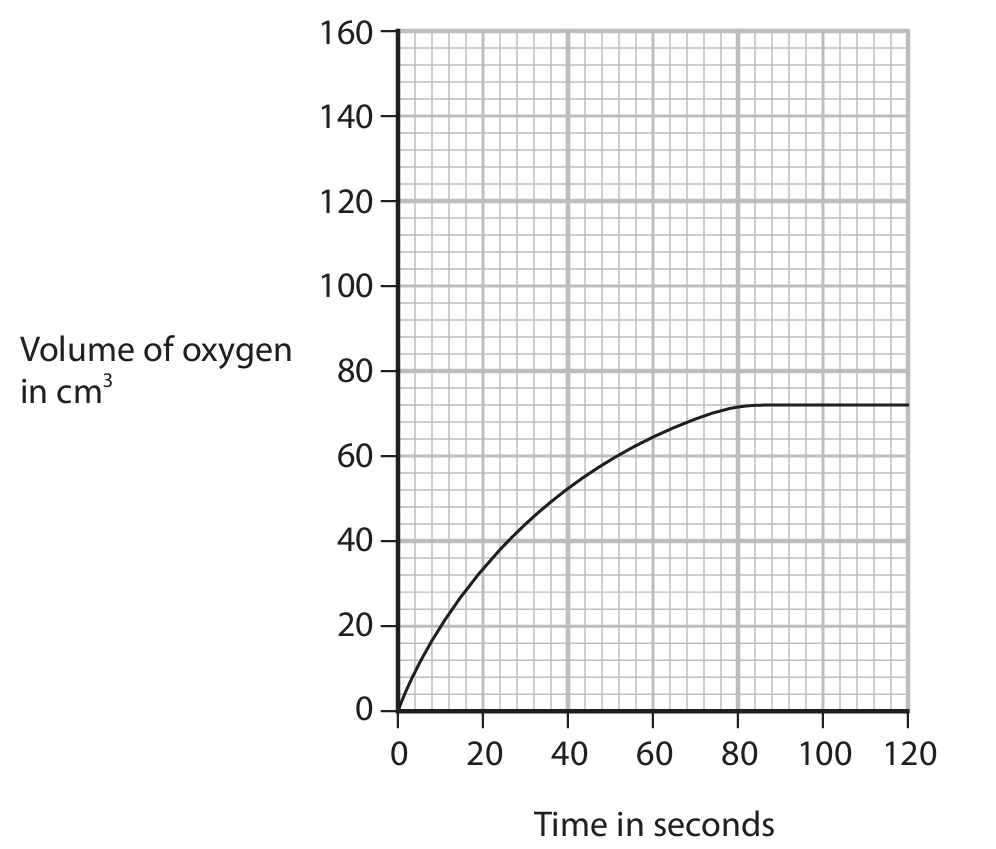
On the grid, draw the curve the student would obtain at a temperature of 40°C when all other conditions are kept the same.
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry 4CH1):
• 3(a): Energetics — part (b) relating to activation energy
• 2(f): Acids, alkalis and titrations / 2(g): Acids, bases and salt preparations — context of hydrogen peroxide decomposition
• Experimental skills & investigations — parts (c) and (d)
▶️ Answer/Explanation
(a) The gas also contains air (displaced from the conical flask).
(b)
- M1: A catalyst provides an alternative (reaction) pathway / route.
- M2: Of lower activation energy.
(c)
M1: Add hydrogen peroxide solution (to the conical flask) and add one of the catalysts.
M2: Record the time taken to collect a fixed volume of gas OR record the volume of gas collected in a fixed time.
AND any 2 from:
M3: Repeat with the same volume / same concentration of hydrogen peroxide solution.
M4: (Repeat at) same temperature.
M5: Use same mass / same surface area of each catalyst.
AND
M6: The most effective catalyst produces the greatest volume of gas per unit time OR takes the least time to produce a fixed volume of oxygen.
Allowable alternatives: “record the time when no more gas produced”, “same amount”, “the least time taken to complete the reaction is the most effective catalyst”.
(d)
M1: Steeper curve starting at the origin.
M2: Same final volume of oxygen produced.
The curve at 40°C should rise more steeply than the 20°C curve but plateau at the same maximum volume.
A teacher uses this apparatus to test the products of the combustion of liquid hydrocarbons.
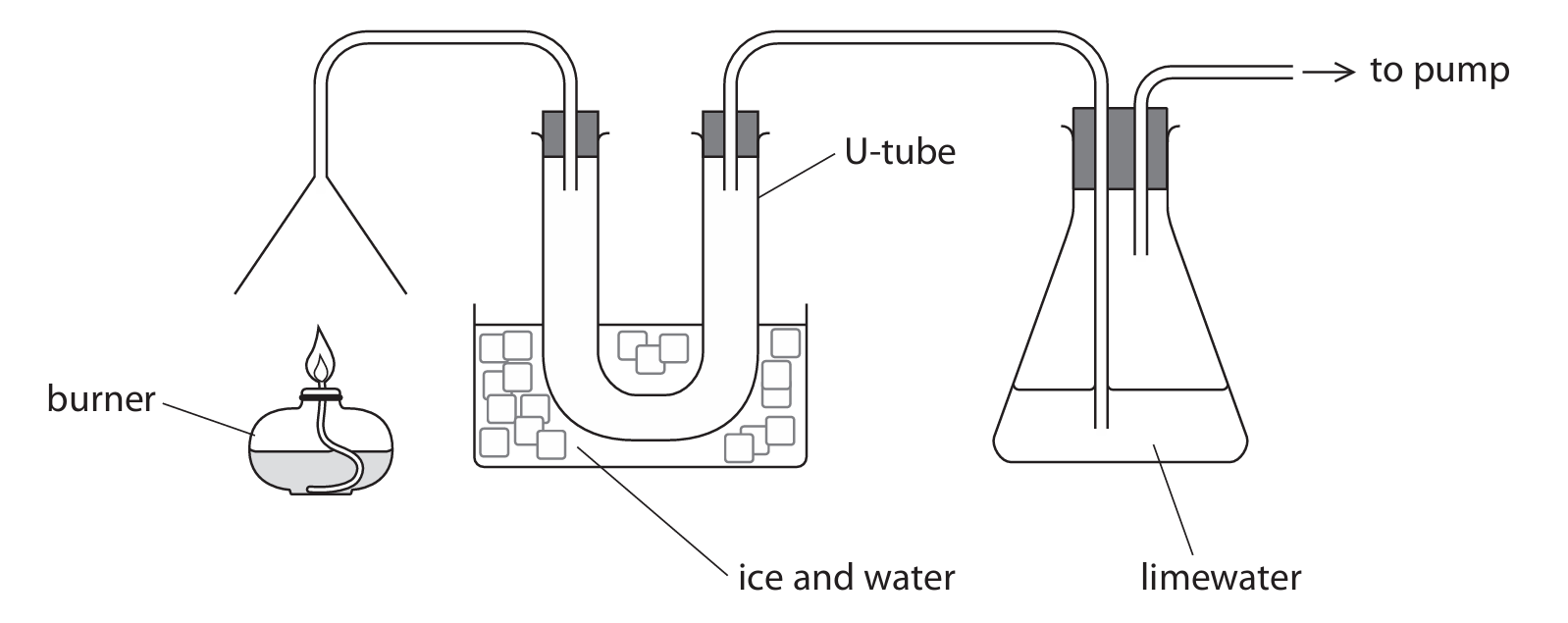
(a) Explain the change in appearance that occurs in the limewater.
(b) The equation represents the complete combustion of an alkene.
\[C_nH_{2n} + \text{oxygen} \rightarrow \text{carbon dioxide} + n\text{H}_2\text{O}\]
Complete combustion of 0.0100 mol of the alkene produces 2.16 g of water.
[for \( H_2O, M_r = 18 \)]
(i) Determine the molecular formula of this alkene.
(ii) Give a reason why the mass of pure water that collects in the U-tube is less than 2.16 g.
(iii) Give a physical test to show that the water that collects in the U-tube is pure.
(c) The teacher burns 30.0 g of heptane.
This is the equation for the complete combustion of heptane.
\[C_7H_{16} + 11O_2 \rightarrow 7CO_2 + 8H_2O\]
Calculate the minimum mass of oxygen needed for the complete combustion of 30.0 g of heptane.
[for \( C_7H_{16}, M_r = 100 \) for \( O_2, M_r = 32 \)]
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 4(b)(c): Crude oil & Alkenes — part (b)(i)
• 4(b): Crude oil – Combustion — parts (b)(ii), (b)(iii), (c)
• 1(e): Chemical formulae, equations and calculations — parts (b)(i), (c)
• 3(a): Energetics (exothermic reactions) — context of combustion
▶️ Answer/Explanation
(a)
M1: limewater turns cloudy / milky
M2: because carbon dioxide / CO₂ is produced / one of the products
M3: which reacts with limewater forming calcium carbonate
ALLOW: chalky / white precipitate; carbon dioxide is present; forming an insoluble product
(b)(i)
M1: amount of water = \(2.16 \div 18 = 0.12\) mol
M2: number of hydrogen atoms = \((0.12 \div 0.01) \times 2 = 24\)
M3: formula of alkene = \(C_{12}H_{24}\)
ALLOW: \(12H_2O\); Correct answer of \(C_{12}H_{24}\) scores 3; \(C_6H_{12}\) scores 2
(b)(ii)
Some steam / water vapour is lost (to the atmosphere) / does not condense.
IGNORE references to incomplete combustion
(b)(iii)
M1: Heat (the water) / measure the boiling point
M2: (If it) boils at \(100^\circ C\) (it is pure water) / boiling point is \(100^\circ C\)
ALLOW: find the freezing point / melting point; freezes / melts at \(0^\circ C\)
REJECT: evaporate
(c)
M1: (moles of heptane) = \(30 \div 100 = 0.30\) mol
M2: (moles of oxygen) = \(0.30 \times 11 = 3.3\) mol
M3: (mass of oxygen) = \(3.3 \times 32 = 106\) g
ALLOW: \(105.6\) g; answer of \(106\) g scores 3; answer of \(9.6\) g scores 2
Diamond, graphite and \( C_{60} \) fullerene are all forms of the element carbon.
The diagram shows the structures of these three substances.
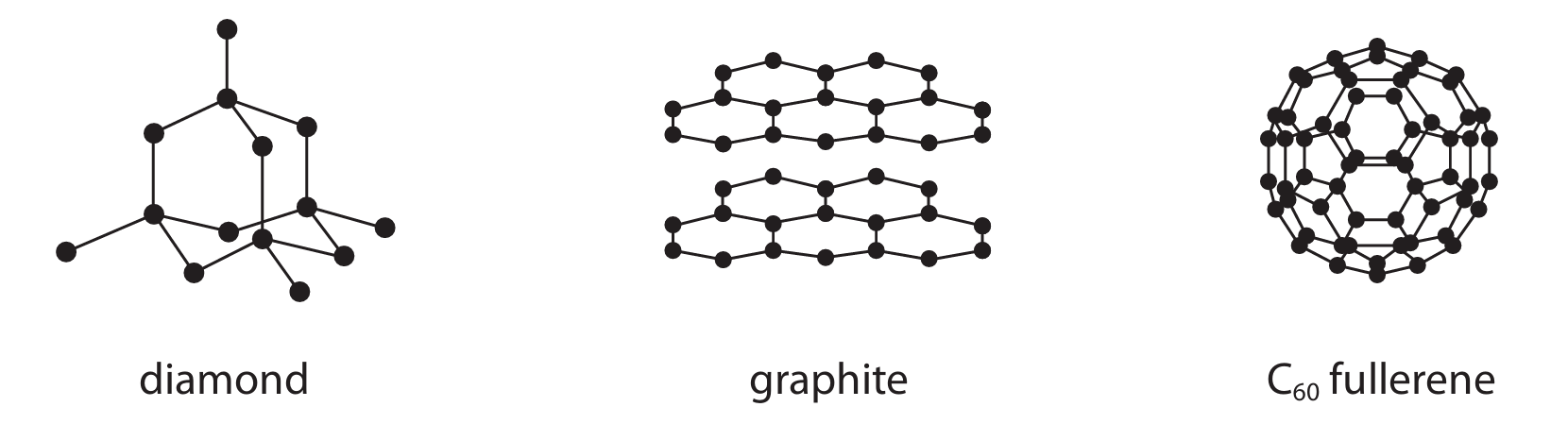
(a) Explain why graphite conducts electricity.
(b) Explain why diamond is hard but graphite is soft.
(c) Doctors use \( C_{60} \) fullerene to deliver medicines to certain parts of the body, so that the medicine does not damage other parts of the body. Suggest why \( C_{60} \) fullerene is suitable for this purpose.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
▶️ Answer/Explanation
(a)
M1: Graphite has delocalised electrons.
M2: These electrons are able to flow/move throughout the structure.
Note: Any mention of ions scores 0.
(b)
M1 (Diamond): Diamond is hard because it has a 3D/tetrahedral/rigid lattice where each carbon atom is bonded to four others by strong covalent bonds.
M2 (Graphite): Graphite is soft because it has a layered structure. The layers are held together by weak forces and can slide over one another.
Note: Reject references to intermolecular forces in diamond.
(c)
Any one from:
• The \( C_{60} \) molecule is inert/unreactive (with blood/medicine).
• The \( C_{60} \) molecule is non-toxic.
• The medicine can be enclosed/fit inside the \( C_{60} \) molecule’s cage-like structure.
This question is about ionic compounds.
(a) Calcium nitrate has the formula \(\text{Ca(NO}_3)_2\).
(i) Give the formula of each ion in calcium nitrate.
calcium ion:
nitrate ion:
(ii) Explain why calcium nitrate has a high melting point. Refer to structure and bonding in your answer. (4)
(iii) Calcium nitrate decomposes when heated. Complete the chemical equation for the decomposition of calcium nitrate. (1)
\(\qquad\)……\(\text{Ca(NO}_3)_2\) → ……\(\text{CaO}\) + ……\(\text{NO}_2\) + ……\(\text{O}_2\)
(b) A student has four unlabelled beakers, each containing a colourless solution of a different salt. These are the four salt solutions:
- calcium bromide
- calcium chloride
- sodium chloride
- sodium sulfate
Describe a series of tests to identify each solution.
Do not refer to safety precautions in your answer.
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry 4CH1):
• 1(f): Ionic bonding — part (a)(ii)
• 2(h): Chemical tests — part (b)
• 2(g): Acids, bases and salt preparations — ionic identification in part (b)
▶️ Answer/Explanation
(a)(i)
Calcium ion: \(\text{Ca}^{2+}\)
Nitrate ion: \(\text{NO}_3^-\)
(a)(ii)
• Calcium nitrate has a giant ionic lattice / structure.
• There are strong electrostatic forces / attractions.
• Between oppositely charged ions / cations and anions.
• A lot of energy is required to overcome these forces / break the bonds.
(a)(iii)
\(2\text{Ca(NO}_3)_2 \rightarrow 2\text{CaO} + 4\text{NO}_2 + \text{O}_2\)
(b)
• Perform a flame test on each solution.
– Sodium chloride and sodium sulfate give a yellow flame (sodium ions).
– Calcium chloride and calcium bromide give an orange-red / brick-red flame (calcium ions).
• To distinguish between the sodium salts and the calcium salts:
– Add acidified silver nitrate (or nitric acid followed by silver nitrate) to each solution.
– Calcium bromide gives a cream precipitate (bromide ions).
– Calcium chloride and sodium chloride give a white precipitate (chloride ions).
• To distinguish between sodium sulfate and sodium chloride (or confirm sulfate):
– Add acidified barium chloride (or barium nitrate) to the solutions.
– Sodium sulfate gives a white precipitate (sulfate ions).
This question is about propene.
(a) (i) The structural formula of propene is \( \text{CH}_3\text{CH} = \text{CH}_2 \).
Complete the dot-and-cross diagram for a molecule of propene.
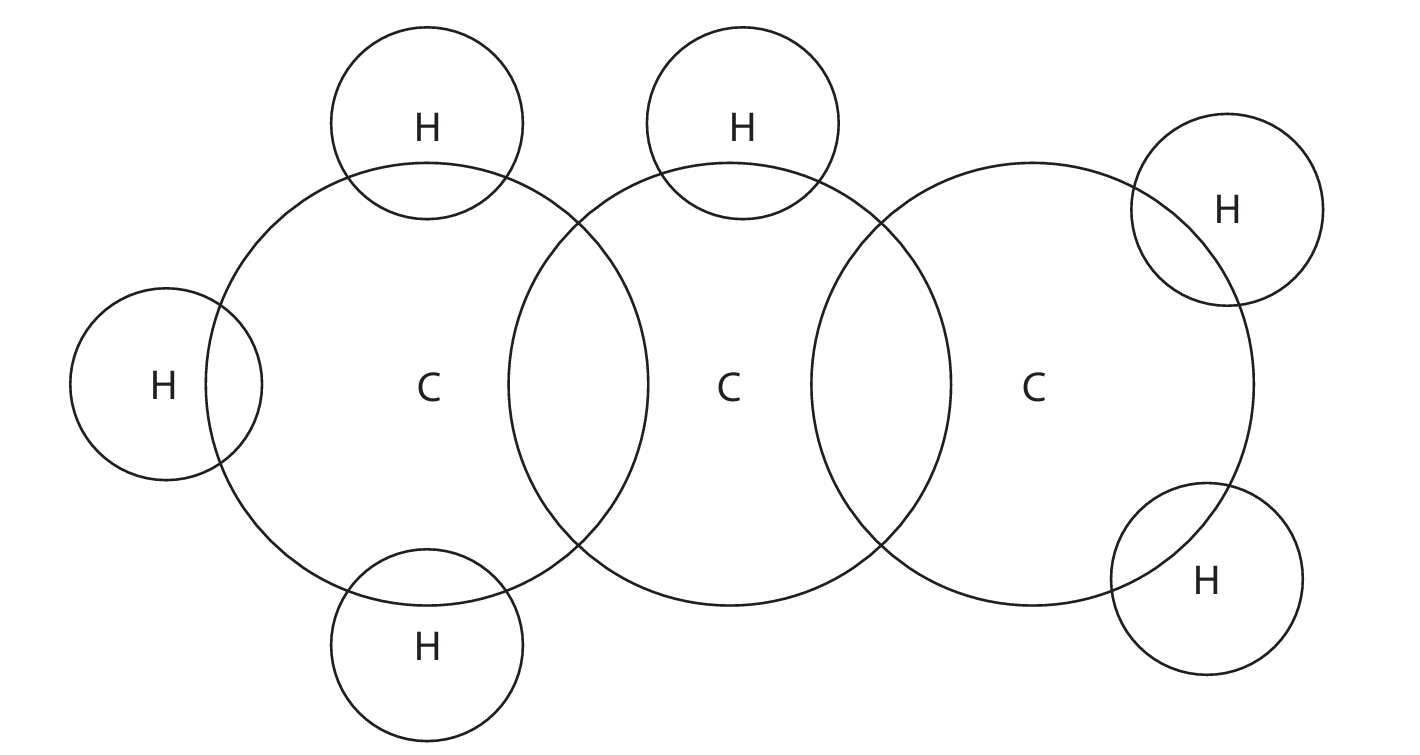
(a) (ii) Describe the forces of attraction that hold the atoms together in a molecule of propene.
(b) The alkane pentadecane has the formula \( C_{15}H_{32} \).
Describe how propene can be produced from pentadecane.
(c) Propene is used to make poly(propylene).
(i) Complete the equation for the formation of poly(propylene).

(ii) Explain why the disposal of poly(propylene) in landfill sites is a problem.
(d) Propene can be converted into compound X.
(i) Compound X contains these percentages by mass:
- carbon 60%
- hydrogen 13.3%
- oxygen 26.7%
Compound X has an \( M_r \) of 60.
Determine the molecular formula of compound X.
(ii) Propene and compound X both have simple molecular structures.
Explain why compound X has a higher boiling point than propene.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 4(b/d): Crude oil / Alkenes — part (b) (cracking)
• 4(h): Synthetic polymers — parts (c)(i), (c)(ii)
• 1(e): Chemical formulae, equations and calculations — part (d)(i)
• 1(g): Covalent bonding (intermolecular forces) — part (d)(ii)
▶️ Answer/Explanation
(a)(i)
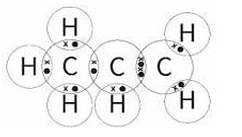
M1: Four electrons (two shared pairs) between carbon 2 and carbon 3 (the double bond) and two electrons (one shared pair) between carbon 1 and carbon 2.
M2: Two electrons (one shared pair) between each carbon and its bonded hydrogen atoms.
ACCEPT any combination of dots and crosses. Max 1 mark if any extra non-bonding electrons are added.
(a)(ii)
M1: Shared pair(s) of electrons.
M2: (Which are) attracted to the (two) nuclei.
OR
M1: (Two) nuclei.
M2: Attracted to the shared pair(s) of electrons.
REJECT singular “nucleus”. Must be plural for the mark.
(b)
Any two from:
M1: Cracking.
M2: Heat to a temperature of 600–700°C.
M3: With a catalyst of silica/alumina (or aluminosilicates/zeolites).
ALLOW a correct cracking equation including propene.
(c)(i)
M1: Correct repeat unit:
M2: Extension bonds, brackets, and ‘n’ subscript to the right.
ALLOW correct repeat unit with or without extension bonds.
(c)(ii)
M1: Poly(propylene) remains for a long time in landfill / takes up a lot of space.
M2: Because it is inert / unreactive / non-biodegradable / does not decompose.
M2 is dependent on M1.
(d)(i)
M1: Calculate moles of each element:
\( \text{C: } \frac{60}{12} = 5 \) \( \text{H: } \frac{13.3}{1} = 13.3 \) \( \text{O: } \frac{26.7}{16} = 1.66875 \)
M2: Divide by smallest (1.66875) to get ratio:
\( \text{C: } \frac{5}{1.66875} \approx 3 \) \( \text{H: } \frac{13.3}{1.66875} \approx 8 \) \( \text{O: } \frac{1.66875}{1.66875} = 1 \)
Empirical formula = \( C_3H_8O \).
M3: \( M_r \text{ of } C_3H_8O = (3 \times 12) + (8 \times 1) + 16 = 60 \). This matches the given \( M_r \), so molecular formula is \( C_3H_8O \).
ACCEPT alternative correct methods. No marks for using atomic numbers.
(d)(ii)
M1: The intermolecular forces (between molecules of compound X) are stronger than those between propene molecules.
AND any one from:
M2: So more energy is required to overcome them.
M3: Because compound X has a higher \( M_r \) / larger surface area.
M4: Because compound X has more electrons (leading to stronger London/dispersion forces).
M2, M3, M4 are dependent on M1.
A student uses this method to investigate the temperature change when solid sodium hydrogencarbonate is added to ethanoic acid solution.
- pour 100 cm\(^3\) of ethanoic acid solution into a polystyrene cup
- record the temperature of the ethanoic acid solution
- add a 1 g portion of sodium hydrogencarbonate to the ethanoic acid solution and stir
- record the new temperature
- add further 1 g portions of sodium hydrogencarbonate, stirring and recording the temperature after each portion is added
The table shows the student’s results.
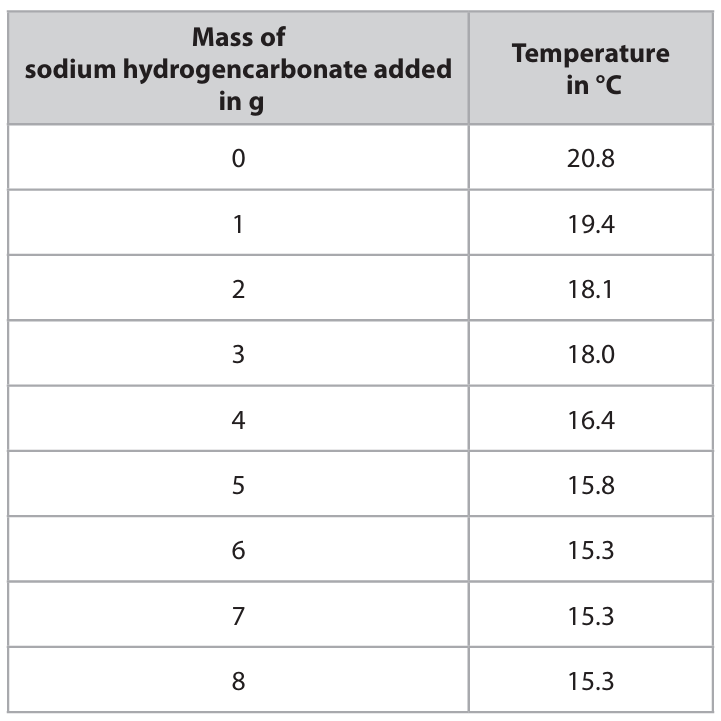
(a) (i) Plot the student’s results on the grid.
(ii) Draw a circle around the anomalous result.
(iii) Draw a curve of best fit.
(b) (i) Explain why using a polystyrene cup makes the results more accurate.
(ii) Suggest a mistake the student might have made to cause the anomalous result.
(iii) State how the results show the reaction is complete.
(iv) State how the results show that the reaction is endothermic.
(c) Use the results to calculate the heat energy change, \( Q \), in joules.
[for 1.0 cm\(^3\) of ethanoic acid solution, mass = 1.0 g]
[for ethanoic acid solution, \( c = 4.2 \, \text{J/g/}^\circ\text{C} \)]
(d) The student repeats the experiment starting with a different volume of ethanoic acid solution.
The student uses 7.0 g of sodium hydrogencarbonate to neutralise the ethanoic acid solution.
The heat energy change, \( Q \), is 3200 J.
Calculate the molar enthalpy change, \( \Delta H \), in kJ/mol.
Include a sign with your answer.
[for sodium hydrogencarbonate, \( M_r = 84 \)]
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry 4CH1):
• 2(g) Acids, bases and salt preparations: Reaction of an acid with a carbonate (context of the experiment)
• Appendix 4: Mathematical skills: Arithmetic computation, handling data, algebra, and substitution (parts c, d)
• Appendix 5: Command word taxonomy: Explain, suggest, state, calculate, use (applied throughout)
• Practical Investigation Skills: Identifying anomalies, improving accuracy, interpreting graphical data (parts a, b-i, b-ii, b-iii)
▶️ Answer/Explanation
(a)
(i), (ii), (iii):
- All 9 points plotted correctly (± half a square).
- Anomalous result circled: the point at mass = 3 g (Temperature = 18.0 °C).
- A smooth curve of best fit is drawn, passing close to all points except the anomalous one.
(b)
(i) Polystyrene is a good insulator / poor conductor of heat. It reduces heat transfer (gain or loss) between the reaction mixture and the surroundings, leading to more accurate temperature measurements.
(ii) The student might have forgotten to stir the mixture properly / took the temperature reading too soon before it stabilized.
(iii) The temperature remains constant (at 15.3 °C) after adding the last few portions of sodium hydrogencarbonate (6g, 7g, 8g).
(iv) The temperature of the mixture decreases, showing that thermal energy is taken in from the surroundings (an endothermic process).
(c)
\[ \Delta T = 20.8 – 15.3 = 5.5 \, ^\circ\text{C} \] \[ m = 100 \, \text{g} \quad (\text{since volume} = 100 \, \text{cm}^3 \text{ and density} \approx 1 \, \text{g/cm}^3) \] \[ Q = m \times c \times \Delta T = 100 \times 4.2 \times 5.5 = 2310 \, \text{J} \] Answer: \( Q = 2310 \, \text{J} \)
(d)
\[ \text{Moles of NaHCO}_3 = \frac{7.0}{84} = 0.0833 \, \text{mol} \] \[ \Delta H = \frac{Q}{\text{moles}} = \frac{3200 \, \text{J}}{0.0833 \, \text{mol}} = 38400 \, \text{J/mol} = 38.4 \, \text{kJ/mol} \] Since the reaction is endothermic (temperature decreased), \( \Delta H \) is positive.
Answer: \( \Delta H = +38.4 \, \text{kJ/mol} \)
