This question is about sulfur.
(a) The diagram represents the electronic configuration of an atom of sulfur.
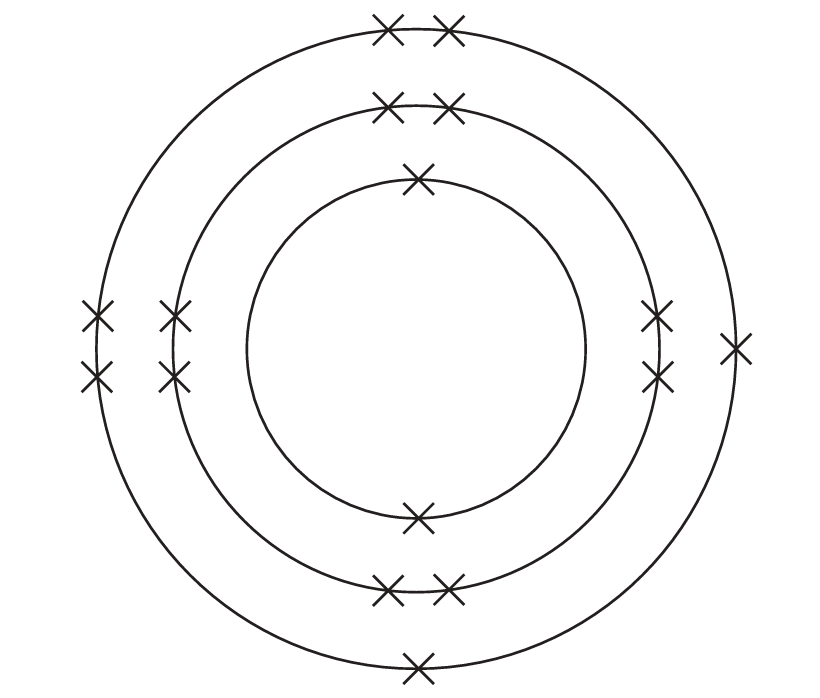
(i) Which period of the Periodic Table contains sulfur?
- A) 2
- B) 3
- C) 4
- D) 6
(ii) What is the charge on a sulfide ion?
- A) \(1+\)
- B) \(2+\)
- C) \(1-\)
- D) \(2-\)
(b) When a mixture of sulfur and zinc is heated to a high temperature a reaction occurs, forming the compound zinc sulfide, \(\text{ZnS}\).
(i) Give a reason why the mixture of sulfur and zinc needs heating before a reaction occurs.
(ii) Calculate the relative formula mass (\(M_r\)) of zinc sulfide.
\[ \text{[for Zn, } A_r = 65 \quad \text{for S, } A_r = 32] \]
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry 4CH1):
• 1(f): Ionic bonding — part (a)(ii)
• 1(e): Chemical formulae, equations and calculations — part (b)(ii)
• 3(b): Rates of reaction — part (b)(i)
▶️ Answer/Explanation
(a)(i) B (3)
A is not the correct answer because sulfur is not in period 2
C is not the correct answer because sulfur is not in period 4
D is not the correct answer because sulfur is not in period 6
(a)(ii) D (\(2-\))
A is not the correct answer because the charge on a sulfide ion is not \(1+\)
B is not the correct answer because the charge on a sulfide ion is not \(2+\)
C is not the correct answer because the charge on a sulfide ion is not \(1-\)
(b)(i) The reaction has a high activation energy.
Alternative acceptable answers:
• To give zinc and sulfur enough energy to react
• Speeds reaction up / increases rate of reaction / reacts quicker
• The reaction is very slow at room temperature
• Gains kinetic energy
(b)(ii) \(M_r(\text{ZnS}) = 65 + 32 = 97\)
No units required.
(a) The diagram represents an atom of an element.
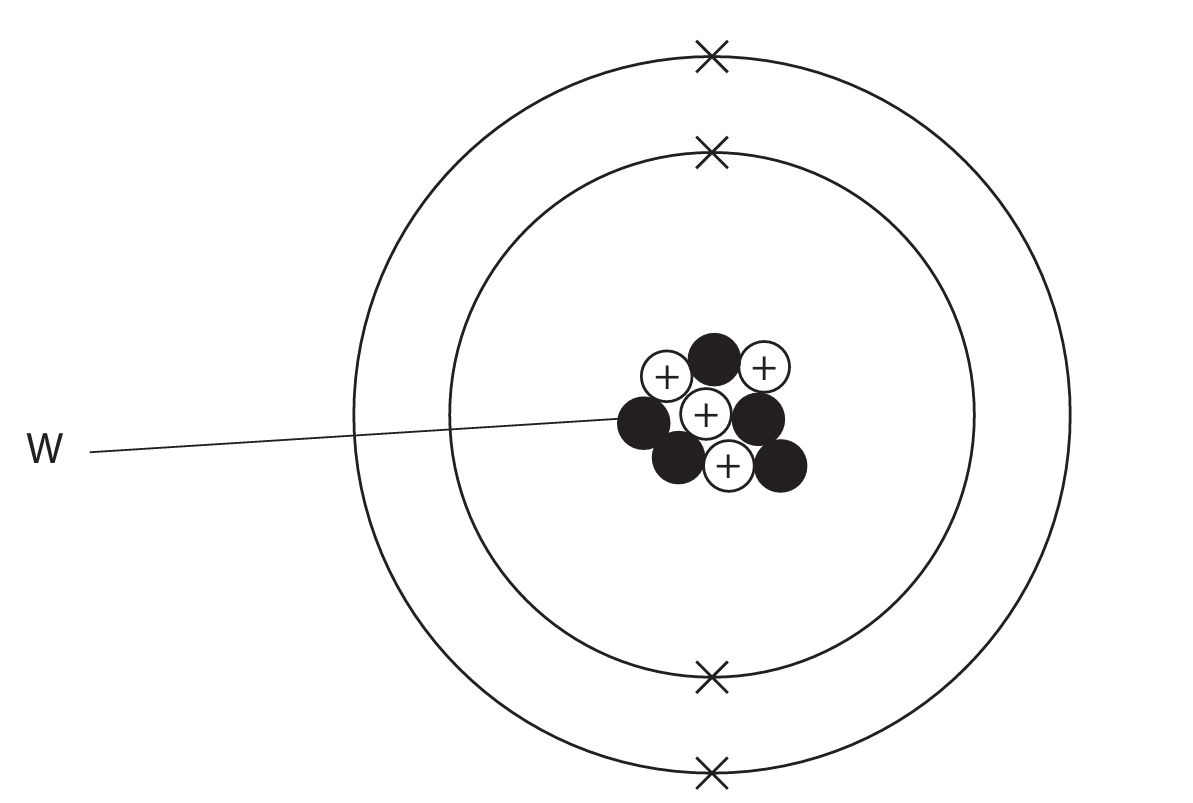
(i) What is the name of the particle labelled W?
- A) electron
- B) ion
- C) neutron
- D) proton
(ii) What is the mass number of this atom?
- A) 4
- B) 5
- C) 9
- D) 13
(b) These are the symbols for the two isotopes of lithium.
\(_3^6Li \quad _3^7Li\)
(i) In terms of sub-atomic particles, give one similarity and one difference between the two isotopes.
(ii) A sample of lithium contains 7.5% of \(_3^6Li\) and 92.5% of \(_3^7Li\)
Calculate the relative atomic mass (\(A_r\)) of this sample of lithium.
Most-appropriate topic codes (Pearson Edexcel IGCSE Chemistry 4CH1):
• 1(c): Isotopes — parts (b)(i), (b)(ii)
• 1(c): Relative atomic mass calculation — part (b)(ii)
▶️ Answer/Explanation
2 (a) (i) C (neutron)
- A is not the correct answer because electrons are not in the nucleus.
- B is not the correct answer as ions are not in the nucleus.
- D is not the correct answer as protons have a positive charge.
(ii) C (9)
- A is not the correct answer because the proton number = 4.
- B is not the correct answer as there are 5 neutrons.
- D is not the correct answer because 13 is the number of protons + neutrons + electrons.
(b) (i)
similarity = number of protons / proton number / atomic number
difference = number of neutrons
ALLOW number of electrons.
ALLOW one has 3 neutrons and the other has 4 neutrons.
IGNORE reference to atomic number and mass numbers.
(ii)
M1 \((7.5 \times 6) + (92.5 \times 7) \div 100\)
M2 \(6.925 / 6.93 / 6.9\)
ALLOW 2 or more significant figures.
Answer \(6.925 / 6.93 / 6.9\) without working scores 2 marks.
Answer of 7 with correct method scores M1.
Answer of 7 without working scores 0.
This question is about magnesium metal and its compounds.
(a) (i) The diagram represents the structure of magnesium.
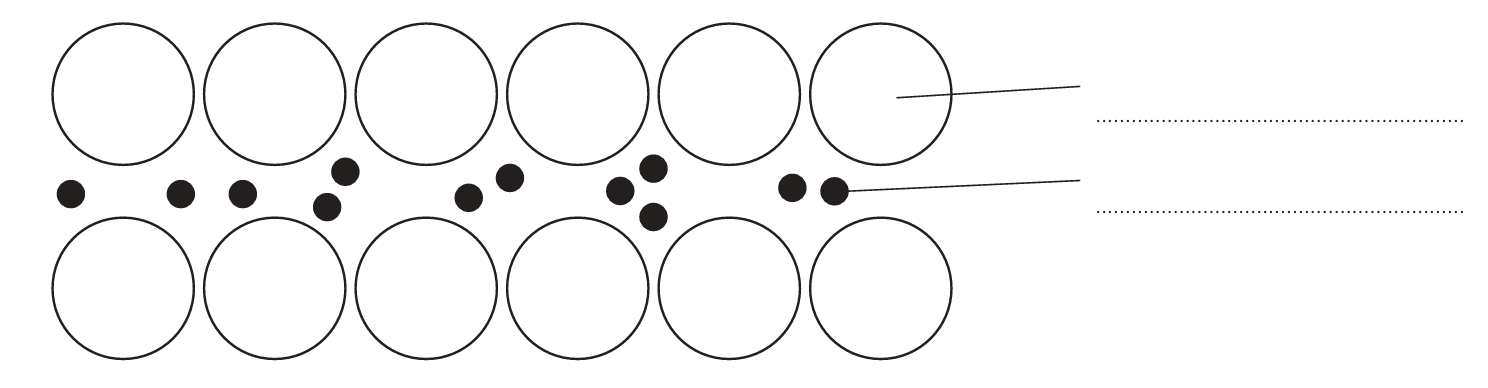
Complete the diagram by adding the labels.
(a) (ii) Explain why magnesium is malleable.
(b) (i) Magnesium burns in oxygen gas to form solid magnesium oxide.
Complete the chemical equation for the reaction by balancing the equation and adding the state symbols.
\[ \text{Mg(s)} + \text{O}_2(\text{……}) \rightarrow \text{MgO}(\text{……}) \]
(b) (ii) Give a reason why magnesium is oxidised in this reaction.
(c) (i) A student uses this method to make a solution of magnesium chloride.
Step 1: measure 25 cm\(^3\) of dilute hydrochloric acid into a beaker
Step 2: add magnesium powder a little at a time
Step 3: keep adding magnesium powder until it is in excess
Step 4: remove the excess magnesium powder by filtration
This is the equation for the reaction.
\[ \text{Mg(s)} + 2\text{HCl(aq)} \rightarrow \text{MgCl}_2(\text{aq}) + \text{H}_2(\text{g}) \]
Give a reason why the student uses magnesium powder rather than magnesium ribbon.
(c) (ii) Give a reason why the student adds an excess of magnesium in step 3.
(c) (iii) Calculate the minimum mass of magnesium needed to react with 25.0 cm\(^3\) of 2.00 mol/dm\(^3\) hydrochloric acid.
[for Mg, \(A_r = 24\)]
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(e): Chemical formulae, equations and calculations — parts (b)(i), (c)(iii)
• 2(d): Reactivity series — part (b)(ii)
• 2(f): Acids, alkalis and titrations — parts (c)(i), (c)(ii), (c)(iii)
• 2(g): Acids, bases and salt preparations — part (c)(i)
▶️ Answer/Explanation
(a)(i)
M1: (positive) ion / cation
M2: (delocalised) electron
(a)(ii)
M1: Layers (of atoms/ions) / particles.
M2: (Atoms/ions) can slide over one another.
(b)(i)
\[ 2\text{Mg(s)} + \text{O}_2(\text{g}) \rightarrow 2\text{MgO(s)} \]
M1: Balancing.
M2: State symbols.
(b)(ii)
Magnesium gains oxygen.
ALLOW magnesium loses electrons.
(c)(i)
To increase the rate of reaction / increases surface area.
(c)(ii)
To ensure all the acid has reacted / to neutralise all the acid.
(c)(iii)
M1: Amount of HCl = \( (25 \times 2) \div 1000 = 0.050 \text{ mol} \)
M2: Amount of Mg = \( 0.050 \div 2 = 0.025 \text{ mol} \)
M3: Mass of Mg = \( 0.025 \times 24 = 0.60 \text{ g} \)
Answer of 0.60 g without working scores 3.
This question is about the manufacture of ethanol by the fermentation of glucose.
(a) Fermentation needs to be done in the absence of air.
Give two other conditions needed for fermentation.
(b) Explain why fermentation needs to be done in the absence of air.
(c) (i) Complete the equation for fermentation.
\[C_6H_{12}O_6 \rightarrow 2C_2H_5OH + 2\]
(c) (ii) A mixture containing 500 mol of glucose is fermented.
A mass of 5750 g of ethanol is obtained.
Calculate the percentage yield.
[For ethanol, \( M_r = 46 \)]
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(e) Chemical formulae, equations and calculations: Mole calculations and percentage yield — part (c)(ii)
• 4(e) Alcohols: Oxidation of ethanol — part (b)
▶️ Answer/Explanation
(a)
1. Temperature of about \(30^\circ C\) / optimum temperature
2. (Enzymes from) yeast / zymase
(b)
An explanation that makes reference to:
• Oxygen in the air would react with / oxidise the ethanol (1)
• To form ethanoic acid (or vinegar) / carbon dioxide and water (1)
(c) (i) \[C_6H_{12}O_6 \rightarrow 2C_2H_5OH + 2CO_2\]
(c) (ii) \(12.5\%\)
Working:
Method 1:
M1: Theoretical moles of ethanol from glucose = \(500 \times 2 = 1000\) mol
M2: Theoretical mass of ethanol = \(1000 \times 46 = 46000\) g
M3: Percentage yield = \(\frac{5750}{46000} \times 100 = 12.5\%\)
Method 2:
M1: Moles of ethanol obtained = \(\frac{5750}{46} = 125\) mol
M2: Theoretical moles of ethanol = \(500 \times 2 = 1000\) mol
M3: Percentage yield = \(\frac{125}{1000} \times 100 = 12.5\%\)
Sodium sulfate can be prepared by the reaction between sodium hydroxide solution and sodium hydrogensulfate \((\text{NaHSO}_4)\) solution.
This is the equation for the reaction.
\[ \text{NaOH(aq)} + \text{NaHSO}_4\text{(aq)} \rightarrow \text{Na}_2\text{SO}_4\text{(aq)} + \text{H}_2\text{O(l)} \]
Sodium hydrogensulfate solution is acidic.
A student adds \(25.0\ \text{cm}^3\) of sodium hydroxide solution to a conical flask and adds two drops of indicator.
The student does a titration.
(a) (i) Name a piece of apparatus that should be used to add \(25.0\ \text{cm}^3\) of sodium hydroxide solution to the conical flask.
(a) (ii) Describe the method the student should use to find the accurate volume of sodium hydrogensulfate solution needed to neutralise the \(25.0\ \text{cm}^3\) of sodium hydroxide solution.
(b) The student repeats the titration without the indicator and forms a solution of sodium sulfate.
Describe how the student can obtain pure, dry crystals of hydrated sodium sulfate from the solution.
(c) Crystals of hydrated sodium sulfate decompose when heated.
This is the equation for the decomposition.
\[ \text{Na}_2\text{SO}_4\cdot x\text{H}_2\text{O} \rightarrow \text{Na}_2\text{SO}_4 + x\text{H}_2\text{O} \]
A student uses this apparatus to find the value of \(x\).
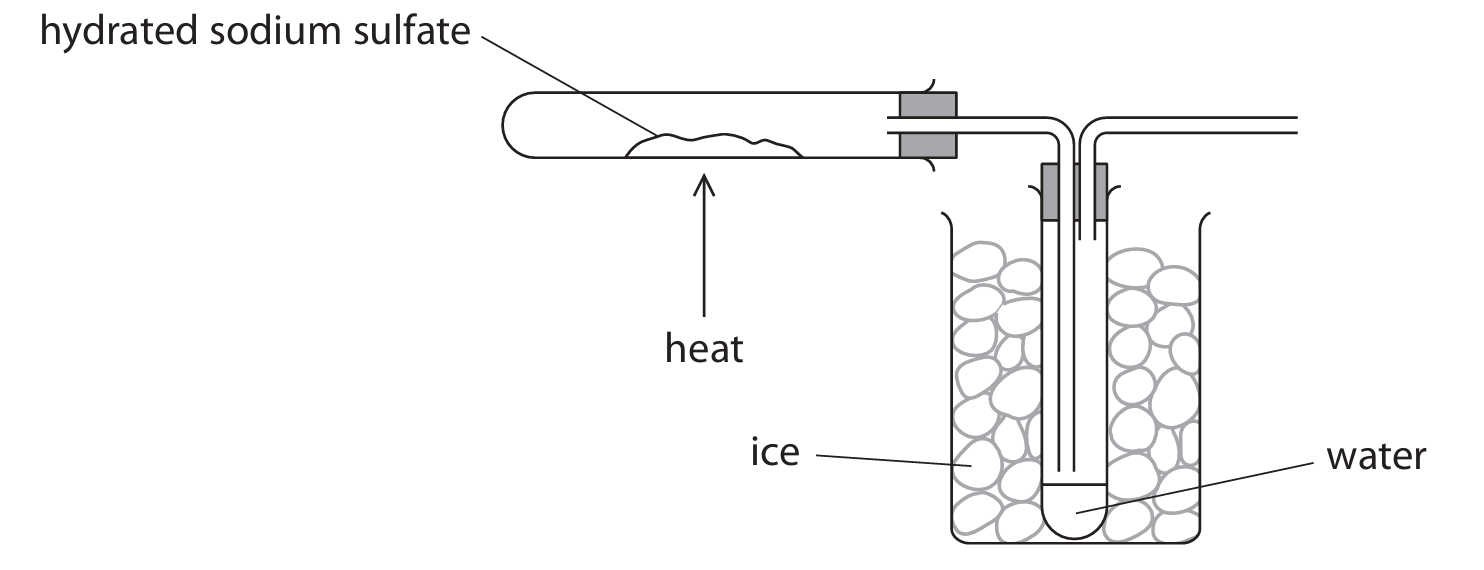
The student heats the crystals until the decomposition is complete.
The table shows the student’s results.

Use the results to calculate the value of \(x\).
[for \(\text{Na}_2\text{SO}_4\), \(M_r = 142\) for \(\text{H}_2\text{O}\), \(M_r = 18\)]
Most-appropriate topic codes (Edexcel International GCSE Chemistry 4CH1):
• 2(g): Acids, bases and salt preparations — part (b)
• 1(e): Chemical formulae, equations and calculations — part (c)
• 1(e): Reacting masses and empirical formulae — part (c)
▶️ Answer/Explanation
(a)(i) (Volumetric) pipette
(a)(ii)
- Fill a burette with sodium hydrogensulfate solution.
- Add the sodium hydrogensulfate solution to the conical flask while swirling it continuously.
- Add the solution dropwise near the endpoint when the indicator colour change is about to occur.
- Stop when the indicator permanently changes colour.
- Record the volume used and repeat the titration to obtain concordant results.
(b)
- Heat the solution until crystals first start to form (or until the solution is saturated).
- Allow the solution to cool and crystallise.
- Filter the mixture to collect the crystals.
- Leave the crystals in a warm place to dry (or dry between filter paper).
(c)
Mass of hydrated salt = \(31.56 – 25.12 = 6.44\ \text{g}\)
Mass of anhydrous \(\text{Na}_2\text{SO}_4\) = \(27.96 – 25.12 = 2.84\ \text{g}\)
Mass of water lost = \(6.44 – 2.84 = 3.60\ \text{g}\)
Moles of \(\text{Na}_2\text{SO}_4\) = \(\frac{2.84}{142} = 0.0200\ \text{mol}\)
Moles of \(\text{H}_2\text{O}\) = \(\frac{3.60}{18} = 0.200\ \text{mol}\)
Ratio \(\text{Na}_2\text{SO}_4 : \text{H}_2\text{O} = 0.0200 : 0.200 = 1 : 10\)
Therefore \(x = 10\).
This question is about electrolysis.
(a) This apparatus is used to collect the gases produced when an electric current passes through dilute sulfuric acid.
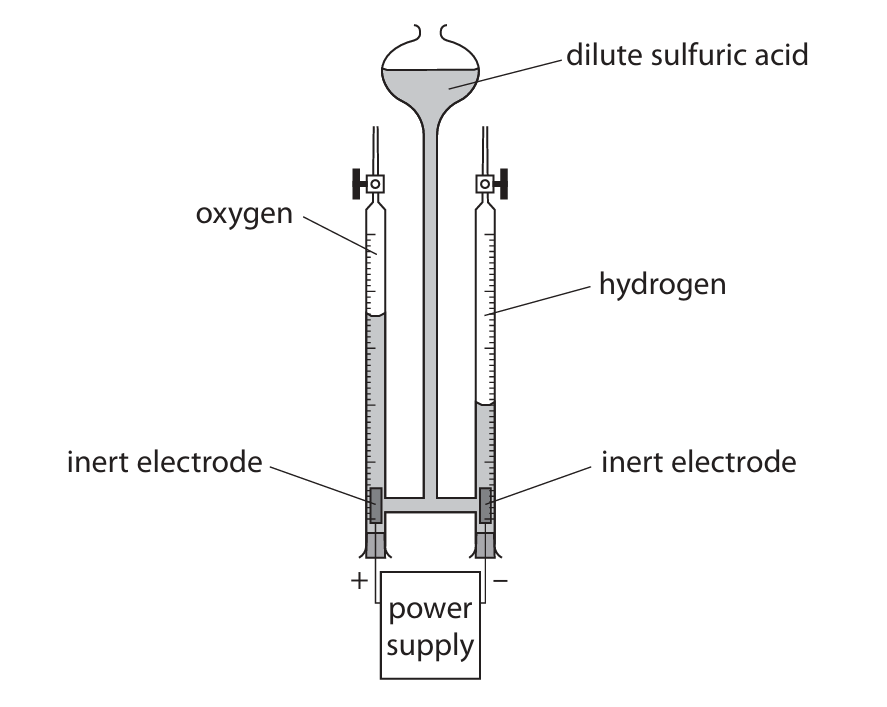
(i) Name a suitable material for the inert electrodes.
(ii) Give a test to show that the gas produced at the positive electrode is oxygen.
(iii) Give a test to show that the gas produced at the negative electrode is hydrogen.
(iv) Give an ionic half-equation for the formation of hydrogen at the negative electrode.
(v) The oxygen gas in the tube has a volume of \( 17.8 \, \text{cm}^3 \) at rtp.
Calculate the mass, in grams, of oxygen gas in the tube.
Give your answer to 3 significant figures.
[For \( O_2 \) at rtp, molar volume = \( 24\,000 \, \text{cm}^3 \)]
(b) This apparatus is used to electrolyse a concentrated solution of sodium chloride.
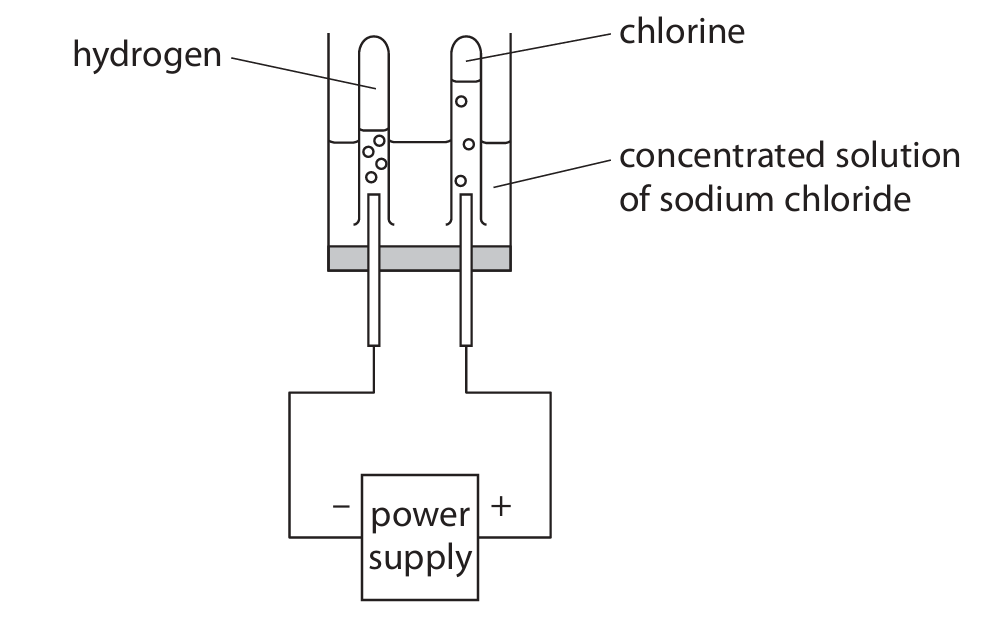
(i) Give a reason why sodium chloride solution conducts electricity.
(ii) The positive ions in the solution are \( H^+ \) and \( Na^+ \).
Give a reason why hydrogen forms at the negative electrode rather than sodium.
(iii) A sample of the solution is taken from near the negative electrode.
The sample is tested with universal indicator.
Explain the final colour of the universal indicator.
Most-appropriate topic codes (Edexcel IGCSE Chemistry 4CH1):
• 1(c): Atomic structure — Part (a)(iv), understanding ions and electron transfer
• 1(e): Chemical formulae, equations and calculations — Part (a)(v), mole and volume calculations
• 2(g): Acids, alkalis and titrations / 2(h): Chemical tests — Parts (a)(ii), (a)(iii), (b)(iii)
• 2(f): Acids, bases and salt preparations — Part (b)(iii), formation of alkali
• 2(d): Reactivity series — Part (b)(ii), relative ease of reduction
▶️ Answer/Explanation
(a)(i) platinum
ALLOW silver / gold / graphite
(a)(ii) glowing splint relights
(a)(iii) lit splint (with a squeaky) pop
(a)(iv) \( 2H^+ + 2e^- \rightarrow H_2 \)
(a)(v) \( 0.0237 \, \text{g} \)
Working:
M1 (moles of oxygen) \( \frac{17.80}{24000} = 0.000741\overline{6} \)
M2 \( M_r \) of oxygen \( (O_2) = 32 \)
M3 Mass = moles × \( M_r = 0.000741\overline{6} \times 32 = 0.02373 \, \text{g} \)
To 3 s.f. = \( 0.0237 \, \text{g} \)
Correct answer to 3 s.f. without working scores 3. ALLOW ecf from M1 and M2; answer must be to 3 s.f. for M3.
(b)(i) ions can move / are mobile (OWTTE)
(b)(ii) Hydrogen is less reactive than sodium / Hydrogen is below sodium in the reactivity series.
ALLOW H⁺ ions are more easily reduced / accept electrons more easily than Na⁺ ions.
(b)(iii)
M1 The universal indicator turns blue/purple.
M2 OH⁻ / hydroxide ions are present in solution.
ALLOW the solution is sodium hydroxide / the solution is alkaline.
Gaseous ethanol is manufactured by reacting ethene gas with steam. This is the equation for the reaction:
\[ \text{C}_2\text{H}_4(g) + \text{H}_2\text{O}(g) \rightleftharpoons \text{C}_2\text{H}_5\text{OH}(g) \]
(a) (i) The pressure on an equilibrium mixture of the three gases is decreased. All other conditions are kept the same. Predict the effect of this change on the yield of ethanol at equilibrium, giving a reason for your answer.
(a) (ii) Predict the effect of adding a catalyst on the yield of ethanol at equilibrium, giving a reason for your answer.
(b)The equation shows the displayed formulae for the reactants and products.
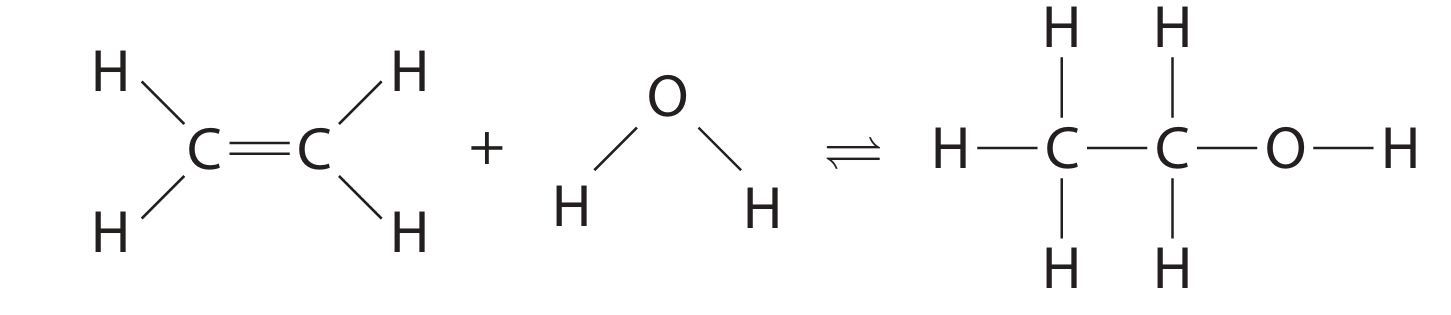
The table gives the bond energies.
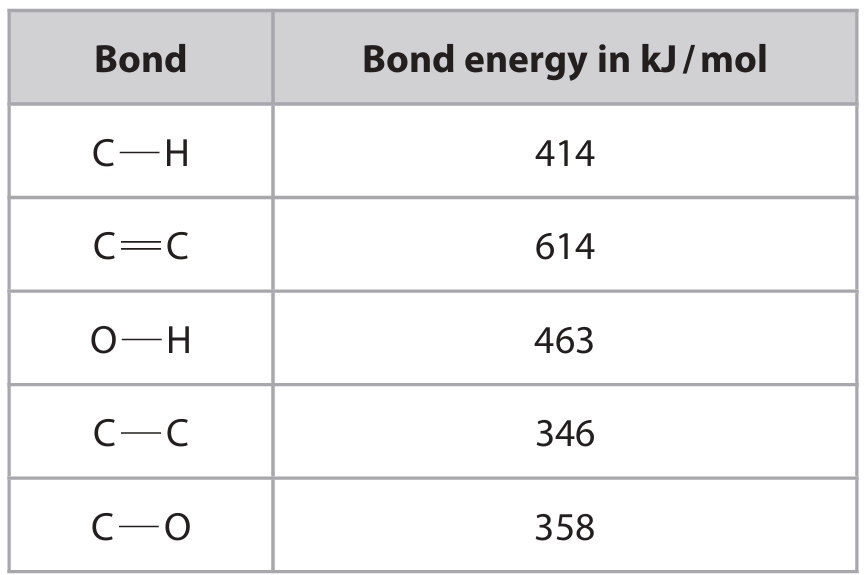
(i) Using the data and the equation, show that the enthalpy change, \(\Delta H\), is approximately \(-40 \, \text{kJ/mol}\).
(ii) Explain, in terms of bonds broken and bonds made, why this reaction is exothermic.
(iii) Draw an energy level diagram for the reaction between ethene and steam. Label the enthalpy change, \(\Delta H\).

(c) Ethanoic acid reacts with ethanol to form ester A and water.
(i) Ethanoic acid reacts with ethanol to form ester A and water. Complete the equation by adding the displayed formula of ester A.
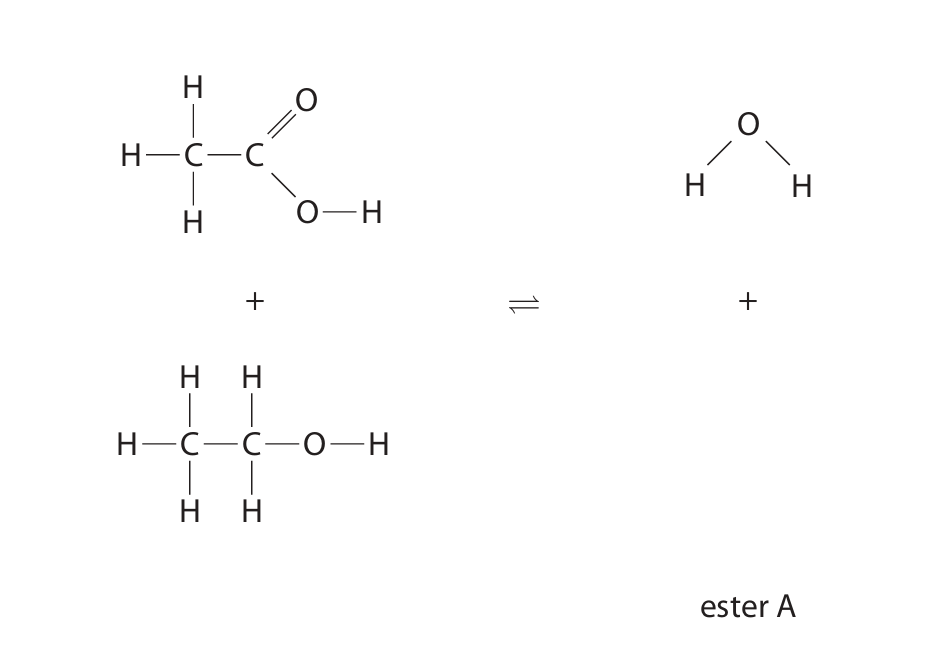
(ii) Give the name of ester A.
Most-appropriate topic codes (Edexcel IGCSE Chemistry):
• 3(a): Energetics — part (b)(i), (b)(ii), (b)(iii)
• 4(g): Esters — part (c)
• 1(g): Covalent bonding — bond energy calculations
• 4(e): Alcohols — ethanol production and reactions
▶️ Answer/Explanation
(a)(i)
• Yield decreases / equilibrium shifts to the left / towards reactants. (M1)
• Because decreasing pressure favors the side with more moles of gas (2 moles on left vs 1 mole on right). (M2)
(a)(ii)
• The yield of ethanol does not change. (M1)
• A catalyst increases the rate of both forward and reverse reactions equally, so equilibrium position is unchanged. (M2)
(b)(i)
• Bonds broken: \(4 \times 414 + 614 + 2 \times 463 = 3196 \, \text{kJ/mol}\) (M1)
• Bonds formed: \(5 \times 414 + 346 + 358 + 463 = 3237 \, \text{kJ/mol}\) (M2)
• \(\Delta H = 3196 – 3237 = -41 \approx -40 \, \text{kJ/mol}\) (M3)
Alternative method using only bonds that change is also acceptable.
(b)(ii)
• More energy is released when new bonds are formed than is absorbed when old bonds are broken. (M1)
• Therefore, the reaction is exothermic. (M2)
(b)(iii)
• Diagram with reactants’ energy level higher than products’. (M1)
• Axes labelled: “Energy” (y), “Reaction progress” (x). (M2)
• \(\Delta H\) shown as a downward arrow labelled “\(\Delta H = -40 \, \text{kJ/mol}\)”. (M3)
(c)(i)
• Ester linkage shown correctly: —O—CO— (M1)
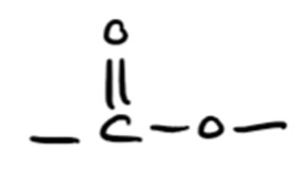
• Full displayed formula: \[ \text{H}—\text{C}—\text{C}—\text{O}—\text{C}—\text{C}—\text{H} \quad \text{with correct H atoms} \] (M2)
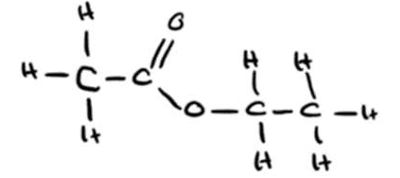
(c)(ii)
• Ethyl ethanoate (M1)
