The table shows the composition of five species that are either atoms or ions.
| Species | Number of protons | Number of neutrons | Number of electrons |
|---|---|---|---|
| A | 1 | 0 | 1 |
| B | 3 | 3 | 3 |
| C | 3 | 4 | 3 |
| D | 9 | 10 | 10 |
| E | 13 | 14 | 13 |
(a) Use the table to answer these questions. You may use each letter once, more than once, or not at all.
(i) Give the letter of the species that is a negative ion.
(ii) Give the letter of the species that has a full outer shell of electrons.
(iii) Give the letter of the species that has an atomic number equal to its mass number.
(b) Explain why species B and C are atoms of the same element with identical chemical properties.
(c) A proton has a mass of \( 1.6726 \times 10^{-24} \, \text{g} \).
A neutron has a mass of \( 1.6740 \times 10^{-24} \, \text{g} \).
Calculate the mass, in grams, of the nucleus of an atom of species C.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(d): The Periodic Table — parts (a)(ii), (b)
• 1(f): Ionic bonding — part (a)(i)
▶️ Answer/Explanation
(a)(i) D
Species D has 9 protons and 10 electrons. The extra electron gives it a negative charge, making it an anion (e.g., F⁻).
(a)(ii) D
Species D has 10 electrons. For an atom with 9 protons (fluorine), a full outer shell is achieved with 10 electrons (2 in the first shell, 8 in the second), corresponding to the electron configuration of neon.
(a)(iii) A
Atomic number = number of protons = 1. Mass number ≈ protons + neutrons = 1 + 0 = 1.
(b)
M1: They are both atoms of the same element because they both have the same number of protons / same atomic number.
M2: They have identical chemical properties because they both have the same electron configuration / same number of electrons.
(c)
Species C has 3 protons and 4 neutrons.
Mass of nucleus = mass of protons + mass of neutrons
\( = (3 \times 1.6726 \times 10^{-24}) + (4 \times 1.6740 \times 10^{-24}) \)
\( = (5.0178 \times 10^{-24}) + (6.6960 \times 10^{-24}) \)
\( = 1.17138 \times 10^{-23} \, \text{g} \)
Answer: \( 1.1714 \times 10^{-23} \, \text{g} \) (to correct standard form and rounding)
This question is about metals and metal compounds.
(a) All metals are malleable and good conductors of electricity.
The diagram shows the structure of a metal.
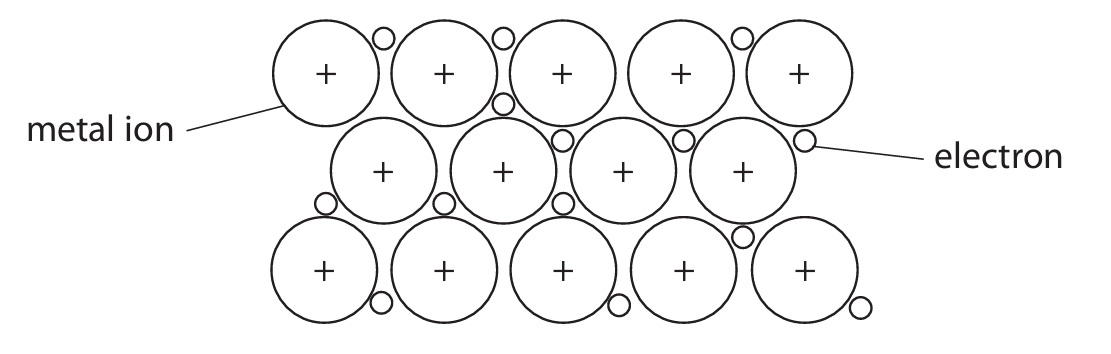
(i) Explain why metals are malleable.
(ii) Explain why metals are good conductors of electricity.
(b) Two methods of obtaining a metal from a compound are electrolysis and extraction using carbon.
Explain, without giving practical details, which method is most suitable for extracting iron from iron(III) oxide, \(\text{Fe}_2\text{O}_3\).
(c) A student has a solution of iron(II) sulfate and a solution of iron(III) sulfate.
Describe a chemical test the student could use to identify which solution is iron(II) sulfate.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(e): Extraction and uses of metals — part (b)
• 2(h): Chemical tests — part (c)
▶️ Answer/Explanation
(a)(i)
M1: Layers/rows/sheets (of atoms/ions/particles) can slide over one another.
M2: (Because) there are no strong directional bonds holding them in fixed positions.
Marking note: Do not credit answers mentioning intermolecular forces or electrons sliding.
(a)(ii)
M1: Electrons are delocalised / free to move.
M2: (These) electrons can flow/carry charge through the structure when a potential difference is applied.
Marking note: Must mention electrons.
(b)
M1: Extraction using carbon / carbon reduction.
M2: Because iron is less reactive than carbon / carbon is more reactive than iron / iron is below carbon in the reactivity series.
Marking note: Electrolysis is used for more reactive metals (e.g., aluminium).
(c)
M1: Add sodium hydroxide solution (NaOH) to both solutions.
M2: The solution that forms a green precipitate is iron(II) sulfate. (Iron(III) sulfate would form a brown precipitate).
Alternative:
• Add potassium hexacyanoferrate(III) solution.
• The solution that forms a dark blue precipitate (Turnbull’s blue) is iron(II) sulfate. (Iron(III) would give a brownish solution).
Marking note: M2 is dependent on M1 or mention of a suitable soluble hydroxide.
A teacher uses this apparatus to separate a mixture of ethanol and water.
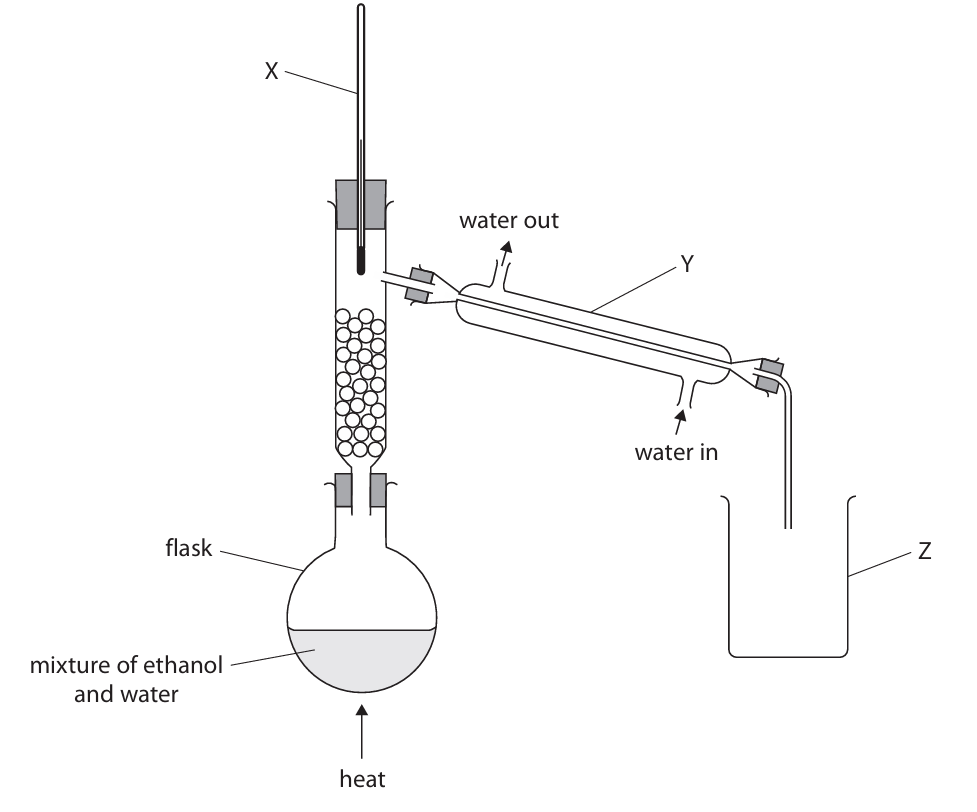
(a) Give the name of this method of separation.
A) chromatography
B) crystallisation
C) filtration
D) fractional distillation
(b) Name the pieces of apparatus labelled X, Y and Z.
(c) Give a physical test that the teacher could do to find out if the ethanol produced is pure.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 1(b): Separation techniques — part (a), part (b)
• 1.10: Describe experimental techniques for the separation of mixtures — part (a), part (b)
• 1.9: Understand that a pure substance has a fixed melting and boiling point — part (c)
▶️ Answer/Explanation
(a) D (fractional distillation)
A is not the answer as this technique is not chromatography
B is not the answer as this technique is not crystallisation
C is not the answer as this technique is not filtration
(b)
X = thermometer
Y = (Liebig) condenser
Z = beaker
(c)
• M1: measure / test the boiling point
• M2: a pure substance has a fixed boiling point / boiling point of 78°C / it will not boil over a range of temperatures
A teacher adds a small piece of lithium to water and collects the gas produced.
(a) (i) Give two observations when lithium is added to water.
(ii) The teacher adds a few drops of universal indicator to the solution at the end of the reaction.
Explain the final colour of the universal indicator.
(b) This is the equation for the reaction between lithium and water.
\[2Li(s) + 2H_2O(l) \rightarrow 2LiOH(aq) + H_2(g)\]
The teacher collects \(550 \text{ cm}^3\) of hydrogen at rtp.
Calculate the mass, in grams, of lithium that the teacher added to the water.
[one mole of gas at rtp has a volume of \(24\,000 \text{ cm}^3\)]
[for lithium, \(A_r = 7\)]
(c) The teacher then adds a small piece of potassium to water.
(i) Give one observation seen with potassium that is not seen with lithium.
(ii) Explain why potassium is more reactive than lithium. Refer to atomic structure in your answer.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 2(a): (Specification point 2.4C) — Explain the trend in reactivity in Group 1 in terms of electronic configurations — part (c)(ii)
• 1(e): Chemical formulae, equations and calculations — part (b)
• 1(e): (Specification point 1.35C) — Calculations involving gas volumes and the molar volume of a gas — part (b)
• 1(e): (Specification point 1.29) — Calculate reacting masses using experimental data and chemical equations — part (b)
▶️ Answer/Explanation
(a)(i) Any two from:
• Effervescence / bubbles / fizzing (of hydrogen) (M1)
• (The piece of lithium) gets smaller / disappears / dissolves (M2)
• (The piece of lithium) moves on the surface / floats (M3)
• White trail (M4)
(Any mention of a flame scores a maximum of 1 mark)
(a)(ii)
• M1: Blue/purple (1)
• M2: Hydroxide ions / lithium hydroxide / an alkaline solution / an alkali / \(\text{OH}^-\) ions is produced / formed (1)
(b) Calculation:
• M1: Moles of \(H_2 = \frac{550}{24000} = 0.0229\) (1)
• M2: Moles of \(Li = 0.0229 \times 2 = 0.0458\) (1)
• M3: Mass of \(Li = 0.0458 \times 7 = 0.321 \text{ g}\) (1)
Allow 0.32 / 0.3206 / 0.32088 / 0.32083 / 0.322 (minimum 2 significant figures)
(c)(i) Any one from:
• (Lilac) flame (M1)
• Melts / turns into a ball (M2)
Reject an incorrect colour of the flame.
(c)(ii)
• M1: (Atoms of potassium) are bigger / have more shells (of electrons) / larger atomic radius / the outer shell electron is further from the nucleus / more shielding (1)
• M2: (Therefore) the outer shell electron is less attracted to the nucleus (1)
• M3: (And therefore the outer shell) electron is more easily lost (1)
Lose 1 mark if ‘outer shell electron’ is not mentioned in M2 or M3.
A student does some titrations to find the volume of dilute nitric acid needed to exactly neutralise \(25.0 \, \text{cm}^3\) of sodium hydroxide solution.
This is the student’s method.
Step 1: add \(25.0 \, \text{cm}^3\) of sodium hydroxide solution to a conical flask
Step 2: add three drops of methyl orange indicator
Step 3: fill a burette with the acid
Step 4: add acid from the burette until the indicator changes colour
Step 5: record the volume of acid added
(a) (i) Give the name of the apparatus that the student should use to measure the volume of sodium hydroxide solution in step 1.
(ii) Give the colour change seen in step 4.
(iii) Give a reason why the student does not use universal indicator in this titration.
(b) The student completes a rough titration and four accurate titrations.
The diagram shows the burette readings from the rough titration.
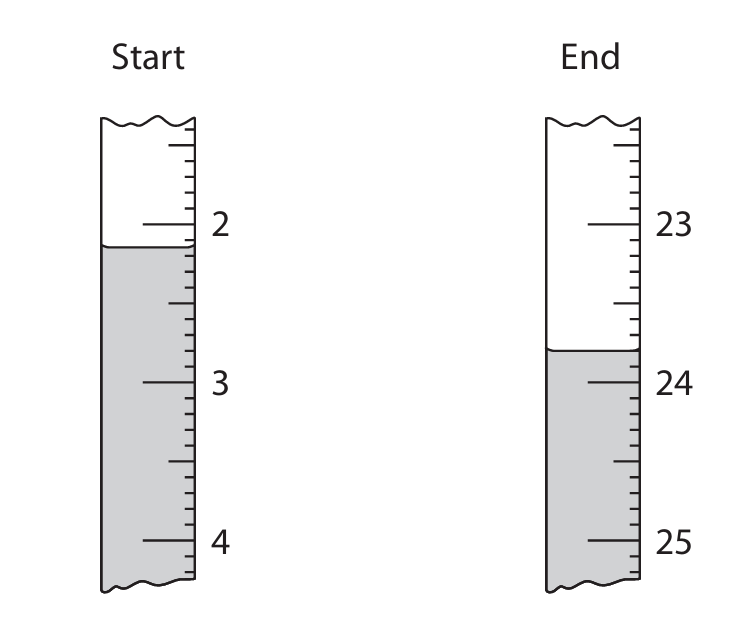
The table shows the student’s results.
| Rough titration | Titration 1 | Titration 2 | Titration 3 | Titration 4 | |
|---|---|---|---|---|---|
| Burette reading at end in cm³ | 21.80 | 22.85 | 21.75 | 24.10 | |
| Burette reading at start in cm³ | 0.50 | 0.15 | 0.25 | 0.10 | |
| Volume added in cm³ | 21.30 | 22.70 | 21.50 | 24.00 |
(i) Complete the table by adding the results from the rough titration.
Record the volumes to the nearest \(0.05 \, \text{cm}^3\)
(ii) Concordant results are results within \(0.20 \, \text{cm}^3\) of each other.
Use the concordant results from the table to calculate the mean volume of acid added.
(c) This is the equation for the reaction between dilute nitric acid and sodium hydroxide solution.
\[ \text{HNO}_3(\text{aq}) + \text{NaOH}(\text{aq}) \rightarrow \text{NaNO}_3(\text{aq}) + \text{H}_2\text{O}(l) \]
After the titration, the student knows the volume of acid needed to neutralise \(25.0 \, \text{cm}^3\) of the sodium hydroxide solution.
Sodium nitrate decomposes at high temperatures.
Describe how the student could obtain pure, dry crystals of sodium nitrate from dilute nitric acid and sodium hydroxide solution.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(g): Acids, bases and salt preparations — Part (c) – preparation of a soluble salt from an acid and alkali (Specification point 2.40C)
• 1(b): Elements, compounds and mixtures — Part (c) – separation technique of crystallisation (Specification point 1.10)
▶️ Answer/Explanation
(a)(i) (Volumetric) pipette.
(a)(ii) Yellow to red / yellow to orange / yellow to pink.
(a)(iii) Universal indicator does not give a sharp colour change / changes colour over a range of pH values / has no clear endpoint.
(b)(i)
Start reading: \(2.15 \, \text{cm}^3\)
End reading: \(23.80 \, \text{cm}^3\)
Volume added: \(21.65 \, \text{cm}^3\) (calculated as \(23.80 – 2.15\))
(b)(ii)
Concordant results (within \(0.20 \, \text{cm}^3\)): Titration 1 (\(21.30\)) and Titration 3 (\(21.50\)).
Mean volume = \(\frac{21.30 + 21.50}{2} = 21.40 \, \text{cm}^3\).
(c)
- Repeat the neutralisation using the calculated volumes of acid (\( \text{HNO}_3 \)) and alkali (\( \text{NaOH} \)) but without adding any indicator.
- Heat the resulting sodium nitrate solution in an evaporating basin to evaporate some water until crystals start to form / until the solution is saturated / until a drop on a glass rod crystallises. (Do NOT heat to dryness as \( \text{NaNO}_3 \) decomposes at high temperatures).
- Allow the solution to cool and crystallise.
- Filter the mixture to collect the crystals, pouring off the excess mother liquor.
- Dry the crystals by leaving them in a warm place / in a desiccator / between filter papers.
This question is about crude oil.
(a) Describe how crude oil is separated into fractions by fractional distillation.
(b) Some of the long-chain alkanes obtained from fractional distillation are cracked, producing shorter-chain alkanes and ethene.
(i) Give the conditions necessary for cracking to occur.
(ii) Explain why shorter-chain alkanes are more useful than longer-chain alkanes.
(iii) Ethene reacts with bromine.
The equation shows the displayed formulae of the reactants and product.
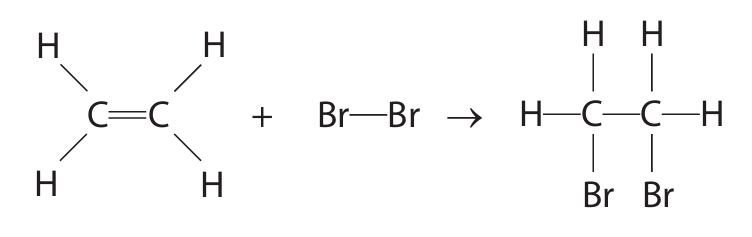
The table shows some bond energies.
| Bond | Bond energy in kJ/mol |
|---|---|
| C=C | 612 |
| C—C | 348 |
| C—H | 414 |
| Br—Br | 193 |
| C—Br | 276 |
Show that the molar enthalpy change, \(\Delta H\), for this reaction is about –100 kJ/mol.
(iv) Explain why the reaction between ethene and bromine is exothermic. Refer to bond energies in your answer.
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry 4CH1):
• 3(a): Energetics — parts (b)(iii), (b)(iv) — Bond energies and enthalpy change calculations
• 1(e): Chemical formulae, equations and calculations — part (b)(iii) — Use of bond energies in calculations
• 4(d): Alkenes — part (b)(iii), (b)(iv) — Reaction of ethene with bromine
▶️ Answer/Explanation
(a)
M1: Crude oil / it is heated
M2: The vapours / gases rise up the column
M3: The column is hotter at the bottom than the top (OWTTE) / The vapours cool as they rise up the column
M4: The vapours condense at their boiling points
(b)(i)
M1: Temperature: \(600 – 700\,^\circ\mathrm{C}\) (allow any number/range within)
M2: Catalyst: silica / alumina / aluminosilicates / zeolites
(b)(ii)
M1: (Shorter chain alkanes) are more flammable / are easier to burn
M2: Therefore are more useful as fuels / petrol / gasoline
(b)(iii)
M1: (Bonds broken) = \(612 + 193 = 805\,\mathrm{kJ\,mol^{-1}}\)
M2: (Bonds formed) = \(348 + (2 \times 276) = 900\,\mathrm{kJ\,mol^{-1}}\)
M3: \(\Delta H = 805 – 900 = -95\,\mathrm{kJ\,mol^{-1}} \approx -100\,\mathrm{kJ\,mol^{-1}}\)
Alternative method (including C–H bonds):
M1: Bonds broken = \((4 \times 414) + 612 + 193 = 2461\,\mathrm{kJ\,mol^{-1}}\)
M2: Bonds formed = \((4 \times 414) + 348 + (2 \times 276) = 2556\,\mathrm{kJ\,mol^{-1}}\)
M3: \(\Delta H = 2461 – 2556 = -95\,\mathrm{kJ\,mol^{-1}} \approx -100\,\mathrm{kJ\,mol^{-1}}\)
(b)(iv)
M1: More energy / heat was given out / released when the bonds (in the products) were formed
M2: Than was taken in / needed to break the bonds (in the reactants)
(Total = 13 marks)
This question is about alcohols and carboxylic acids.
(a) These are the structural formulae of alcohol A and carboxylic acid B.
Alcohol A: CH3CH2CH2OH
Carboxylic acid B: CH3(CH2)nCOOH
(i) Name alcohol A.
(ii) Draw the displayed formula of alcohol A.
(iii) Carboxylic acid B has a chain of carbon atoms with no branches. The number of CH2 units is represented by the letter n. Calculate the value of n. [for carboxylic acid B, \( M_r = 242 \)]
(iv) Alcohol A and carboxylic acid B react together to form an ester. Give the other product of the reaction.
(b) These are the structural formulae of dicarboxylic acid C and diol D.
Dicarboxylic acid C: HOOCCH2CH2COOH
Diol D: HOCH2CH2OH
(i) This is the equation for the reaction between dicarboxylic acid C and sodium hydroxide solution.
HOOCCH2CH2COOH + 2NaOH → NaOOCH2CH2COONa + 2H2O
25.0 cm3 of 0.150 mol/dm3 sodium hydroxide solution is completely neutralised by 17.5 cm3 of a solution of dicarboxylic acid C.
Calculate the concentration, in mol/dm3, of the solution of dicarboxylic acid C. Give your answer to three significant figures.
(ii) Dicarboxylic acid C and diol D react to form a polyester. Draw the displayed formula of the repeat unit of this polyester.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 4(f): Carboxylic acids — part (a)(iii)
• 1(e): Chemical formulae, equations and calculations — parts (a)(iii), (b)(i)
• 4(g): Esters — part (a)(iv)
• 4(h): Synthetic polymers — part (b)(ii)
• 2(f) / 2(g): Acids, alkalis and titrations / Acids, bases and salt preparations — part (b)(i)
▶️ Answer/Explanation
(a)(i) Propan-1-ol / 1-propanol
(a)(ii) Displayed formula of propan-1-ol:
H H H
| | |
H - C - C - C - O - H
| | |
H H H
(a)(iii) Calculation for \(n\):
General formula: CH3(CH2)nCOOH
\(M_r = 12 + 3 + 14n + 12 + 32 + 1 = 242\)
\(14n + 60 = 242\)
\(14n = 182\)
\(n = 13\)
(a)(iv) Water / H2O
(b)(i) Calculation for concentration of dicarboxylic acid C:
Moles of NaOH = \(0.150 \times 0.025 = 0.00375 \text{ mol}\)
From equation: 1 mol acid reacts with 2 mol NaOH
Moles of acid = \(0.00375 \div 2 = 0.001875 \text{ mol}\)
Volume of acid = \(17.5 \text{ cm}^3 = 0.0175 \text{ dm}^3\)
Concentration = \(\frac{0.001875}{0.0175} = 0.10714 \text{ mol/dm}^3\)
Answer = \(0.107 \text{ mol/dm}^3\) (to 3 s.f.)
(b)(ii) Displayed formula of the polyester repeat unit:
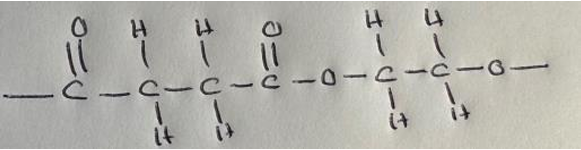
(The repeat unit is formed from one molecule of dicarboxylic acid C and one molecule of diol D, with the loss of water.)
