This question is about atomic structure.
(a) The table shows the number of protons, neutrons and electrons in five species, V, W, X, Y and Z. The letters represent the species but are not symbols from the Periodic Table.
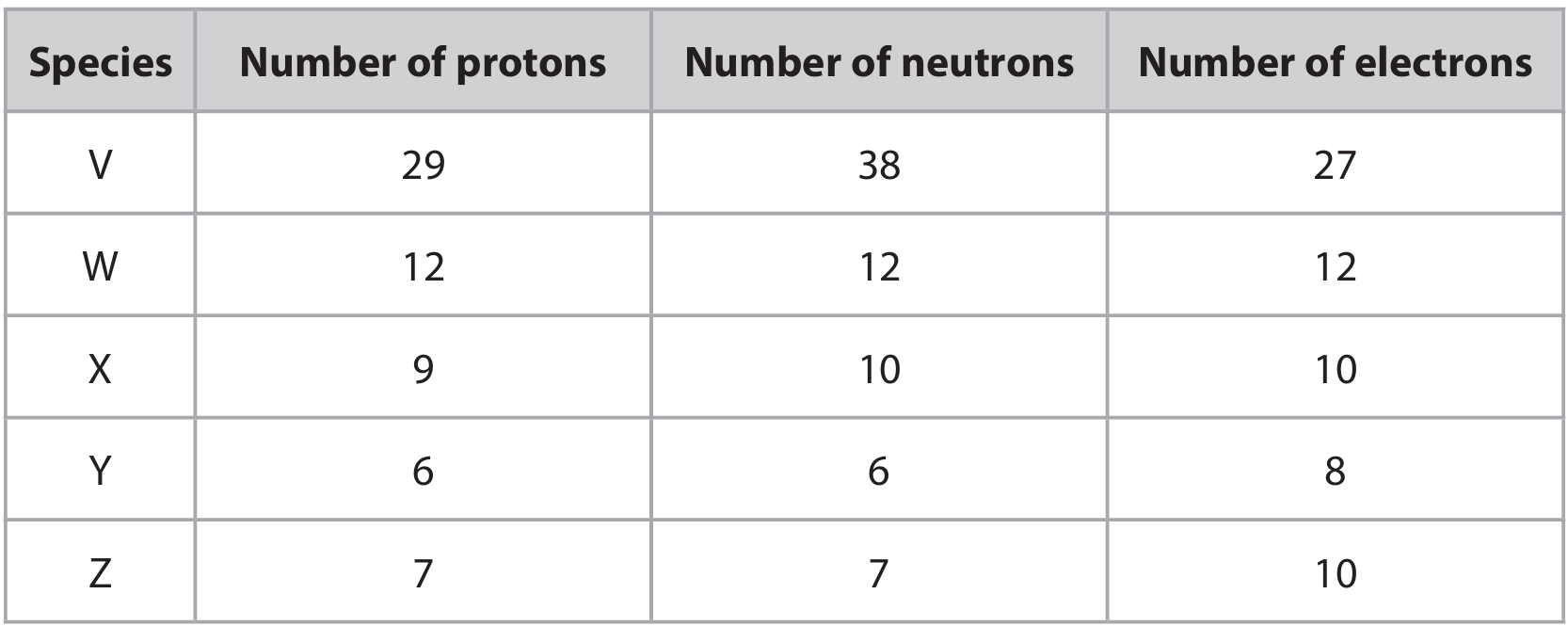
Choose letters from the table to answer these questions. Each letter may be used once, more than once or not at all.
(i) Which species is an atom?
(ii) Which species is an ion with a positive charge?
(iii) Which species is an ion with a \(3-\) charge?
(b) (i) State what is meant by the term atomic number.
(ii) State what is meant by the term mass number.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(d): The Periodic Table (basis of arrangement) — linked to proton number
▶️ Answer/Explanation
(a)(i) W
An atom has equal numbers of protons and electrons. Only W satisfies this (12 protons, 12 electrons).
(a)(ii) V
A positive ion (cation) has fewer electrons than protons. V has 29 protons and 27 electrons, giving a charge of \(2+\).
(a)(iii) Z
A \(3-\) ion has three more electrons than protons. Z has 7 protons and 10 electrons, giving a charge of \(3-\).
(b)(i) The number of protons in the nucleus (of an atom).
(b)(ii) The sum of the number of protons and neutrons in the nucleus (of an atom).
This question is about methane, \(\text{CH}_4\). The diagram shows a Bunsen burner that uses methane.
(a) During combustion, methane reacts with a gas in the air. Give the name of this gas.
(b) Give the two products of the complete combustion of methane.
(c) During the incomplete combustion of methane, carbon monoxide forms.
(i) Give a reason why carbon monoxide forms during incomplete combustion.
(ii) State why carbon monoxide is poisonous.
(d) The equation shows the reaction of methane with bromine.
\[ \text{CH}_4 + \text{Br}_2 \rightarrow \text{CH}_3\text{Br} + \text{HBr} \]
Give the name of this type of chemical reaction.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 4(b): Crude oil / Fuels / Combustion — parts (a), (b), (c) – combustion of hydrocarbons
▶️ Answer/Explanation
(a) oxygen
ALLOW \(\text{O}_2\)
(b)
M1: carbon dioxide / \(\text{CO}_2\)
M2: water / \(\text{H}_2\text{O}\)
ACCEPT water vapour, steam. Answers can be in either order.
(c)(i) There is a limited supply of air / oxygen. (OWTTE)
(c)(ii) Carbon monoxide reduces the capacity of blood to transport oxygen. (OWTTE)
ACCEPT correct references to haemoglobin / produces carboxyhaemoglobin.
(d) substitution
ALLOW redox (reaction).
This question is about elements, mixtures and compounds.
(a) The box gives some methods used to separate mixtures.

Choose methods from the box to answer these questions.
(i) Identify a method to remove sand from a mixture of sand and seawater.
(ii) Identify a method to separate a mixture of liquids with different boiling points.
(b) The diagram shows part of the structure of silicon dioxide.
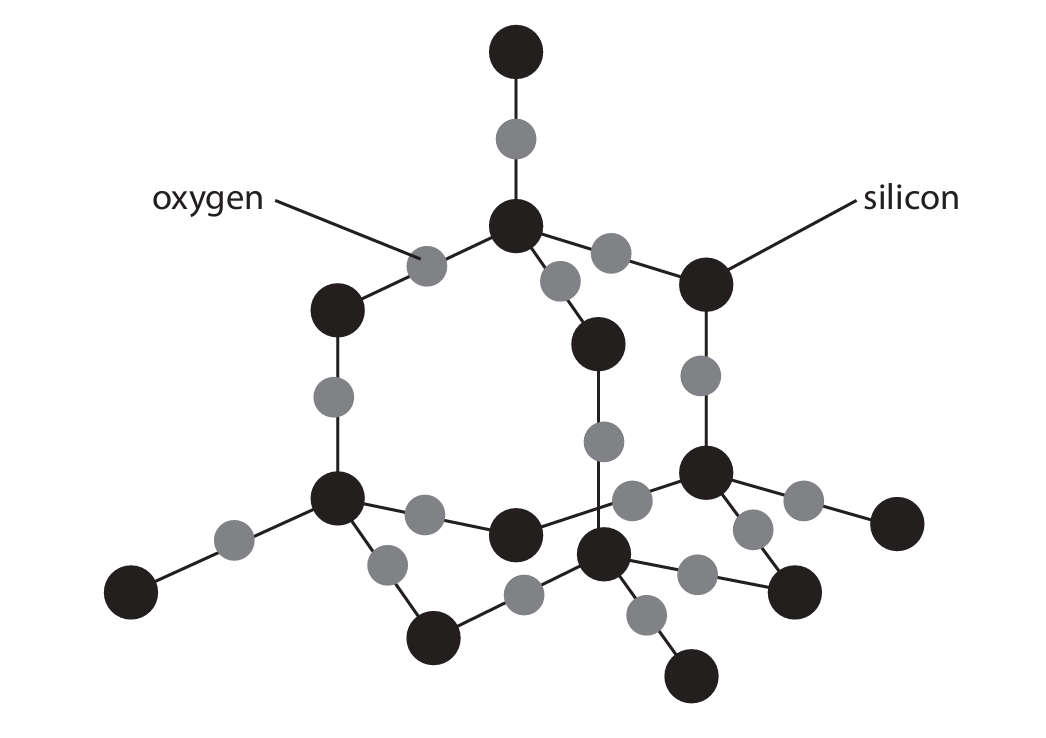
Explain why silicon dioxide is a compound.
(c) The molecular formula of the compound insulin is \( C_{257}H_{383}N_{65}O_{77}S_6 \).
(i) Determine the number of different elements in \( C_{257}H_{383}N_{65}O_{77}S_6 \).
(ii) Determine the number of atoms in a molecule of \( C_{257}H_{383}N_{65}O_{77}S_6 \).
(d) Magnalium is a mixture of magnesium atoms and aluminium atoms. The diagram shows a sample of magnalium.
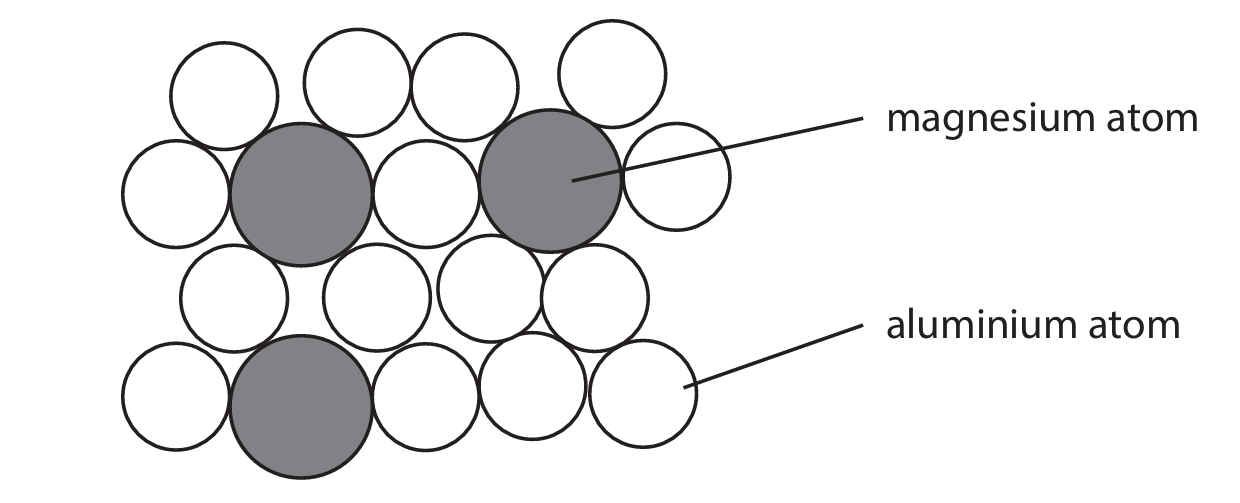
Calculate the percentage of magnesium atoms in this sample.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(e): Chemical formulae, equations and calculations — parts (c), (d)
• 1(b): States of matter / Separation techniques — part (a)
▶️ Answer/Explanation
(a)
| (i) | filtration |
| (ii) | fractional distillation |
(b) An explanation that makes reference to the following:
- It is made up of two different types of atom/element (silicon and oxygen).
- The atoms are chemically bonded/joined together.
(c)
| (i) | 5 |
| (ii) | 788 (Calculation: 257 + 383 + 65 + 77 + 6 = 788) |
(d)
Number of magnesium atoms = 3
Total number of atoms = 15
Percentage of magnesium atoms = \( \left( \frac{3}{15} \right) \times 100 = 20\% \)
Note: The mark scheme from the image indicates (3 ÷ 18) × 100 = 16.7%, but based on the provided diagram (Screenshot 2026-01-21 125211.png) which shows 15 total atoms (3 Mg + 12 Al), the correct calculation is 20%. The discrepancy is likely due to a different sample diagram in the original question.
This question is about the alkali metals.
A teacher demonstrates the reaction between sodium and water.
The teacher fills a trough with water and then adds a piece of sodium.
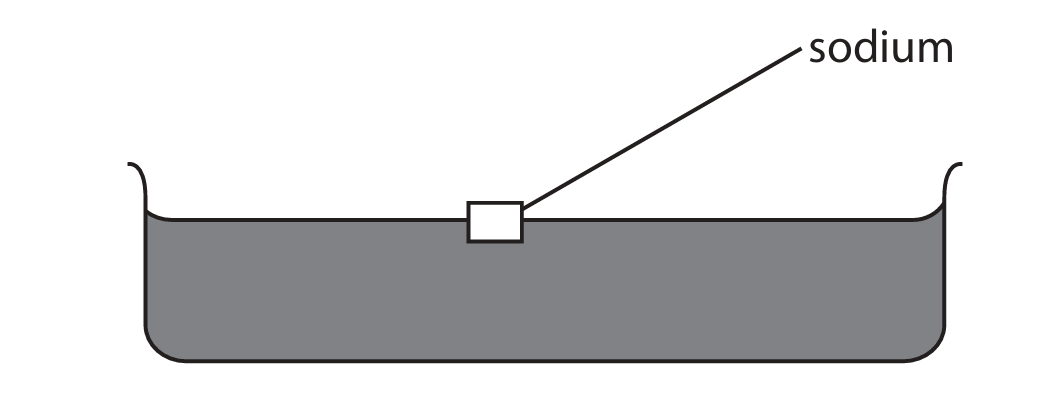
(a) The sodium reacts with the water, forming bubbles of hydrogen gas and a colourless solution.
State two other observations that would be made.
(b) Give a test to show that, at the end of the reaction, the solution contains sodium ions.
(c) Lithium, sodium and potassium react in a similar way when added to water.
(i) State, with reference to the electronic configurations of atoms, why these elements have similar reactions.
(ii) The table shows the atomic radius of a lithium atom, a sodium atom and a potassium atom.
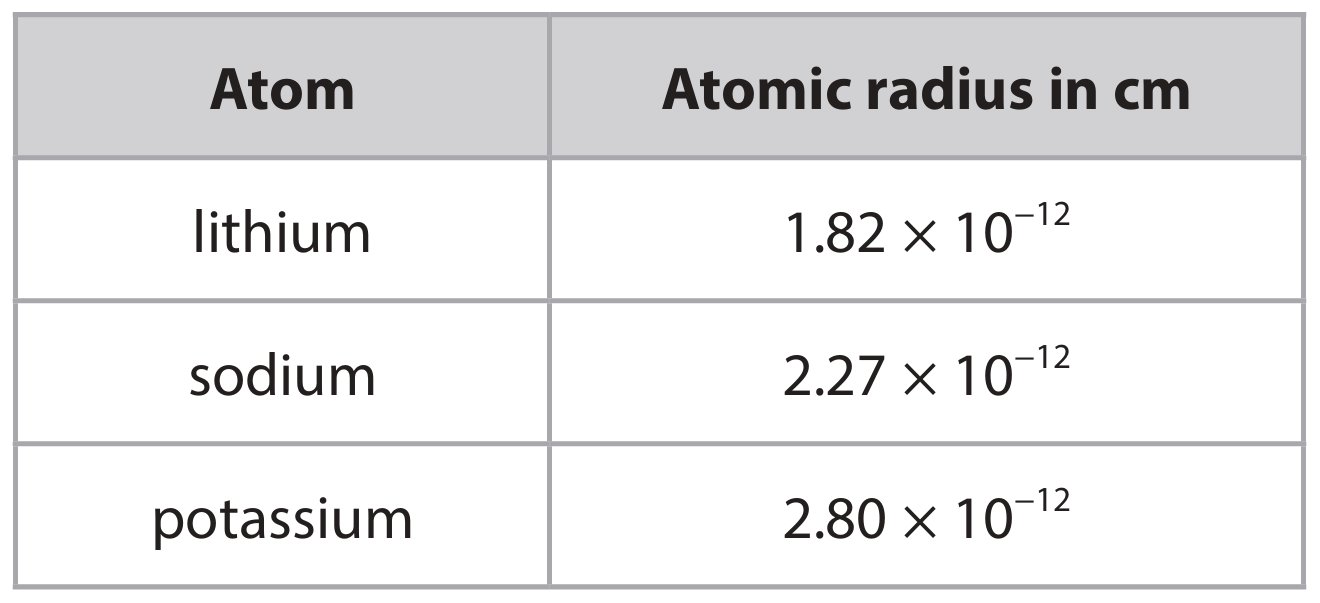
Deduce the relationship between the atomic radius and the reactivity of the metals.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(d): The Periodic Table — parts (c)(i), (c)(ii)
• 2(d): Reactivity series — part (c)(ii)
• 2(h): Chemical tests — part (b)
▶️ Answer/Explanation
(a) Any two from:
• (The sodium) floats
• (The sodium) moves (on the surface)
• (The sodium) melts / turns into a ball / sphere
• (The sodium) gets smaller / disappears
• (There is a) white trail
(b)
• Do a flame test (on the solution) (1)
• (A) yellow flame (is observed) (1)
Allow orange flame.
(c) (i)
• (Because they all have) the same number of (electrons in their) outer shell / one outer shell electron (1).
(c) (ii)
• As the atomic radius increases, the reactivity increases / the larger the atomic radius, the more reactive the metal (1).
5 Chromatography is used to separate the components in a mixture.
(a) Diagram 1 shows the apparatus used to separate the different dyes in a food colouring.
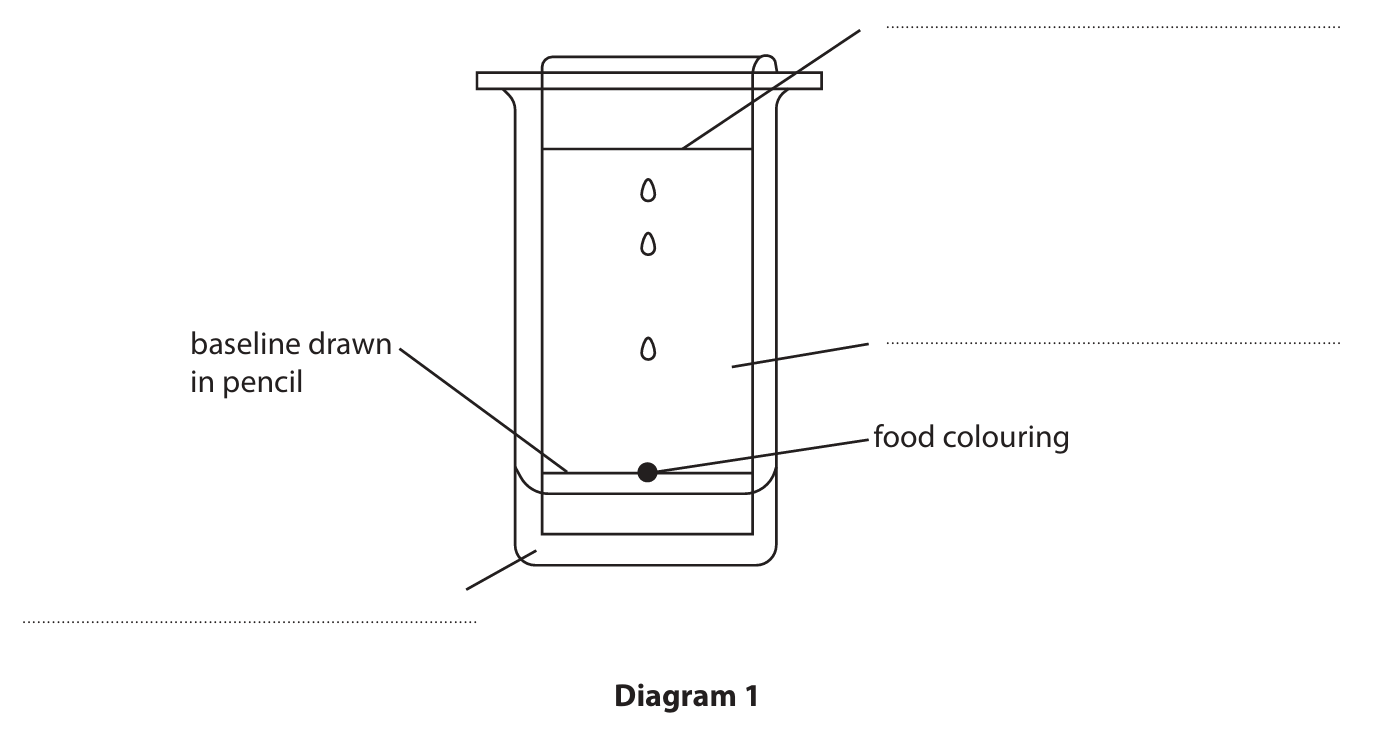
(i) Complete the diagram by adding the missing labels.
(ii) Give a reason why the baseline is drawn in pencil.
(b) Diagram 2 shows a chromatogram produced from four different food colourings, W, X, Y and Z.
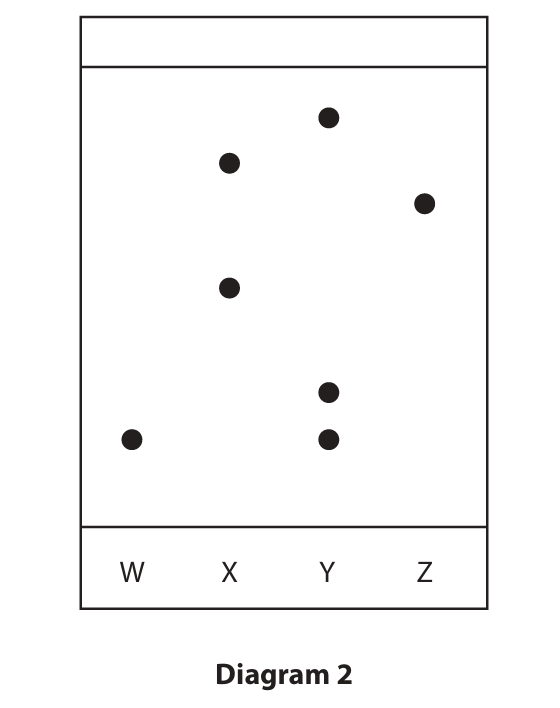
(i) Which two food colourings contain the same dye?
- A) W and X
- B) W and Y
- C) X and Z
- D) Y and Z
(ii) Calculate the \( R_f \) value of the dye in food colouring W.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(b) States of matter: Chromatography — parts (a)(i), (a)(ii), (b)(i), (b)(ii)
▶️ Answer/Explanation
(a)(i) Labels (clockwise from bottom left):
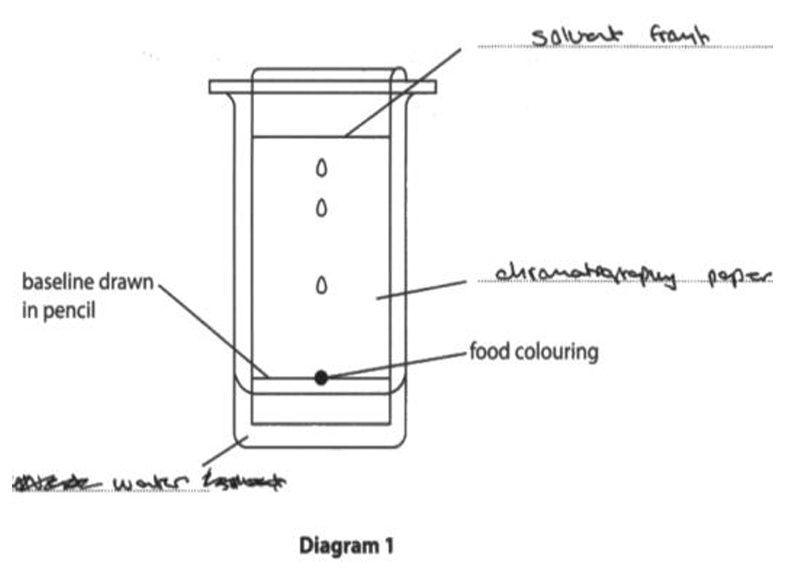
M1: solvent / water
M2: solvent front
M3: chromatography paper / paper / chromatogram
Marking Guidance (from image): ALLOW water, ALLOW paper, ALLOW chromatogram.
(a)(ii) Pencil is not soluble / insoluble.
Marking Guidance: ACCEPT pencil will not dissolve. ALLOW pencil will not run (up the chromatogram).
(b)(i) B (W and Y)
Marking Guidance: A is not the correct answer because W and X do not have a spot at the same height. C is not the correct answer because X and Z do not have a spot at the same height. D is not the correct answer because Y and Z do not have a spot at the same height.
(b)(ii)
M1: Distance moved by the dye = 1.1 to 1.4 cm (e.g., 1.25 cm). Distance moved by the solvent = 6.5 cm.
M2: \( R_f = \frac{\text{distance moved by dye}}{\text{distance moved by solvent}} = \frac{1.25}{6.5} \approx 0.19 \).
Marking Guidance: ALLOW any number of significant figures as long as it is correctly rounded. ALLOW ECF (error carried forward) from M1.
Example acceptable answers: 0.17, 0.18, 0.20, 0.22.
A student uses this apparatus to find the heat energy released by the combustion of liquid fuels.
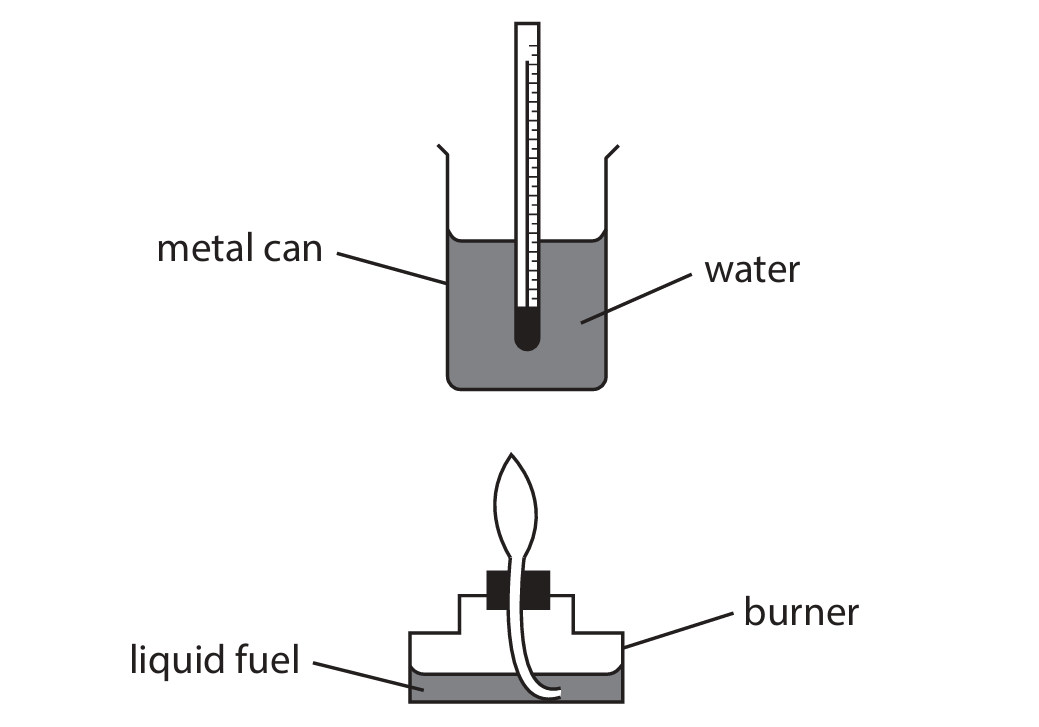
(a) Explain what is meant by the term fuel.
(b) (i) In one experiment, the student uses liquid ethanol as the fuel. The thermometer shows the temperature of the water at the start of the experiment.
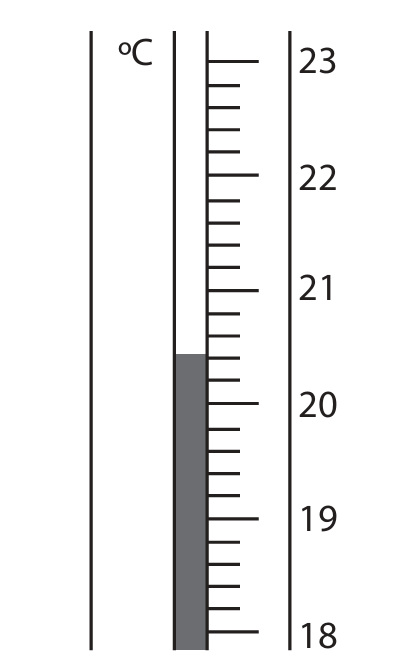
Complete the table by giving the temperatures to the nearest 0.1 °C.

(ii) The metal can contains water of mass 150 g. Show, by calculation, that the heat energy change (\(Q\)) for this reaction is approximately 36 000 J. (For water, \(c = 4.2 \, \text{J g}^{-1} \text{°C}^{-1}\))
(iii) In the experiment, 2.3 g of ethanol (\(M_r = 46\)) is burned. Calculate the molar enthalpy change (\(\Delta H\)), in \(\text{kJ mol}^{-1}\), for the combustion of ethanol, \(\text{C}_2\text{H}_5\text{OH}\). Include a sign in your answer. Give your answer to two significant figures.
(c) In this experiment, the calculated value of \(\Delta H\) is less than the value given in a data book. Give a possible reason for the difference in values.
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry 4CH1):
• 4(e): Alcohols — part (b)(iii) context (ethanol combustion)
• 1(e): Chemical formulae, equations and calculations — part (b)(iii) (mole calculation)
• Experimental Skills — parts (b)(i), (c) (practical considerations, heat loss)
▶️ Answer/Explanation
(a)
M1: A (substance/fuel that) when burned / undergoes combustion.
M2: Releases heat (energy) / thermal energy.
(b) (i)
Temperature at the start = \(20.4 \, \text{°C}\) (reading from the thermometer scale).
Highest temperature reached = \(20.4 + 57.2 = 77.6 \, \text{°C}\).
(b) (ii)
Using \(Q = mc\Delta T\):
\(Q = 150 \times 4.2 \times 57.2\)
\(Q = 36036 \, \text{J} \approx 36000 \, \text{J} \) (to 2 significant figures).
(b) (iii)
Amount of ethanol = \(\frac{2.3}{46} = 0.05 \, \text{mol}\).
Heat energy change in kJ = \(\frac{36036}{1000} = 36.036 \, \text{kJ}\).
Molar enthalpy change, \(\Delta H = \frac{36.036}{0.05} = 720.72 \, \text{kJ mol}^{-1}\).
Since combustion is exothermic: \(\Delta H = -720 \, \text{kJ mol}^{-1}\) (to 2 significant figures).
(c)
Any one from:
• Heat absorbed by the metal can.
• Incomplete combustion (of ethanol).
• Heat lost to the surroundings / heat loss.
A student uses this apparatus to investigate the rate of reaction between dilute sulfuric acid and an excess of small pieces of zinc.
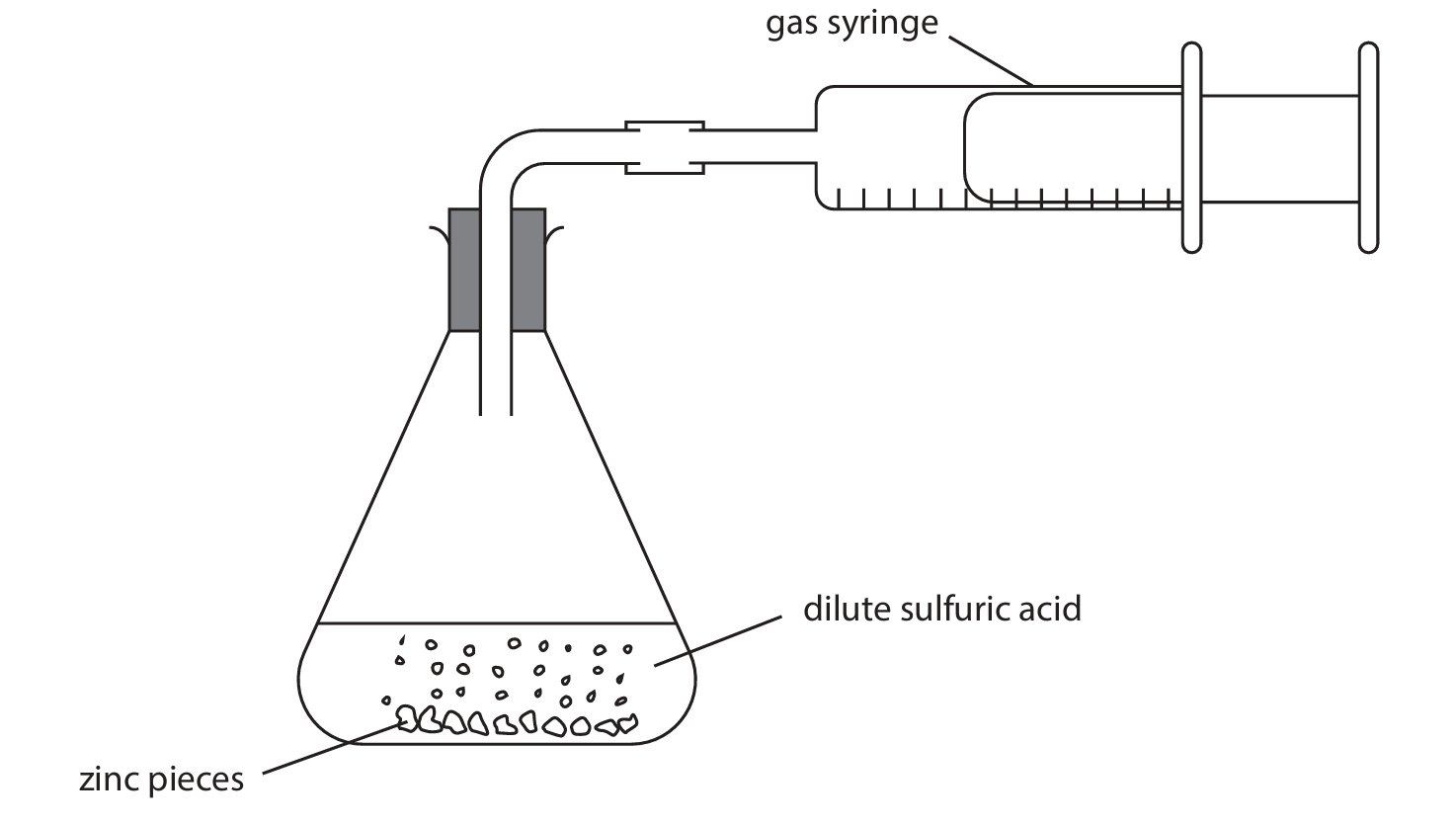
This is the student’s method.
- Step 1: use 50 cm\(^3\) of dilute sulfuric acid
- Step 2: add approximately 5 g of small zinc pieces to the sulfuric acid
- Step 3: quickly connect the gas syringe
- Step 4: record the reading on the gas syringe every 30 seconds until the reaction stops
(a) (i) Name a suitable piece of apparatus to measure the volume of sulfuric acid.
(ii) Give a reason why the mass of zinc pieces does not need to be measured accurately.
(iii) Give a reason why the student quickly connects the gas syringe in step 3.
(iv) State how the student would know when the reaction stops.
(b) The graph shows the volume of gas collected in the syringe during the experiment.
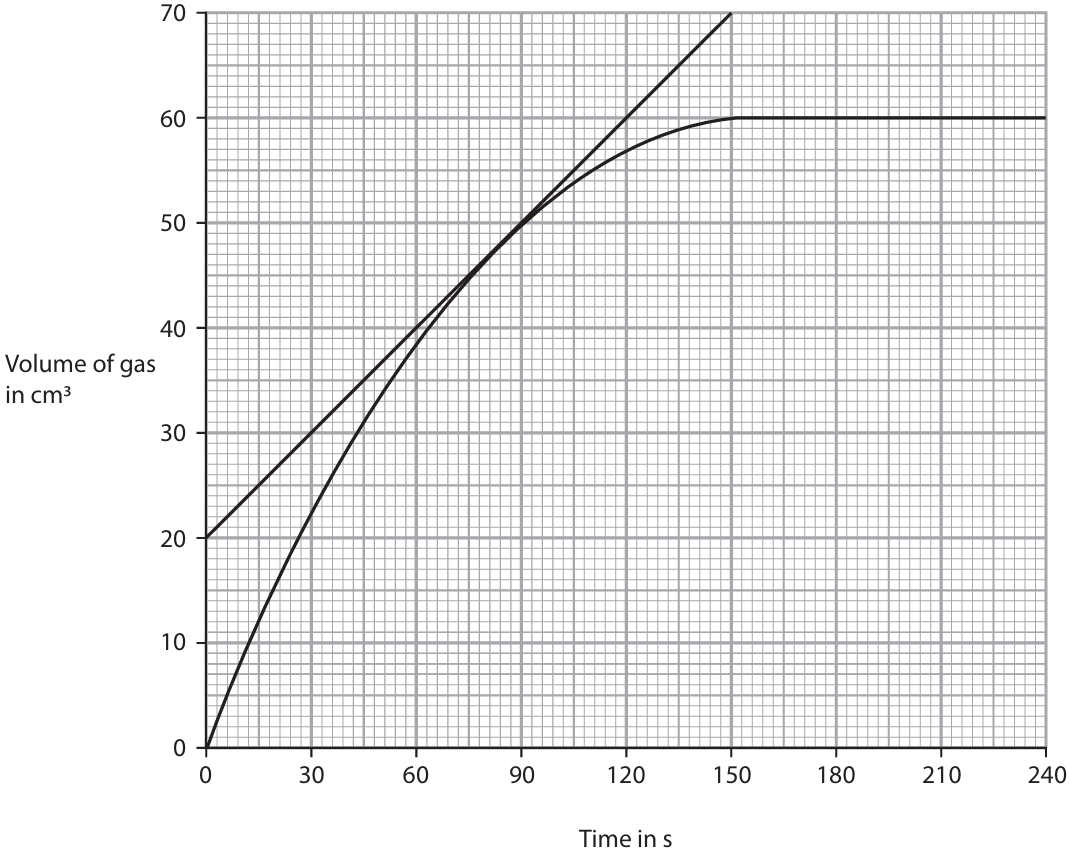
(i) A tangent to the curve has been drawn at a time of 80 s.
Use the tangent to calculate the rate of reaction at 80 s.
Show your working on the graph.
Give the unit.
(ii) Explain the shape of the graph in these regions.
• from 0 s to 60 s
• from 60 s to 150 s
• from 150 s to 240 s
Most-appropriate topic codes (Pearson Edexcel International GCSE Chemistry (4CH1)):
• 3(b) Physical Chemistry / Rates of reaction — 3.9–3.11: Factors affecting rate; 3.15: Practical investigation; Graph interpretation and tangent calculation
• 3(a) Physical Chemistry / Energetics — 3.3: Calculation of energy change \( Q = mc\Delta T \) (mathematical skill applied)
• Appendix 4: Mathematical skills — Graph plotting, gradient calculation, units
• Appendix 5: Command word taxonomy — “Name”, “Give a reason”, “State”, “Calculate”, “Explain”
• Experimental skills (Section 3 & Appendix 6) — Practical method, apparatus selection, data collection, evaluating experimental design
▶️ Answer/Explanation
(a)(i) Measuring cylinder
ALLOW burette / pipette / syringe. REJECT gas syringe.
(a)(ii) The zinc is in excess.
ACCEPT “zinc / Zn / it is in excess”.
(a)(iii) To prevent / minimise loss of gas / so as little gas as possible escapes.
ACCEPT “to keep as much gas as possible”, “to avoid loss of gas”.
(a)(iv) No further effervescence / bubbles / fizzing OR No more gas collects in the syringe / gas syringe reading stops increasing.
ALLOW “gas syringe does not move”.
(b)(i)
M1: Correct gradient calculation using tangent (e.g., \( \frac{\Delta V}{\Delta t} = \frac{50\ \text{cm}^3}{150\ \text{s}} \))
M2: \( 0.33 \) (allow \( 0.333 \))
M3: Unit: \(\text{cm}^3/\text{s}\) or \(\text{cm}^3 \text{ s}^{-1}\)
Triangle must be drawn on graph for M1. ECF from M1.
(b)(ii)
From 0 s to 60 s:
• Gradient is steepest / rate is highest.
• Because the concentration of acid is highest / most acid particles per unit volume / most frequent collisions.
From 60 s to 150 s:
• Curve becomes less steep / rate decreases.
• Because acid concentration decreases / fewer acid particles per unit volume / fewer collisions.
From 150 s to 240 s:
• Curve levels off / becomes flat / plateaus / reaction stops.
• Because the (sulfuric) acid has been used up / limiting reactant exhausted.
ACCEPT “volume of gas becomes constant”.
Mark scheme based on provided examiner comments.
This question is about crude oil.
(a) Describe how crude oil is separated into fractions by fractional distillation.
(b) Some of the products of fractional distillation are then cracked. This equation represents a reaction that occurs during cracking:
\[C_{15}H_{32} \rightarrow C_8H_{18} + C_3H_6 + 2C_2H_4\]
Explain why cracking is an important process in the oil industry.
(c) Fuels obtained from crude oil may contain impurities. Explain how an impurity found in fuels can cause an environmental problem.
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry – 4CH1):
• 4(c): Alkanes — implicit in (a)
• 4(d): Alkenes — implicit in (b)
• 4(b): Fuels and combustion — part (c)
• 4(b): Environmental chemistry — part (c)
▶️ Answer/Explanation
(a) A description that links any 4 of the following points:
M1: Crude oil is heated / vaporised. (ALLOW: boiled)
M2: The vapour enters the lower part / bottom of the column. (ACCEPT: cooler at the top and hotter at the bottom)
M3: There is a temperature gradient in the column. (ALLOW: the fractions are separated according to their boiling point)
M4: The vapours rise up the column until they condense.
M5: At a height where the boiling point of the vapour is lower than the temperature in the column.
(b) An explanation that links any 4 of the following points:
M1: Fractional distillation of crude oil produces more long chain hydrocarbons than can be used directly. (ALLOW: is a lower demand for long chain hydrocarbons / a higher demand for short chain hydrocarbons)
M2: (Cracking) produces shorter (chain) alkanes. (ALLOW: petrol / gasoline)
M3: Which are more flammable / more useful as fuels. (M3 dep on M2)
M4: (Cracking) produces alkenes.
M5: Which are used to make polymers/plastics. (M5 dep on M4)
(c) An explanation that makes reference to the following:
M1: Fuels contain sulfur.
M2: Which burns producing sulfur dioxide.
M3: Causing acid rain. (M3 dep on M1 or M2)
(IGNORE: C, CO, CO\(_2\) and any reference to global warming. REJECT: nitrogen for M1. ALLOW: effects of acid rain.)
(a) The table shows the formulae of some positive and negative ions, and the formulae of some compounds containing these ions.
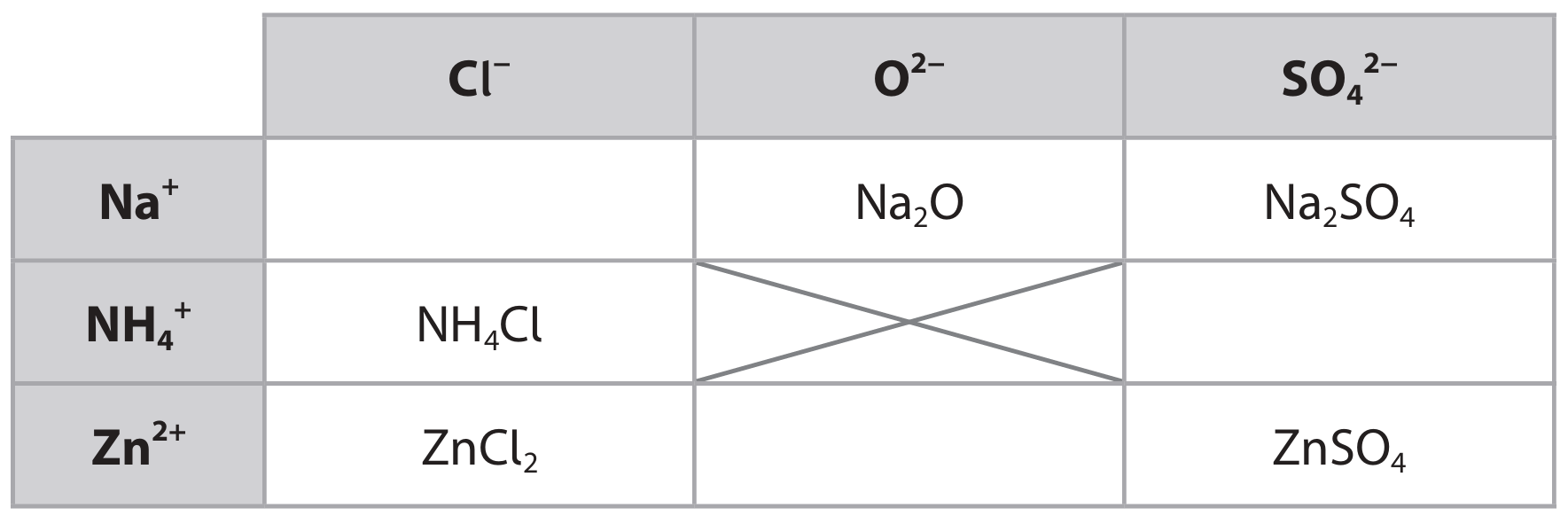
(i) Complete the table by giving the formulae of the missing compounds.
(ii) Give the name of the compound with the formula ZnSO₄.
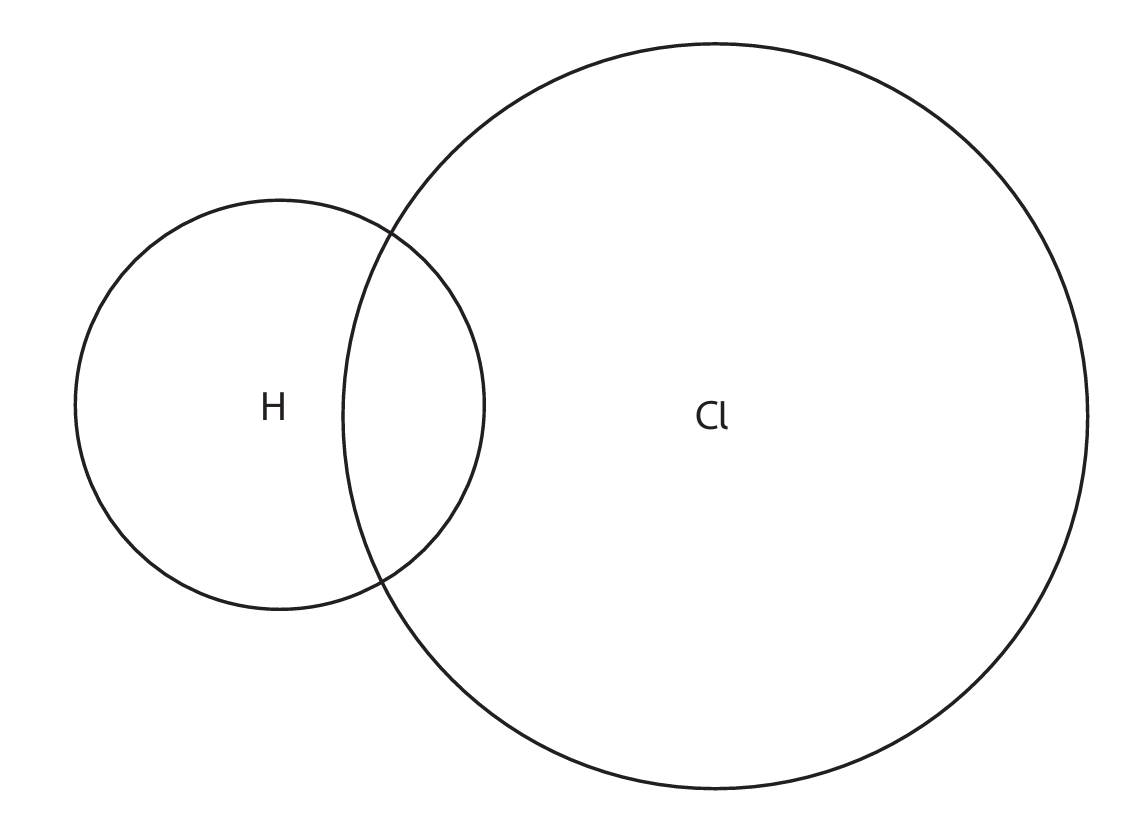
(b) Hydrogen chloride and magnesium chloride have different types of bonding and have different structures.
(i) Complete the dot-and-cross diagram to show the outer shell electrons in a molecule of hydrogen chloride.
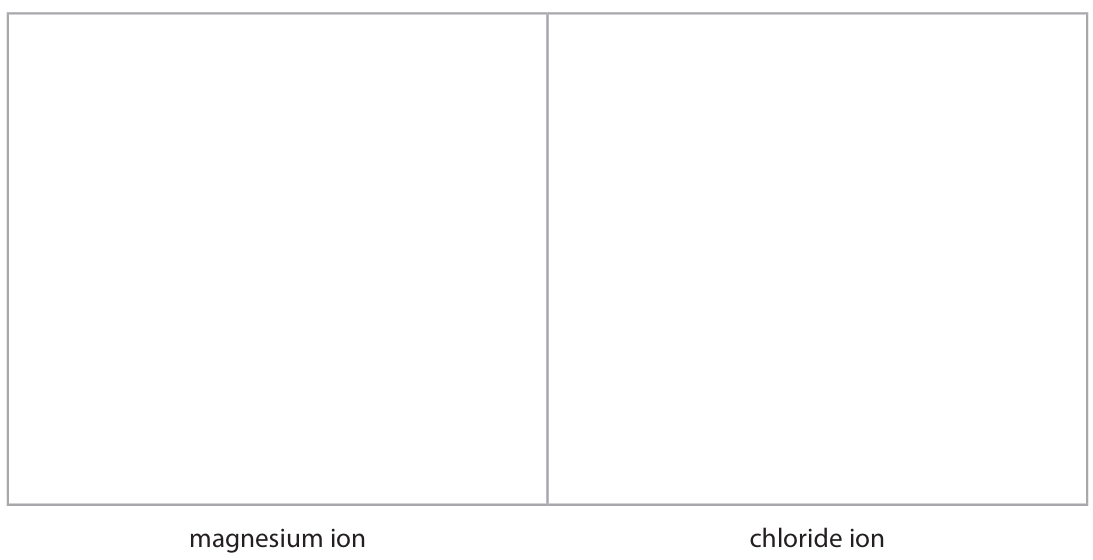
(ii) The diagram shows the electronic configuration of a magnesium atom and of a chlorine atom.
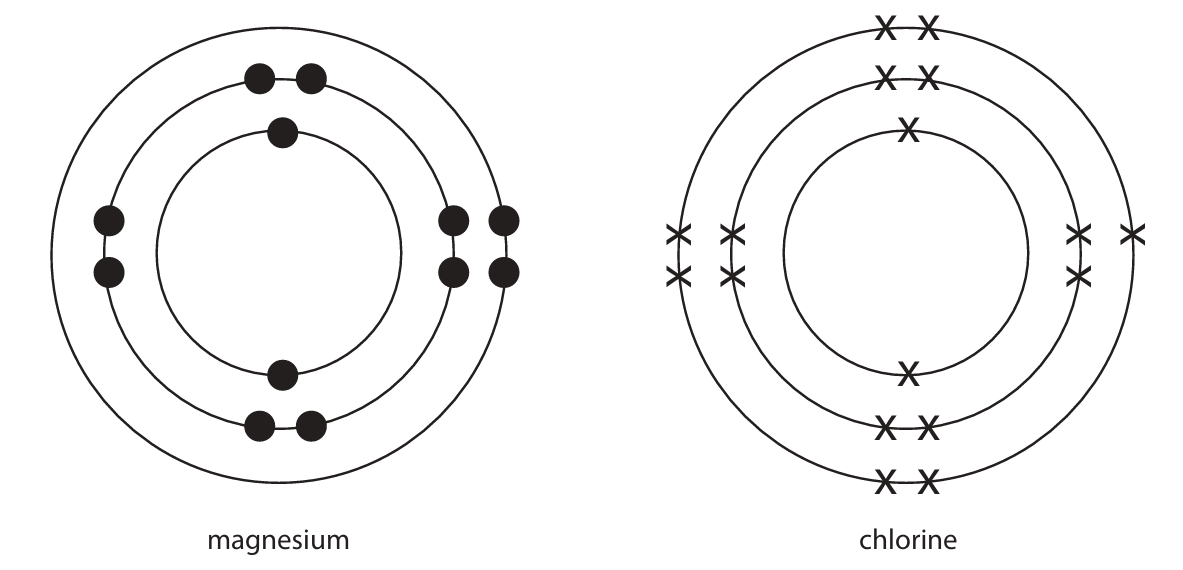
Draw the electronic configuration of a magnesium ion and of a chloride ion in the boxes. Show the charge on each ion.
(iii) Explain why magnesium chloride has a much higher melting point than hydrogen chloride. Refer to structure and bonding in your answer.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(f): Ionic bonding — parts (b)(ii), (b)(iii)
• 1(g): Covalent bonding — parts (b)(i), (b)(iii)
• 1(h): Metallic bonding — context for structure-property relationships
▶️ Answer/Explanation
(a)(i)
- M1: NaCl
- M2: ZnO
- M3: (NH4)2SO4
(a)(ii) Zinc sulfate
(b)(i)
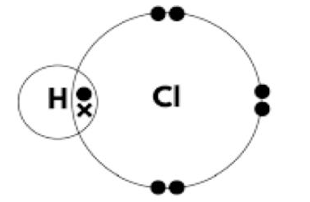
The dot-and-cross diagram should show:
- M1: 2 bonding electrons (shared pair) between H and Cl.
- M2: The remaining 6 outer electrons on Cl shown correctly (3 lone pairs).
(b)(ii)
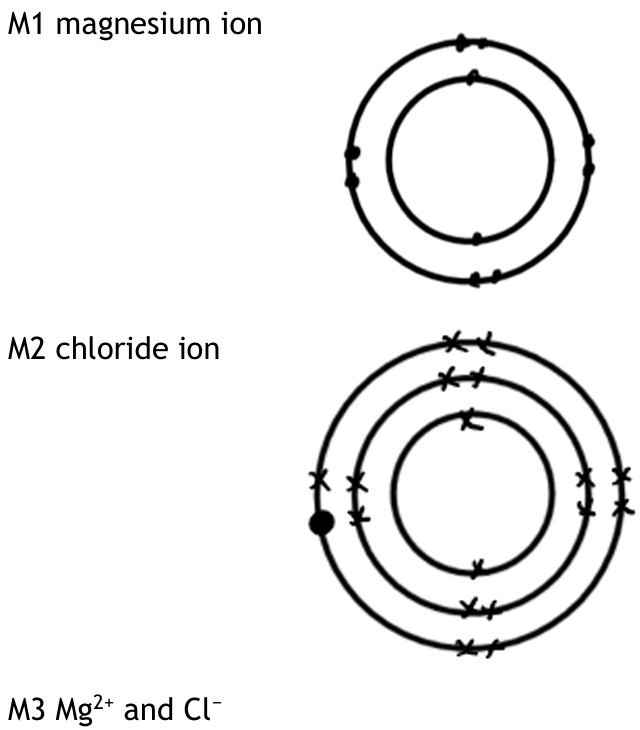
- M1: Magnesium ion (Mg²⁺) diagram: electronic configuration 2,8 (no electrons in the third shell).
- M2: Chloride ion (Cl⁻) diagram: electronic configuration 2,8,8.
- M3: Correct charges shown: Mg²⁺ and Cl⁻.
(b)(iii) An explanation that links any 5 of the following points:
- M1: Hydrogen chloride is simple molecular / simple covalent.
- M2: Magnesium chloride is giant ionic / ionic lattice.
- M3: Strong electrostatic attraction between (oppositely charged) ions in MgCl₂.
- M4: In hydrogen chloride there are weak intermolecular forces / weak forces between molecules.
- M5: (Much) more energy is required to break the (ionic) bonds in MgCl₂ than to overcome the (intermolecular) forces in HCl.
This is the equation for the decomposition of hydrogen peroxide.
\[2H_2O_2(aq) \rightarrow 2H_2O(l) + O_2(g)\]
The rate of reaction increases when a catalyst of manganese(IV) oxide is added.
(a) Describe how a catalyst increases the rate of a reaction.
(b) A student adds 50 cm\(^3\) of hydrogen peroxide solution to a glass container and then adds 1.0 g of manganese(IV) oxide.
The diagram shows the apparatus the student uses.
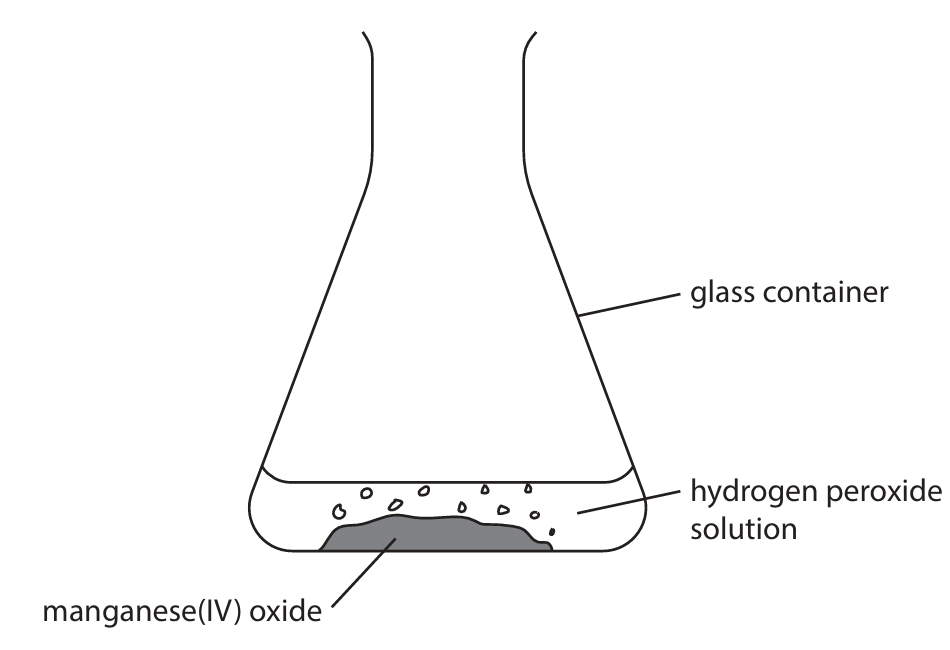
(i) Name the glass container the student uses.
(ii) The student waits until the hydrogen peroxide solution completely decomposes.
Describe how the student could then show that the manganese(IV) oxide was a catalyst and not a reactant.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 3(b): Rates of reaction (Practical investigation) — part (b)(ii)
• 1(i): Electrolysis (Definitions) — part (a) context of catalysts
• Appendix 6: Suggested practical investigations — experimental context in part (b)
▶️ Answer/Explanation
(a)
M1: A catalyst provides an alternative pathway / route (1 mark)
M2: of lower activation energy (1 mark)
(b) (i)
Conical flask
ALLOW: flask (1 mark)
(b) (ii)
M1: Filter out the manganese(IV) oxide (1 mark)
M2: Allow it to dry (1 mark)
M3: Reweigh the catalyst; the same mass should be left / mass is still 1 g (1 mark)
Diamond and graphite are both forms of the element carbon. Diamond and graphite both have covalent bonds and giant covalent structures. The diagram represents the structure of diamond and the structure of graphite.
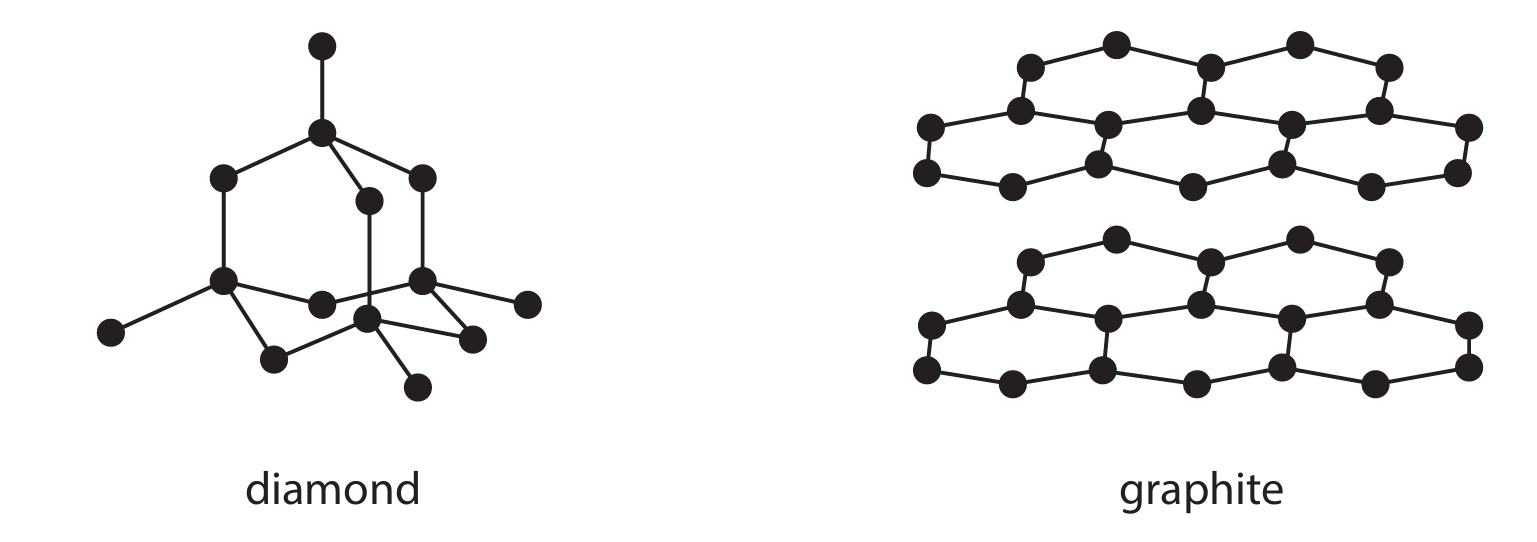
(a) Give a reason why diamond is an element.
(b) Describe the forces of attraction in a covalent bond.
(c) (i) Explain why graphite conducts electricity.
(c) (ii) Explain why diamond is hard but graphite is soft.
(d) Another form of carbon has molecules with the formula \( C_x \).
\( x \) represents the number of carbon atoms in each molecule.
Each molecule of \( C_x \) has a mass of \( 1.40 \times 10^{-21} \, \text{g} \).
One mole of \( C_x \) contains \( 6.02 \times 10^{23} \) molecules.
Calculate the \( M_r \) of \( C_x \) and the value of \( x \).
[for carbon, \( A_r = 12 \)]
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry 4CH1):
• 1(g): Structures and properties of diamond and graphite — parts (a), (c)(i), (c)(ii)
• 1(e): Chemical formulae, equations and calculations — part (d)
• 1(c): Atomic structure — part (a)
▶️ Answer/Explanation
(a) Diamond is an element because it is made up of only one type of atom / only carbon atoms.
(b) In a covalent bond, the force of attraction is between a shared pair of electrons and the nuclei of the bonded atoms.
(c) (i) Graphite conducts electricity because it has delocalised electrons (between the layers). These electrons are free to move / flow through the structure, carrying charge.
(c) (ii) Diamond is hard because it has a giant three-dimensional tetrahedral lattice / rigid structure where each carbon atom is bonded to four others by strong covalent bonds. These bonds require a lot of energy to break.
Graphite is soft because it has a layered structure. The layers are held together by weak intermolecular forces and can slide over one another easily.
(d)
Step 1: Calculate the molar mass \( M_r \) of \( C_x \).
Mass of one molecule = \( 1.40 \times 10^{-21} \, \text{g} \).
Number of molecules in one mole = \( 6.02 \times 10^{23} \).
\( M_r = (1.40 \times 10^{-21}) \times (6.02 \times 10^{23}) \)
\( M_r = 842.8 \, \text{g/mol} \) (or \( 843 \) to 3 s.f.)
Step 2: Calculate \( x \).
\( A_r \) of carbon = 12.
\( x = \frac{M_r}{A_r} = \frac{842.8}{12} \approx 70.23 \)
Since \( x \) must be an integer (number of atoms), \( x = 70 \).
Therefore, \( M_r = 843 \) and \( x = 70 \).
Mark Scheme Reference:
- (a): ACCEPT “only made up of carbon atoms”.
- (b): M1 for “attraction between a shared pair of electrons”; M2 for “and nuclei”. (Nuclei must be plural).
- (c)(i): M1 for “delocalised electrons”; M2 for “(electrons) can move / flow”.
- (c)(ii): M1 for 3D/rigid/tetrahedral lattice in diamond; M2 for bonds requiring a lot of energy to break; M3 for layered structure in graphite; M4 for layers can slide. (M4 dependent on M3).
- (d): M1 for calculating \( M_r \); M2 for dividing by 12; M3 for integer answer (70).
This question is about the metal tantalum, Ta.
Tantalum metal can be produced by heating tantalum chloride (\( \text{TaCl}_5 \)) and hydrogen gas in a furnace. The other product of the reaction is hydrogen chloride.
(a) Complete the equation for the reaction.
\[ \text{TaCl}_5(s) + \text{H}_2(g) \rightarrow \text{Ta}(s) + \text{HCl}(g) \]
(b) As tantalum chloride is heated, the mass of solid in the furnace decreases leaving tantalum as the only solid product.
The table shows the mass of solid in the furnace at one-hour intervals.
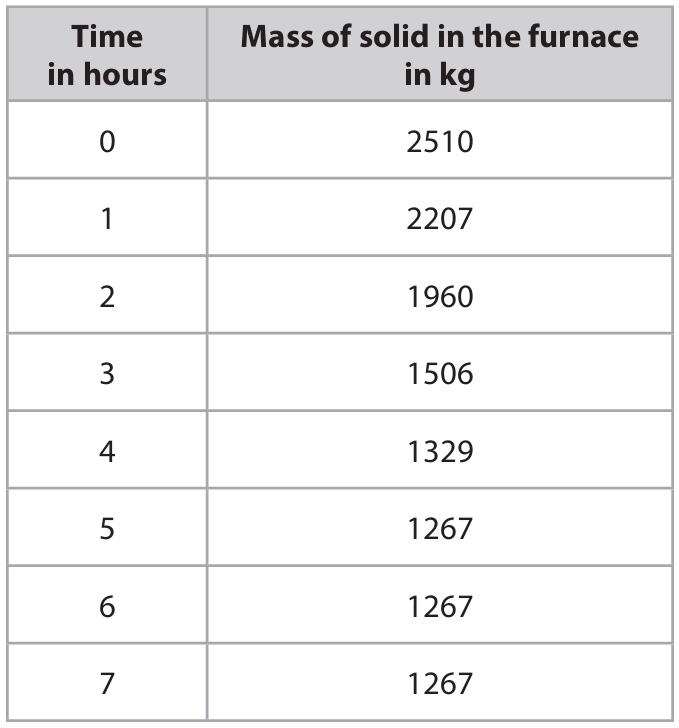
(i) State how the data in the table shows that the reaction is complete.
(ii) Use the data to show that the formula of tantalum chloride is TaCl\(_5\).
[for tantalum, \(A_r = 181\); for chlorine, \(A_r = 35.5\)]
(c) Another method of extracting tantalum(V) oxide uses carbon. This is the equation for the reaction.
\[ \text{Ta}_2\text{O}_5(\text{s}) + 5\text{C}(\text{s}) \rightarrow 2\text{Ta}(\text{s}) + 5\text{CO}(\text{g}) \]
(i) Explain why this is a redox reaction.
(ii) 2000 mol of tantalum(V) oxide is heated with 500 000 g of carbon. Show by calculation that the carbon is in excess.
[for carbon, \( A_r = 12 \)]
(iii) Calculate the maximum mass, in grams, of tantalum that can be obtained from 2000 mol of tantalum(V) oxide.
[for tantalum, \( A_r = 181 \)]
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(d) Reactivity series — parts (b)(i), (c)(i)
• 2(e) Extraction and uses of metals — part (c)(i), (c)(ii), (c)(iii)
• 1(f) Ionic bonding & 1(g) Covalent bonding — background for understanding compound formation
▶️ Answer/Explanation
(a) Balanced equation:
\[ \text{TaCl}_5(s) + \frac{5}{2}\text{H}_2(g) \rightarrow \text{Ta}(s) + 5\text{HCl}(g) \]
Allow multiples.
(b)(i) The last three masses (at 5, 6, 7 hours) are the same / the mass does not change / mass becomes constant.
(b)(ii) Calculation to show TaCl\(_5\):
Final mass of solid (Ta) = 1267 kg = \(1.267 \times 10^6\) g
Mass loss = \(2510 – 1267 = 1243\) kg (mass of chlorine in TaCl\(_5\)).
Moles of Ta = \(\frac{1.267 \times 10^6}{181} \approx 7000\) mol.
Moles of Cl = \(\frac{1.243 \times 10^6}{35.5} \approx 35014\) mol.
Ratio Cl : Ta ≈ \(35014 : 7000 \approx 5 : 1\).
Hence formula is TaCl\(_5\).
(c)(i) This is a redox reaction because:
• Carbon gains oxygen to form CO → carbon is oxidised.
• Tantalum(V) oxide loses oxygen → tantalum is reduced.
OR Oxidation numbers: C from 0 to +2 (oxidation), Ta from +5 to 0 (reduction).
(c)(ii) Moles of C needed = \(5 \times 2000 = 10\,000\) mol.
Mass of C needed = \(10\,000 \times 12 = 120\,000\) g.
Carbon available = 500 000 g, which is > 120 000 g → carbon is in excess.
(c)(iii) From equation: 1 mol Ta\(_2\)O\(_5\) → 2 mol Ta.
Moles of Ta = \(2 \times 2000 = 4000\) mol.
Mass of Ta = \(4000 \times 181 = 724\,000\) g (or 724 kg).
