Use the Periodic Table to answer these questions.
(a) (i) Give the name of the element with atomic number 16.
(a) (ii) Give the name of the element with relative atomic mass 9.
(a) (iii) Give the name of the element in Group 3 and Period 2.
(a) (iv) Give the electronic configuration of an atom of silicon.
(b) Explain, in terms of electron configuration, why sodium is more reactive than lithium.
Most-appropriate topic codes (Edexcel IGCSE Chemistry – 4CH1):
• 1(c): Atomic structure — parts (a)(iv), (b)
• 1(d) / 2(a): Understanding how electronic configuration relates to position in the Periodic Table and group reactivity trends — part (b)
▶️ Answer/Explanation
(a)(i) sulfur
(a)(ii) beryllium
(a)(iii) boron
(a)(iv) \( 2,8,4 \) or \( 2.8.4 \)
(b) An explanation that links the following points:
- The outer electron in sodium is in a shell further from the nucleus / sodium has more electron shells than lithium / sodium has a larger atomic radius.
- There is more shielding of the outer electron from the nucleus in sodium.
- Therefore, the attraction between the nucleus and the outer electron is weaker in sodium.
- This makes it easier for sodium to lose its outer electron in reactions, making it more reactive than lithium.
Allow alternative correct wordings that convey the same comparative reasoning about distance, shielding, and nuclear attraction.
Structures made of iron can rust.
(a) (i) Name the two substances that cause iron to rust.
(a) (ii) State a barrier method that prevents rusting.
(b) Explain how sacrificial protection prevents the rusting of iron.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
▶️ Answer/Explanation
(a)(i)
| M1 | oxygen | ALLOW air / \(\text{O}_2\) |
| M2 | water | ALLOW moisture / water vapour / \(\text{H}_2\text{O}\) |
(a)(ii)
painting / oiling / coating with plastic / galvanising / electroplating / waxing / greasing
REJECT sacrificial protection
(b)
An explanation that links the following two points:
| M1 | a more reactive metal is connected to / coated on the iron | ACCEPT a suitable metal, e.g. zinc / magnesium / aluminium IGNORE an element |
| M2 | the more reactive metal will react / oxidise / corrode instead of iron | REJECT a more reactive metal rusts instead of iron |
The diagram shows the separation of crude oil into fractions.
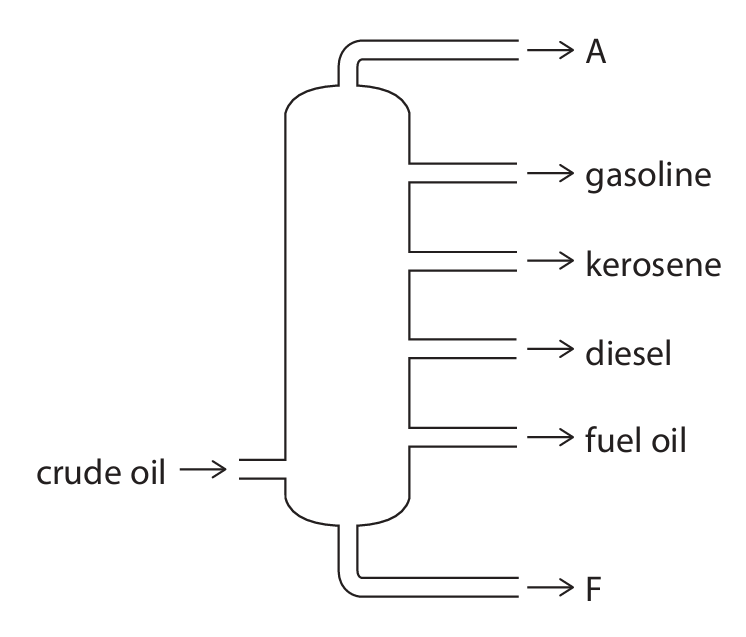
(a) Give the name of fraction A and the name of fraction F.
(b) One of the fractions is kerosene.
(i) Give one use of kerosene.
(ii) Describe how kerosene can be obtained from crude oil.
(c) Catalytic cracking is a process used to break down fractions containing long-chain molecules.
(i) Give the name of the catalyst and the temperature used for catalytic cracking.
(ii) Explain why catalytic cracking is useful.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 4(c): Alkanes & 4(d): Alkenes (related to cracking) — part (c)(i), (c)(ii)
▶️ Answer/Explanation
(a)
Fraction A: refinery gases
Fraction F: bitumen
(b)(i)
One use of kerosene: e.g., aircraft fuel / jet fuel.
(b)(ii)
A description that refers to three of the following points:
• Crude oil is heated / vaporised.
• The vapour passes into / rises up the fractionating column.
• Kerosene (the fraction) is tapped off / removed at its boiling point range (condenses and is removed).
(Allow: kerosene is removed at the 3rd or 4th level)
(c)(i)
Catalyst: silica / alumina / zeolite / aluminosilicate \((\text{SiO}_2 / \text{Al}_2\text{O}_3)\).
Temperature: any value or range between 600 and 700 \(^\circ\text{C}\).
(c)(ii)
An explanation that links three of the following points:
• There is a surplus supply / less demand for larger fractions / molecules / hydrocarbons.
• There is not enough supply / greater demand for smaller fractions / molecules / hydrocarbons (e.g., for petrol/gasoline).
• Alkenes are produced which are needed to make polymers / plastics.
• Smaller alkanes / hydrocarbons are needed for fuel.
The table shows the maximum mass of potassium nitrate \(\left( \text{KNO}_3 \right)\) and the maximum mass of sodium nitrate \(\left( \text{NaNO}_3 \right)\) that dissolves in \(25\ \text{cm}^3\) of water at different temperatures.
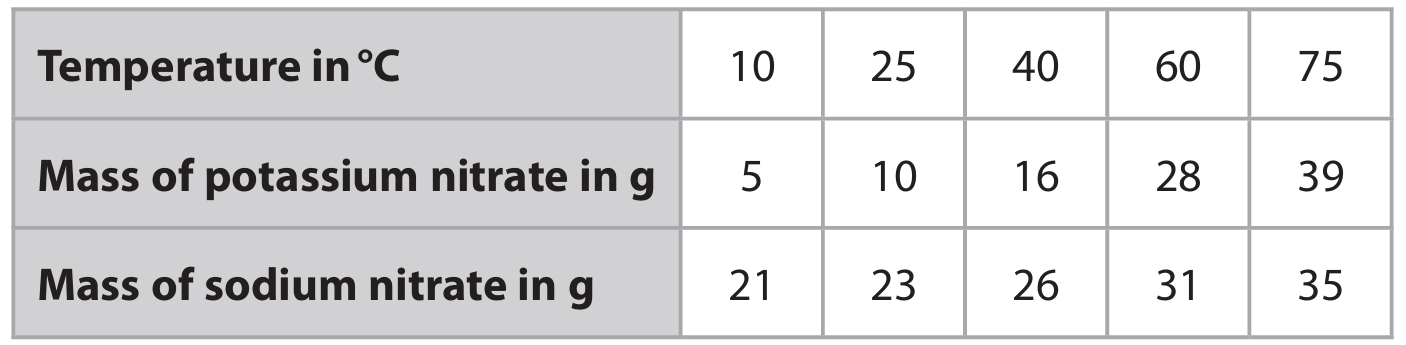
(a) (i) Plot the data for potassium nitrate and for sodium nitrate on the grid.
(a) (ii) Draw and label a curve of best fit for \(\text{KNO}_3\) and for \(\text{NaNO}_3\).
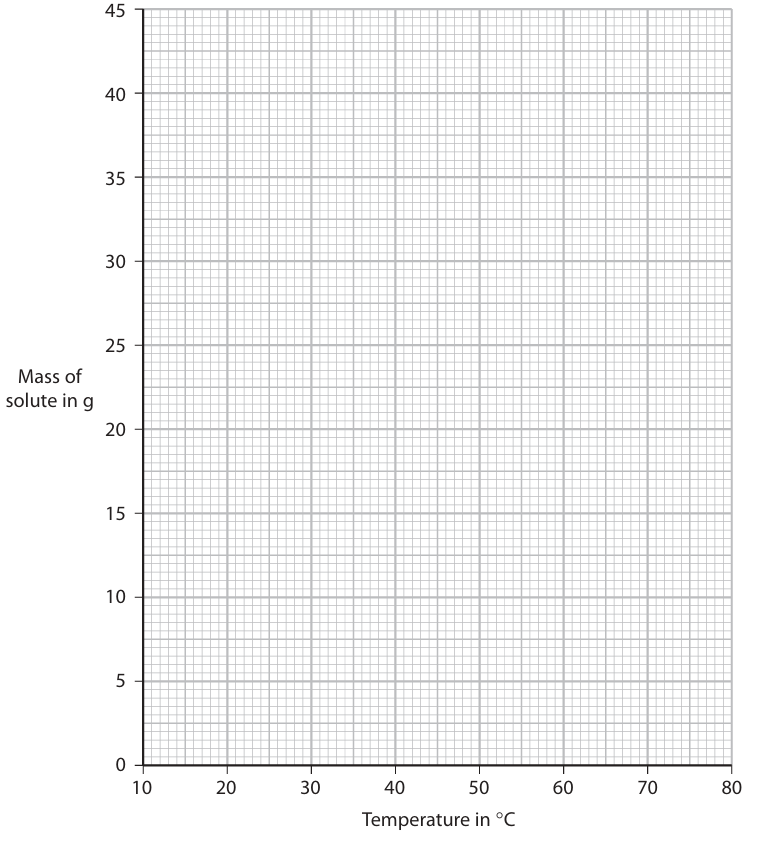
(b) The graph shows the temperature at which the maximum mass dissolved is the same for each solute. Determine this temperature.
(c) Use your graph to calculate the solubility, in g per 100 g of water, of sodium nitrate at 30 °C.
\(\left[ 1.0\ \text{cm}^3\ \text{of water has a mass of } 1.0\ \text{g} \right]\)
(d) \(25\ \text{cm}^3\) of a saturated solution of potassium nitrate is cooled from 50 °C to 20 °C.
Use your graph to determine the mass, in grams, of potassium nitrate that crystallises.
Show your working on the graph.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1.5C, 1.6C: Know what is meant by solubility (g per 100 g of solvent) and understand how to plot and interpret solubility curves — parts (a), (b), (c), (d)
• 1.7C practical: Investigate the solubility of a solid in water at a specific temperature — context of the question
• Mathematical Skills (Appendix 4): Plotting graphs, reading values, performing calculations — throughout
▶️ Answer/Explanation
(a) (i)
• M1: All points for \(\text{KNO}_3\) plotted correctly (to within ±½ small square).
• M2: All points for \(\text{NaNO}_3\) plotted correctly (to within ±½ small square).
(a) (ii)
• M1: Smooth curve of best fit drawn for \(\text{KNO}_3\).
• M2: Smooth curve of best fit drawn for \(\text{NaNO}_3\) and both curves clearly labelled.
Note: If curves are unlabelled or incorrectly labelled, lose 1 mark.
(b)
• Temperature where the two curves intersect.
• Expected value ≈ \(68^\circ \text{C}\).
(c)
• M1: Read mass of \(\text{NaNO}_3\) dissolved in \(25\ \text{cm}^3\) water at \(30^\circ \text{C}\) from graph. Expected ≈ \(24\ \text{g}\).
• M2: Convert to solubility in \( \text{g per 100 g water} \):
Since \(25\ \text{cm}^3\) water = \(25\ \text{g}\),
Solubility = \( \text{Mass from graph} \times 4 \).
Expected ≈ \(24 \times 4 = 96\ \text{g per 100 g water}\).
(d)
• M1: Read mass of \(\text{KNO}_3\) dissolved in \(25\ \text{cm}^3\) water at \(50^\circ \text{C}\) from graph. Expected ≈ \(21\ \text{g}\).
• M2: Read mass of \(\text{KNO}_3\) dissolved in \(25\ \text{cm}^3\) water at \(20^\circ \text{C}\) from graph. Expected ≈ \(8\ \text{g}\).
• M3: Mass crystallised = \( \text{M1} – \text{M2} \).
Expected ≈ \(21 – 8 = 13\ \text{g}\).
Note: Working must be shown on the graph for M1 and M2 to score.
Methanol, \( \text{CH}_3\text{OH} \), is the first member of the homologous series of alcohols.
(a) Give two characteristics of a homologous series.
(b) Methanol is heated with potassium dichromate(VI) and one other reagent. The methanol is oxidised to methanoic acid, HCOOH.
(i) Give the formula of the other reagent.
(ii) Give the colour change that occurs during the reaction.
(iii) Draw the displayed formula for methanol and for methanoic acid in the boxes.

(c) Methanol reacts with methanoic acid to form an ester.
Complete the equation for this reaction.
\[ \text{CH}_3\text{OH} + \text{HCOOH} \rightarrow \_\_\_\_\_\_\_\_\_\_ + \_\_\_\_\_\_\_\_\_\_ \]
(d) The structural formula of an ester is \( \text{CH}_3\text{COOCH}_2\text{CH}_2\text{CH}_2\text{CH}_3 \).
(i) What is the name of this ester?
- A. butyl ethanoate
- B. butyl methanoate
- C. ethyl butanoate
- D. methyl butanoate
(ii) Deduce the molecular formula of this ester.
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry 4CH1):
• 4(e): Alcohols — parts (b)(i), (b)(ii), (b)(iii) (Oxidation of alcohols, colour change with potassium dichromate(VI), displayed formulae)
• 4(g): Esters — parts (c), (d)(i), (d)(ii) (Esterification reaction, naming esters, molecular formula)
▶️ Answer/Explanation
(a) Any two from:
M1 same general formula
M2 same functional group
M3 similar chemical properties / characteristics
M4 trend in physical properties / characteristics
M5 consecutive members differ by a \(\text{CH}_2\) group
IGNORE same chemical properties; accept any trend in specified physical property.
(b)(i) \(\text{H}_2\text{SO}_4\) (sulfuric acid)
(b)(ii)
M1 (from) orange
M2 (to) green
(b)(iii)
M1 (methanol):
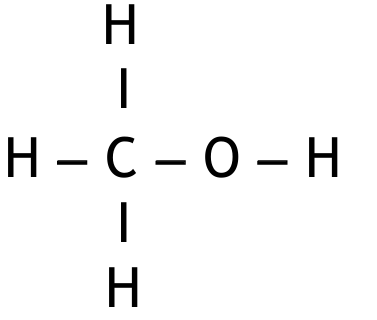
M2 (methanoic acid):
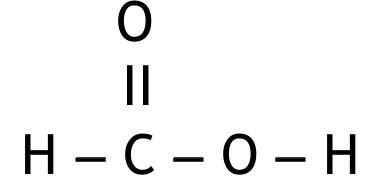
Must be in the correct order. Penalise once only if O–H bond not shown and both structures correct.
(c)
\[ \text{CH}_3\text{OH} + \text{HCOOH} \rightarrow \text{HCOOCH}_3 + \text{H}_2\text{O} \]
ALLOW multiples (e.g., 2CH\(_3\)OH). ALLOW \(\text{CH}_3\text{OOCCH}_3\). IGNORE state symbols even if incorrect. REJECT \(\text{CH}_3\text{COOH}\) and \(\text{C}_2\text{H}_4\text{O}_2\).
(d)(i) A (butyl ethanoate)
• B is not the answer because butyl methanoate would be \(\text{HCOOCH}_2\text{CH}_2\text{CH}_2\text{CH}_3\).
• C is not the answer because ethyl butanoate would be \(\text{CH}_3\text{CH}_2\text{CH}_2\text{COOCH}_2\text{CH}_3\).
• D is not the answer because methyl butanoate would be \(\text{CH}_3\text{CH}_2\text{CH}_2\text{COOCH}_3\).
(d)(ii) \(\text{C}_6\text{H}_{12}\text{O}_2\)
ALLOW symbols in any order.
A scientist reacts lead(II) nitrate solution with sodium bromide solution.
This is the equation for the reaction.
\[ \text{Pb(NO}_3)_2(aq) + 2\text{NaBr}(aq) \rightarrow \text{PbBr}_2(s) + 2\text{NaNO}_3(aq) \]
(a) Describe how the scientist could obtain a pure dry sample of lead(II) bromide (\(\text{PbBr}_2\)) from the reaction mixture.
(b) The scientist reacts an excess of lead(II) nitrate solution with 25 cm\(^3\) of sodium bromide solution of concentration 2.0 mol/dm\(^3\).
(i) Show that the amount of sodium bromide used is 0.050 mol.
(ii) Show that the maximum theoretical mass of lead(II) bromide is approximately 9 g. [for \(\text{PbBr}_2\), \(M_r = 367\)]
(c) The scientist electrolyses molten lead(II) bromide using this apparatus.
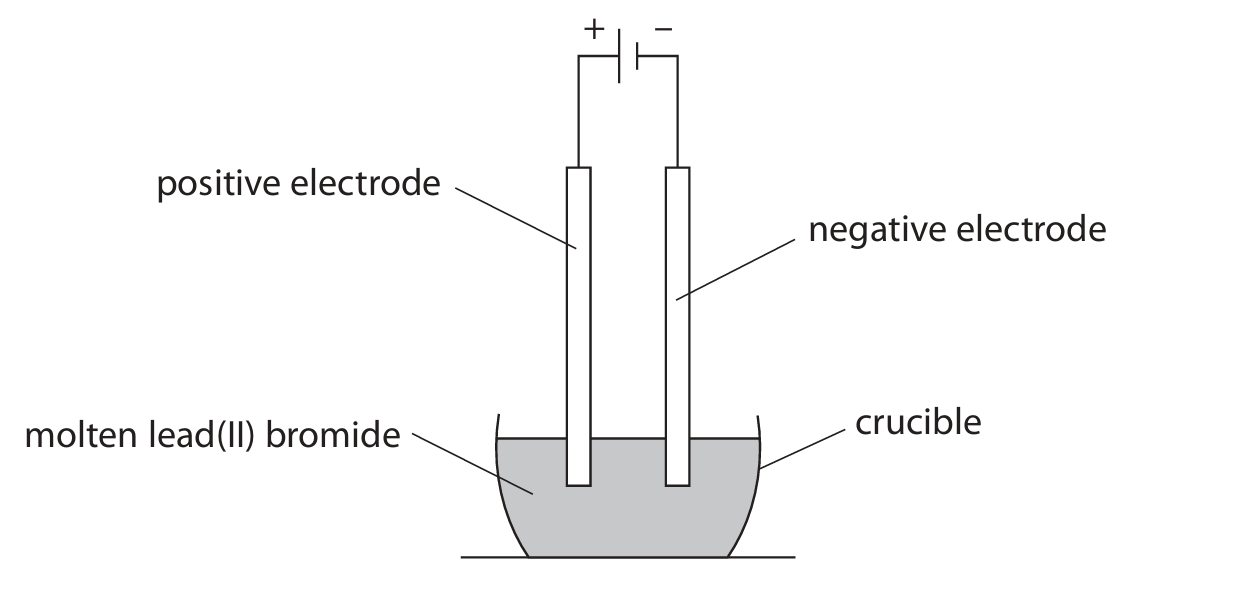
(i) Explain why lead(II) bromide needs to be molten rather than solid for electrolysis to occur.
(ii) The electrolyte is at a temperature of 400 °C. Explain a suitable material for the electrodes.
(iii) Give the half-equation that occurs at the negative electrode.
(d) Bromine forms at the positive electrode. This is the half-equation for the reaction at the positive electrode.
\[ 2\text{Br}^- \rightarrow \text{Br}_2 + 2\text{e}^- \]
(i) State what is observed at the positive electrode.
(ii) State why the half-equation represents an oxidation reaction.
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry 4CH1):
• 1(e): Chemical formulae, equations and calculations — parts (b)(i), (b)(ii)
• 1(i): Electrolysis — parts (c)(i), (c)(ii), (c)(iii), (d)
• 1(f): Ionic bonding — part (c)(i)
• 2(h): Chemical tests — part (d)(i)
• 1(e) & 2(d): Redox concepts — part (d)(ii)
▶️ Answer/Explanation
(a) An answer that refers to the following three points:
- M1: Filter (the mixture).
- M2: Wash (the precipitate/solid/lead(II) bromide with distilled water).
- M3: Suitable drying method, e.g., dry with filter paper / leave to dry / dry in a desiccator / dry in an oven.
Reject M3 if they attempt to crystallise the filtrate. If any attempt to evaporate the solution, allow MAX 1 mark.
(b)(i)
\[ \text{Amount} = \text{concentration} \times \text{volume} = 2.0 \, \text{mol/dm}^3 \times 0.025 \, \text{dm}^3 = 0.050 \, \text{mol} \]
Working must be shown.
(b)(ii)
From the equation, 2 mol NaBr produce 1 mol \(\text{PbBr}_2\).
Moles of \(\text{PbBr}_2 = 0.050 \div 2 = 0.025 \, \text{mol}\).
Mass = moles \(\times M_r = 0.025 \times 367 = 9.175 \, \text{g} \approx 9 \, \text{g}\).
Alternative calculation: \(0.050 \times 367 = 18.35 \, \text{g}\), then \(18.35 \div 2 = 9.175 \, \text{g}\). All working must be shown.
(c)(i) An explanation that links the following three points:
- M1: When solid, the ions are in fixed positions/in a lattice.
- M2: So there are no ions/charged particles free to move.
- M3: When molten, the ions are free to move so can conduct electricity/carry a current.
Ignore “carry charge”. Reject “solution” for M3. Reject “electrons moving/delocalised electrons” for M3.
(c)(ii) An explanation that links the following two points:
- M1: Graphite (or platinum).
- M2: It has a high melting point / is resistant to high temperature / is inert / conducts electricity.
M2 is dependent on M1 being a suitable material.
(c)(iii)
\[ \text{Pb}^{2+} + 2\text{e}^- \rightarrow \text{Pb} \]
Ignore state symbols.
(d)(i) Brown vapour/gas/fumes.
Allow red-brown vapour/gas/fumes. Reject orange/orange-brown/red alone.
(d)(ii) Bromide ions (\(\text{Br}^-\)) lose electrons.
Allow “electrons are lost”. Reject “bromine loses electrons”.
This question is about hydrogen chloride (HCl). Hydrogen chloride is a covalent compound.
(a) State, in terms of electrostatic attraction, what is meant by a covalent bond.
(b) When hydrogen chloride gas is dissolved in an organic solvent, the hydrogen chloride remains a covalent molecule.
When hydrogen chloride gas is dissolved in water, ions are formed.
Explain what happens when dry blue litmus paper is dipped into separate samples of each solution.
(c) In the presence of ultraviolet radiation, hydrogen reacts with chlorine to form hydrogen chloride. This is the equation for the reaction:
\[ \text{H}_2(\text{g}) + \text{Cl}_2(\text{g}) \rightarrow 2\text{HCl}(\text{g}) \]
The table shows the bond energies.
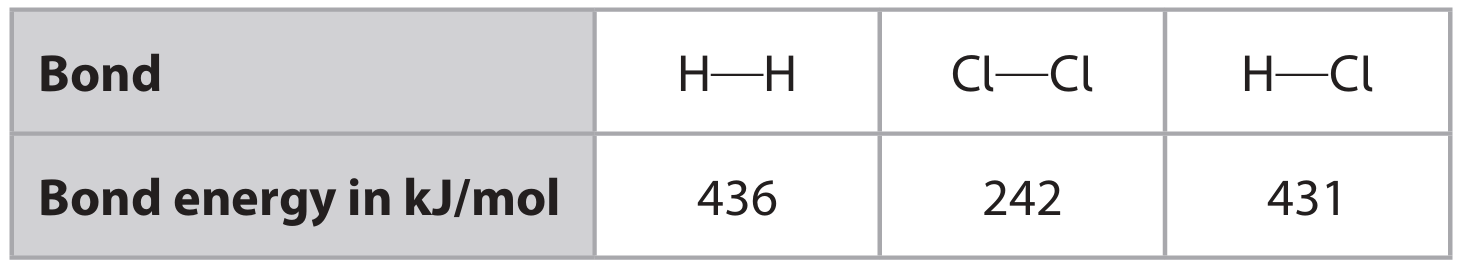
(i) Calculate the enthalpy change (\(\Delta H\)), in kJ/mol, for the reaction. Include a sign in your answer.
(ii) Draw a reaction profile for the reaction. Label the reactants, the products, \(\Delta H\) and the activation energy (\(E_a\)).
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 2(f)(g): Acids, alkalis and titrations / Acids, bases and salt preparations — part (b)
• 3(a): Energetics — parts (c)(i) and (c)(ii)
• 1(i): Electrolysis — part (b) – related to ionic dissociation in water
▶️ Answer/Explanation
(a)
M1: (Electrostatic) attraction between nuclei (of both atoms) [nuclei must be plural]
M2: and a shared/bonding pair of electrons
OR
M1: (Electrostatic) attraction between a shared/bonding pair of electrons
M2: and nuclei (of both atoms) [nuclei must be plural]
(b)
An explanation that links the following four points:
M1: (In the organic solvent) litmus paper stays blue/has no change [No M1 or M2 if litmus paper turns red]
M2: because there are no (H⁺) ions / the solution is not acidic / does not dissociate in an organic solvent
M3: (In the aqueous solution) litmus paper turns red [REJECT if litmus is bleached or turns white]
M4: because H⁺ ions are formed / hydrochloric acid forms [M4 dependent on litmus turning red initially]
(c)(i)
M1: Reactants bond energy = \(436 + 242 = 678\) kJ
M2: Products bond energy = \(2 \times 431 = 862\) kJ
M3: \(\Delta H = 678 – 862 = -184\) kJ/mol [Correct answer –184 with or without working scores 3; (+)184 scores 2]
(c)(ii)
Reaction profile should include:
M1: Correct positions of horizontal lines for reactants and products, with an activation energy hump in between.
M2: Correct labelling of reactants (\(H_2 + Cl_2\)) and products (\(2HCl\)).
M3: A vertical line in the correct position labelled \(\Delta H\) (arrow pointing down or double-headed arrow; REJECT arrow pointing up).
M4: A vertical line in the correct position labelled \(E_a\) or activation energy (arrow pointing up or double-headed arrow; REJECT arrow pointing down).
Note: ECF (error carried forward) allowed for incorrect values in (c)(i).
