Topic: 2.1
(a) Animal cells, plant cells and prokaryotic cells have similarities and differences in their structure.
Table 1.1 lists five organelles found in cells.
Complete Table 1.1 by placing a tick (✓) to show whether the organelle is present in animal cells, plant cells and prokaryotic cells or a cross (✗) if the organelle is absent.
Put a tick (✓) or a cross (✗) in every box.
The first row has been completed for you.
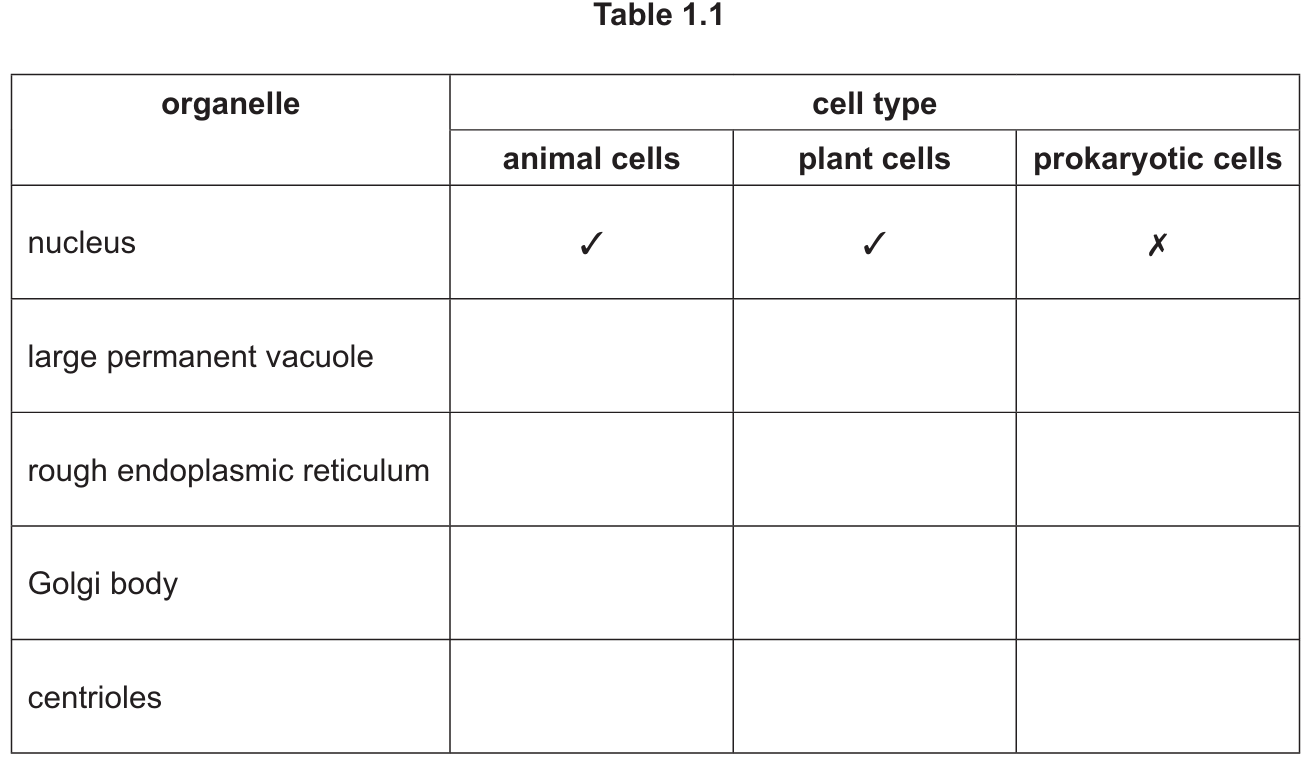
(b) Fig. 1.1 shows a section through part of an epithelial cell found in the digestive system of an animal.
The cell is specialised for absorption of digested food.
The structures labelled P and Q in Fig. 1.1 are involved in the absorption of digested food.
(i) Name the structures labelled P.
(ii) Explain how the organelle labelled Q in Fig. 1.1 is involved in this process.
▶️ Answer/Explanation
(a)
Completed Table 1.1:
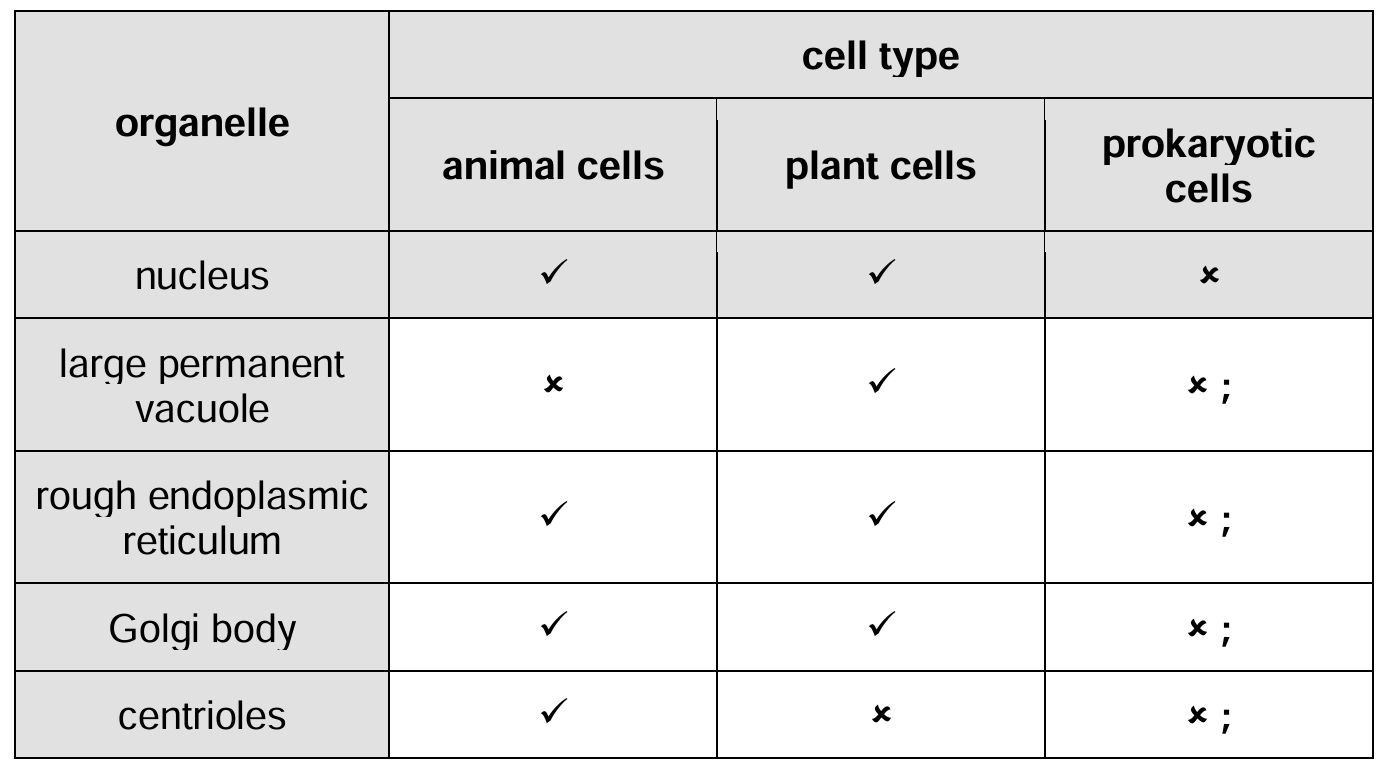
Explanation: The table shows the presence or absence of organelles in different cell types:
- Large permanent vacuole: Present only in plant cells (helps maintain turgor pressure and stores nutrients)
- Rough endoplasmic reticulum: Present in eukaryotic cells (animal and plant) but absent in prokaryotes (site of protein synthesis)
- Golgi body: Present in eukaryotic cells (modifies and packages proteins) but absent in prokaryotes
- Centrioles: Present only in animal cells (involved in cell division), absent in plants and prokaryotes
(b)(i) Microvilli
Explanation: Structure P is identified as microvilli, which are finger-like projections on the surface of epithelial cells in the digestive system. These greatly increase the surface area for absorption of digested food molecules.
(b)(ii)
Explanation: Organelle Q is the mitochondrion, which plays a crucial role in absorption through two main mechanisms:
- Mitochondria are the site of aerobic respiration, producing ATP which provides energy for active transport of digested food molecules against concentration gradients.
- The ATP generated is used for processes like endocytosis or pinocytosis, where larger molecules are transported into the cell through bulk transport mechanisms.
Without sufficient mitochondria, the epithelial cell wouldn’t have enough energy to efficiently absorb nutrients from the digestive tract into the bloodstream.
Topic: 8.1
(a) In the mammalian circulatory system, red blood cells travel through different types of blood vessel as they pass from the heart to respiring tissues and back to the heart.
Fig. 2.1 shows the types of blood vessels through which red blood cells travel in the circulatory system.
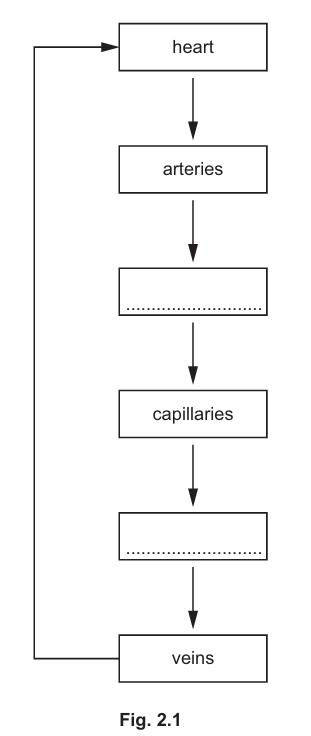
Complete Fig. 2.1 by writing the names of the missing types of blood vessels through which red blood cells travel.
(b) Water is the main component of blood. It has an important role in the transport of substances around the body.
Fig. 2.2 shows the ionic compound sodium chloride dissolving in water.
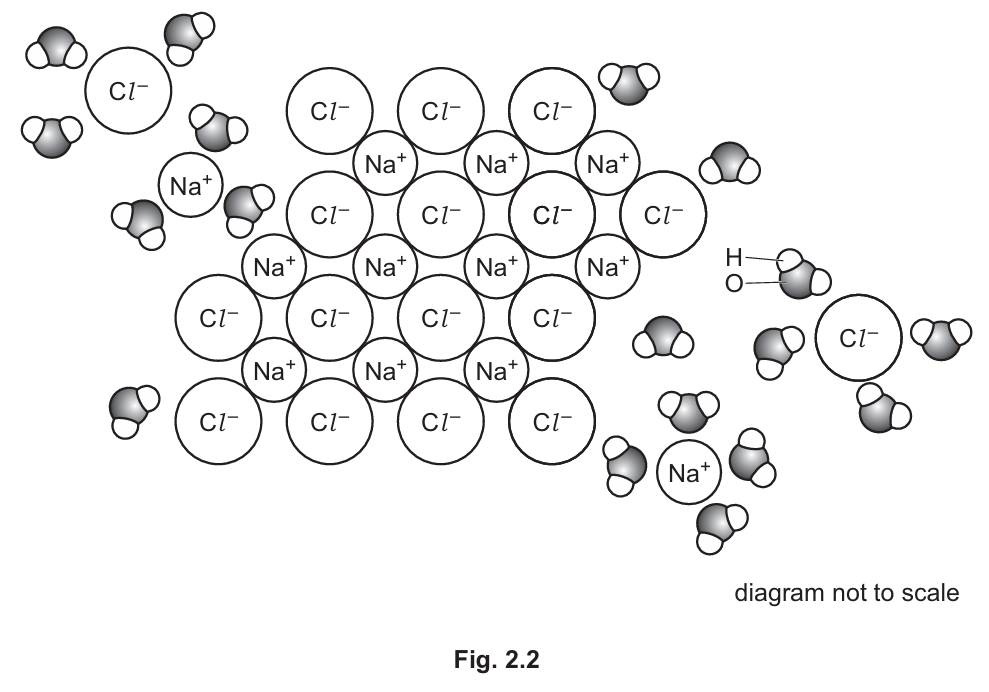
With reference to Fig. 2.2, explain how water acts as a solvent for sodium chloride.
(c) Fig. 2.3 shows a Galapagos penguin, Spheniscus mendiculus, swimming in the water.

Penguins are birds that live on land but spend a lot of time swimming underwater hunting for food. Penguins can remain underwater for up to twenty minutes. During this time they do not breathe but their tissues continue to respire.
Haemoglobin in the red blood cells of penguins has a higher affinity for oxygen than haemoglobin in other birds that do not swim underwater.
Fig. 2.4 shows the oxygen dissociation curve for a bird that does not swim underwater.
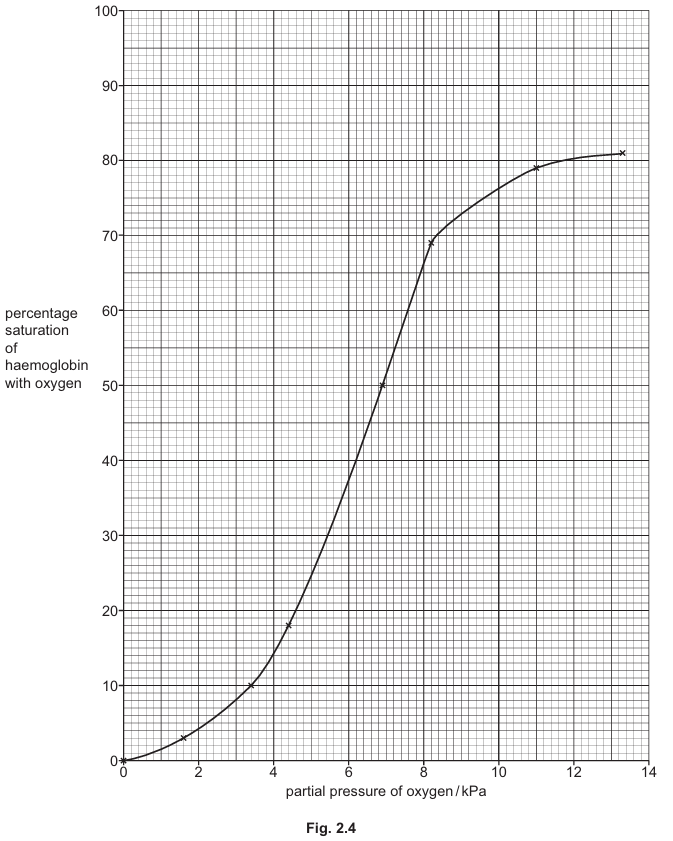
(i) Draw a line on Fig. 2.4 to suggest the position of the oxygen dissociation curve for penguin haemoglobin.
(ii) Penguin haemoglobin is very sensitive to a decrease in pH caused by an increase in the carbon dioxide concentration in the blood.
Explain how a decrease in pH affects penguin haemoglobin, and suggest how this helps the penguin to swim underwater for a long time.
(d) The heart rate of a penguin decreases while it is swimming underwater.
Heart rate is regulated by a group of specialised cells in the wall of the right atrium. The activity of these cells is modified by nerve impulses.
Name the group of specialised cells in the wall of the right atrium that regulates heart rate.
▶️ Answer/Explanation
(a) The missing blood vessels are arterioles (between arteries and capillaries) and venules (between capillaries and veins).
Explanation: The complete pathway is: heart → arteries → arterioles → capillaries → venules → veins → heart. Arterioles are smaller branches of arteries that lead to capillaries, while venules are small vessels that collect blood from capillaries and join to form veins.
(b) Water acts as a solvent for sodium chloride because:
- Water is a polar molecule with δ+ hydrogen atoms and δ- oxygen atoms
- The δ+ hydrogen atoms attract and surround the chloride ions (Cl⁻)
- The δ- oxygen atoms attract and surround the sodium ions (Na⁺)
- This breaks the ionic bonds between Na⁺ and Cl⁻, separating the ions
- The separated ions become evenly distributed throughout the water
Explanation: Water’s polarity enables it to dissolve ionic compounds like NaCl by surrounding and separating the ions. The partial charges on water molecules interact strongly with the charged ions, overcoming the ionic bonds and creating a hydration shell around each ion.
(c)(i) The penguin’s oxygen dissociation curve would be shifted to the left of the normal bird’s curve, maintaining the same sigmoid shape starting at (0,0).
Explanation: The left shift indicates higher oxygen affinity, meaning penguin haemoglobin binds oxygen more readily at any given partial pressure. This adaptation allows penguins to extract more oxygen from their lungs before diving and release it more slowly to tissues during dives.
(c)(ii) A decrease in pH (Bohr effect):
- Causes haemoglobin to release oxygen more readily (reduced affinity)
- H⁺ ions bind to haemoglobin, changing its shape and promoting oxygen release
- This is beneficial as active muscles produce CO₂, lowering pH
- More oxygen is delivered to respiring tissues when needed most
- Allows penguins to maintain aerobic respiration longer during dives
Explanation: The Bohr effect ensures oxygen is released precisely where it’s needed most – in active muscles during diving. The pH sensitivity is particularly important for penguins as it allows efficient oxygen unloading during prolonged dives when breathing isn’t possible.
(d) The sinoatrial node (SAN).
Explanation: The sinoatrial node is the heart’s natural pacemaker, located in the right atrium wall. It initiates electrical impulses that regulate heart rate. In penguins, nerve impulses can modify its activity to slow the heart during dives (dive reflex), conserving oxygen for vital organs.
Topic: 10.1
(a) Fig. 3.1 is a photomicrograph of a transverse section through a region of the wall of the bronchus in the gas exchange system.
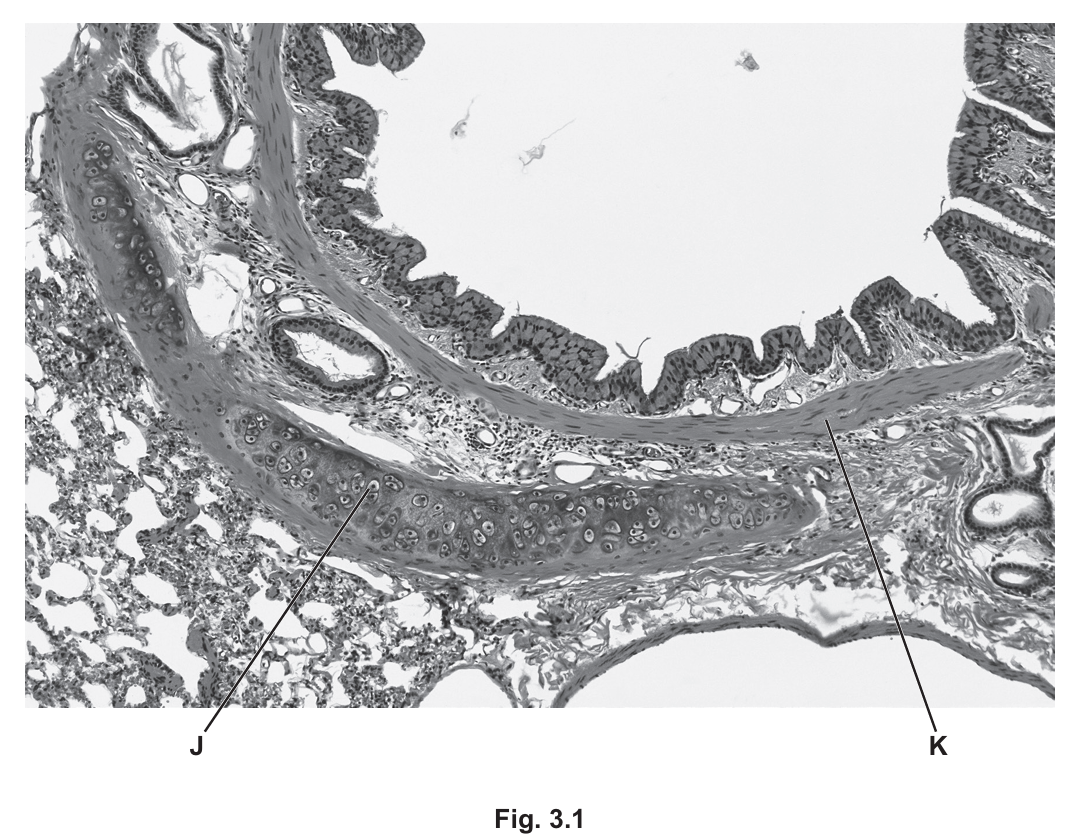
Identify the tissues J and K shown in Fig. 3.1, and suggest how the wall of a bronchiole differs from the wall of the bronchus for these two tissues.
(b) Tuberculosis (TB) is an infectious disease that affects the human gas exchange system.
The pathogen that causes TB secretes a protein that can be detected in saliva.
Early diagnosis of TB is important in reducing the transmission of the pathogen.
Scientists have developed a test strip for TB that uses monoclonal antibodies. Monoclonal antibodies are specific in their action.
This test strip contains:
- mobile monoclonal antibodies that bind to one part of the protein secreted by the pathogen
- immobilised monoclonal antibodies.
Fig. 3.2 shows a simplified diagram of the test strip.
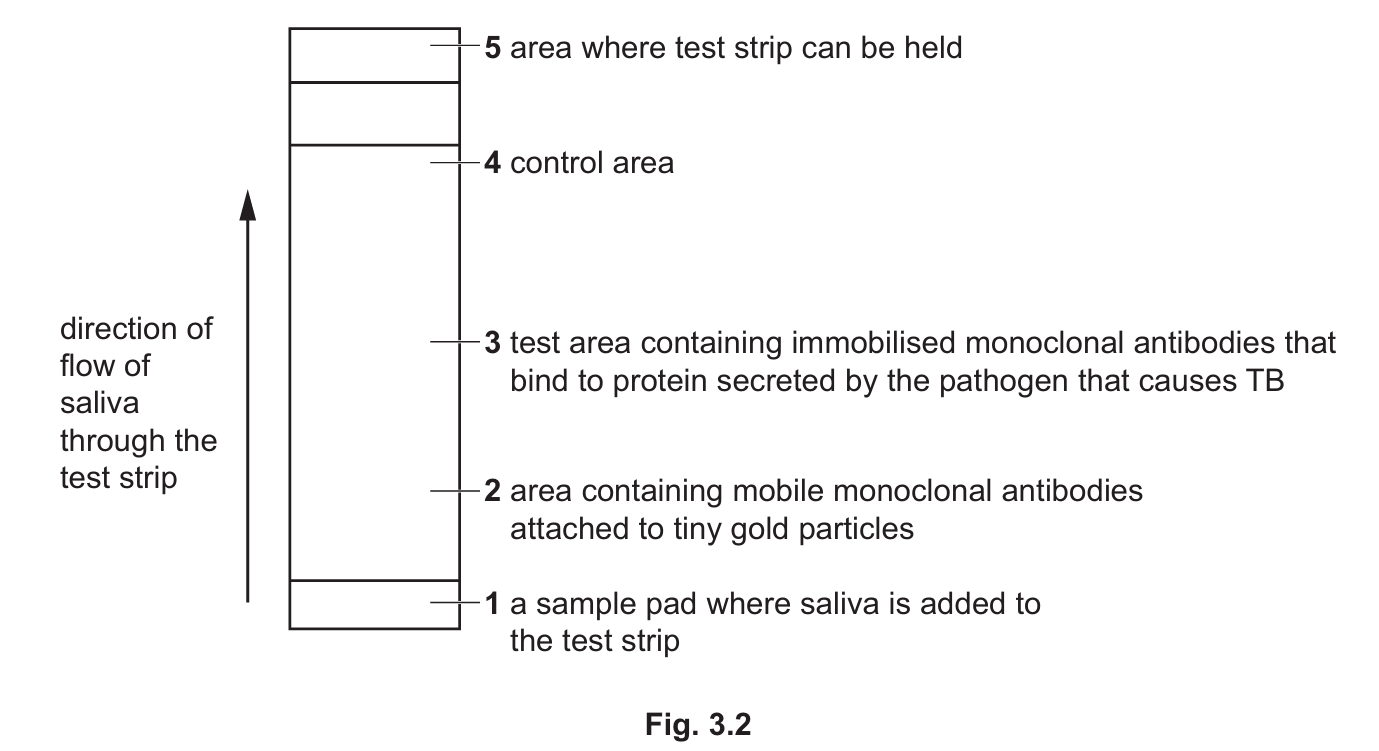
A sample of saliva is collected and put onto the sample pad in the test strip.
The saliva moves up the test strip through area 2.
The mobile monoclonal antibodies are attached to tiny gold particles. If these antibodies collect in test area 3, a gold line becomes visible on the test strip.
A gold line that becomes visible in area 4 confirms that the test strip is working and that the results are valid.
(i) State the name of the pathogen that causes TB.
(ii) Name the part of the monoclonal antibody that binds to the protein from the pathogen.
(iii) Saliva is added to a test strip to test for the presence of the protein secreted by the TB pathogen.
Fig. 3.3 is a diagram showing some of the molecules in area 3 of the test strip when a positive result for TB is obtained.
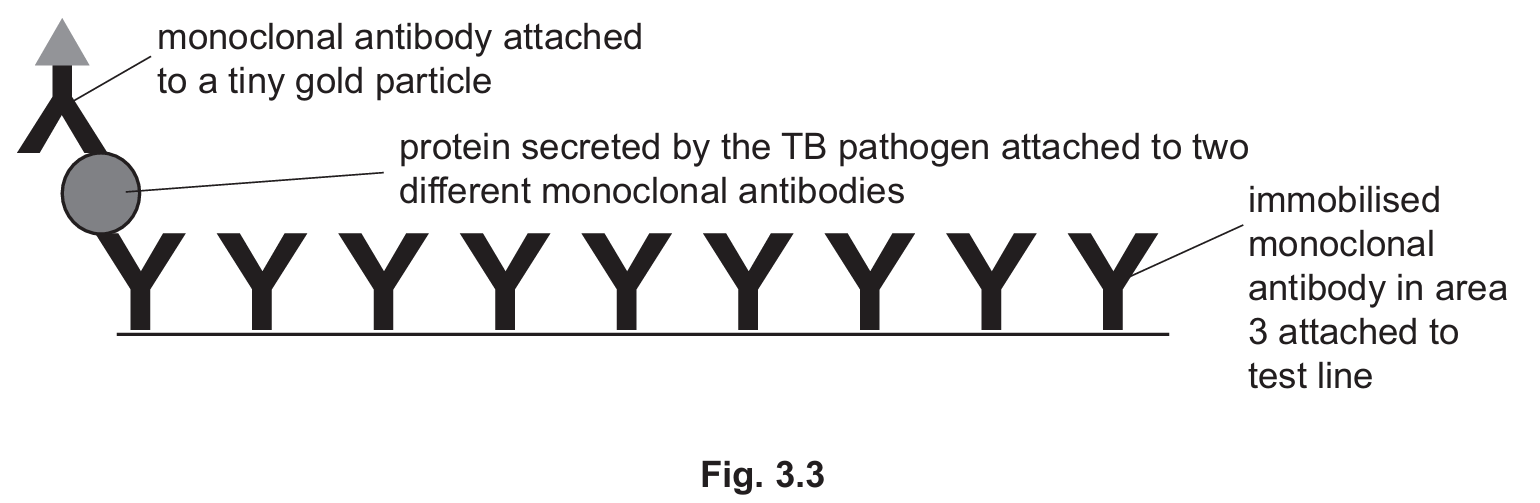
Use the information in Fig. 3.3 to suggest and explain why this test is specific for TB.
(iv) Area 4 contains different immobilised antibodies to those in area 3.
The mobile monoclonal antibodies bound to tiny gold particles will bind to these immobilised monoclonal antibodies in area 4.
If the test has functioned correctly, a gold line will be visible in area 4.
Suggest how the structure of immobilised monoclonal antibodies in area 3 differs from the structure of the immobilised monoclonal antibodies in area 4.
(c) Vaccination is another way of reducing the transmission of infectious diseases such as TB. The BCG vaccine is used to help control the spread of TB. This vaccine contains a weakened strain of the pathogen that causes TB. The BCG vaccine stimulates the development of antigen-specific memory T-lymphocytes.
Explain how memory T-lymphocytes provide protection from TB in a person who has been given a BCG vaccination.
(d) The bladder is the organ in the body used to store urine.
When cells divide uncontrollably in the bladder, a tumour develops. This can lead to bladder cancer.
The BCG vaccine has been used to treat bladder cancer.
The BCG vaccine is introduced into the bladder. The tumour cells take up the weakened pathogens in the vaccine and act as antigen-presenting cells.
(i) Name the process used by the tumour cells to take up the weakened pathogens.
(ii) Suggest how antigen presentation by tumour cells stimulates an immune response that leads to the destruction of the tumour cells.
▶️ Answer/Explanation
(a)
J: Cartilage
K: Smooth muscle
Difference: The bronchus contains cartilage in its wall which provides structural support, while bronchioles lack cartilage. Bronchioles have proportionally more smooth muscle tissue in their walls compared to bronchi, allowing for greater control over air flow.
Explanation: The bronchus is a larger airway that requires cartilage rings to prevent collapse during breathing. As airways become smaller (bronchioles), the cartilage disappears but smooth muscle becomes more prominent to regulate airflow through contraction and relaxation.
(b)(i) Mycobacterium tuberculosis / Mycobacterium bovis
Explanation: These are the bacterial species known to cause tuberculosis in humans. Mycobacterium tuberculosis is the primary causative agent, while Mycobacterium bovis is a less common cause typically associated with cattle.
(b)(ii) Antigen binding site(s)
Explanation: The variable region of the monoclonal antibody contains the antigen binding site that is specifically shaped to recognize and bind to a particular epitope on the TB protein.
(b)(iii) The test is specific for TB because the monoclonal antibodies are designed to bind only to the protein secreted by the TB pathogen. The immobilised monoclonal antibodies in the test area have binding sites that are complementary in shape to specific epitopes on the TB protein, ensuring they won’t bind to proteins from other pathogens.
Explanation: The specificity comes from the precise molecular complementarity between the antibody’s binding site and the TB protein’s epitope. This is like a lock-and-key mechanism where only the correct key (TB protein) will fit the lock (antibody binding site). The gold particles only become visible in the test line if this specific binding occurs.
(b)(iv) The immobilised monoclonal antibodies in area 3 have a different variable region/antigen binding site structure compared to those in area 4. They have different primary structures (amino acid sequences) leading to different tertiary structures and binding specificities.
Explanation: While both sets of antibodies are monoclonal, they are produced to recognize different epitopes. The antibodies in area 3 bind the TB protein, while those in area 4 are designed to bind the mobile antibodies (acting as a control). This difference in function requires different molecular structures in their binding sites.
(c) Memory T-lymphocytes provide long-term immunity by remaining in the body after vaccination. If the actual TB pathogen later enters the body, these memory cells can mount a rapid, strong secondary immune response. They quickly recognize the TB antigens, activate other immune cells, and help destroy the pathogen before it can cause disease.
Explanation: The BCG vaccine primes the immune system by exposing it to weakened TB bacteria. This stimulates the production of memory T-cells that “remember” the TB antigens. Upon subsequent exposure, these memory cells can proliferate quickly and coordinate immune defenses much faster than during the initial exposure, often preventing infection from becoming established.
(d)(i) Endocytosis
Explanation: The tumor cells engulf the weakened pathogens from the vaccine through endocytosis, a process where the cell membrane invaginates to form a vesicle containing the external material.
(d)(ii) When tumor cells present the BCG antigens on their surface, T-lymphocytes with complementary receptors recognize these foreign antigens. This activates the T-cells, which then multiply and differentiate into cytotoxic T-cells that can directly kill the tumor cells. Helper T-cells also stimulate other immune cells like macrophages to attack the tumor.
Explanation: The presentation of BCG antigens by tumor cells essentially marks them as targets for the immune system. Cytotoxic T-cells recognize these antigen-presenting tumor cells and destroy them through various mechanisms like releasing perforins and granzymes that induce apoptosis. This immune response can lead to significant tumor shrinkage.
Topic: 4.1
(a) Fig. 4.1 shows the structure of sucrose, a disaccharide produced by plant cells.
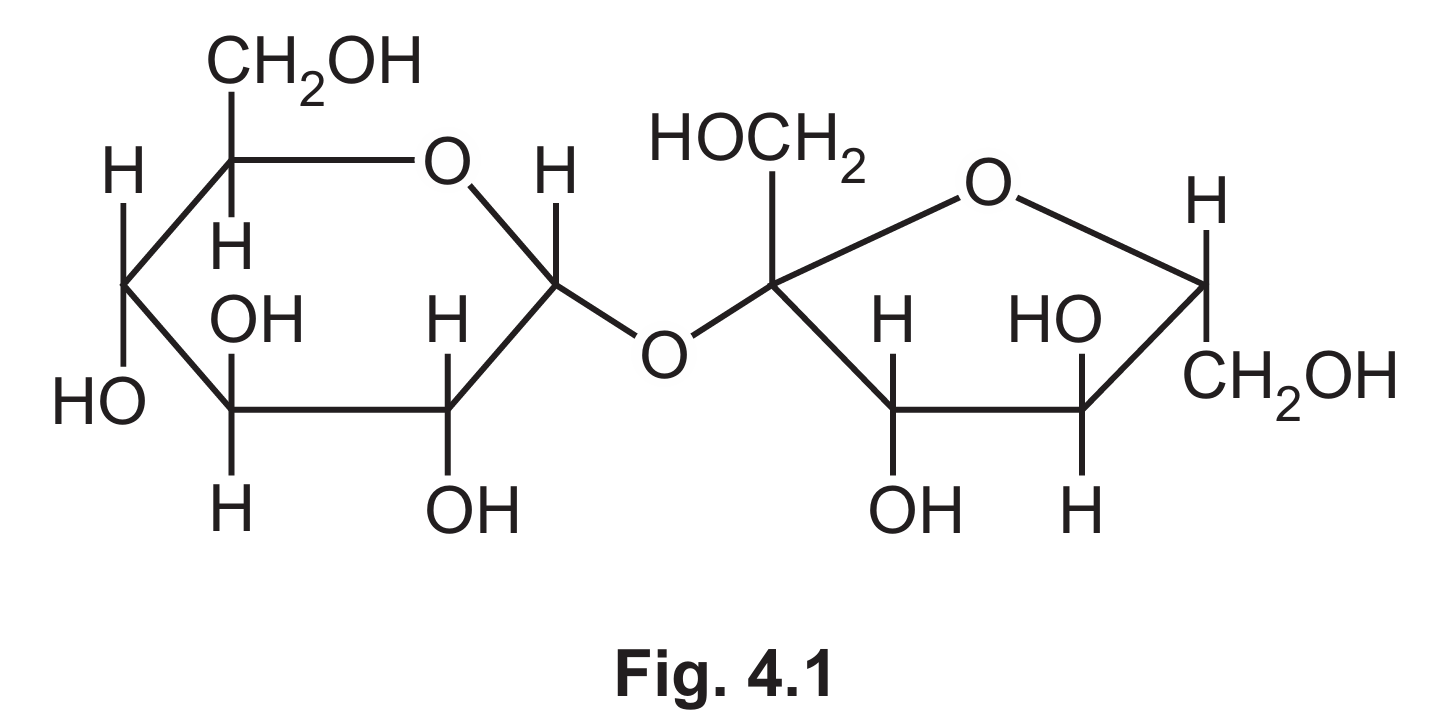
(i) Name the covalent bond that joins the two monomers in sucrose.
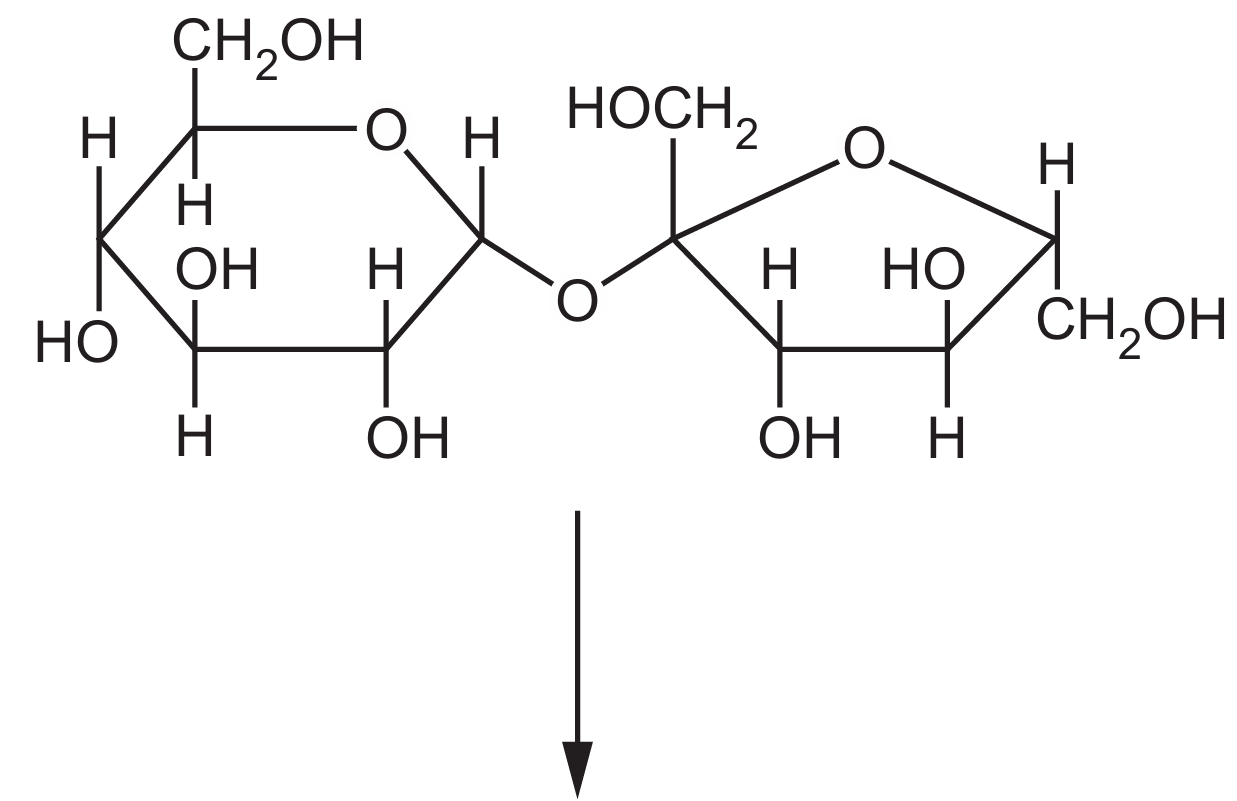
(ii) Sucrose is hydrolysed by the enzyme sucrase in the human digestive system. The products of this hydrolysis reaction are the monosaccharides α-glucose and fructose.
Complete the diagram to show the hydrolysis of sucrose to form α-glucose and fructose.
(b) Plants transport sucrose from a source to a sink. Fig. 4.2 is a scanning electron micrograph (SEM) of a transverse section through a plant tissue used to transport sucrose.
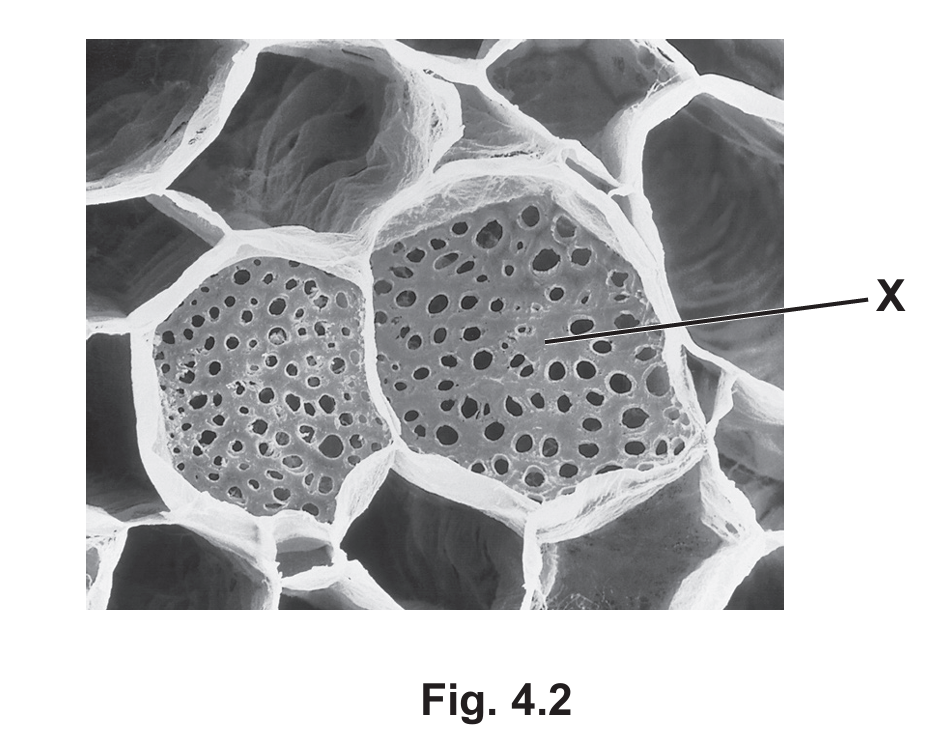
(i) Name the structure labelled X in Fig. 4.2.
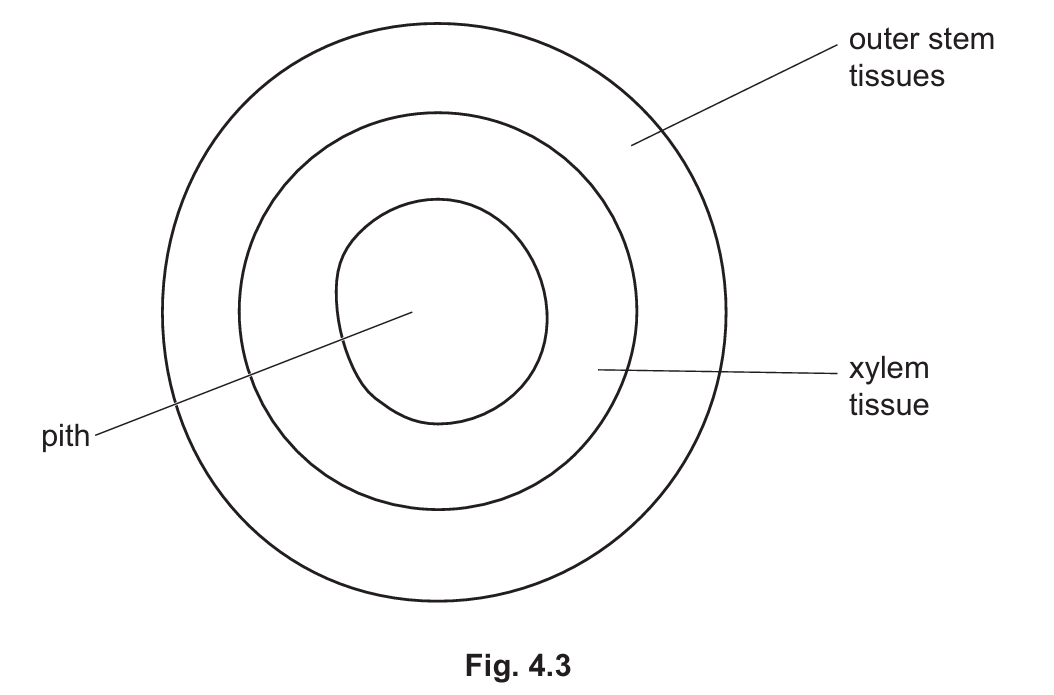
(ii) A scientist carried out an experiment to study carbohydrate transport in the stem of a woody plant. Fig. 4.3 shows a plan diagram of a transverse section of the stem studied by the scientist. The position of the xylem tissue in the stem is shown.
Fig. 4.3 shows a plan diagram of a transverse section of the stem studied by the scientist. The position of the xylem tissue in the stem is shown.
The scientist carried out a set of experiments using plants of the same species.
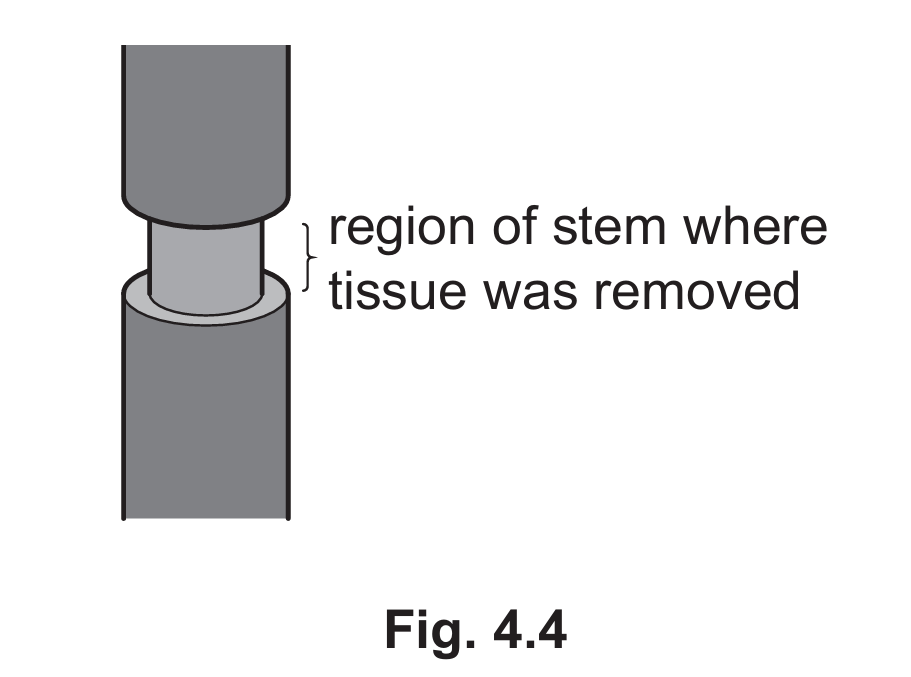
In each experiment, a ring of tissue was removed from the outer stem of the plant, but the xylem tissue was left intact. This is shown in Fig. 4.4.
The mass of carbohydrate transported to the lower part of the stem in 24 hours was recorded.
In each experiment a different percentage of the outer stem tissue was removed.
All other variables remained constant.
Table 4.1 shows the results of this investigation.
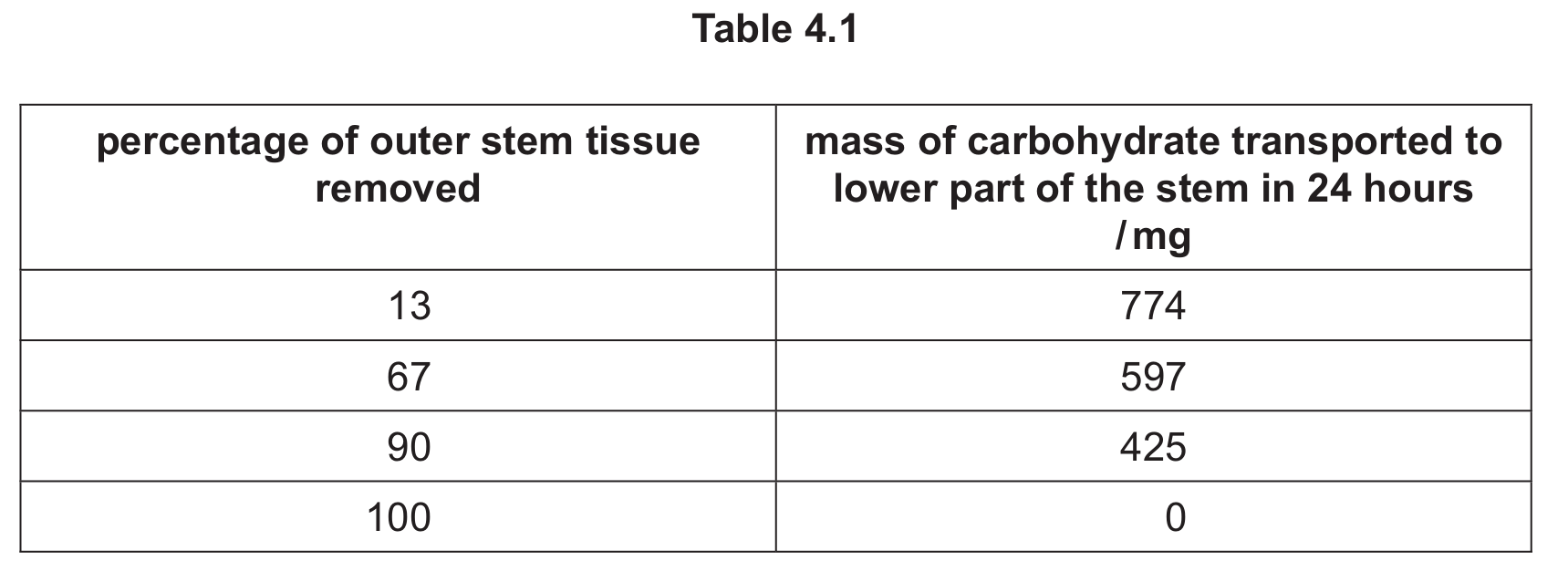
Explain the results shown in Table 4.1.
(c) Sucrose is a sweet-tasting sugar found in many foods.
Some people become ill when they have sucrose in their diet. These people have a gene mutation in the gene coding for sucrase and cannot hydrolyse sucrose in the digestive system.
Scientists studying the DNA of people with this condition identified a deletion mutation in the gene coding for sucrase.
Suggest and explain why a person with this deletion mutation cannot digest sucrose.
▶️ Answer/Explanation
(a)(i) glycosidic bond
Explanation: The covalent bond that joins the two monosaccharide units (glucose and fructose) in sucrose is called a glycosidic bond. Specifically, it’s a 1-2 glycosidic bond because it forms between carbon 1 of glucose and carbon 2 of fructose.
(a)(ii)
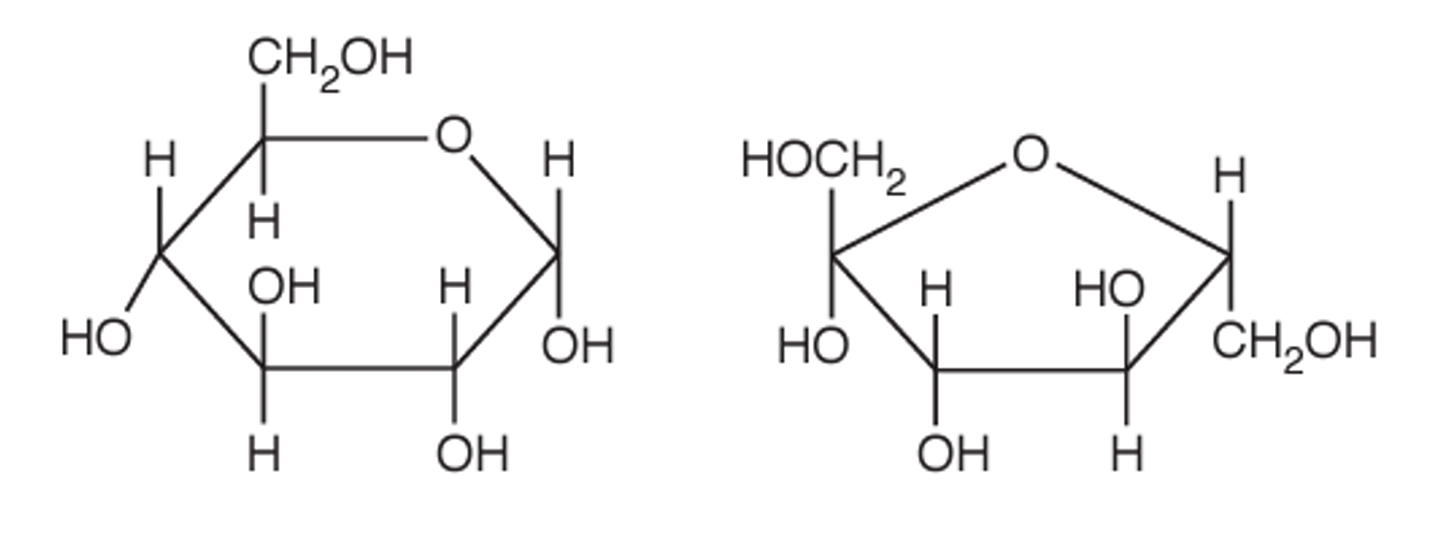
Explanation: During hydrolysis, the glycosidic bond between glucose and fructose is broken by the addition of a water molecule. This results in the formation of two separate monosaccharides: α-glucose (with a hydroxyl group on C1) and fructose (with a hydroxyl group on C2). The enzyme sucrase catalyzes this reaction in the small intestine.
(b)(i) sieve plate
Explanation: Structure X is identified as a sieve plate, which is a characteristic feature of phloem sieve tube elements. These perforated end walls allow for the flow of sap between adjacent sieve tube elements during translocation.
(b)(ii)
Explanation: The results show that as more outer stem tissue is removed, less carbohydrate is transported. This is because:
- The phloem tissue responsible for carbohydrate transport (sucrose) is located in the outer stem.
- When outer tissue is removed, the phloem sieve tubes are destroyed, reducing the pathway for carbohydrate transport.
- At 100% removal, no carbohydrate is transported because all phloem has been removed, despite the xylem remaining intact.
- The xylem tissue (which remains intact) doesn’t transport carbohydrates, only water and minerals.
- The decreasing amounts show that carbohydrate transport is proportional to the amount of remaining phloem tissue.
(c)
Explanation: A person with this deletion mutation cannot digest sucrose because:
- The deletion mutation alters the DNA sequence of the sucrase gene, potentially causing a frameshift if not a multiple of three nucleotides.
- This leads to production of abnormal mRNA that may be degraded or produce a truncated protein.
- If a protein is produced, it likely has an altered tertiary structure due to changes in amino acid sequence.
- The active site of the enzyme would be misshapen, preventing sucrose from binding properly.
- Without functional sucrase, the glycosidic bond in sucrose cannot be hydrolyzed, leaving sucrose undigested in the gut.
- This undigested sucrose can cause osmotic diarrhea and fermentation by gut bacteria, leading to discomfort.
Topic: 5.1
Trypsin is an enzyme which catalyses the hydrolysis of casein, a protein found in milk.
Milk that contains casein has a cloudy, white appearance. As the casein is hydrolysed by trypsin, the milk changes in appearance to a clear (transparent), colourless solution.
A student carried out an experiment to investigate the effect of enzyme concentration on the rate at which trypsin hydrolyses casein.
The student added a solution of trypsin to a sample of milk and recorded the time taken for the milk to become transparent. The student repeated the experiment with different concentrations of trypsin. All other variables were kept constant.
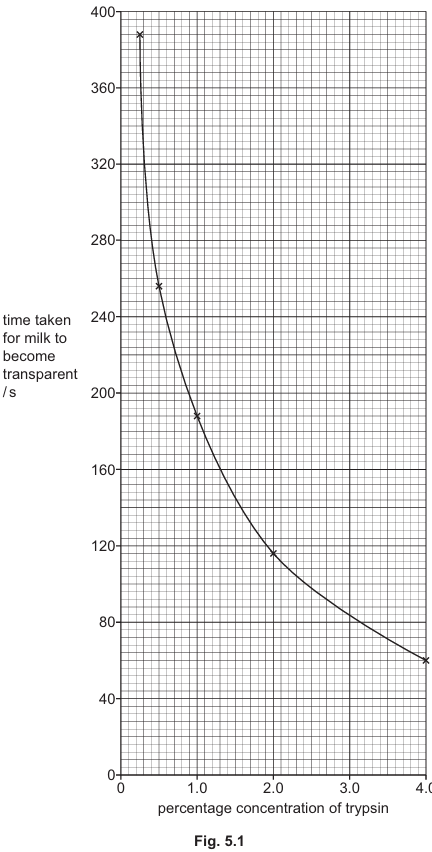
Fig. 5.1 shows the results from the experiment.
(a) (i) When the concentration of trypsin increases from 2.0% to 4.0%, the time taken for the milk to become transparent decreases by 48%.
Calculate the percentage decrease in the time taken for milk to become transparent when the concentration of trypsin increases from 0.25% to 0.5%.
Write your answer to the nearest whole number.
(ii) Explain the results shown in Fig. 5.1.
(b) Trypsin has the potential to be used in a wide range of industrial processes.
The use of immobilised enzymes in industrial processes has many advantages.
Scientists investigated the effect of temperature on the activity of trypsin immobilised on the surface of a material and trypsin free in solution.
Table 5.1 shows the results of the investigation.
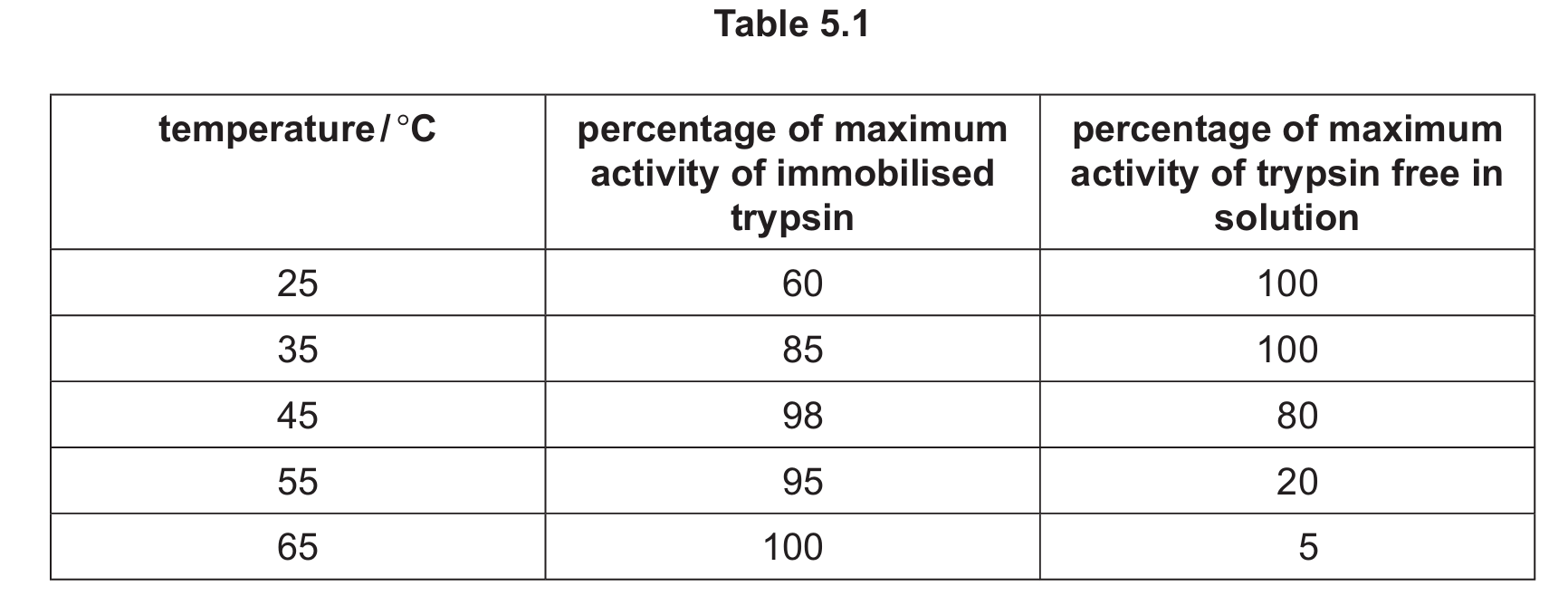
(i) State a reason for the difference in percentage of maximum activity of immobilised trypsin and trypsin free in solution at 25°C.
(ii) Suggest and explain why the percentage of maximum activity of immobilised trypsin at 55°C is higher than the percentage of maximum activity of trypsin free in solution at 55°C.
▶️ Answer/Explanation
(a)(i) 34%
Explanation: To calculate the percentage decrease, we compare the time difference between the two concentrations relative to the original time. The exact calculation involves determining the proportional decrease when moving from 0.25% to 0.5% trypsin concentration, resulting in a 34% decrease in time taken for the milk to become transparent.
(a)(ii)
Explanation: The results show that as trypsin concentration increases, the time taken for milk to become transparent decreases. This occurs because:
- At lower concentrations, there are fewer enzyme molecules available, limiting the number of active sites for casein to bind to.
- As concentration increases, more enzyme molecules are present, providing more active sites for the substrate (casein) to bind to.
- This leads to more frequent successful collisions between enzyme and substrate, forming more enzyme-substrate complexes per unit time.
- The increased number of active sites allows for faster breakdown of casein into smaller peptides and amino acids, causing the milk to clear more quickly.
- At very high concentrations, the rate increase may plateau as substrate availability becomes the limiting factor rather than enzyme concentration.
(b)(i)
Explanation: The difference at 25°C could be because immobilizing the enzyme may have slightly altered the enzyme’s tertiary structure or active site shape. Alternatively, the immobilization material might partially block some active sites or reduce enzyme-substrate collision frequency compared to free enzymes in solution.
(b)(ii)
Explanation: At 55°C, the immobilised trypsin shows higher activity because:
- Immobilization stabilizes the enzyme’s structure, protecting it from thermal denaturation that affects free enzymes.
- The support material may restrict molecular vibrations that could lead to denaturation in free enzymes.
- Immobilization maintains the active site’s shape at higher temperatures where free enzymes would begin to unfold.
- The immobilized enzyme’s tertiary structure is preserved better, with hydrogen and ionic bonds maintaining stability despite increased thermal energy.
Topic: 6.1
Fig. 6.1 shows a plant cell in a stage of mitosis.
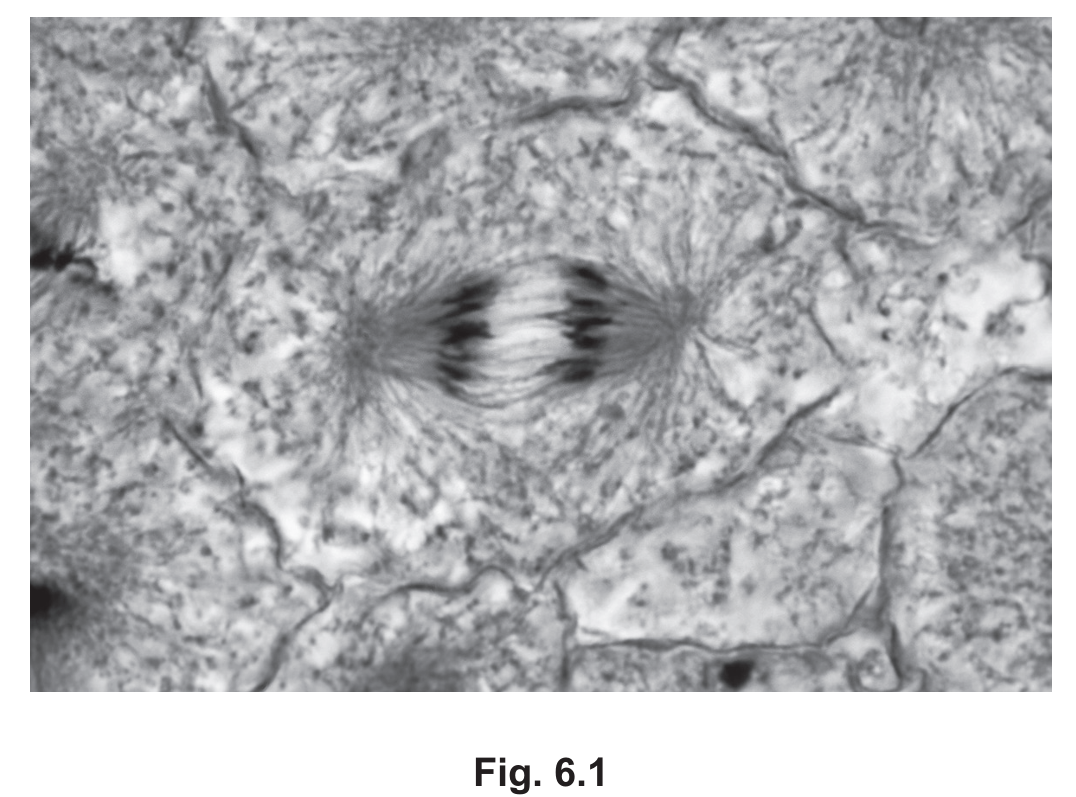
(a) Some of the structures shown in Fig. 6.1 contain DNA.
Use a line labelled D on Fig. 6.1 to indicate one of these structures.
(b) Name the stage of mitosis shown in Fig. 6.1.
(c) Colchicine is a chemical used by scientists to study mitosis. This chemical inhibits the organisation of the microtubules in prophase of mitosis.
The cell shown in Fig. 6.1 had not been treated with colchicine.
Explain the evidence in Fig. 6.1 that shows the cell had not been treated with colchicine.
▶️ Answer/Explanation
(a) Line D should point to any chromosome
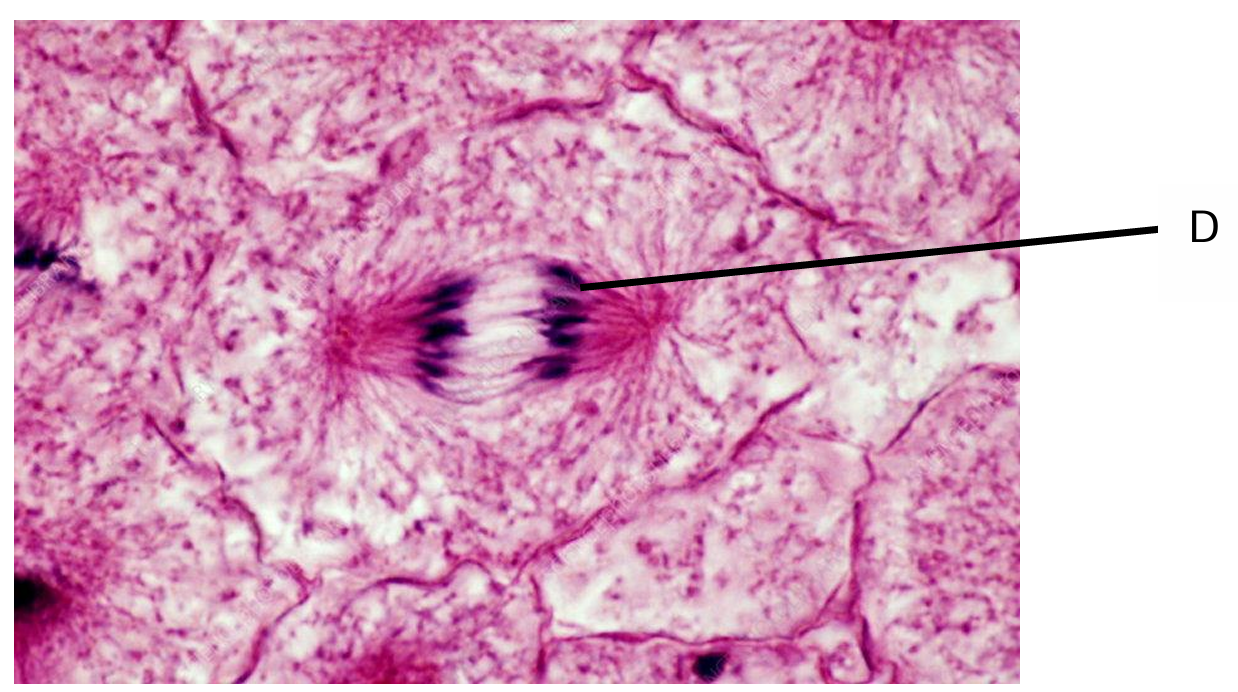
Explanation: During mitosis, the DNA is condensed into visible chromosomes. In the figure, these would appear as distinct, rod-shaped structures that have already replicated (each consisting of two sister chromatids). These chromatids contain the DNA that will be separated during cell division.
(b) Anaphase.
Explanation: The stage shown is anaphase because we can observe that the sister chromatids (now called chromosomes) have separated and are being pulled toward opposite poles of the cell. This is characteristic of anaphase, which follows metaphase and precedes telophase in the mitotic process.
(c)
1. The presence of a fully formed spindle apparatus is visible in Fig. 6.1.
2. The chromatids are clearly attached to spindle fibers and are being pulled apart.
Explanation: Colchicine works by inhibiting microtubule formation, which prevents the development of the mitotic spindle. Since the cell in Fig. 6.1 shows:
- A complete spindle structure with microtubules clearly visible
- Chromatids properly attached to spindle fibers and moving toward opposite poles
- Successful separation of chromosomes
This demonstrates that microtubule formation and organization occurred normally, which wouldn’t happen if colchicine had been present. The drug would have stopped the process in prophase, preventing spindle formation and subsequent chromosome movement.
