Topic: 9.1
Copper is used in electrical equipment. It has a melting point of 1085°C.
(a) (i) Identify the lattice structure of copper.
(ii) Draw a labelled diagram to show the bonding present in copper.
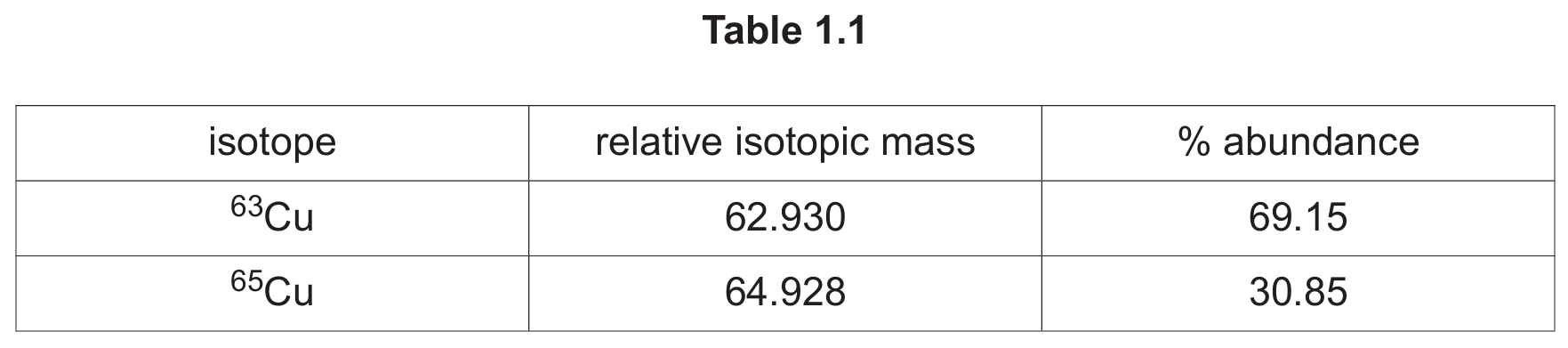
(b) The relative isotopic masses and natural abundances of the two isotopes in a sample of copper are shown in Table 1.1.
(i) Define the unified atomic mass unit.
(ii) Define relative atomic mass, \( A_r \), in terms of the unified atomic mass unit.
(iii) Calculate the relative atomic mass, \( A_r \), of copper in this sample using the data in Table 1.1. Show your working.
(c) The mass spectrum of a sample of pure copper is shown in Fig. 1.1.
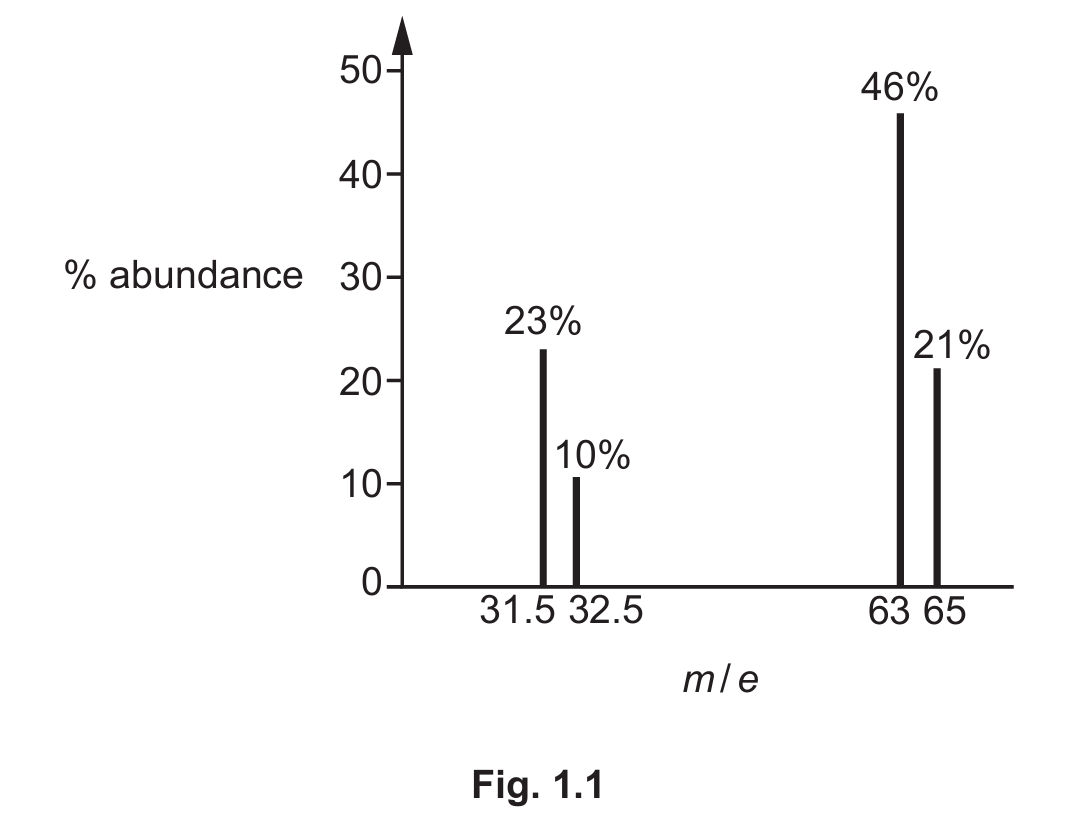
Identify the ion with an abundance of 23% in the sample.
(d) When KI(aq) is added to CuSO\(_4\)(aq) the blue-coloured solution turns brown and a white precipitate of CuI(s) is seen. The reaction between copper ions and iodide forms only two products.
(i) Complete the equation for this reaction.
\[……Cu^{2+} + ……I^- \rightarrow ……CuI + ……\]
(ii) Identify the oxidising agent in this reaction. Explain your answer in terms of electron transfer.
(iii) State the full electronic configuration of Cu\(^{2+}\).
▶️ Answer/Explanation
(a)(i) Giant metallic.
Explanation: Copper is a metal. Metals form giant metallic lattice structures, where positive metal ions are arranged in a regular pattern and are surrounded by a ‘sea’ of delocalised electrons.
(a)(ii)
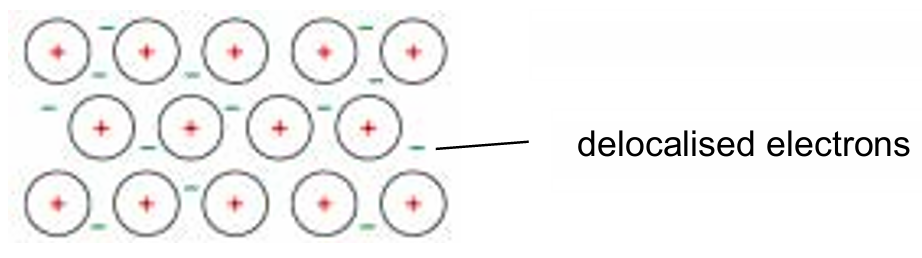
Explanation: The diagram should show a regular arrangement of positive ions (labelled as Cu²⁺ ions or positive ions). These ions are surrounded by delocalised electrons, which are mobile and not associated with any single atom. This sea of delocalised electrons is responsible for metallic bonding, which allows copper to conduct electricity and have a high melting point.
(b)(i) The unified atomic mass unit is defined as one twelfth of the mass of a carbon-12 atom.
Explanation: This is the standard definition. It provides a consistent scale for measuring the masses of atoms and subatomic particles. One unified atomic mass unit (u or Da) is exactly 1/12 of the mass of a single atom of carbon-12.
(b)(ii) The relative atomic mass (\(A_r\)) is the average mass of the atoms of an element, measured on a scale where the mass of an atom of carbon-12 is exactly 12 unified atomic mass units.
Explanation: This definition emphasizes that \(A_r\) is a weighted average that takes into account the different isotopes of an element and their natural abundances. It is a dimensionless quantity (a number) because it is a ratio compared to the standard carbon-12 mass.
(b)(iii) \[ A_r = \frac{(62.930 \times 69.15) + (64.928 \times 30.85)}{100} = \frac{(4351.6095) + (2003.0488)}{100} = \frac{6354.6583}{100} = 63.55 \] \(A_r = 63.55\)
Explanation: To calculate the relative atomic mass, you find the weighted average of the isotopic masses. For each isotope, you multiply its relative isotopic mass by its percentage abundance (expressed as a decimal, which is percentage/100). Then, you add these values together for all isotopes. The calculation is: \((62.930 \times 0.6915) + (64.928 \times 0.3085) = 43.516 + 20.030 = 63.546\), which rounds to 63.55.
(c) \(^{65}\text{Cu}^+\)
Explanation: In mass spectrometry, the m/e ratio (mass-to-charge ratio) is measured. The peak at m/e = 65 with 23% abundance corresponds to the \(^{65}\text{Cu}\) isotope that has lost one electron to form a 1+ ion, \(^{65}\text{Cu}^+\). The height of the peak represents the relative abundance of that ion.
(d)(i) \[2\text{Cu}^{2+} + 4\text{I}^- \rightarrow 2\text{CuI} + \text{I}_2\]
Explanation: This is a disproportionation reaction where copper is both reduced and oxidized. The white precipitate is copper(I) iodide (CuI). The brown colour is due to iodine (\(\text{I}_2\)) being formed. Balancing the equation requires 2 Cu²⁺ ions to gain 2 electrons (to become 2 Cu⁺ in CuI) and 2 I⁻ ions to lose 2 electrons (to become I₂). Since each I⁻ only provides one electron, 4 I⁻ ions are needed in total: 2 to reduce the copper and 2 to form the I₂ molecule.
(d)(ii) The oxidising agent is \(\text{Cu}^{2+}\). Copper(II) ions gain electrons from iodide ions to form copper(I) iodide. \(\text{Cu}^{2+} + e^- \rightarrow \text{Cu}^+\).
Explanation: An oxidising agent accepts electrons and is itself reduced. In this reaction, some of the \(\text{Cu}^{2+}\) ions are reduced to \(\text{Cu}^+\) (as seen in the product CuI). Therefore, \(\text{Cu}^{2+}\) is acting as the oxidising agent because it is gaining electrons from the I⁻ ions, which are the reducing agents.
(d)(iii) \(1s^2 2s^2 2p^6 3s^2 3p^6 3d^9\)
Explanation: The atomic number of copper is 29, so a neutral copper atom has the electron configuration [Ar] 4s¹ 3d¹⁰. When it forms a Cu²⁺ ion, it loses two electrons. The 4s electron is lost first, followed by one electron from the 3d subshell. This results in the configuration \(1s^2 2s^2 2p^6 3s^2 3p^6 3d^9\).
Topic: 10.1
(a) The reaction of pure aluminium is only observed if the aluminium oxide layer is removed first. When pure aluminium is added to cold water, bubbles of gas are seen.
(i) State one property of aluminium oxide that explains why an aluminium object does not react with cold water until the aluminium oxide layer is removed.
(ii) Write an equation, with state symbols, for the reaction of aluminium oxide with an excess of NaOH(aq).
(iii) Name one other Period 3 element that also produces bubbles of gas when added to cold water.
(b) Aluminium nitrate is a white soluble salt. On heating aluminium nitrate, thermal decomposition occurs and a brown gas is seen.
State the formula of the salt of another element in Period 3 which also decomposes on heating to produce a brown gas.
(c) Aluminium chloride and phosphorus chloride are both white solids.
(i) State the maximum oxidation number of aluminium and of phosphorus in these solid chloride salts.
(ii) State why the maximum oxidation number of aluminium is different from that of phosphorus.
(iii) Write an equation for the reaction of solid phosphorus chloride and excess water.
(iv) Name the type of reaction that occurs when aluminium chloride is added to water.
(v) Explain why the solution produced after aluminium chloride is added to water has a pH of 1–2.
▶️ Answer/Explanation
(a)(i) It / aluminium oxide is insoluble (in water) OR it / aluminium oxide does not react (with water).
Explanation: Aluminium naturally forms a thin, protective layer of aluminium oxide (Al₂O₃) on its surface when exposed to air. This layer is chemically inert and physically robust. Its key property here is its insolubility in water and its lack of reactivity with water. This impervious layer acts as a barrier, preventing the underlying pure aluminium metal from making contact with and reacting with the cold water. Once this layer is removed (e.g., by scratching or amalgamation), the reactive aluminium metal is exposed and can react.
(a)(ii) Al₂O₃(s) + 2NaOH(aq) + 3H₂O(l) → 2NaAl(OH)₄(aq) OR Al₂O₃(s) + 2NaOH(aq) → 2NaAlO₂(aq) + H₂O(l)
Explanation: Aluminium oxide is an amphoteric oxide, meaning it can react with both acids and bases. When reacted with an excess of sodium hydroxide, it forms a soluble complex ion. The most common reaction is the formation of sodium aluminate (NaAlO₂ or NaAl(OH)₄, which is often written as NaAlO₂·2H₂O). The equation with state symbols is: Al₂O₃(s) + 2NaOH(aq) → 2NaAlO₂(aq) + H₂O(l). An alternative representation showing the tetrahydroxoaluminate complex is also acceptable: Al₂O₃(s) + 2NaOH(aq) + 3H₂O(l) → 2NaAl(OH)₄(aq).
(a)(iii) Sodium
Explanation: Sodium (Na) is another element in Period 3 that reacts vigorously with cold water. This reaction produces hydrogen gas (seen as bubbles) and sodium hydroxide: 2Na(s) + 2H₂O(l) → 2NaOH(aq) + H₂(g).
(b) Mg(NO₃)₂
Explanation: Magnesium nitrate, Mg(NO₃)₂, is another Period 3 nitrate salt that decomposes on heating to produce a brown gas. The brown gas is nitrogen dioxide (NO₂). The decomposition reaction is: 2Mg(NO₃)₂(s) → 2MgO(s) + 4NO₂(g) + O₂(g).
(c)(i) maximum oxidation number of aluminium +3
maximum oxidation number of phosphorus +5
Explanation: The maximum oxidation number an element can achieve is usually equal to the number of valence electrons it has. Aluminium (Al) is in group 13 and has 3 valence electrons. Therefore, its maximum oxidation number is +3, as in AlCl₃. Phosphorus (P) is in group 15 and has 5 valence electrons. Therefore, its maximum oxidation number is +5, as in PCl₅.
(c)(ii) Different number of valence / outer shell electrons.
Explanation: The maximum oxidation number is determined by the total number of electrons an atom can lose or share to form bonds, which is fundamentally based on the number of electrons in its outer shell. Aluminium has 3 valence electrons, while phosphorus has 5. This difference in their electronic configurations is the reason for the difference in their maximum oxidation states.
(c)(iii) PCl₅(s) + 4H₂O(l) → H₃PO₄(aq) + 5HCl(aq)
Explanation: Phosphorus(V) chloride reacts vigorously and hydrolyzes with excess water. The products are phosphoric(V) acid and hydrochloric acid. The balanced equation for this reaction is: PCl₅(s) + 4H₂O(l) → H₃PO₄(aq) + 5HCl(aq).
(c)(iv) Hydrolysis OR substitution
Explanation: When aluminium chloride is added to water, it undergoes a chemical reaction where water molecules act as a nucleophile and break the bonds in AlCl₃. This type of reaction, where a compound is split by water, is specifically called hydrolysis. It can also be viewed more generally as a substitution reaction where chloride ions are replaced by hydroxide ions from water.
(c)(v) HCl(g) dissolves in water to make (hydrochloric) acid.
Explanation: Aluminium chloride (AlCl₃) has a covalent character with significant polarisation. When it is added to water, it doesn’t just simply dissolve; it hydrolyzes. The reaction produces hydrogen chloride gas: AlCl₃(s) + 3H₂O(l) → Al(OH)₃(s) + 3HCl(g). This hydrogen chloride gas is highly soluble and dissolves in the water to form hydrochloric acid (HCl(aq)), which is a strong acid. The high concentration of H⁺ ions from the dissociation of HCl is what causes the resulting solution to be strongly acidic, hence the low pH of 1-2.
Topic: 5.1
A neutralisation reaction occurs when NaOH(aq) is added to H2SO4(aq).
equation 1: \[ 2NaOH(aq) + H_2SO_4(aq) \rightarrow Na_2SO_4(aq) + 2H_2O(l) \]
(a) Define enthalpy change of neutralisation, \(\Delta H_{\text{neut}}\).
(b) In an experiment, 50.0 cm3 of 2.00 mol dm–3 NaOH(aq) is added to 60.0 cm3 of 1.00 mol dm–3 H2SO4(aq) in a polystyrene cup and stirred. Both solutions have a temperature of 21.4 °C before mixing. The maximum temperature of the mixture is measured.
(i) Use equation 1 to calculate the amount, in mol, of H2SO4(aq) that is neutralised in the experiment.
(ii) Calculate the theoretical maximum temperature of the mixture in this experiment.
Assume that:
- enthalpy change of neutralisation, \(\Delta H_{\text{neut}}\) of NaOH(aq) and H2SO4(aq) is –57.1 kJ mol–1
- full dissociation of H2SO4(aq) occurs
- the specific heat capacity of the final solution is 4.18 J g–1 K–1
- 1.00 cm3 of the final solution has a mass of 1.00 g
- there is no heat loss to the surroundings
- the experiment takes place at constant pressure.
(c) The enthalpy change of neutralisation of CH3COOH(aq) and NaOH(aq) is –55.2 kJ mol–1.
(i) Complete the equation for the reaction.
(ii) Values for the enthalpy change of neutralisation, \(\Delta H_{\text{neut}}\), are shown in Table 3.1.
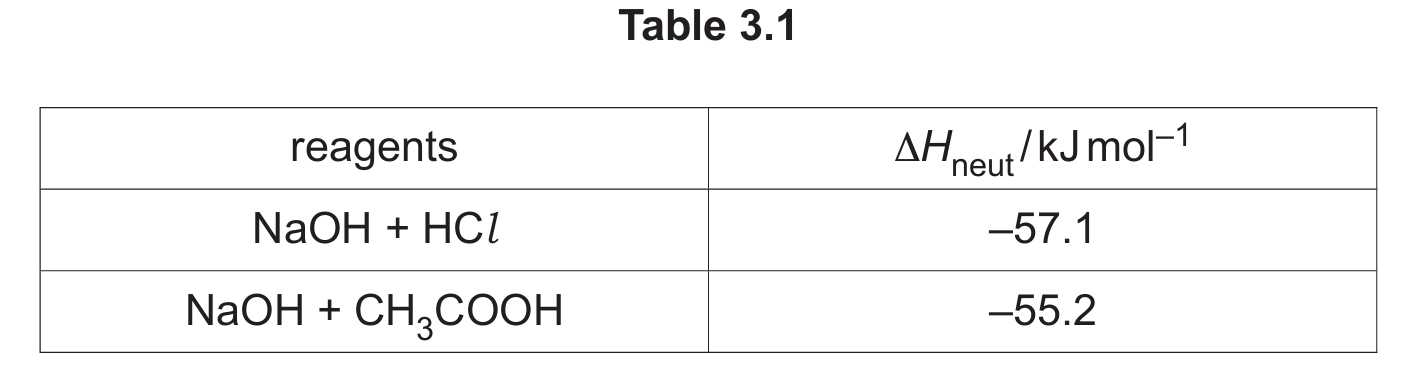
Suggest why the value for \(\Delta H_{\text{neut}}\) of the weak acid, CH3COOH, reacting with NaOH is different to the value obtained using the strong acid, HCl. Assume that the values are determined under the same conditions.
▶️ Answer/Explanation
(a) The enthalpy change of neutralisation, \(\Delta H_{\text{neut}}\), is defined as the energy change when one mole of water is formed from the reaction between an acid and a base (or alkali) under standard conditions.
Explanation: This definition specifies the key components: the formation of one mole of water as the product and the reactants being an acid and a base. The reaction is typically carried out in aqueous solutions.
(b)(i) amount of H2SO4 neutralised = 0.05 mol
Explanation: From the stoichiometry of equation 1, 2 moles of NaOH react with 1 mole of H2SO4.
Moles of NaOH used = Concentration × Volume (in dm³) = 2.00 mol dm–3 × 0.050 dm³ = 0.100 mol.
Therefore, moles of H2SO4 neutralised = (Moles of NaOH) / 2 = 0.100 / 2 = 0.050 mol.
(b)(ii) theoretical maximum temperature = 33.8 °C
Explanation: The calculation involves several steps:
Step 1: Calculate the heat energy released (Q). The enthalpy change is given for the formation of 1 mole of water. From equation 1, 2 moles of water are formed when 1 mole of H2SO4 is neutralised. Therefore, for 0.05 mol of H2SO4, the moles of water formed are 0.10 mol.
Heat energy released, Q = moles of water × |\(\Delta H_{\text{neut}}\)| = 0.10 mol × 57.1 kJ mol–1 = 5.71 kJ. Convert to Joules: Q = 5710 J.
Step 2: Calculate the total mass of the solution. The total volume is 50.0 cm³ + 60.0 cm³ = 110.0 cm³. Since the density is 1.00 g cm–3, the total mass, m = 110.0 g.
Step 3: Use the formula Q = m × c × ΔT to find the temperature change.
5710 J = 110.0 g × 4.18 J g–1 K–1 × ΔT
ΔT = 5710 / (110.0 × 4.18) = 5710 / 459.8 ≈ 12.42 °C (or K, as the scale is the same for change).
Step 4: The maximum temperature is the initial temperature plus the temperature rise.
Tmax = 21.4 °C + 12.42 °C = 33.82 °C, which rounds to 33.8 °C considering significant figures.
(c)(i) \[ CH_3COOH + NaOH \rightarrow CH_3COONa + H_2O \]
Explanation: This is the standard neutralization reaction between a weak acid (ethanoic acid) and a strong base (sodium hydroxide), producing sodium ethanoate (a salt) and water.
(c)(ii) The value for \(\Delta H_{\text{neut}}\) is less exothermic for ethanoic acid because some energy is absorbed to ionize the weak acid fully before it can react with the base.
Explanation: Strong acids like HCl are fully dissociated in water. Weak acids like CH3COOH are only partially dissociated. The neutralization reaction for a weak acid involves two steps: first, the energy-absorbing step of dissociating the weak acid into H+ ions (which is endothermic), and second, the exothermic reaction of H+ with OH– to form water. The overall \(\Delta H_{\text{neut}}\) is the sum of these two steps, making it less exothermic than for a strong acid, where only the second, highly exothermic step occurs.
Topic: 7.1
(a) Hydrogen chloride gas is made in the laboratory by adding concentrated sulfuric acid to potassium chloride.
(i) Construct an equation for this reaction.
(ii) Explain why hydrogen iodide is not prepared by adding concentrated sulfuric acid to sodium iodide.
(b) A sample of HI(g) is added to a 2.00 dm3 sealed vessel at 764 K and allowed to reach equilibrium.
reaction 1: \[ 2HI(g) \rightleftharpoons H_2(g) + I_2(g) \quad K_c = 0.0217 \, \text{at} \, 764 \, \text{K} \]
At equilibrium the mixture contains 1.70 mol of HI(g).
(i) State one difference in the appearance of the initial reaction mixture compared to the mixture at equilibrium.
(ii) Deduce the expression for equilibrium constant \( K_c \) for reaction 1.
(iii) Calculate the concentration of \( I_2 \) present in the reaction mixture at equilibrium. Show your working.
(c) The experiment is repeated at 500K. The value of \( K_c \) under these conditions is 0.00625.
(i) Describe the difference in the composition of the equilibrium mixture at 500K compared to 764K.
(ii) Use Le Chatelier’s principle to deduce whether the decomposition of HI(g) is endothermic or exothermic. Explain your answer.
▶️ Answer/Explanation
(a)(i) \[ 2KCl + H_2SO_4 \rightarrow K_2SO_4 + 2HCl \]
OR
\[ KCl + H_2SO_4 \rightarrow KHSO_4 + HCl \]
Explanation: Concentrated sulfuric acid reacts with ionic chlorides to produce hydrogen chloride gas. The reaction can produce the normal sulfate (K2SO4) or the hydrogen sulfate (KHSO4), depending on the stoichiometry and conditions. Both equations are acceptable.
(a)(ii) Hydrogen iodide is not prepared this way because the concentrated sulfuric acid oxidizes the iodide ion (I–), producing other products like iodine (I2), sulfur dioxide (SO2), or hydrogen sulfide (H2S), instead of pure HI gas.
Explanation: Sulfuric acid is a strong oxidizing agent. Iodide ions (I–) are strong enough reducing agents to be oxidized by hot, concentrated H2SO4. The reaction does not simply produce HI(g); instead, it leads to a mixture of products like purple I2(s/g), SO2(g), and H2S(g), making it impossible to obtain pure HI. HCl and HBr can be prepared this way because Cl– and Br– are weaker reducing agents and are not oxidized under these conditions.
(b)(i) The initial mixture is colourless, whereas the equilibrium mixture is purple.
Explanation: Initially, only colourless HI(g) is present. At equilibrium, one of the products, I2(g), is purple, which would give the mixture a purple colouration.
(b)(ii) \[ K_c = \frac{[H_2][I_2]}{[HI]^2} \]
Explanation: The equilibrium constant expression, Kc, is defined as the product of the concentrations of the products raised to the power of their stoichiometric coefficients, divided by the product of the concentrations of the reactants raised to the power of their stoichiometric coefficients.
(b)(iii) concentration of I2 = 0.125 mol dm–3
Explanation:
Step 1: Calculate the equilibrium concentration of HI.
[HI] = moles / volume = 1.70 mol / 2.00 dm³ = 0.85 mol dm–3.
Step 2: Let the equilibrium concentration of I2 be x mol dm–3. From the stoichiometry of the reaction (2HI ⇌ H2 + I2), the concentration of H2 will also be x mol dm–3.
Step 3: Substitute the values into the Kc expression.
\[ K_c = \frac{[H_2][I_2]}{[HI]^2} \]
\[ 0.0217 = \frac{(x)(x)}{(0.85)^2} \]
\[ 0.0217 = \frac{x^2}{0.7225} \]
Step 4: Solve for x.
\[ x^2 = 0.0217 \times 0.7225 \]
\[ x^2 = 0.01567825 \]
\[ x = \sqrt{0.01567825} \]
\[ x \approx 0.1252 \]
Therefore, the concentration of I2 at equilibrium is 0.125 mol dm–3 (to 3 significant figures).
(c)(i) At the lower temperature (500 K), the equilibrium mixture will have a lower concentration of H2 and I2 (and a higher concentration of HI) compared to the mixture at 764 K.
Explanation: The value of the equilibrium constant Kc decreases from 0.0217 (at 764 K) to 0.00625 (at 500 K). A smaller Kc means the position of equilibrium lies further to the left, favouring the reactants (HI) over the products (H2 and I2).
(c)(ii) The decomposition of HI is endothermic.
Explanation: According to Le Chatelier’s principle, if a system at equilibrium is subjected to a change, the system will shift to counteract that change. When the temperature is decreased from 764 K to 500 K, the equilibrium shifts towards the left (towards the reactants, HI), as indicated by the smaller Kc value. This means the reverse reaction (the formation of HI from H2 and I2) is exothermic. Therefore, the forward reaction (the decomposition of HI) must be endothermic. A decrease in temperature favours the exothermic direction.
Topic: 18.2
Y is formed from X in a single-step reaction, as shown in Fig. 5.1.
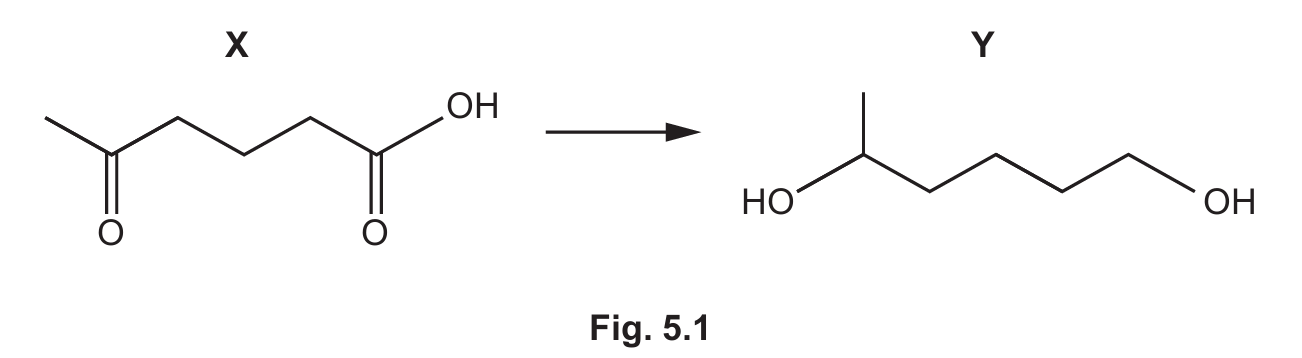
(a) Deduce the empirical formula of Y.
(b) The formation of Y from X requires the addition of a suitable reducing agent.
(i) Construct an equation using molecular formulae and [H] for the reaction in Fig. 5.1. Use [H] to represent one atom of hydrogen from the reducing agent.
(ii) Identify a suitable non-gaseous reducing agent for the formation of Y from X.
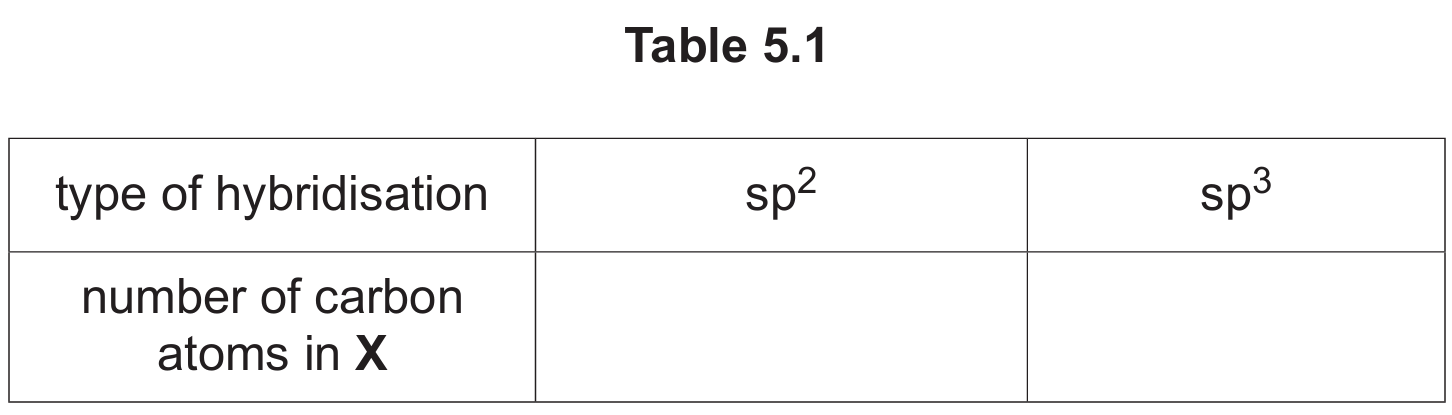
(c) Complete Table 5.1 to show the number of sp2 and sp3 hybridised carbon atoms in a molecule of X.
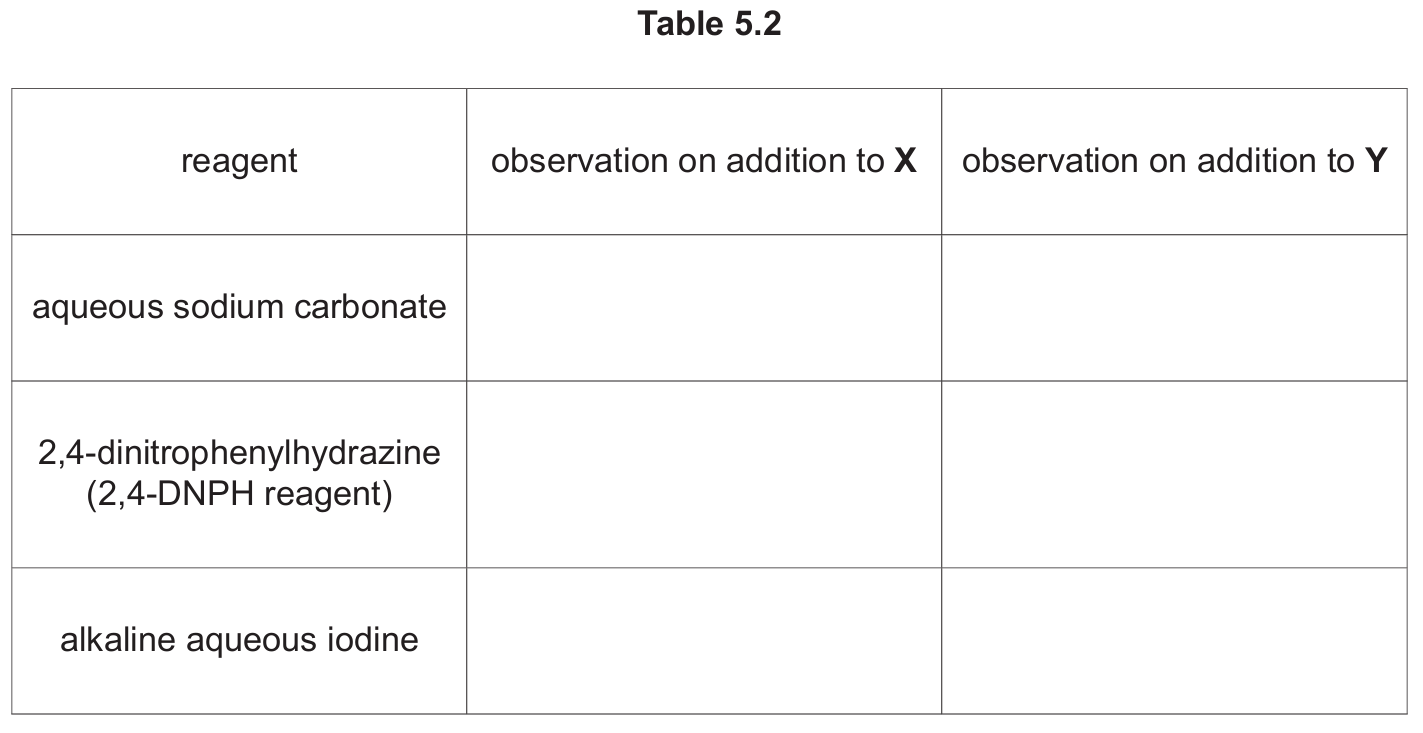
(d) Complete Table 5.2 with the expected observations that occur when the reagents shown are added to separate solutions of X and Y. Do not refer to temperature changes in your answer.
▶️ Answer/Explanation
(a) C6H12O
Explanation: To find the empirical formula, we need to determine the simplest whole number ratio of atoms in compound Y. Looking at the molecular structure of Y (which would be shown in Fig. 5.1), we can count the atoms: 6 carbon atoms, 12 hydrogen atoms, and 1 oxygen atom. The ratio is already in its simplest form (6:12:1), so the empirical formula is C6H12O.
(b)(i) C6H10O3 + 6[H] → C6H12O2 + H2O
Explanation: The reaction involves the reduction of compound X (C6H10O3) to form compound Y (C6H12O2). We use [H] to represent one hydrogen atom from the reducing agent. Comparing the molecular formulas, we see that X has 2 fewer hydrogen atoms and 1 more oxygen atom than Y. This suggests a reduction process where the oxygen is removed as water. To balance the equation, we need 6 hydrogen atoms from the reducing agent, resulting in the formation of one water molecule.
(b)(ii) LiAlH4 (lithium aluminium hydride) in dry ether
Explanation: Lithium aluminium hydride is a powerful reducing agent commonly used in organic chemistry for reducing carbonyl compounds. It’s particularly suitable here because it’s a non-gaseous reducing agent that can provide the necessary hydride ions (H–) for the reduction process. The reaction is typically carried out in anhydrous conditions like dry ether to prevent the reducing agent from reacting with water.
(c)

Explanation: To determine hybridization, we examine each carbon atom in compound X. Carbon atoms with double bonds are sp2 hybridized, while those with only single bonds are sp3 hybridized. In the structure of X (which would be shown in Fig. 5.1), we can identify 2 carbon atoms that are part of carbonyl groups (C=O) or other double bonds, making them sp2 hybridized. The remaining 4 carbon atoms are saturated with single bonds only, making them sp3 hybridized.
(d)
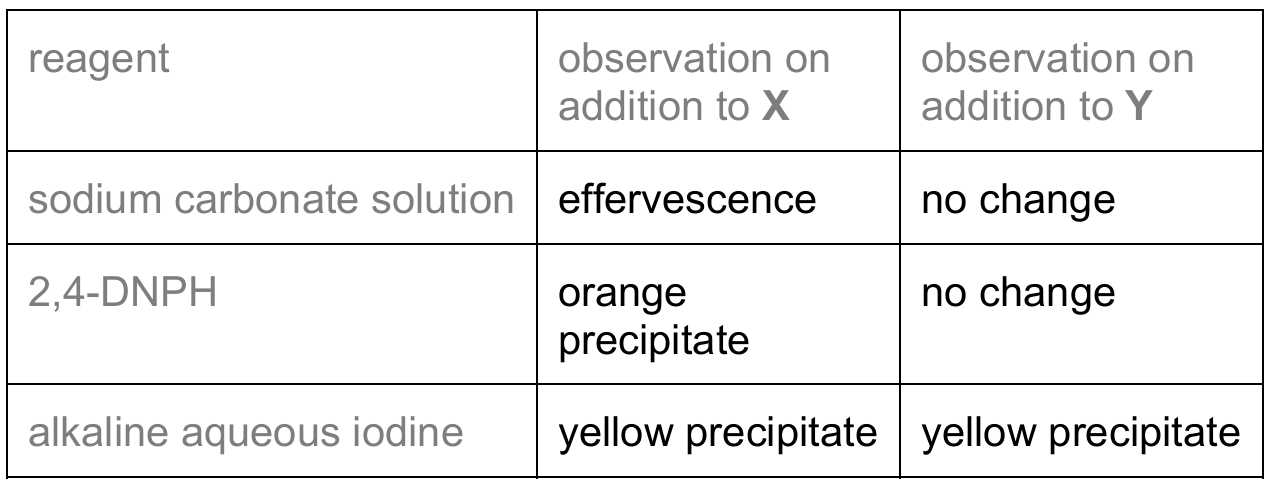
Explanation:
Aqueous sodium carbonate: X shows effervescence because it contains carboxylic acid groups that react with carbonate to produce CO2 gas. Y shows no change because it lacks acidic protons and doesn’t react with carbonate.
2,4-DNPH reagent: X gives an orange precipitate because it contains carbonyl groups (C=O) that react with 2,4-DNPH to form hydrazones. Y shows no change because it lacks carbonyl groups after reduction.
Alkaline aqueous iodine: Both X and Y give yellow precipitates because they contain methyl ketone or acetaldehyde groups that undergo the iodoform reaction, producing yellow iodoform precipitate.
Topic: 20.1
Compound W, CH2=CHCN, is used to make an addition polymer which is present in carbon fibres.
(a) Draw one repeat unit of the addition polymer of W.
(b) CH3CHO is used in a two-step synthetic route to form W, as shown in Fig. 6.1. In step 1, CH3CHO is heated with HCN in the presence of KCN.
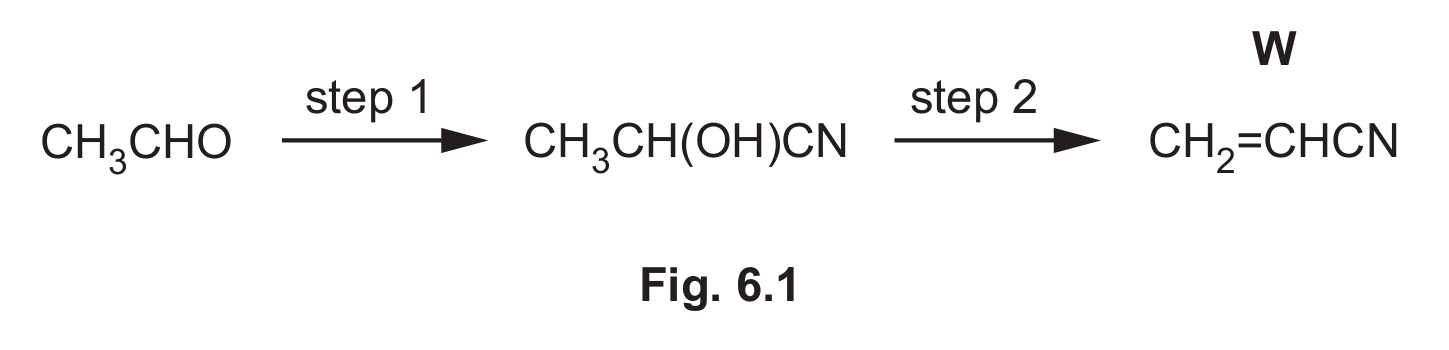
(i) Name the mechanism for the reaction in step 1 in Fig. 6.1.
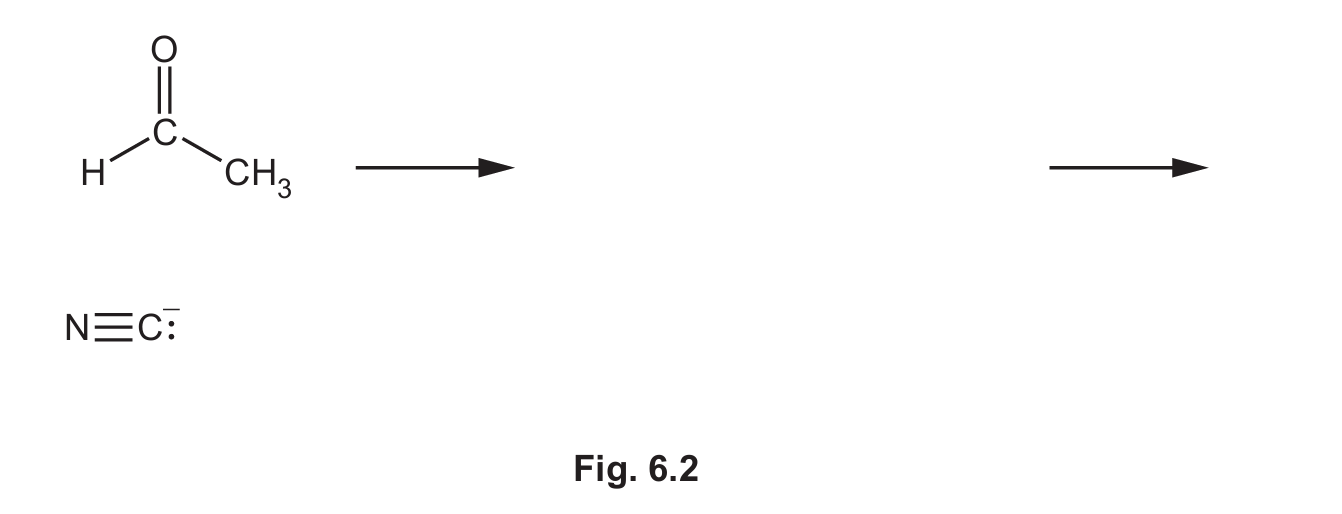
(ii) Complete Fig. 6.2 to show the mechanism for the reaction in step 1. Include all products, charges, dipoles, lone pairs of electrons and curly arrows, as appropriate.
(iii) Suggest a suitable reagent and conditions for step 2 in Fig. 6.1.
(iv) Fig. 6.3 shows the infrared spectrum of W, CH2=CHCN.
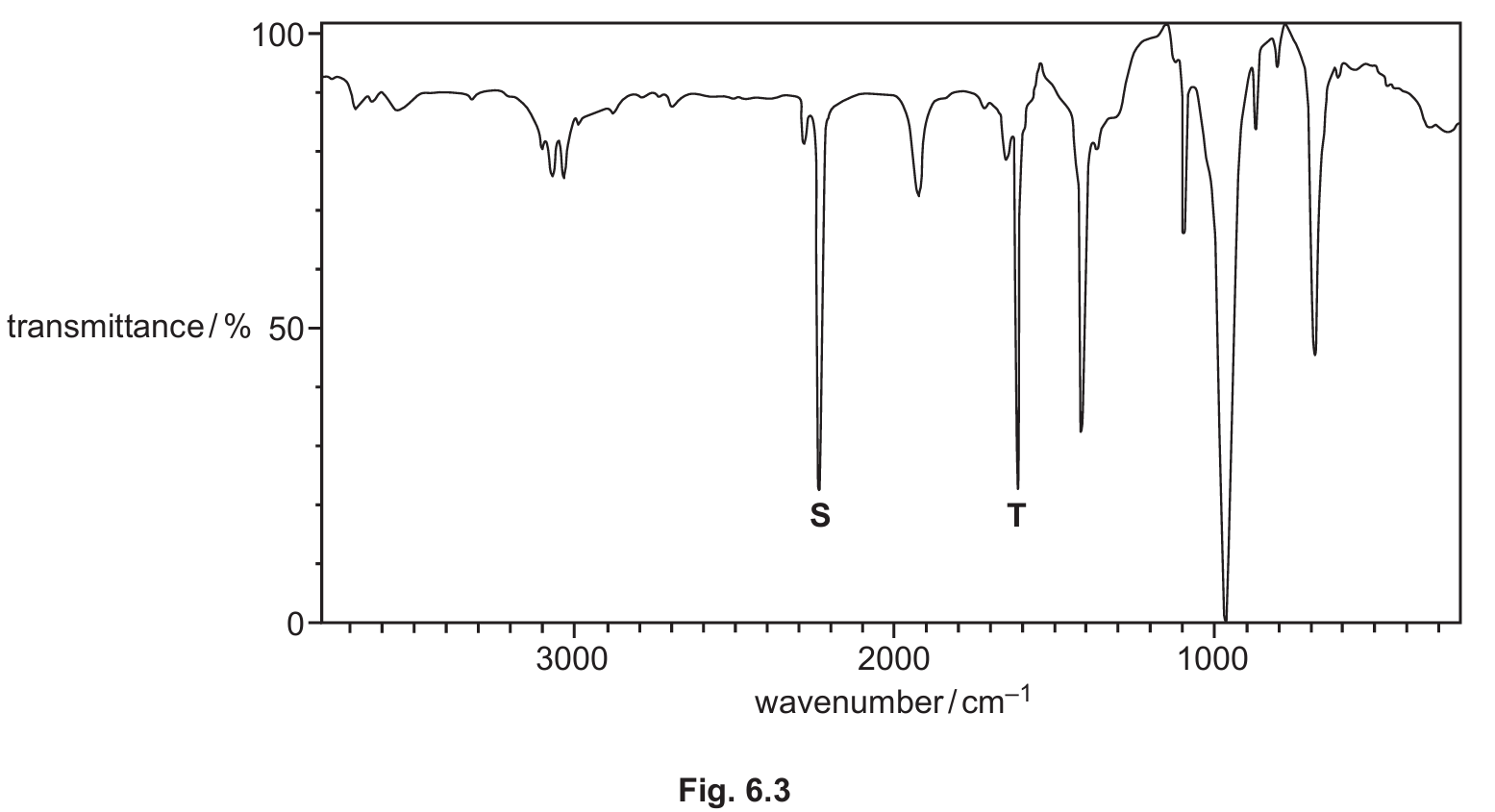
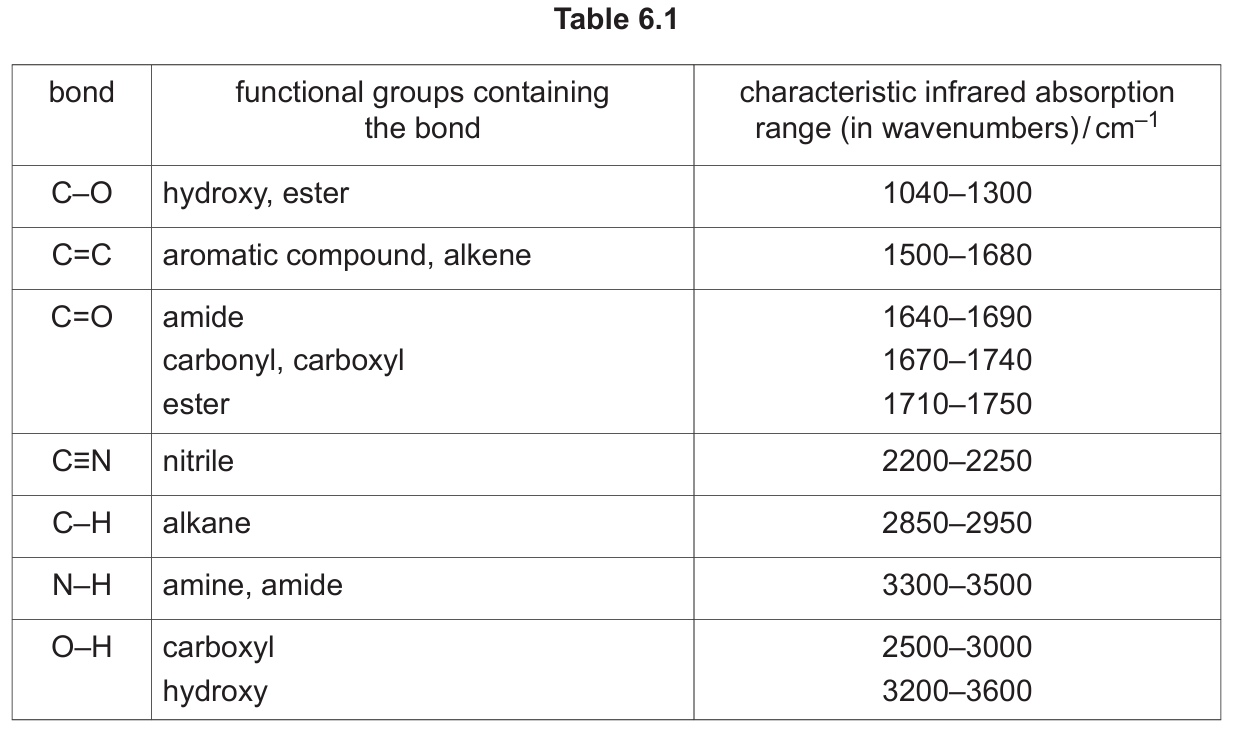
Use Table 6.1 to identify the bonds responsible for the absorptions marked S and T on Fig. 6.3.
(c) Molecules of W, CH2=CHCN, do not show stereoisomerism.
(i) Describe stereoisomerism.
(ii) Describe the two essential features of an alkene molecule that cause it to show geometrical stereoisomerism.
(d) Molecules of CH3CH(OH)CN exist as a pair of optical isomers.
Draw three-dimensional diagrams in the boxes to show the optical isomers of CH3CH(OH)CN.

(e) Propanenitrile is heated with hydrogen gas and a platinum catalyst. The only product is propylamine.
Construct an equation for this reaction.
(f) Propylamine can also be formed in a two-step synthesis from propan-1-ol, as shown in Fig. 6.4.
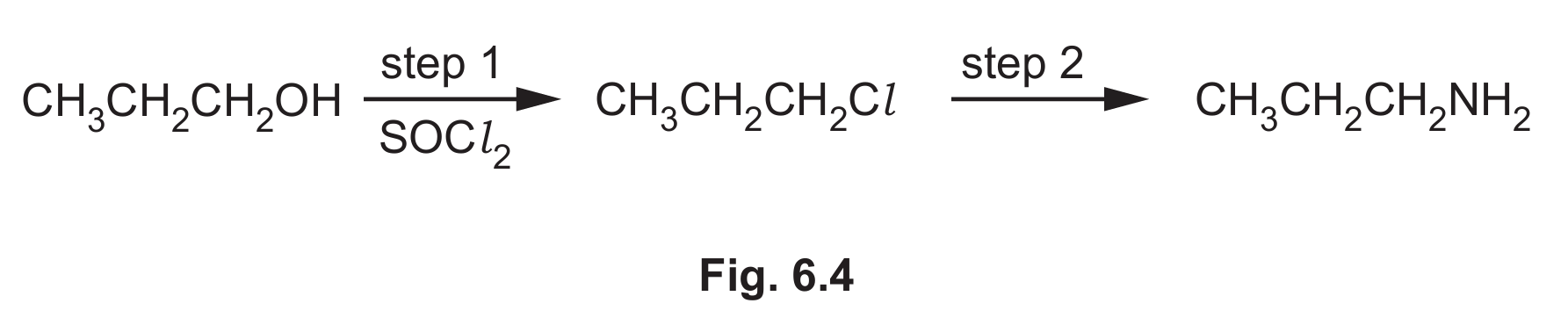
(i) Name the type of reaction in step 1 in Fig. 6.4.
(ii) Identify the reagent and conditions for step 2 in Fig. 6.4.
▶️ Answer/Explanation
(a)
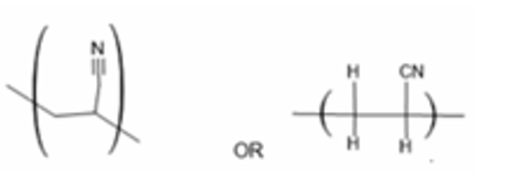
One repeat unit: \(\text{-}[\text{CH}_2\text{-CH}(\text{CN})]\text{-}\)
Explanation: The addition polymer of CH2=CHCN (acrylonitrile) is polyacrylonitrile (PAN). The double bond opens up, and the monomers add together. The repeat unit is derived from the monomer by replacing the double bond with a single bond and extending the bonds on either side.
(b)(i) Nucleophilic addition
Explanation: The reaction involves the nucleophile cyanide ion (:CN–) attacking the electrophilic carbon of the carbonyl group (C=O) in ethanal. This is the defining characteristic of a nucleophilic addition reaction.
(b)(ii)
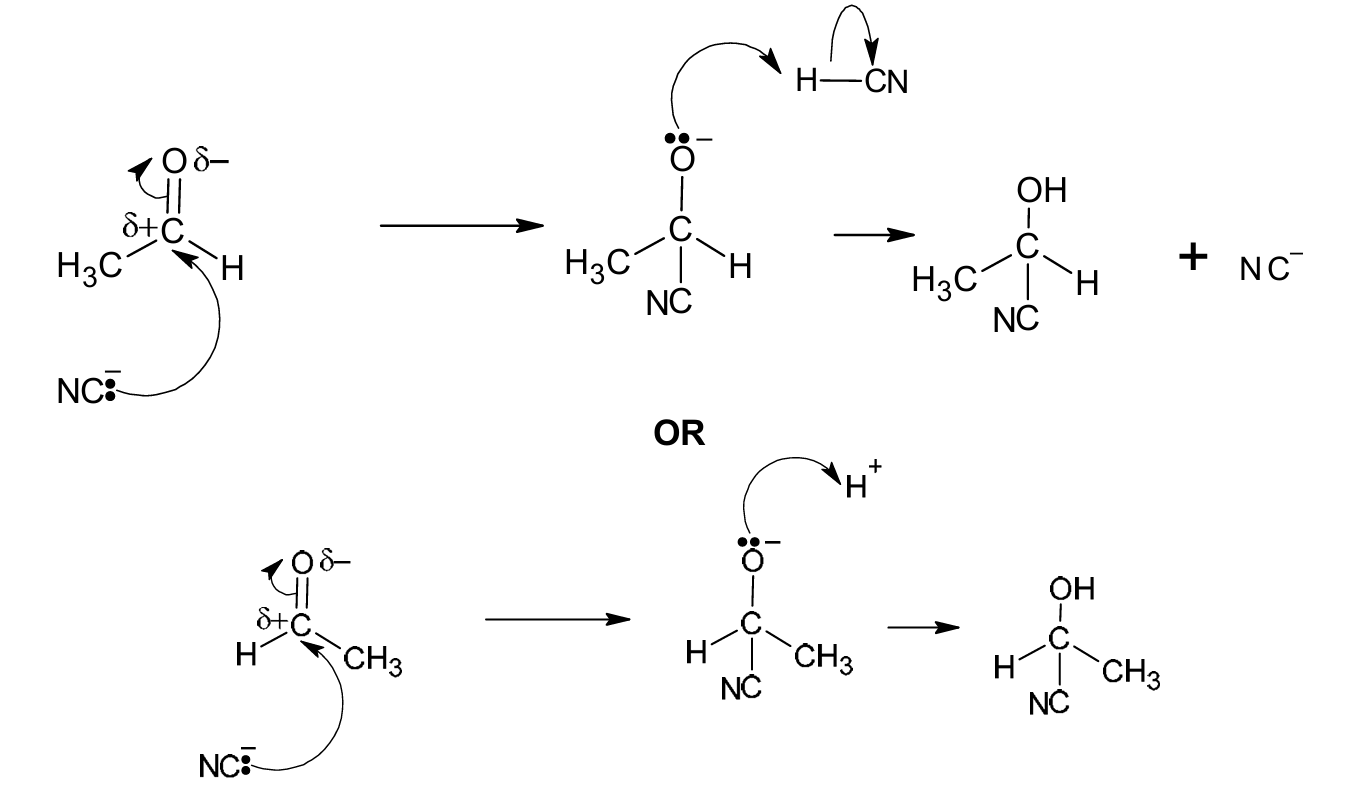
Explanation: The mechanism involves three key steps:
1. (M1) A curly arrow must show the lone pair from the carbon of the cyanide ion (the nucleophile) attacking the δ+ carbon atom of the polar carbonyl group. A dipole must be shown on the C=O bond (δ+ on C, δ- on O).
2. (M2) This forms a tetrahedral intermediate with a negative charge on the oxygen atom.
3. (M3) A curly arrow must show a lone pair from the negatively charged oxygen atom abstracting a proton (H+) from the environment (often represented as HCN or H3O+), resulting in the final product, 2-hydroxypropanenitrile (CH3CH(OH)CN).
(b)(iii) Reagent: Concentrated sulfuric acid OR concentrated phosphoric acid OR Al2O3 catalyst. Condition: Heat.
Explanation: Step 2 is a dehydration reaction, removing a molecule of water from the hydroxy-nitrile (CH3CH(OH)CN) to form the unsaturated nitrile (CH2=CHCN). Strong acids like H2SO4 or H3PO4 catalyze this elimination, as does heated aluminium oxide.
(b)(iv) S: C≡N AND T: C=C
Explanation: Referring to Table 6.1, the nitrile group (C≡N) has a characteristic strong, sharp absorption in the range 2200–2250 cm-1. The alkene group (C=C) has an absorption in the range 1500–1680 cm-1. These are the two key functional groups in the structure of W (CH2=CHCN).
(c)(i) Stereoisomerism is when molecules/compounds have the same structural formula (and same molecular formula) but a different arrangement of atoms/groups in space.
Explanation: This is the fundamental definition. Structural isomers have different atom connectivity, whereas stereoisomers have the same connectivity but differ in how their atoms are oriented in three-dimensional space.
(c)(ii)
1. (M1) There must be restricted rotation around the C=C double bond.
2. (M2) Each carbon atom involved in the double bond must have two different atoms or groups attached to it.
Explanation: The double bond’s pi bond prevents free rotation, locking the structure. If both carbons have two identical groups (e.g., in ethene, H2C=CH2), rotating the ends doesn’t create a distinct isomer. For geometrical isomerism (E/Z isomerism) to exist, each carbon of the double bond must have two different substituents, allowing for two distinct spatial arrangements (e.g., the groups can be on the same side, cis/Z, or opposite sides, trans/E).
(d)
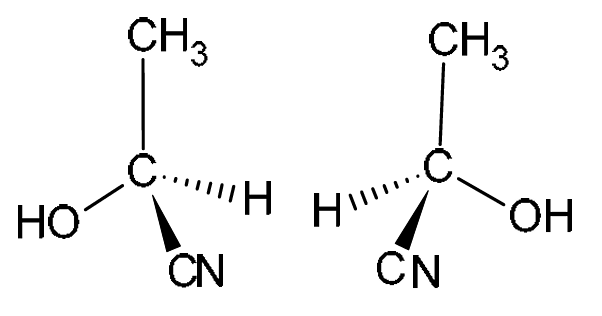
Explanation: CH3CH(OH)CN (2-hydroxypropanenitrile) has a chiral center (the carbon atom with the H, OH, CH3, and CN groups attached). This means it can exist as two non-superimposable mirror images. The 3D diagrams must show this, using wedges (coming out of the plane) and dashed lines (going into the plane) to represent the tetrahedral arrangement around the chiral carbon. The two isomers must be mirror images of each other.
(e) \(\text{CH}_3\text{CH}_2\text{CN} + 2\text{H}_2 \rightarrow \text{CH}_3\text{CH}_2\text{CH}_2\text{NH}_2\)
Explanation: This is a reduction reaction. The nitrile group (-C≡N) is reduced to a primary amine group (-CH2-NH2) using hydrogen gas and a metal catalyst like platinum (Pt), nickel (Ni), or palladium (Pd). Two moles of hydrogen are required for this conversion.
(f)(i) Substitution
Explanation: In step 1, the hydroxyl group (-OH) of propan-1-ol is replaced (substituted) by a chlorine atom from SOCl2 (thionyl chloride) to form chloro propane. This is a nucleophilic substitution reaction.
(f)(ii)
Reagent: Ammonia (NH3)
Conditions: Heat, and the reaction is often carried out in a sealed tube/under pressure or in ethanol as a solvent.
Explanation: Step 2 is another nucleophilic substitution reaction. The ammonia molecule (a nucleophile) attacks the chloroalkane, displacing the chloride ion and forming a primary amine (propylamine). Excess ammonia is often used to minimize further substitution (e.g., forming secondary and tertiary amines). Heating is required to overcome the activation energy for this substitution, and performing the reaction under pressure or in a sealed tube helps contain the reactants, especially since ammonia is a gas.
